2-Butanone: Substance, Science, and Shifting Demands
Historical Roots and Chemical Backdrop
Looking back at how 2-Butanone turned into a staple for chemists and manufacturers tells as much about the story of modern chemistry as it does about the substance itself. Known for decades as methyl ethyl ketone (MEK), this clear, fast-evaporating liquid started out as a by-product within early petroleum refining. The drive to synthesize better coatings and adhesives gave 2-Butanone a starring role in formulations across North America, Europe, and Asia as chemical industries boomed in the 20th century. Folks in manufacturing recognized that, compared to the likes of acetone, MEK often delivered stronger solvency power and a moderate evaporation rate, letting paints or adhesives dry evenly and fast, which made production lines move faster and products look and work better. Technological leaps and environmental rules shaped its use but never sidelined it—this chemical still matters.
Physical & Chemical Properties That Matter in Real Life
Anyone who’s opened a drum of 2-Butanone in a plant knows its sharp, fruity odor. With a boiling point around 80°C, this liquid vaporizes quickly, sometimes too quickly if ventilation isn’t up to snuff. Its strong polarity compared with other industrial solvents makes it quite good at dissolving resins, plastics, and gums. What I’ve always found interesting is its low water solubility, which means it can strip away tough residues while leaving some water behind—helpful when you want selective cleaning in electronics or automotive plants. Its flammability poses real risks: vapors ignite without much provocation, much like other low-molecular-weight ketones. Technicians value its low viscosity and clarity, especially in labs where a clouded solution means a bad batch or botched results.
Technical Standards and Labeling
Regulatory agencies have hammered out specifications so users know exactly what they’re getting. Industrial-grade MEK usually exceeds 99% purity. Strict rules call for a water content limit, often below 0.1%, plus restrictions on aldehydes, acids, and other organic impurities. Labels state hazards straight out: highly flammable, can build explosive mixtures with air, harmful by inhalation. Standard pictograms warn users at a glance. In practice, the drum labels and data sheets give a rundown that plant managers review before opening up large orders—nobody wants surprises in processes that involve operators and open flame.
Synthesizing 2-Butanone: Industrial Practice
The world’s supply of 2-Butanone leaves refinery and chemical plant reactors daily, made mainly by two methods. The older process relied on dehydrogenating 2-butanol in the presence of copper, zinc, or bronze catalysts, with temperatures running hot to drive off water and get pure MEK. In another common approach, some plants crack secondary butyl acetate or process n-butene in oxygen over catalysts to yield MEK. Each method has different strengths: dehydrogenation lets operators use renewable feedstocks, while oxidation taps into petrochemicals. Raw material prices, regional regulation on emissions, and needs for process efficiency steer producers toward the best route for their scale and local supply chain.
Chemical Reactions and Looking for Added Value
2-Butanone isn’t just a solvent. Chemists use its carbonyl group to build more complex molecules. In research and industry, it serves as a launching pad for synthesis by reacting with nucleophiles—sourcing things from pharmaceuticals to specialty plastics. Under basic or acidic conditions, it undergoes aldol condensation, linking up with itself or other compounds, a reaction pathway every organic chemist faces in the first chapters of advanced textbooks. Some research points to using MEK as an intermediate in producing methyl isobutyl ketone (MIBK) and diacetone alcohol, both valuable in their own right. For labs and factories alike, having flexible synthetic options lets them pivot with changing demands.
Synonyms and Product Names
Industry insiders don’t get tripped up by the parade of names. 2-Butanone and methyl ethyl ketone (MEK) show up in the same context, whether you’re ordering bulk for a coatings plant or grabbing a sample for lab work. Other language variants—like ethyl methyl ketone or butan-2-one—pop up in regulatory or academic circles. This variety in naming can trip up regulatory reviews or shipping paperwork. That’s why experienced handlers pay close attention to labeling and inventory management to keep confusion and misdelivery at bay.
