2-Chloroethanol: A Chemical with a Complex Legacy and Future
Looking Back: How 2-Chloroethanol Entered the Modern World
Most people don’t spend much time thinking about the story behind a single chemical, but 2-chloroethanol has followed a path worth knowing. Early chemists pursuing ways to transform simple alcohols into more reactive compounds found that introducing chlorine into the molecule could unlock a host of new reactions. Industrial production scaled up in the first half of the twentieth century, as markets for textiles, solvents, and insecticides expanded. The innovation came with risks and repercussions. At several points across the decades, researchers and workers faced unexpected episodes of exposure, spurring real changes in how manufacturing safety was handled. Governments responded with regulations. Industries gradually pivoted toward cleaner, safer technologies. That history continues to teach lessons about unintended consequences and the enduring tension between progress and safety.
The Chemical in a Nutshell
2-Chloroethanol appears as a clear, colorless liquid with a faint, sometimes unpleasant odor. The chemical formula, C2H5ClO, hints at its structure: two carbons, five hydrogens, a chlorine, and an oxygen holding it all together. What jumps out in the lab is how readily it mixes with water and organic solvents, lending flexibility to its use in both aqueous and non-aqueous systems. Its boiling point sits just above water’s, and with a density higher than pure ethanol, it signals its unique mix of alcohol and halogen properties. The molecule’s reactive sites invite chemists to push the boundaries of chemical synthesis, though working with it demands respect because of both its chemical potential and health impact.
Getting Specific: Technical Details and Labels
In daily operations, purity and concentration always come under scrutiny. Reliable suppliers deliver 2-chloroethanol with tight margins on contaminants like water or aldehydes, reflecting the serious need for consistency in industrial or research environments. Clear, durable labeling communicates the dangers as well as specifications. Each container leaves little ambiguity: the words “toxic” and “harmful if inhaled or absorbed through skin” remind everyone that complacency leads to accidents. These handling guidelines have emerged not from theory, but from hard experience in plants and labs. Standards bodies have codified these rules to keep workers safe and processes predictable, but even so, vigilance is never optional.
From Synthesis to Shelf: How It’s Made
Most production routes center around either ethylene oxide or ethylene chlorohydrin as the critical starting point. One method runs ethylene through a reaction with chlorine and water, producing the target compound in a pretty direct fashion. Another common path modifies ethylene oxide with hydrogen chloride gas, opening a different set of operational parameters but yielding essentially the same product. Plant operators balance cost, energy use, and byproduct management to decide which approach makes the most sense in a given context. The chemical industry’s shift toward greener chemistry has nudged producers to cut down on waste and improve capture of byproducts, though perfection remains elusive.
Reactivity: Expanding Possibilities and Hazards
Chemists value 2-chloroethanol both as a reacting agent and as a springboard to build more complex molecules. The chlorine atom is an open invitation for substitution reactions, while the alcohol group gives it a hydrophilic punch. Through these pathways, it plays a role in synthesizing plasticizers, pharmaceuticals, herbicides, and dyes. This versatility feeds both innovation and risk, since those same reactive properties mean it interacts with moisture, bases, and nucleophiles in sometimes unpredictable ways. The hazards shine a spotlight on the importance of secure storage, leak prevention, and containment. Those practical concerns overshadow any theoretical curiosity in the daily grind of real-world chemistry.
What’s in a Name? More Than Just 2-Chloroethanol
2-Chloroethanol appears in records and catalogs under several synonyms. Take “ethylene chlorohydrin”—a name that hints at its dual heritage from ethylene chemistry and chlorinated solvents. Some labels will also show “beta-chloroethanol,” “ethylene glycol monochlorohydrin,” or “chloroethyl alcohol.” Each moniker brings its own baggage, with certain names more common in the textile industry, others in agriculture or laboratory supply chains. This tangle of synonyms doesn’t just confuse new handlers; it underscores the spread of the compound across a spectrum of applications and industrial traditions.
