2-Butanol: Looking Beyond Its Lab Roots
Historical Development
Long before labs used synthetic routes to make 2-butanol by the kilogram, people found it showing up in trace amounts in nature—though not in a form anyone would bother collecting. Sometime in the early part of the twentieth century, chemists started taking a serious look at secondary alcohols like 2-butanol as industries went from using simple alcohols for drinking and fuels to seeing the value in more nuanced organic molecules. With petroleum processing opening new pathways, 2-butanol found itself pulled from crude oil fractions and then, by mid-century, made more efficiently through hydrating butenes coming out of cracking plants. Anyone who spent time in a refinery during those days heard plenty about the push to wring more value from byproducts, and 2-butanol was squarely in their sights.
Product Overview
2-Butanol, sitting between ethanol and tert-butanol in complexity, stands out for its secondary alcohol structure. It’s not the first thing people reach for at the hardware store—that spot belongs to isopropanol and ethanol—but in the right hands, it solves a specific set of problems. It brings some of the solubility and reactivity people expect from an alcohol but adds enough difference from the simple ones that chemists often prefer it over the others for distinct applications. From time to time, people even run across it in flavorings, though not intentionally as an ingredient, more as a trace byproduct in fermented goods.
Physical & Chemical Properties
People used to simple alcohols like ethanol notice quickly that 2-butanol’s smell stands out—a slightly sweet, almost banana-like odor, though not something most folks would call pleasant. It's a liquid at room temperature and clear, so unless you know what you’re looking for, you could easily mistake it for water. Its boiling point sits just over 99 degrees Celsius, which makes distillation tricky but feasible with regular lab setups. It mixes with water to a degree, but unlike smaller alcohols, you can see phase separation if you try to push it into high concentrations with non-polar solvents. Its volatility sometimes surprises lab beginners, leading to headaches or nausea if ventilation isn’t up to par. Having spent long hours in university labs for organic preparations, I've learned to keep plenty of air moving anytime a solvent with this kind of vapor pressure shows up.
Technical Specifications & Labeling
Anyone handling 2-butanol in an industrial or lab setting checks the container for percent purity and residual water content—every bit as critical as the physical look or feel. Regulations in many countries require clear hazard markings tied to its flammable nature and potential for irritation; these labels are not just bureaucratic overhead. Labels typically show the CAS number, warning pictograms, and sometimes the enantiomeric makeup as the chemical can show up as either the racemate or a specific optical isomer. People in the chemical industry respect the paperwork as much as the glassware—mixing up isomers or missing a hazard warning can set a project back by days or weeks.
Preparation Method
Old textbooks talk about classic fermentation and biosynthetic methods, but those routes rarely make commercial quantities. In the real world, most 2-butanol comes from industrial hydration of 1-butene or 2-butene, usually using acid catalysts. The choice between direct and indirect hydration often comes down to equipment and feedstock purity—plants near large petroleum crackers will opt for direct methods to save on catalyst costs. In the lab, a common way to synthesize it starts from 2-butanone with reduction by sodium borohydride. This route gives reasonably pure secondary alcohol without heavy byproducts, and that convenience makes it popular for students and researchers working on small scales.
Chemical Reactions & Modifications
What makes 2-butanol flexible is its ability to act as both a substrate and a reagent. As a secondary alcohol, it oxidizes easily to create 2-butanone (MEK), a common solvent, by using mild oxidizers. People have used Jones oxidation or PCC for this, though safety concerns now push many labs toward less toxic choices. On the reduction side, it converts cleanly to butane with strong hydrogenation or dehydration and hydrogenation steps. In substitution reactions, chemists sometimes convert it to halides for use as alkylating agents in organic synthesis. Its chiral center gives rise to enantiomers, opening doors for asymmetric synthesis and optical purity testing.
Synonyms & Product Names
Most chemists recognize 2-butanol from a dozen other names, especially secondary butanol or sec-butanol. Sometimes it comes labeled by its IUPAC name, but anyone who spends enough time in stockrooms learns to search by all its names, including methyl ethyl carbinol, or its enantiomer-specific terms like (S)-2-butanol or (R)-2-butanol. In supply catalogs, it can turn up as SBA or simply as butan-2-ol—there’s always a good reason to double-check before mixing that first solution.