Safety Red Flags and Keeping Operations on Track
Workplaces relying on 2-Butanone face real safety risks—fire takes the top slot. This chemical builds up vapor fast, and with an ignition point barely above room temperature, one small spark can escalate to disaster. OSHA and other agencies set occupational exposure limits at levels often below 200 ppm averaged over an eight-hour shift. Factories use explosion-proof systems, custom exhaust setups, and non-sparking tools. Regular air monitoring and personal protection controls—like tight-sealing respiratory masks—keep workers from breathing concentrated fumes. Spills call for quick containment and expert cleanup, not just tossing sawdust and hoping for the best. Supporting safety standards means fewer accidents, which resonates through insurance claims and employee well-being, not just compliance checklists.
Main Application Fields and Industry Practice
Demand for 2-Butanone traces back to its role dissolving specialty resins and polymers better than most competitors. Paint shops and marine coating facilities rely on it for thinners and cleaning solutions. The adhesives market appreciates the bite it brings to glue formulations, helping bond hard-to-stick materials. Electronics manufacturers reach for MEK during circuit board fabrication, especially for precision cleaning after soldering and before encapsulating. Leather processing and textile facilities use it to degrease or prep surfaces before finishing treatments. Even outside the walls of factories, its ability to dissolve stubborn marks lands it in some graffiti removers and heavy-duty cleaners.
Research, Innovation, and Real-World Observations
Academic and corporate labs see 2-Butanone as both a mainstay and a challenge. Chemists dig into new reactions on the MEK backbone, while engineers investigate greener production paths. There’s a strong push to recycle MEK-rich waste streams and design catalysts that cut greenhouse gas emissions during synthesis. Recycling solvents from industrial cleaning tanks helps companies save money and meet legal standards on waste disposal. On the research front, analysts dug into the detection of trace MEK in air and water for regulatory monitoring. There’s active work exploring hybrid production—blending bio-derived alcohols with petro-processes—in hopes of future-proofing supply chains and shaving down environmental impact.
Toxicity and Worker Well-being
Years of toxicology study make the dangers of 2-Butanone clear. Short-term inhalation brings headaches, dizziness, and nausea; higher doses trigger central nervous system effects. Chronic exposure sparks debate about subtle impacts—memory, cognition, organ health—but the consensus points to clear risks if limits aren’t enforced. The solvent’s ability to cut oils makes skin contact a problem, sometimes leading to dryness or dermatitis. Regulators flag MEK use around pregnant workers based on animal studies showing possible developmental harm, though human data stays less clear. Routine air testing, skin protection protocols, and workplace medical surveillance go a long way to keeping operators out of the danger zone.
What Comes Next for 2-Butanone?
Environmental regulations keep squeezing the solvent market, pushing facilities to find lower-emission and less toxic alternatives. Yet MEK stays stubbornly relevant thanks to its strong performance and relatively affordable price. Some innovators turn to bio-based feedstocks for lower-carbon MEK or blend it with less hazardous co-solvents to reduce environmental and worker impact. Research teams dig into safer, more efficient catalysts to make MEK with less waste and lower energy spending. Sensors measuring tiny airborne traces become more affordable and widespread—critical for enforcing tighter workplace exposure standards in the coming years. Down the road, stricter bans could reshape the market, favoring vendors who can document sustainable, low-risk origins and handling. Manufacturers, engineers, and regulators all face the same challenge: balancing practical utility, health, and the changing demands of regulators and consumers.
What is 2-Butanone commonly used for?
What 2-Butanone Does in Our Lives
2-Butanone shows up in more places than most people realize. It’s easy to overlook, but this clear liquid goes by another name—methyl ethyl ketone (MEK)—and finds its way into everything from paint strippers to industrial cleaners. I remember opening a can of paint thinner in my father’s garage and getting hit by that sharp, almost sweet odor. That’s the smell of 2-Butanone doing its job, helping dissolve paints and coatings so surfaces come clean or old paint layers lift off easier.
Factories Rely on MEK
Manufacturing plants depend on MEK for more than just cleaning. Businesses use it in the process that produces plastic, textiles, and rubber. It’s prized for its strong solvent action. The chemical breaks down glue and adhesives, giving manufacturers a way to adjust or rework bonded joints. Car shops choose MEK to prep surfaces before painting because it cuts through oils and residues. Nail polish removers sometimes lean on 2-Butanone too, though less often than acetone these days.
