2-Chlorobutane: A Look into Its Life, Lab, and Impact
Historical Development
The tale of 2-chlorobutane stretches back to the earliest explorations into halogenated hydrocarbons. During the 19th century, as chemists dove headfirst into substitution reactions, compounds like 2-chlorobutane showed up in organic chemists’ glassware and lab notes. Many students first brush up against this compound as they study alkyl halides and learn how chemistry’s building blocks often hold transformations both flashy and deeply practical. Even after decades, research never really turned away from these simple molecules, given their unique structure and reactivity. As a secondary alkyl halide, 2-chlorobutane lands smack in the middle of classic substitution and elimination reactions in organic textbooks. Its existence ties together the foundational moments of chemical synthesis and the steady growth of applied chemistry.
Product Overview
At first glance, 2-chlorobutane might seem like just another small molecule, but spend a few years in the lab, and you'd recognize its potential. Structurally simple — just a four-carbon chain with a chlorine sticking off the second carbon — yet this molecular tweak turns it into a chameleon in the realm of organic chemistry. Its uses range from a straightforward teaching model in SN1 and SN2 mechanisms to spawning more complex intermediates for pharmaceuticals, agrochemistry, and dyes. In commercial spaces, you find 2-chlorobutane used as a reactant more often than as an end product, but that foundational position in a synthetic route makes it surprisingly influential.
Physical and Chemical Properties
2-Chlorobutane stands as a liquid under normal lab conditions. The smell gives away its volatile character — you open the bottle, and there’s an immediate rush of that familiar sharp odor. Under a fume hood, its clear colorless liquid swishes easily; the boiling point hovers around 68°C, and freezing comes much lower, so it doesn’t solidify even under most storage conditions. Its density is a bit heavier than water, a nod to the chlorine’s influence. The molecule is less polar than many expect, leading to low solubility in water but fair compatibility with organic solvents. In chemical terms, its reactivity traces to the chloride on the secondary carbon, making it preferential for substitution and elimination, often serving as an enjoyable exercise in reaction predictability for organic chemistry students and seasoned chemists alike.
Technical Specifications & Labeling
Pick up a drum or a bottle of 2-chlorobutane, and the label always focuses on purity and concentration. In labs, chemists standardly use grades that reflect synthetic or analytical needs, often 98 percent or better, since impurities can steer reactions off track. Labeling standards don't just cover purity — proper hazard symbols and regulatory cues set the stage for safe handling. With any halogenated hydrocarbon, transport and storage pay careful attention to flammability and health risk, and these details should always be crystal clear in any workspace.
Preparation Method
In most commercial settings, the main route to 2-chlorobutane comes through the reaction of 2-butanol with hydrochloric acid. This traditional synthesis — the classic Lucas reaction — is a staple in college-level labs for a reason: it demonstrates both the underlying ease and the careful balance needed for selective substitution. The process depends on the conversion of a secondary alcohol to the corresponding chloride, often with a focus on minimizing byproducts and maximizing yield. Industrially, modifications sometimes use catalysts to push both speed and selectivity, helping manufacturers meet both efficiency requirements and environmental guidelines.
Chemical Reactions & Modifications
2-Chlorobutane’s chemical utility shows in its behavior under different reaction conditions. Subject it to nucleophilic substitution, and you can make a range of new molecules just by swapping that chlorine for other nucleophiles. Push a strong base into the solution, and elimination kicks in, yielding butenes — a simple illustration of E2 reactivity, something many chemists have witnessed firsthand. It’s this versatility that keeps the molecule relevant; whether you aim to build larger, functional, or more active molecules, 2-chlorobutane makes the journey possible. Sometimes, it even acts as a branching point for more elaborate rearrangements or coupling reactions, both in industrial scale-ups and smaller laboratory syntheses.
Synonyms & Product Names
It’s rare for chemicals to have just one calling card. 2-Chlorobutane also answers to sec-butyl chloride and secondary butyl chloride. Every catalog and literature batch might switch between these terms, but they all connect to the same structure. Each name carries its own legacy, with “sec-butyl chloride” highlighting the branching on the butane skeleton and “2-chlorobutane” sticking to modern IUPAC convention. These synonyms help researchers navigate the literature and avoid confusion, especially when diving into older publications or safety data sheets.