Raising the Bar: Safety and Daily Practice
Working closely with 2-chloroethanol tests every safety protocol set down in chemical handling guides. Acute toxicity shows up quickly. Unprotected skin, unfiltered air, or accidental spills can produce headaches, dizziness, nausea, or worse. Chronic exposure raises the stakes. Health authorities have flagged its potential to cause cancer in animal studies, sparking a drive for tight occupational exposure limits and heavy investment in personal protective equipment. Plant managers now mandate chemical-resistant gloves, goggles, fume hoods, and closed systems. Emergency response plans don’t gather dust, and training sessions underline the stakes for both personal and community well-being. That level of care came from hard lessons learned over too many years, a living record of small and large tragedies.
The Reach of 2-Chloroethanol: Applications Old and New
This chemical flows into more products and processes than most people realize. The textile industry leans on it for cross-linking agents and dye intermediates. Some pharma labs rely on it to help build antiviral drugs or anesthetic compounds. Makers of pesticides turn to its chemical reactivity to assemble molecules that persist in the environment or break down with a well-timed trigger. Even in research labs, it serves as a crucial test case for exploring new pathways in organic chemistry. Its role in these fields highlights both the ingenuity of industrial design and the continuing puzzle of legacy chemical footprints.
The Ongoing Challenge: Research and Refinement
Researchers keep pushing the boundaries, searching for safer alternatives, greener synthetic strategies, and better ways to destroy or recycle 2-chloroethanol once its job is done. Analytical chemists have homed in on sensitive detectors to track minute quantities in the environment. Toxicologists dig deeper into the mechanisms of cell damage, aiming to identify biomarkers that flag exposure long before symptoms appear. Process engineers are busy designing equipment that seals away vapors and cuts down on waste. The drive toward “benign by design” influences both government funding and private investment. Yet, legacy uses linger, and the chemical’s utility means it won’t disappear overnight.
Living with Risk: What We Know About Toxicity
Toxicity research tells a tough story. Work in rodents shows that even short-term exposure can lead to liver and kidney effects, while high levels quickly cross over into fatal territory. In humans, acute exposures most often happen through skin contact or inhalation—each carrying its own risks, from mild irritation up to convulsions or coma in the worst-case scenarios. These findings triggered worldwide legal limits on workplace levels and relentless campaigns to reduce unintentional releases into air or water. As portable testing gear and online monitoring improve, the gap between identifying, measuring, and controlling exposure continues to shrink. But the drive to keep workers, neighbors, and customers safe demands never-ending vigilance.
Looking Ahead: A Chemical in Transition
Change is probably the best word for the future of 2-chloroethanol. The chemical industry is not blind to the damage older processes have done, and new legal frameworks push every producer toward cleaner, safer approaches. Environmental activists keep up the pressure, sparking innovation in how waste streams are treated and driving investment into replacements for compounds with difficult safety histories. Some companies are investigating enzymatic pathways and biomass-based feedstocks to reduce reliance on chlorine and ethylene. Others develop targeted destruction methods that turn leftover 2-chloroethanol into safer byproducts. As consumer demand for transparency grows, traceability and full life-cycle assessments have started steering research priorities. In this landscape, 2-chloroethanol stands as a symbol: useful, potentially dangerous, and constantly evolving under public, scientific, and industrial scrutiny.
What is 2-Chloroethanol used for?
Real-World Roles of 2-Chloroethanol
2-Chloroethanol finds its place in some corners of chemical manufacturing where not many people think to look. Factories use it to make glycol ethers. I worked summers in a paint lab in college, and managers talked about solvents that help paints dry smooth and spread even color. Glycol ethers gave that balance of fast evaporation and proper texture, and much of it starts from 2-chloroethanol. These ethers go into cleaners, varnishes, even printing inks, making daily items perform better.
Its other life comes in producing ethylene oxide. This one, I’ve noticed, runs deep in industry. Ethylene oxide spins off into antifreeze, detergents, polyester, and some foams. Companies want pure starting chemicals, and 2-chloroethanol often gives that punch because it’s highly reactive. It allows chemists to steer molecules into something valuable. Chemical safety data from the CDC and NIOSH show how carefully operators watch this process, knowing the hazards in case anything leaks.