Impact on Human Health
You might not think much about the health side, but it deserves attention. The sharpness of MEK isn’t just about smell. Inhaling too much can cause dizziness, headaches, and nausea. Factory workers and folks doing home renovation jobs run the highest risk, especially without proper ventilation. The U.S. Environmental Protection Agency points to short-term exposure being most dangerous, with long-term studies still ongoing. Gloves and goggles matter—skin contact might cause dryness or irritation and splashes can sting eyes.
Environmental Effects Matter
2-Butanone doesn’t stick around in the environment quite like some heavy chemicals. It evaporates quickly, but that doesn’t mean it gives a free pass. The fumes end up in the air, contributing to ground-level ozone pollution. Rivers and lakes catch runoff too, although studies suggest breakdown occurs faster in water than in soil. I once saw a local hardware store switch to “green” solvents after a community group raised concerns over river pollution—people chose products with fewer environmental impacts when given options.
Thinking About Safer Alternatives
Plenty of workers and hobbyists have moved toward water-based or low-VOC products as a safer option. Companies have invested in alternatives after years of pressure from scientists and consumer groups. My own experience with “eco” paint strippers makes one thing clear: performance and safety often trade places. Some water-based cleaners take more elbow grease, but the trade-off for cleaner air and easier breathing feels worth it. Workers deserve better safety guidelines and more affordable alternatives, and government regulations help push companies in the right direction.
MEK’s Future in Industry
Many industries face a balancing act: stick with MEK and keep overhead low, or invest in safer practices. As awareness grows, demand for greener chemistry rises. The paint and coatings market already talks about moving away from strong solvents where possible. Real change means more research and incentives. Better public information also matters. If consumers keep asking questions, stores might offer more options, just like my local hardware shop did. It’s one way to cut risks and protect both health and the environment—without giving up on the tasks that keep homes and businesses running.
Is 2-Butanone hazardous to health?
Understanding 2-Butanone in Daily Life
I’ve spent time in workshops, garages, and busy kitchens, and the sharp smell of chemicals sometimes hangs in the air. That scent often comes from 2-Butanone, better known as methyl ethyl ketone or MEK. It’s everywhere: paint thinners, cleaners, adhesives, even nail polish remover. People trust these products, using them at work and around the home, not always thinking about what’s in the air or soaking into their skin.
Immediate Effects of Exposure
After spending only a few minutes in a freshly painted room or around certain glues, my nose tingles, eyes water, and sometimes my head feels cloudy. This isn’t just me being sensitive. Health studies and regulatory agencies say short-term exposure to 2-Butanone can irritate eyes, nose, and throat. It can leave people feeling dizzy, tired, or give them a headache. One study published by the National Institute for Occupational Safety and Health found high vapor levels can even make people feel sick or disoriented.
Long-Term Risks and How the Body Handles It
Most folks don’t realize the body can handle a little bit of many chemicals, including 2-Butanone, by breaking them down and getting rid of them. But the trouble starts with breathing high concentrations day after day. Workers in manufacturing or auto repair can get this kind of exposure. Chronic contact with the vapor can affect lung function and the central nervous system. Research from the American Conference of Governmental Industrial Hygienists draws a link between workplace exposure and a higher rate of chronic headaches, memory trouble, and even nerve damage. So it’s not just short-lived discomfort; over time, repeated exposure adds up.
Making Safer Choices
Regulation can only go so far, and companies often train staff to follow safety rules, but in my experience, people rush through jobs without proper masks or ventilation. In busy settings, it’s easy to skip gloves or leave windows closed. Venting a workspace and using gloves stops most everyday dangers. NIOSH and OSHA both agree that eye protection and proper airflow cut risk for almost everyone using products that contain this solvent.
Alternatives and Next Steps
The chemical industry keeps advancing, and the push for safer substitutes has picked up speed. Low-toxicity cleaners and water-based products now fill store shelves, offering choices that don’t come with 2-Butanone’s risks. Some workplaces have moved toward stricter exposure limits and modern ventilation systems. This progress comes down to awareness. The best strategy is to read product labels, push for better safety at work, and choose less-toxic products at home.