Safety & Operational Standards
Anyone who’s ever spilled a bit of 2-chlorobutane knows that it’s not something to take lightly. Its volatility means even small spills clear out a room if there’s no proper ventilation. Skin contact brings that sharp irritation, and you never want to breathe the vapors for any extended period. Stick strictly to the fume hood, always reach for chemical splash goggles, and gloves come standard. Regulatory guidance flags 2-chlorobutane as flammable and hazardous to health, often clubbing it with other chlorinated solvents in safety briefings. Standard operating procedures focus on spill containment, controlled storage away from ignition sources, and airtight labeling to prevent both accidental exposure and long-term health effects among lab workers.
Application Area
You won’t find 2-chlorobutane on pharmacy shelves, but its presence is felt up and down the industrial chemical chain. In organic synthesis, it’s often a stepping stone — the molecule you build, convert, and move past as reactions drive toward more complex targets like pharmaceuticals, crop protection agents, or specialty materials. I’ve watched it anchor student experiments in substitution and elimination labs, then turn up in bigger flasks during commercial synthesis of flavors, intermediates, and sometimes even as a component in the production of dyes and surfactants. These applications never stand alone; they ripple into broader contributions in fuel chemistry, custom chemical manufacture, and the preparation of more exotic halogenated compounds.
Research & Development
Innovations don’t always start with flashy breakthroughs; sometimes, steady progress emerges from finding cleaner, more efficient routes to traditional molecules. R&D teams continue to play with reaction conditions for 2-chlorobutane, searching for catalysts that minimize waste and for recycling strategies that fit green chemistry targets. Research stretches into better analytical methods for detecting residuals and monitoring production, crucial for keeping environmental impact low and product quality high. Organic chemists leverage its reactivity to develop new reaction methodologies, testing it across different fields — from total synthesis efforts to developing new functional materials.
Toxicity Research
Halogenated hydrocarbons demand attention when it comes to toxicity. Decades of occupational health studies underline the short-term consequences of skin or respiratory tract irritation from 2-chlorobutane. It doesn’t rise to the notoriety of some heavier solvents, but that doesn’t make it benign. Safety assessments point out chronic concerns, including impacts on central nervous system functioning after prolonged exposure. Even now, toxicology groups work to better understand how breakdown products might play into long-term environmental or health effects, especially if releases go unmanaged. In practice, strict engineering controls, personal protective equipment, and real training make the bulk of these risks manageable, but they only work as long as people keep safety in mind every step of the way.
Future Prospects
Looking ahead, 2-chlorobutane won’t make headlines, but its role shows no sign of fading. Chemical manufacturers depend on steady supplies and reliable reactivity, pushing for ever greener production routes. The race for sustainability keeps research teams busy, particularly with lifecycle analyses focused on solvent recycling, cleaner catalytic processes, and faster, less wasteful syntheses. As educational labs adapt to newer pedagogical models, it’s fair to expect even greener alternatives for standard reactions, but 2-chlorobutane’s place as a teaching tool and an industrial building block keeps it in the conversation for years to come. Keeping a watchful eye on emerging toxicity research will continue to shape guidelines and handling standards, ensuring both progress and safety have equal weight in the future of this humble yet impactful molecule.
What is 2-Chlorobutane used for?
Understanding What 2-Chlorobutane Brings to the Table
Not every chemical you meet in a lab or read about in textbooks is destined for public fame. 2-Chlorobutane grabs a modest audience compared to the mainstage act of global chemical trade, yet its work behind the scenes keeps several industries humming. I remember my early days in organic chemistry lab, being handed a bottle of 2-chlorobutane with a quick mention that, if I spilled it, everyone would know. The sharp scent lingers and so does its significance, even if most people outside a lab have never heard of it.
A Useful Tool for Making Other Compounds
2-Chlorobutane helps researchers and manufacturers build useful things. Its biggest role? Becoming a building block. This clear, flammable liquid with a sweetish smell gives chemists a straightforward route to create other, more complex molecules. In my classes, we’d use it to make alkenes through elimination reactions—textbook stuff, but the kind of work that sets you up to produce more valuable products.