Beyond Factories—Old and New Uses
Pharmaceutical production has leaned on 2-chloroethanol for decades, mostly as a starter for making specialty drugs or as a piece in reaction chains. I saw a case in the late 2010s, hearing from a friend in pharma, where tiny batches led into heart drug research. Old textbooks mention agricultural chemicals too: 2-chloroethanol covered seeds as a fungicide long ago, though tighter safety laws pushed those uses aside. Its presence in specialty adhesives sometimes pops up in small electronics, where glue has to bind components that heat up and cool down without fail.
Risks Hidden in Clean Labs
The bad side doesn’t hide for long. 2-chloroethanol can irritate skin, damage eyes, and, in the wrong hands, cause serious harm through inhalation. Accident reports show workers in specialty chemical plants wear thick gloves and heavy hoods for a reason. My lab’s chemical inventory ran training every quarter to make sure no one mixed this one up with safer alternatives. Public health records trace some environmental spills, highlighting risks for groundwater and aquatic life. No wonder environmental teams keep tight logs if they handle any.
Room for Safer Progress
The grip of 2-chloroethanol over chemical supply chains keeps research centers busy hunting for safer or greener options. More companies test out bio-based solvents or focus on closed-loop systems that trap vapor before it hits the air. I’ve read pilot reports from university labs, showing that less toxic substitutes can sometimes drop right into solvent formulas. Proper ventilation, better leak sensors, and strong workplace protocols turn up in many industry best practices. If companies and regulators work with insight from real-world spills and accidents, industries can keep their output high without feeding extra risk to workers or neighbors.
Looking Further
2-Chloroethanol reminds us that behind modern products live chemicals carrying both value and risk. My years in and around labs point toward vigilance, innovation, and open sharing of best practices as keys to safe use. Industry progress will depend on honest reviews, creative experiments, and clear communication so coming generations meet higher standards by choice, not just regulation.
What are the hazards and safety precautions for handling 2-Chloroethanol?
Understanding the Risks
2-Chloroethanol often shows up in chemical manufacturing and research labs because of its role as a building block for other chemicals. It looks harmless at a glance, but this liquid brings plenty of serious health and safety concerns right into the workplace. Take it from anyone who has handled volatile liquids: this one demands respect.
Health risks sit front and center with 2-Chloroethanol. It's got a reputation for being toxic. Swallowing even a small amount or breathing in its vapors can quickly lead to headaches, dizziness, confusion, and—at higher concentrations—hard-hitting impacts like convulsions or respiratory failure. Prolonged skin contact has left plenty of workers regretting any slip-up, with stories of severe burns, rashes, and blisters. The eyes stand no chance, burning instantly with just a splash. It's absorbed right through the skin, so simple contact adds up to a real hazard.
Turning to its flammability, 2-Chloroethanol doesn't waste time catching fire. An open flame, static electricity, or even a spark from machinery can kick off a nasty blaze that spreads fast. It also gives off toxic fumes when heated or burned, so fighting such fires is no picnic for any crew on site.
Best Practices in Direct Handling
Pouring, measuring, or transferring 2-Chloroethanol asks for a clear plan and strict attention to detail. I’ve seen the best results in labs that use fully sealed containers, chemical fume hoods, and clear labeling on every bottle or drum. Workers who take shortcuts end up with exposure incidents or accidental spills, and those mistakes hang heavy over their careers.
Spills happen in real labs, so nobody shrugs off the importance of having absorbent materials and neutralizing agents nearby. Standard soap and water do nothing here—specialty spill kits built for hazardous chemicals belong at arm’s reach, and staff should actually know how to use them. Learning cleanup procedures in calm training sessions saves panic during an emergency.