Why Awareness Still Matters
Ignoring the risks just keeps people in harm’s way. From my own hands-on work, small steps like airing out a room or swapping out harsh chemicals can change health outcomes over time. Reports from the U.S. Environmental Protection Agency echo this: even if 2-Butanone doesn’t cause cancer, it still triggers symptoms that hurt quality of life. Taking personal safety seriously—especially for children or people with asthma—goes a long way toward staying healthy in environments where this chemical shows up.
What are the storage requirements for 2-Butanone?
Don’t Treat Flammable Liquids Like Regular Chemicals
2-Butanone, better known as methyl ethyl ketone, ranks high on the list of solvents that spark safety debates in labs and factories. Anyone who’s handled it recognizes the sharp, sweet smell, sometimes so strong it makes your eyes water a bit. What too many folks don’t realize: storing this stuff is almost as important as how you use it.
Fire Risks Demand Respect
Walk into any chemical storeroom and the warning labels on 2-butanone containers jump out—dangerous, flammable liquid. Vapors catch fire fast, and you don’t get much warning. Toss a 2-butanone drum near a heat source, and you’re asking for trouble. Many fire marshals, myself included, just shake our heads when we see it sharing space with open electrical panels or poorly ventilated rooms. Historical data from the U.S. Chemical Safety Board list multiple warehouse fires tracing back to carelessness around solvents like this.
OSHA and NFPA spell it out clear: only use ventilated, fire-resistant storage cabinets and keep those far from anything that could spark. Metal cans and drum storage units save lives. Never skimp on grounding either—static discharge is a sneaky risk that doesn’t get enough attention. One tiny spark, and the vapor flash is near-instant. Fire department incident reports often mention static from transferring solvents as the root cause in storage-area accidents.
Ventilation Keeps Workers Breathing Easy
If you’ve ever worked in a summer lab with poor airflow, you know how fast solvent vapors can make a room feel stuffy and workers dizzy. Chronic exposure to 2-butanone leads to headaches and nausea. The answer is targeted ventilation: exhaust hoods, ducted cabinets, and fans built for chemical fumes, not just ordinary box fans. I’ve seen storage rooms re-engineered with smart airflow, and the result is night-and-day—air stays fresher, and workers stick around for the long haul without health complaints.
Labeling and Segregation Are Not Useless Habits
Folks roll their eyes at the call for clear labels and chemical separation, but the worst chemical accidents happen in poorly labeled stores. You want to keep 2-butanone well away from oxidizers, acids, and bases. In real life, this means a separated section on purpose-built shelving, with clear strips on the floor marking hazard zones. Most industry pros use color-coded tags and keep a current inventory in plain sight. It’s not paranoia; it’s experience from cleaning up spills where someone didn’t know what was on the next shelf over.
Spill Plans Require Real Commitment
No one expects a drum to leak, but as I learned early in my career, you discover every shortcut during a spill. Keeping absorbent pads, specialized solvents, and neutralizing agents at arm’s reach (ideally in a spill kit right by the storage area) saves hours and may prevent permanent damage to property and lungs. Workers trained on what do in seconds—not after thumbing through a manual—stop disasters before they escalate.
Building a Culture that Prioritizes Safety
The companies best at handling 2-butanone treat safe storage as part of their daily routine, not an afterthought. Trainings stay fresh, signs stay up, inspections get done, and no one trusts just one set of eyes. Relying on memory or informal habits makes mistakes more likely. Strong procedures, regular walkthroughs, and worker buy-in build safer sites and prevent both fires and injuries.
Simple steps make a big impact: fireproof cabinets, well-labeled containers, grounded metal racks, active ventilation, and ready-to-go spill kits. Each detail matters—every safe day spent with 2-butanone is the payoff for real effort and real vigilance.
What is the chemical formula of 2-Butanone?