Industries lean on it for synthesizing pharmaceuticals, flavors, fragrances, and even agrochemicals. Toss 2-chlorobutane into a reaction and you can end up with butenes, which then open the door to making synthetic rubbers or certain plastics. Not many people outside industrial circles realize these transformations kick off with such humble roots.
Stereo Drama: Why its Structure Matters
The shape of 2-chlorobutane deserves a look—for such a small molecule, it packs a punch with its ability to form two mirror-image forms, or enantiomers. Living systems often “care” about this detail because one enantiomer can work differently than its twin. Think of painkillers, flavors, or pesticides. I’ve seen projects slow to a crawl when the wrong form of a molecule was present. 2-Chlorobutane’s structure pushes researchers to develop cleaner, safer ways to make only the right enantiomer.
Handling Risks and Environmental Responsibility
Anyone who’s used 2-chlorobutane knows the safety notes by heart. This stuff evaporates quickly and irritates the eyes, even in a well-ventilated fume hood. Chemical companies and labs set strict rules to avoid breathing it in or letting it splash on skin. Stories from colleagues recounting spills or careless handling still circulate as cautionary tales. Following these precautions matters to both lab workers and neighbors downwind of manufacturing sites.
Spillage and improper disposal can cause issues. Companies using 2-chlorobutane typically recover or safely burn waste rather than dumping it, meeting strict laws that apply across most developed countries. Accidents happen, but clear protocols and quick cleanup help limit the harm.
Room for Safer Alternatives and Greener Methods
Newer technology keeps pushing chemical makers to rethink old habits with 2-chlorobutane. Green chemistry programs push for less hazardous substitutes for synthesis or for reactions that avoid using chlorine chemistry. On the academic side, students run simulated reactions with digital tools to cut back on physical risks. Workplaces focused on sustainability invest in better ventilation, personal protective equipment, and real-time air monitoring.
Keeping an eye on both the science and the impact matters. Every improvement, from stricter rulebooks to smarter process design, keeps people safer and helps protect the local environment. These changes, though slow at times, show a real, lasting benefit.
What are the safety precautions for handling 2-Chlorobutane?
2-Chlorobutane: Why Care About Safety?
A clear liquid like 2-chlorobutane might seem harmless on the surface, but working with it gets tricky fast. Breathing in its fumes or letting it touch skin is risky business. Just a whiff in a lab left me nauseous, reminding me nothing beats vigilance for personal safety. Overexposure often leads to headaches, dizziness, and even more serious nervous system issues. The stuff also burns quick—catching fire from a small spark. Keeping safety front and center isn’t about paranoia; it’s about protecting your lungs, your skin, and possibly your life.
Reliable Protective Gear Every Time
No shortcuts. Nitrile gloves, tightly fitting goggles, and a lab coat form my first line of defense. Latex gloves don’t give the same chemical protection—stick to nitrile. For eyes, side-shielded goggles block stray splashes. Loose sleeves or open shoes make you a sitting duck for accidents. A spill several years back taught me never to work bare-armed, not even for a “quick transfer.” Too many people skip face shields or full-length aprons, thinking they’ll get in the way. Every extra layer matters, especially when you’re pouring or transferring between containers.
Ventilation and Handling: Don’t Chance It
A cramped storeroom might look fine for storage, but fresh air keeps fumes from building up. Fume hoods—properly maintained, with active airflow—cut down inhalation risk. I remember a graduate assistant getting lazy and working on the bench because “it was only five milliliters.” A day later, the faint sweet smell lingered, proving the hood wasn’t optional. Moving or mixing 2-chlorobutane always means working inside the hood. Never handle it in small, poorly ventilated spaces, no matter how brief the task.
Storage Smarts: Small Steps, Big Impact
Separate 2-chlorobutane from acids, oxidizers, and direct sunlight. Keep it in tightly sealed glass containers. I mark bottles with the opening date, and check expired reagents often. Cool, dry lockers away from heat sources make a huge difference for stability. Always double-check that lids haven’t cracked, since fumes can slowly escape and put workers at risk for low-level, long-term exposure. Store a spill kit right nearby—delaying response only increases danger.