Safety & Operational Standards
Anyone who has worked around volatile solvents knows the importance of respecting 2-butanol’s hazards. Its flash point is low enough that flames and static electricity pose real fire risks. Exposure can lead to drowsiness or dizziness; in poorly ventilated areas, higher concentrations in the air can lead to serious intoxication. Keeping personal protective equipment handy—goggles, gloves, and sometimes even a face shield—makes a difference when handling liters instead of milliliters. Safety data sheet reviews and local training are not just box-ticking activities; spending a night dealing with fumes drives home the reason those rules matter. Emergency showers and eyewash stations deserve their place near any bench where secondary alcohols are on the menu.
Application Area
2-Butanol’s bread and butter comes from its use as a solvent in chemical manufacturing and pharmaceutical synthesis. Synthetic routes often include alcohols as intermediaries or solvents, and in particular, 2-butanol finds its way into the making of butanone and as a reagent in making esters and other fine chemicals. Its industrial role doesn’t end there; it sees use in the production of cleaning agents, adhesives, and as a flotation agent in ore processing. On the research side, folks investigating new catalysts and studying stereochemistry often turn to its unique structure to test new analytical techniques or build specialty reagents.
Research & Development
Academic researchers keep 2-butanol in their toolbox, not just for routine syntheses but also as a testbed for chiral resolution and optical activity experiments. As green chemistry projects gain momentum, there’s increasing interest in biocatalytic production of secondary alcohols to avoid heavy metal waste and cut fossil feedstock use. Even in established industries, this alcohol shows up in process optimization studies, where improved yields and safer reaction conditions are the order of the day. In my own work, I've seen 2-butanol used to train new graduate students on chromatographic resolution of enantiomers, since its relatively simple structure provides an approachable case study.
Toxicity Research
Studying the toxicology of 2-butanol means looking beyond just immediate flammability hazards. Researchers tracking metabolic pathways have identified its breakdown to methyl ethyl ketone, which itself has CNS effects at high exposures. Animal studies have shown effects at high doses on the liver and kidney, but at levels found in controlled industrial environments, the risks come primarily from acute intoxication and chronic inhalation. Industries have taken steps to minimize exposure by improving ventilation control and providing monitoring for occupational exposure. Poison control centers note that most accidental exposures come from lack of ventilation or improper storage, pushing for better education in small labs and teaching settings.
Future Prospects
Growth in specialty chemicals hinges on finding safer, cleaner, and often more sustainable ways to make building blocks like 2-butanol. With demand for greener solvents and pushback against petroleum-based processes, companies and research labs are exploring fermentation pathways using engineered microbes, even though yields and costs still trail the chemical routes. Advances in analytical chemistry open the door for finer control of optical purity, which in turn benefits pharmaceuticals and specialty reagents. If new catalysts can drive selective reactions without harsh acids or precious metals, expect to see more processes calling for 2-butanol as either a product or key intermediate. Seeing its long-term future depends on how chemists solve the age-old problem of balancing economics, safety, and environmental impact. In my view, those solutions come from listening to operators and researchers who actually work with these chemicals each day, who see firsthand where the process falls short or where new methods change the safety calculus.
What are the main uses of 2-Butanol?
Finding 2-Butanol Beyond the Lab
Step into a garage, a print shop, or a chemical warehouse, and chances are you’ve crossed paths with 2-butanol. This liquid sits among a family of alcohols best known for being more than just solvents but as workhorses across industries. I’ve watched a friend in commercial printing swear by the stuff for cleaning presses, cutting through ink like it’s nothing. He’d probably say 2-butanol holds its place because it gets the job done safely and quickly.
Solvent That Keeps Work Moving
Solvents often make or break efficiency on a production line. 2-butanol shows up in paints, varnishes, and coatings because it dissolves dyes, oils, waxes, and resins better than plenty of alternatives. Thanks to its short carbon chain, it evaporates at a speed that balances drying time with coverage—an open secret among painters and refinishers. The careful use of 2-butanol also stems from its performance with adhesives, helping glue set without becoming brittle.
Folks in labs rely on its cleaning power. Research from the Journal of Chemical Education points out how it’s a go-to for crystallization or separation steps in organic chemistry. I remember moments during my university days where skipping higher-cost solvents meant relying on 2-butanol for a tight budget and decent results.
Building Block for Bigger Chemistry
Synthetic processes run better with reliable reagents. 2-butanol acts as a starting material for the production of methyl ethyl ketone, a chemical that powers up cleaning agents, lacquers, and some modern plastics. That chain of use doesn’t end there—it contributes to perfumes, rubbers, and various butyl esters found in the plastics industry.
Current figures from global chemical manufacturing show demand steadily rising as emerging markets push for coatings and adhesives that meet higher standards. Responsible sourcing and improved manufacturing lower risks, keep costs under control, and encourage wider application.