Personal Protective Equipment
A lab coat and basic gloves just don’t cut it with 2-Chloroethanol. Chemical-resistant gloves—like those made from butyl rubber—keep this liquid from seeping through. Full splash goggles protect eyes from vapors and splashes better than safety glasses. Long sleeves, impervious aprons, and closed-toe shoes round out the right gear. It helps to have an eyewash station and safety shower right nearby, and checking that these stations work should happen before any work begins for the day.
Engineering Controls and Workplace Habits
Ventilation plays a huge role. I’ve worked in spaces that made every use of ducted chemical hoods, giving fumes no chance to build up. Places that settle for open windows or simple fans usually pay for that decision in the long run. Regular air quality checks and clear walkways reduce trips, falls, and the risk of exposure to vapors that sometimes settle lower to the floor.
Secure storage keeps this chemical from causing more trouble. Manufacturers ship 2-Chloroethanol in tightly sealed bottles—storing those in cool, well-labeled cabinets away from oxidizers and acids prevents dangerous reactions. Double-checking shelf labels and chemical compatibility goes well beyond compliance; everyone remembers the time a leaky cap led a rookie to grab the wrong bottle, and luckily, double-checking saved the day.
Training and Emergency Response
Solid training builds a strong safety culture. Workers need to recognize the symptoms of exposure to 2-Chloroethanol and understand emergency procedures without having to fumble through manuals in a crisis. Fire drills and spill response exercises should be as routine as morning coffee in settings where this chemical sees regular use. If something does go sideways, having external emergency contacts already posted and memorized shortens response times.
Reducing Risk Over Time
Good recordkeeping, clear incident reporting, and frequent inspections strengthen workplace protection against 2-Chloroethanol. Substituting less hazardous chemicals whenever possible makes a long-term impact, but in cases where its use remains critical, steady attention, thoughtful engineering, and trained hands make daily operations much safer for everyone in the room.
What is the chemical formula and structure of 2-Chloroethanol?
The Formula Behind the Name
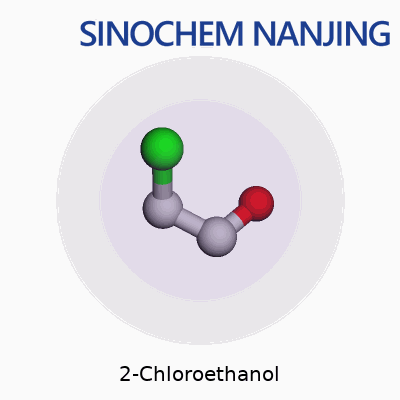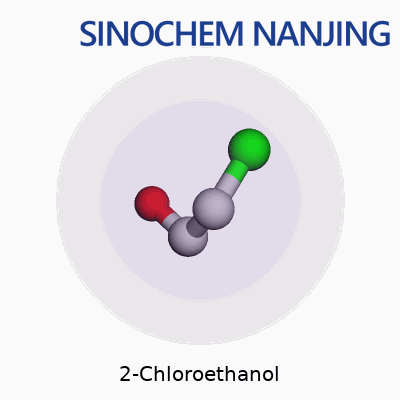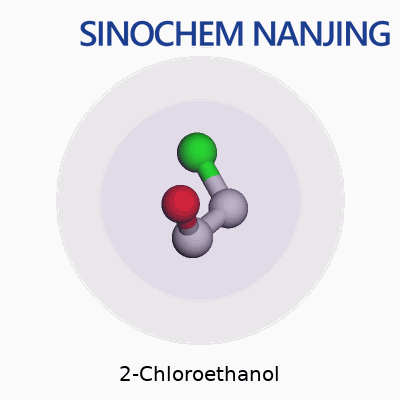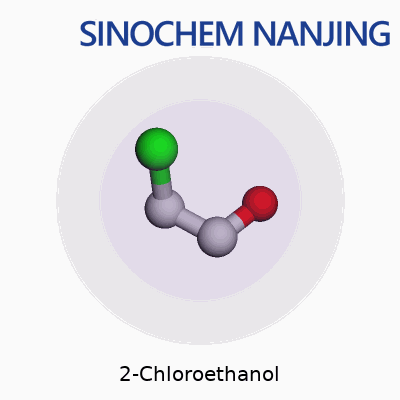
2-Chloroethanol carries the chemical formula C2H5ClO. On paper, it looks like a small combination of carbon, hydrogen, chlorine, and oxygen atoms. In practice, it plays a bigger role in chemistry and industry than people often realize. The molecule’s core structure places a chlorine atom and a hydroxyl group on a two-carbon chain. Written out, you’d see it as HOCH2CH2Cl, showing both the alcohol and chlorine functional groups clearly.