Understanding 2-Butanone, Plain and Simple
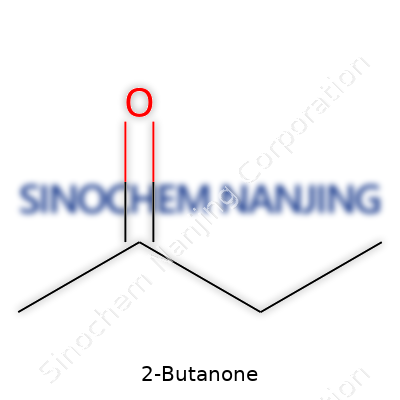
2-Butanone, known out in the world as methyl ethyl ketone, kicks around with the chemical formula C4H8O. That formula tells you: here’s a molecule with four carbon atoms, eight hydrogen atoms, and one oxygen atom. This isn’t just trivia for a test—real lives and plenty of industries bank on understanding what even a small molecule like this can do.
Getting to Know This Solvent’s Role
My experience working in a factory taught me that chemicals like 2-butanone show up far more often than most folks imagine. Walk through a warehouse stacked with paints, adhesives, or cleaning supplies—chances are strong that this stuff is tucked behind the scenes powering the process. Scientists and workers trust its strong dissolving power to get tough jobs done. The formula C4H8O unlocks its identity.
Take industrial coatings as an example. A lot of us use things every day that rely on tough, glossy finishes. Those coatings don’t just stick by magic—solvents like 2-butanone set the stage. They help dissolve resins, spread the mix evenly, and vanish at the right moment so the finish can harden up. Knowing what you’re working with keeps workers safe and helps factories pick the right ingredient.
Health and Environment—Why Formula Transparency Counts
I can’t stress enough what happens if workplaces turn a blind eye to the chemistry. 2-Butanone evaporates fast, so anyone handling it breathes in some vapor unless the space is set up right. People who don’t know their chemicals might figure, “if it’s in the paint, it’s harmless.” But a formula doesn’t lie; C4H8O means a compound that can irritate eyes, skin, and lungs. A few years back, coworkers and I relied on reading safety data sheets. The right formula let us link symptoms to the solvent and switch up protections to match the risk.
Beyond personal health, the molecule finds its way into water and soil if spills go unchecked. Regulators keep a watchful eye on formulas, directing proper disposal and keeping communities safer. A chemical formula turns into a story about stewardship—it means painting companies don’t just slap stuff on a label. It’s public knowledge for users and emergency crews who need to act fast in a spill.
Solutions Involve Smarter Choices
Quality information empowers each person in the chain. If you’re a student, don’t pass off C4H8O as just one more fact to memorize. If you use solvents at work or at home, push for training that gets hands-on about risks and uses. Watch for new research, too. Scientists keep hunting for safer, greener alternatives. Switching to a low-toxicity replacement means checking up on those formulas—if it’s less volatile, you can breathe easier in the workplace.
Understanding the formula means grasping the real-world power and pitfalls of even a simple molecule. Knowing the simple makeup of 2-butanone, C4H8O, adds up to smarter decisions every single day across shop floors and family garages alike.
How should 2-Butanone be handled safely?
The Reality of Handling 2-Butanone
If you’ve ever spent time in a lab, paint shop, or a spot where solvents flow, you may know 2-Butanone by another name—methyl ethyl ketone (MEK). Many workers hear the phrase “strong solvent” and think sticky residue on hands and headaches from harsh vapor. But the substance can do more than irritate skin or eyes. Long exposure or careless handling often leads to more serious effects like nervous system issues, liver and kidney stress, or trouble breathing.
Why Safe Handling Matters in Everyday Work
On a regular workday, rushing through a task around open containers or with poor ventilation builds risks quickly. Chemistry textbooks warn against direct skin contact or inhaling fumes for a reason—this stuff evaporates fast, and its vapor hangs near the floor, just waiting to be breathed in. When you get that sharp smell, you’re already catching more than a whiff. It only takes one splash or forgetful step to turn a manageable job into an emergency room visit.
Simple Steps That Protect Health
Gloves and goggles come first, but not every glove stands up to solvents. Nitrile holds up longer than latex. Thick neoprene ones add another layer if splashes might happen, especially with busy hands or big batches. For eye safety, wraparound goggles beat safety glasses—less chance of a stray drop getting through. Lab coats and good shoes do more than keep clothes clean; they create a barrier that helps if a container tips or a spill runs along the floor.