Spills and Emergencies: Prepare, Don’t Panic
One day, a co-worker knocked over a half-full bottle. Quick action prevented real harm—everyone had rehearsed cleanup steps and wore gloves. A sand-based absorbent trapped the chemical, and cleanup worked smoothly. Never use sawdust or paper towels. Have a neutralizing agent (like sodium bicarbonate) and proper waste containers ready at arm’s length. After handling a spill, immediately wash any exposed skin and seek medical evaluation if irritation or symptoms persist.
Training and Team Effort
Nothing replaces a solid briefing before new staff handle 2-chlorobutane. Walkthroughs, practical drills, and refreshers boost confidence. We use routine safety talks and practice drills. I’ve seen less experienced team members freeze during spills. Debriefing each incident, sharing stories, and correcting mistakes helps reinforce best practices.
Final Words: Shared Responsibility
Nobody protects your health and well-being as much as you do. Simple habits—good gloves, storage checks, and fresh air circulation—keep teams safe and shops running. Respect for chemicals like 2-chlorobutane comes from experience and learning from close calls. Safety isn’t a burden; it’s just how work gets done right.
What is the chemical formula and structure of 2-Chlorobutane?
Getting to Know 2-Chlorobutane
2-Chlorobutane stands out as a simple but important organic compound found in labs and discussed by chemists in classrooms worldwide. Its chemical formula, C4H9Cl, points to a structure made of four carbon atoms, nine hydrogen atoms, and a single chlorine atom. In my own early days studying organic chemistry, I remember sketching out its backbone and learning how swapping out just one atom can lead to new reactivity. The experience sticks with you: a clear reminder that changing small parts of a molecule can totally change how it behaves.
Structure: More Than Just a Drawing
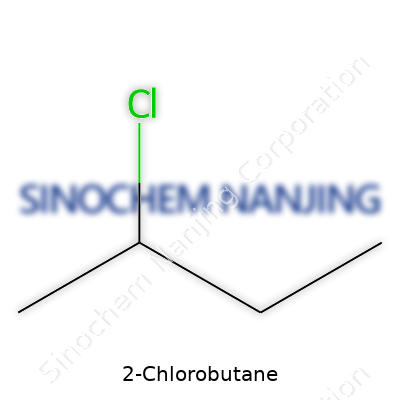
In 2-chlorobutane, the chlorine atom attaches to the second carbon in a straight chain of four. The simplest way to visualize this: start with butane, a well-known hydrocarbon used in lighters and fuel canisters—its structure is CH3-CH2-CH2-CH3. Replace a hydrogen on the second carbon with a chlorine, and the structure becomes CH3-CHCl-CH2-CH3. This switch gives the molecule its defining character. A chlorine atom brings some weight and changes the way the molecule reacts, especially with bases or nucleophiles.
Back in a university lab, a bottle of 2-chlorobutane didn't look like much—just a colorless liquid. Once you try to run a reaction with it, the story changes. The molecule sits ready for substitution and elimination, two fundamental paths in organic chemistry. Take a strong base and you get butene; use a mild nucleophile and the chlorine can slip away for something else. The chlorine on that second carbon doesn’t just define the name—it shapes everything about the compound’s chemistry.
Real-World Impact
Most people never encounter 2-chlorobutane in daily life, yet its applications fuel processes from pharmaceuticals to agrochemicals. Because that chlorine serves as a leaving group, the molecule plays a starring role in synthesizing more complex compounds. While teaching high school advanced chemistry, I watched students struggle with the idea that swapping out a single atom unlocks a path to completely different sets of molecules. You notice how a simple compound helps build bigger worlds—this is foundational in chemical manufacturing.
Safety steps are critical. Chlorinated hydrocarbons can trigger both environmental and personal health problems if mishandled. Years working in labs force you to respect gloves, fume hoods, and labeling. Chemical companies use closed systems and careful monitoring to limit leaks and exposure during production.
Learning from 2-Chlorobutane
Working with 2-chlorobutane illustrates something essential about chemistry: structure equals behavior. The placement of a single chlorine determines how it acts in a reaction, which shapes the end product you get. When educators use 2-chlorobutane as an example, students see firsthand that structure isn’t just a textbook picture—it defines possibilities and limitations. As curriculum developers and teachers, we can highlight these lessons early. Visual aids, hands-on experiments, and open discussion help show how the arrangement of atoms opens up opportunities and risks.