The molecule’s structure isn’t only important to chemists. Many folks working in labs become familiar with 2-Chloroethanol’s place in organic synthesis early in their careers. The molecule acts as a gateway—its dual nature as both an alcohol and halide means it reacts with a wide variety of other chemicals. You often find it in textbooks as an intermediate for synthesizing dyes, pharmaceuticals, and certain plasticizers. Its chemical makeup lends it flexibility for building larger, more complex compounds.
Hands-On Lessons: Safety and Effects
Experience in a university research lab taught me to respect 2-Chloroethanol’s hazards. The very structure that makes it useful also makes it dangerous. Exposure through skin or inhalation can affect the nervous system or lead to respiratory problems. This risk comes directly from the molecule’s ability to disrupt biological pathways, not unlike similar chlorinated alcohols. The Centers for Disease Control and Prevention (CDC) labels it as toxic, and proper safety training always includes strict protocols for storage, use, and disposal.
Its toxicity means facilities rely on good ventilation and personal protective equipment to keep workers safe. Once, during a routine cleanup, a small spill led to evacuation until it was dealt with. These are the sorts of variables that only seem abstract until you see colleagues reacting to a real-world hazard. Learning by doing clears up how theory connects to real safety stakes.
Industrial Uses and Oversight
Many industries see 2-Chloroethanol as a workhorse. It’s a building block for producing ethylene oxide and ethylene glycol, both essential in making antifreeze and polyester fibers. Demand for these products puts pressure on plants to keep large supplies on hand. To limit environmental impact, producers need robust containment and treatment systems. Accidental release into water sources, for example, could impact aquatic life or make its way into drinking water if not handled with care.
Strict regulation helps keep misuse in check. The United States Environmental Protection Agency (EPA) keeps a close watch, and compliance officers frequently monitor for safe disposal and proper reporting. In the European Union, classification under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) forces companies to register and assess risks thoroughly before using or selling it in bulk.
Better Solutions and Safer Practices
The chemical sector has started exploring less hazardous substitutes and greener processes, but in some reactions, 2-Chloroethanol remains unmatched. Whenever possible, process engineers look for modifications that capture emissions, treat wastewater, and recycle by-products. Simple steps, like implementing double-walled storage tanks and thorough employee safety training, reduce risks without sacrificing productivity. In academic settings, teaching responsible handling and disposal builds a culture of safety from the ground up.
My own experiences led me to appreciate that chemistry never operates in a vacuum. Every structure, like the arrangement in 2-Chloroethanol, carries ripple effects—from the molecular scale to the community surrounding a plant. Attention to both technical details and daily practice makes the difference between safe use and dangerous incident.
How should 2-Chloroethanol be stored and disposed of?
Facing the Real Risks: Why 2-Chloroethanol Matters
If you’ve worked in a chemistry lab, you’ve probably heard about 2-chloroethanol. This stuff doesn’t mess around. It’s clear, it smells strong, and it turns dangerous without much warning. What hits home with me is how just a tiny slip-up can lead to some serious health scares. The Centers for Disease Control calls it toxic if inhaled, swallowed, or touched. At one facility where I interned, a spilled bottle sent an employee to urgent care after just a few seconds of exposure.
Storing and disposing of this compound isn’t just another checkbox for compliance paperwork. It’s more like having a loaded mousetrap on your workbench—don’t look away, and don’t let down your guard. Let’s talk about doing this right and about the habits that truly keep people safe.