A mask is more than a prop. Vapor from 2-Butanone can’t be dodged by working fast. Charcoal-filter respirators or fume hoods are a must if you’re dealing with open containers or pouring the stuff in big volumes. A window cracked open won’t cut it, and fans can make things worse by blowing vapors into other parts of the workspace.
Good Habits Outlast Warning Signs
Reading a label or a safety sheet gives clues, but habits shape outcomes. Training for anyone working with solvents pays off for both the new folks and those with years under their belt. Spills happen. Cut down on drama: keep an absorbent handy, bottle tops on tight, and store old rags in marked metal cans. Washing hands before eating or heading out is boring advice, but it saves trouble nobody sees until much later.
Storage talks always sound dry until you see what happens if a bottle leaks or the wrong item sits nearby. Keep 2-Butanone away from anything flammable or reactive, especially around acid fumes or oxidizers. Shelving that lets leaks drip downward turns a small problem into a bigger mess, so all containers go low and alone.
A Culture That Looks Out for Everyone
The real safety net comes from looking after each other. If a coworker starts moving slower, stumbles, or gets lightheaded, you step in. Get outside fast, call for help, and don’t shrug it off. Posting emergency numbers and keeping spill kits visible sets a tone. It tells everyone in the room, from the boss to the new hire, that short cuts don’t cut it.
Those who’ve worked around these chemicals for a living remember times when safety gear seemed like a hassle. But time and again, the workers who made it home safe and healthy did so because they respected the risks, not because they ignored them.
Leading With Practical Experience
Facing 2-Butanone with clear eyes and a bit of preparation keeps daily dangers to a dull roar. Most rules boil down to respect—for the solvent, for health, and for coworkers. Build those into routines, and the work not only gets done, but everyone stands a better chance of seeing the job site again tomorrow.
| Names | |
| Preferred IUPAC name | Butan-2-one |
| Pronunciation | /ˈbjuː.təˌnoʊn/ |
| Identifiers | |
| CAS Number | 78-93-3 |
| Beilstein Reference | Beilstein 1209249 |
| ChEBI | CHEBI:28398 |
| ChEMBL | CHEMBL42876 |
| ChemSpider | 727 |
| DrugBank | DB01912 |
| ECHA InfoCard | 100.003.278 |
| EC Number | 203-550-1 |
| Gmelin Reference | Gm. 59 |
| KEGG | C00432 |
| MeSH | D001975 |
| PubChem CID | 6569 |
| RTECS number | EL6475000 |
| UNII | K6953XJ6JJ |
| UN number | UN1193 |
| Properties | |
| Chemical formula | C4H8O |
| Molar mass | 72.11 g/mol |
| Appearance | Colorless liquid with a sharp, sweet odor |
| Odor | Sweet, mint-like |
| Density | 0.805 g/cm³ |
| Solubility in water | Fully miscible |
| log P | 0.29 |
| Vapor pressure | 10 kPa (at 20 °C) |
| Acidity (pKa) | 20.0 |
| Basicity (pKb) | pKb = 7.81 |
| Magnetic susceptibility (χ) | -8.67 × 10⁻⁶ |
| Refractive index (nD) | 1.378 |
| Viscosity | 0.43 mPa·s (at 20 °C) |
| Dipole moment | 2.734 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 200.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -249.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2435 kJ/mol |
| Pharmacology | |
| ATC code | V03AB18 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | Flame, Exclamation mark |
| Signal word | Warning |
| Hazard statements | H225, H319, H336 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P261, P271, P280, P301+P312, P303+P361+P353, P304+P340, P305+P351+P338, P312, P337+P313, P370+P378, P403+P235, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | -9 °C |
| Autoignition temperature | 515 °C |
| Explosive limits | 1.8% - 11.5% |
| Lethal dose or concentration | LD50 Oral Rat 2737 mg/kg |
| NIOSH | KET 35 |
| PEL (Permissible) | 200 ppm |
| REL (Recommended) | 200 ppm |
| IDLH (Immediate danger) | 800 ppm |