Approaching chemicals like 2-chlorobutane with respect and curiosity bridges the gap between learning and real-world responsibility. Recognizing not only the chemical makeup but also the potential applications and risks puts safe and smart science within reach for everyone.
How should 2-Chlorobutane be stored?
Why Proper Storage Matters
Anyone who's spent a little time around a chemistry lab quickly learns that some chemicals need more care than others. 2-Chlorobutane can catch even seasoned folks off guard. This chemical brings a few risks — flammability, potential to irritate skin or lungs, not to mention that old laboratory standby, the fire hazard. Skipping safe storage just because “nothing has ever happened” rarely ends well.
Understanding What You're Dealing With
2-Chlorobutane comes as a colorless liquid, often with a sharp smell. Consider this—its flash point is less than 0°C. That means it catches fire at fridge temperatures. Safe handling starts with knowing how unpredictable it can be if left sitting in the wrong spot. NIOSH and OSHA both point out that vapors from this substance, even at room temperature, can form explosive mixtures with air. That paints a pretty clear picture: a little leak or the wrong spark, and things could go very wrong.
Store Away from Heat and Flames
Flammable liquids don't mix well with heat or open flames — this rings especially true for 2-Chlorobutane. Shelves near radiators, electrical panels, or windows with direct sunlight should get ruled out immediately. I always keep flammable materials in a metal safety cabinet rated for such chemicals, and I'd recommend the same. These cabinets often come with vents and self-closing doors, adding one more level of control.
Keep the Lid Tight
Leaking fumes can fill up a poorly ventilated storage room faster than you'd think. I once opened a poorly sealed bottle and could smell the difference right away. Good-quality, airtight containers with properly fitted screw-tops help avoid accidental evaporation or leaks. Polyethylene or glass bottles often work fine, though checking compatibility charts never hurts. Labels must be clear and current — nothing beats being able to spot exactly what you have at a glance.
Ventilation Matters
Even with sturdy containers, no setup is perfect. Good air circulation and exhaust hoods can take care of stray vapors that might escape. Never tuck these chemicals into cabinets without some airflow. In my university lab days, just a tiny fume leak behind a stack of boxes lingered for hours and irritated multiple people. Routine checks of vents and fans quickly became part of our daily safety walk.
Separate from Incompatibles
Mixing oxidizers, acids, or strong bases with many organics can start chain reactions. 2-Chlorobutane fits that bill. Segregating storage shelves saves a lot of trouble, since even a dropped bottle can set off a nasty surprise if the chemicals hit one another. Flammable liquids should sit far from oxidizers or acids—no exceptions.
Don’t Overlook Emergency Tools
Having a fire extinguisher nearby makes a real difference. For liquid chemical fires, a CO2 or dry powder extinguisher works best. Spill kits sit right inside our storage room, stocked with absorbent materials and gloves meant for strong solvents.
Knowledge and Routine Keep You Safe
Handling chemicals like 2-Chlorobutane always means giving respect to the dangers they bring. Regular training updates, routine checks, and sharp labeling mean you're less likely to fumble in a pinch. Small steps — closing the lid, double-checking the cabinet, making sure the air moves — stack up to create real safety.
What are the physical and chemical properties of 2-Chlorobutane?
The Basics: What 2-Chlorobutane Looks and Feels Like
2-Chlorobutane sits in the family of halogenated alkanes. The name almost says it all—four carbons in a chain, with a chlorine atom attached to the second carbon. Out in the open, you’ll spot it as a clear, colorless liquid. Anyone working near it will catch a whiff of its sweet, sharp odor pretty quickly. It boils right around 68–69°C, which means it evaporates easily at room temperature, making good ventilation a must. Pour it in water and the story changes. 2-Chlorobutane barely wants to mix, but it dives right in alongside many organic solvents, like alcohol and ether.
Why Chemical Structure Matters
Pull a model kit apart and piece this compound together. Carbon two holds the chlorine, flipping the molecule into a chiral state. That simple switch makes 2-chlorobutane crucial for chemists who care about stereochemistry. Racemic mixtures of this compound pop up in university labs time and again, letting students see the theory hit the real world. In industries that tailor pharmaceuticals, plastics, or specialty chemicals, having access to a straightforward secondary alkyl halide like this one speeds up synthesis. The polarity created by the carbon-chlorine bond makes it react faster in some settings than its more stubborn cousin, 1-chlorobutane.