Safe Storage: Details That Save Lives
Two words matter most here: isolation and ventilation. The best chemists I know don’t just stack 2-chloroethanol in any cabinet. They pick steel safety cabinets, away from direct sunlight and sparks, and keep it locked up tight. Moisture and heat speed up reactions, and this chemical doesn’t need much encouragement to go from stable to explosive. One poorly grounded fridge in a back room can ignite vapors, so only explosion-proof refrigerators make the cut for cold storage.
Labeling counts, too. Big, clear, chemical-resistant labels mean nobody grabs the wrong bottle. Spills happen quickest after someone fumbles with faded names or hand-scrawled tape. Safety always leans on solid labeling and regular inspections. A shelf that’s too full, or bottles without a date, makes mistakes all but certain.
Handling Disposal Without Regret
Throwing away 2-chloroethanol takes more thought than an average lab solvent. It doesn’t go down the drain or in the trash. According to the Environmental Protection Agency, this chemical qualifies as hazardous waste under federal law. In my college lab, every drop got poured into a specifically marked waste jug. Miss the mark, and you’re risking fines, fires, and worse.
Finding the right disposal service pays off. Reputable hazardous waste companies handle pick-up and incineration. Skipping steps or using corner-cutting contractors rarely ends well. Stories surface every year of labs fined six figures for illegal dumping or improper paperwork. These aren’t rare errors—they’m the fallout from rushing or ignoring the details.
Training and Habit Build a Culture of Safety
No sign or sticker replaces training. Old-timers at my first job made it a rule: every new chemist practiced incident response for spills and leaks. During a drill, someone tipped a bottle into a spill tray—by mistake. The response showed who knew their stuff and who needed a refresher. Watching people scramble burns in the lesson: know the safety shower’s spot, and never handle this chemical alone.
The Real Solution: Respect and Routine
Respect comes from routine, not just rules. Safe storage cuts risk, and strict disposal finishes the job. Any shortcut ends up being somebody’s problem down the line. Keeping 2-chloroethanol under control, protecting air quality, and getting everyone from student to senior chemist on the same page means fewer scary stories—and better health outcomes. The best labs fix problems before accidents take away that option.
Is 2-Chloroethanol toxic or carcinogenic?
The Nature of 2-Chloroethanol
2-Chloroethanol pops up in the chemical industry, especially during the manufacture of dyes, pharmaceuticals, and certain pesticides. It looks like a clear liquid with a noticeable smell, and it carries some risk for workers who deal with it often. Questions come up about its safety, especially its potential toxicity and carcinogenicity.
Toxic Effects Seen in Practice
2-Chloroethanol can easily enter the body through breathing, swallowing, or skin contact. Once it enters, it affects the nervous system, which can show up as headaches, dizziness, or lack of coordination. Those working in facilities that use this chemical see skin and eye irritation as the earliest warning signs. Studies in animals show that high doses have led to convulsions and even death, pointing toward significant risk if people aren’t careful.
Regulatory agencies, including the U.S. Occupational Safety and Health Administration (OSHA), mark 2-chloroethanol as hazardous. There’s a reason behind those tight exposure limits. Breathing its vapors in large amounts can knock someone down fast. Liquid spills on skin create burns and blistering. Long-term exposure, even at lower doses, brings up questions about chronic effects, though most research so far focuses on short-term, high-level incidents.
Cancer Risk: Where Science Stands
Information about whether 2-chloroethanol causes cancer remains limited. The International Agency for Research on Cancer (IARC) has not officially classified this chemical. Scientists have conducted some animal studies involving its breakdown products, and some metabolites—like ethylene oxide—are known carcinogens in humans. That tells us we shouldn’t brush off the risk. In my own experience reviewing similar compounds, what we learn from related chemicals often applies, even if direct human data is thin on the ground.
For people in chemical plants and laboratories, safety experts don’t just let this chemical linger in open trays. Personal protective gear and strong ventilation systems become mandatory, partly because the long-term risks still raise concern. Health agencies call for careful handling, even in places where legal limits might seem generous. The chemical’s potential breakdown in the body and environment could mean hidden dangers we don’t fully appreciate yet.