Concerns About Health and Environment
Any chemical with wide reach brings questions about safety. Small amounts of 2-butanol in controlled settings don’t set off alarms. Still, regular handling at industrial levels needs respect. Inhaling vapors or long-term skin exposure can lead to dizziness, skin irritation, or more serious problems. The U.S. National Institute for Occupational Safety and Health lays out exposure guidelines that protect workers, but enforcement varies. Local oversight, routine air quality checks, and strong ventilation go a long way.
Wastewater and accidental spills introduce another angle. 2-Butanol does break down relatively fast in the environment, but not instantly. Municipal systems need to watch for overload to prevent harm to aquatic life. Encouraging closed systems, better leak detection, and on-site recycling can push industries ahead without losing sight of safety.
Pushing Toward Greener Chemistry
Demand for chemicals with a lower environmental footprint only grows. Researchers in Europe and Asia look for alternative sources, such as using renewables or biological processes, to cut fossil-fuel dependence. The shift takes time, but real progress shows up as factories try out cleaner production lines or tweak waste handling.
Tougher regulations and customer demand both drive the move for change, building a future where solvents like 2-butanol do the job without leaving a mess behind. Watching manufacturing improve over the years brings hope that workers, communities, and ecosystems can all benefit.
Is 2-Butanol hazardous to health?
Looking at Risks and Common Sense
People start getting nervous when chemicals with names like 2-Butanol crop up in the workplace or laboratory. The name alone gives off a sort of ominous, laboratory vibe. Truth is, 2-Butanol isn’t all that exotic—it’s a secondary alcohol, pops up during fermentation, and chemists see it a lot. That doesn’t mean it’s harmless.
Inhaling vapors of 2-Butanol feels a lot like breathing in rubbing alcohol. When someone breathes it in, there’s a risk of headaches, nausea, even dizziness. Extended or repeated exposure has sent people to occupational health with mental fog and eye irritation. If you splash it on your skin, you’ll notice fast enough—a burning, drying sensation. I still remember a minor splash in my undergrad days; the redness lingered for days. Not fun. Drink it by accident, and you’re looking at central nervous system depression, confusion, loss of coordination. The fact that it’s in the same chemical family as ethanol and methanol should set off alarms—the body breaks it down in similar ways, and that process can go sideways fast.
Anyone handling the stuff needs to keep some basic safety gear in reach. Disposable gloves, eye protection, maybe a lab coat. Ventilation matters, too. I’ve worked in labs where the smell lingers in your nostrils long after you’ve put things away. A good fume hood keeps you from taking that vapor home in your lungs. There’s no badge of honor in skipping procedures—take it from the students who spent the rest of lab barely able to think straight after a spill.
Data People Shouldn’t Ignore
The U.S. National Institute for Occupational Safety and Health (NIOSH) lists 2-Butanol as a potential hazard. Their guidelines show the recommended airborne exposure limit sits at 100 ppm over an eight-hour shift. Above that, health effects get more severe. The International Agency for Research on Cancer (IARC) hasn’t nailed down clear links between 2-Butanol and cancer, though data keeps growing. Chronic high exposure hasn’t played out in huge case studies—at least not yet. Still, even simple acute symptoms create enough disruption for anybody sharing airspace with this chemical to take it seriously.
Most cases of trouble come from workplaces that don’t respect basic safety practices. I’ve seen left-behind open containers on benches or faulty fume hoods that no one reports. These sloppy moments add up to unnecessary risks. No one expects to end up at a doctor’s office from routine work, but every year people do. Sometimes it’s just headaches or skin rashes, but it’s proof that taking short-cuts with chemicals rarely pays off.
Better Habits, Fewer Hazards
Training drives safer labs and factories, plain and simple. Every new student or worker should walk through real-life spill scenarios, not just check off online tests. Keeping material safety data sheets (MSDS) handy can help refresh memory on symptoms and steps. Eye washes and showers shouldn’t gather dust—they save vision if a splash does happen. Labeling every bottle and keeping inventory cuts down on stray containers that get forgotten and pose risks long after their usefulness ends.
On a bigger scale, industries looking for greener processes now try to swap out hazardous solvents wherever possible. Substitution isn’t always easy, but it can prevent chronic exposure in the first place. Until better options are everywhere, attention to detail, protective gear, and routine maintenance on safety equipment stay at the front of the line. Most accidents with 2-Butanol follow repeated patterns that enough people have warned about. Pay attention, stick with what’s proven to work, and ordinary days stay ordinary—no need for drama or injury in the name of science or productivity.