Not Just Numbers: Key Physical Details
Holding a handful of basic figures sets the scene. 2-Chlorobutane weighs in with a molecular mass around 92.57 g/mol. Its density clocks in at about 0.87 g/cm³, which keeps it floating above water. Pour it in a cold room and you'll see it solidify around −139°C—a temperature rare even in hard winters. Toss a match near it and the liquid catches fire, making safety planning crucial during storage and use. On the skin, contact burns, and inhaling too much vapor turns into a real hazard fast.
How 2-Chlorobutane Reacts and Transforms
This chemical tells an interesting story once it meets a good nucleophile. That secondary carbon opens the doors to both SN1 and SN2 reactions, a real treat for anyone teaching organic chemistry. It lets you walk through the difference between unimolecular and bimolecular substitution without fancy equipment. In fact, the competition between these two reactions becomes a springboard to deep chemical insight for new students.
2-Chlorobutane reacts with strong base just as well. Introduce potassium hydroxide and the molecule sheds its chlorine, forming butene through elimination. That property underlines its value in research and development settings where making new scaffolds is the main goal. The way it can swap out the chlorine makes it a workhorse for building anything from anesthetics to agrochemicals.
Why People Pay Attention
Physical and chemical properties do not just feed curiosity—they tell you how to handle the stuff. 2-Chlorobutane flows out of bottle easy, its vapor clings low to the floor, and a hot afternoon can drive vapors dangerously high indoors. Understanding boiling point, flash point, and volatility means a lot more than memorizing numbers. In university classes, these details teach safety and respect.
These facts remind us to store it tight, keep ignition sources far away, and work where fresh air keeps risks low. In my experience around research labs, ignoring one detail usually brings trouble. The best researchers never skip reading the labels or skimming safety data. It all comes down to knowing your materials—2-chlorobutane tests memory and discipline at every turn.
| Names | |
| Preferred IUPAC name | 2-Chlorobutane |
| Other names |
sec-Butyl chloride 2-Chlorobutan sec-Butylchloride Chloro-sec-butane Butane, 2-chloro- 1-Methylpropyl chloride |
| Pronunciation | /tuːˈklɔːr.əˌbjuː.teɪn/ |
| Identifiers | |
| CAS Number | 78-86-4 |
| 3D model (JSmol) | `JSmol.loadInline("data:text/plain,3D;2-Chlorobutane;C4H9Cl;ClCC(C)C")` |
| Beilstein Reference | 1720561 |
| ChEBI | CHEBI:33161 |
| ChEMBL | CHEMBL15338 |
| ChemSpider | 7277 |
| DrugBank | DB13855 |
| ECHA InfoCard | 100.007.727 |
| EC Number | 602-038-00-5 |
| Gmelin Reference | 7788 |
| KEGG | C01578 |
| MeSH | D002777 |
| PubChem CID | 7913 |
| RTECS number | EJ6475000 |
| UNII | 2917JX673A |
| UN number | UN1127 |
| Properties | |
| Chemical formula | C4H9Cl |
| Molar mass | 92.57 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet odor |
| Density | 0.887 g/mL |
| Solubility in water | Insoluble |
| log P | 1.85 |
| Vapor pressure | 46 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 50 |
| Basicity (pKb) | The basicity (pKb) of 2-Chlorobutane is **"–"**. |
| Magnetic susceptibility (χ) | -8.38 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.403 |
| Viscosity | 0.38 mPa·s (20 °C) |
| Dipole moment | 2.10 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -173.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2713.8 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H225, H319, H336 |
| Precautionary statements | P210, P261, P305+P351+P338, P304+P340, P403+P235 |
| NFPA 704 (fire diamond) | 2-3-0 |
| Flash point | -6 °C |
| Autoignition temperature | 210 °C |
| Explosive limits | Lower 1.8%, Upper 8.0% |
| Lethal dose or concentration | LD50 (oral, rat): 2730 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 2700 mg/kg |
| NIOSH | CN9625000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 100 ppm |
| IDLH (Immediate danger) | 800 ppm |
| Related compounds | |
| Related compounds |
2-Chloropropane 1-Chlorobutane 2-Bromobutane 2-Iodobutane 2-Chloropentane |