Improving Safety Inside and Outside Factories
Over the years, I’ve seen how small process changes improve safety. Storing 2-chloroethanol in airtight containers, using it in closed systems, and giving workers effective gloves and eye protection go a long way. Plant managers use emergency drenching showers and eye-wash stations in case of accidents. Rigorous training, especially for new employees, has played a huge part in lowering incidents. Organizations can invest in continuous air quality monitors for any space where the chemical might escape.
Community awareness matters too. Emergency plans need to be clear, and the local fire department has to know what’s in the building. Authorities should share information about chemical hazards with neighbors in case of accidents. Drawing on my experience in risk communication, communities trust companies more when they see an obvious dedication to transparency and safety—not just compliance with the letter of the law.
The Bottom Line for 2-Chloroethanol
Chemicals like 2-chloroethanol demand respect. Even though we don’t have a simple answer on its cancer risk, the track record for acute harm is clear. Companies keep their workers safer when they recognize the danger, keep exposure down, and stay up-to-date on evolving science. Health and safety protocols, along with public awareness and government oversight, remain the backbone of real-world protections for anyone who might cross paths with this chemical.

| Names | |
| Preferred IUPAC name | 2-chloroethan-1-ol |
| Other names |
Ethylene chlorohydrin 2-Chloro-1-ethanol Glycol chlorohydrin Ethylene chlorohydrin Chloroethyl alcohol |
| Pronunciation | /tuː ˌklɔːr.oʊˈɛθ.ə.nɒl/ |
| Identifiers | |
| CAS Number | 107-07-3 |
| Beilstein Reference | 1209227 |
| ChEBI | CHEBI:16049 |
| ChEMBL | CHEMBL1358 |
| ChemSpider | 12176 |
| DrugBank | DB06733 |
| ECHA InfoCard | 200-830-5 |
| EC Number | 200-830-5 |
| Gmelin Reference | Gmelin 8227 |
| KEGG | C00589 |
| MeSH | D002774 |
| PubChem CID | 7909 |
| RTECS number | KJ5775000 |
| UNII | 9G82FYW8BF |
| UN number | 1135 |
| CompTox Dashboard (EPA) | DJ29I46IWD |
| Properties | |
| Chemical formula | C2H5ClO |
| Molar mass | Molar mass: 80.52 g/mol |
| Appearance | Colorless liquid |
| Odor | mild pleasant odor |
| Density | 1.20 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | -0.15 |
| Vapor pressure | 14 mmHg (20°C) |
| Acidity (pKa) | 14.3 |
| Basicity (pKb) | 1.47 |
| Magnetic susceptibility (χ) | '-7.22 × 10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.439 |
| Viscosity | 2.54 mPa·s (20 °C) |
| Dipole moment | 1.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 194.5 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -161.6 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -792.8 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | D8AX08 |
| Hazards | |
| Main hazards | Toxic if swallowed, in contact with skin or if inhaled; causes severe skin burns and eye damage; may cause genetic defects; may cause cancer. |
| GHS labelling | GHS02, GHS06, GHS05 |
| Pictograms | GHS02, GHS06 |
| Signal word | Danger |
| Hazard statements | H302, H312, H332, H351 |
| Precautionary statements | P210, P260, P261, P264, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P311, P321, P330, P362+P364, P405, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-2-2 |
| Flash point | -> 43 °C (109 °F; 316 K) |
| Autoignition temperature | 360 °C |
| Explosive limits | 5.9% - 16% (in air) |
| Lethal dose or concentration | LD50 oral rat 140 mg/kg |
| LD50 (median dose) | LD50 (median dose): 140 mg/kg (oral, rat) |
| NIOSH | PB6125000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Chloroethanol: 1 ppm (3 mg/m³) |
| REL (Recommended) | 20 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Ethylene oxide Ethylene glycol Ethylene chlorohydrin Vinyl chloride 1,2-Dichloroethane |