What is the chemical formula of 2-Butanol?
Breaking Down 2-Butanol
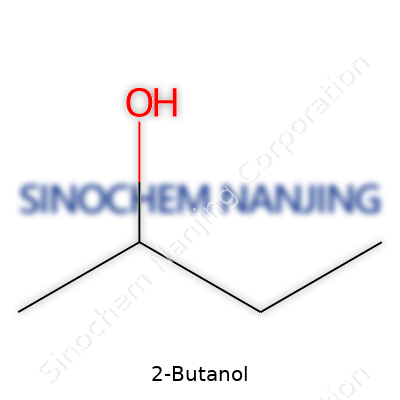
2-Butanol carries the chemical formula C4H10O. Anyone who’s set foot in a chemistry lab or picked up a bottle of rubbing alcohol might have come across names like butanol, iso-butanol, or sec-butanol—these small changes tell a lot about structure and function. 2-Butanol places its hydroxyl group (-OH) on the second carbon atom, which brings out unique properties. This isn’t a trivial difference. Shifting a functional group by just one spot along a chain alters physical behavior, toxicity, and how the molecule interacts in larger chemical reactions.
Straightforward Chemistry with Practical Impact
My own foray into organic chemistry courses introduced me to the world of isomers, where two compounds can share a formula but behave nothing alike. In the case of 2-Butanol, that presence of the -OH group sets the stage for its identity as a secondary alcohol. Not all alcohols are created equal. The secondary alcohol stands apart because its main carbon connects to two other carbons—giving it specific reactivity, especially in oxidation reactions. Chemically, it reads as CH3CHOHCH2CH3; you end up with four carbon atoms, ten hydrogens, and a single oxygen—balancing the formula as C4H10O.
It pops up in many industrial processes, acting as a solvent for paints and coatings, and serving as an intermediate in making plastics, rubbers, and even pharmaceuticals. This makes accuracy in labeling and knowledge about its chemical structure critical for anyone working with it—whether in the lab or on the shop floor. Incorrect handling or misunderstanding of its properties has, in the past, resulted in chemical spills and unsafe exposures. This isn’t the stuff you want poured down a drain or stored carelessly.
Real Life and Safety Concerns
Safety isn’t just a theoretical concern. A friend of mine who worked in chemical manufacturing once shared a story—someone misread a label, assuming “butanol” always meant the same thing, which led to a costly production halt. Turns out, knowing whether you have 1-butanol or 2-butanol makes a big difference in equipment compatibility and safety practices. Though both have the same ratio of elements, their boiling points, toxicity, and even smells can throw off equipment or product outcomes.
Supporting Accurate Handling and Education
One practical way to minimize errors comes through clear, detailed training and mandatory labeling standards. Chemical companies and educational labs benefit from hands-on safety workshops that don’t just recite formulas but talk through the consequences of getting it wrong. Technology has also made it easier to access chemical data sheets quickly, but fostering an environment where asking questions or double-checking information is encouraged might be the missing piece in many workplaces.
Moving Beyond the Formula
At its core, the chemical formula tells you how many carbon, hydrogen, and oxygen atoms exist in one molecule of 2-butanol. That handful of letters and numbers reflects decisions in manufacturing, influences lab safety, and shapes what comes out the other end of a production line. The world of chemicals rarely deals in abstract ideas—small differences affect real people and real outcomes every day.
How should 2-Butanol be stored safely?
Understanding the Real Hazards
Think of 2-Butanol as more than a typical alcohol. This stuff can catch fire easier than people realize, and if careless handling comes into play, it can mean trouble fast. It isn’t only flammable—a spill in a stockroom or lab could send fumes through the air, bringing headaches or dizziness to anyone without solid ventilation. More than once in small academic labs, poor labeling or sloppy sealing has resulted in strong smells wafting down the hall, making everyone tense about the fire risk.
Getting Storage Right from the Start
2-Butanol belongs in a dedicated chemical cabinet built for flammables. That means solid metal construction, doors that close securely, and no wood shelves. Usually, I’ve noticed labs cut corners by mixing solvents in regular cupboards or even under benches. All it takes is one forgotten open container or damaged jar for things to go sideways.
Keep containers in a cool, dry corner away from any sparks, heat, or open flames. Lighting plays a big role—hot lights or exposed bulbs close to shelves increase risk. Temperature fluctuations also speed up evaporation, ramping up pressure inside containers, which leads to leaks. Folks running older facilities without climate control often underestimate summer heat: vapor pressure goes up, and bottles hiss or warp under stress.
Choosing and Labeling Containers
Always go for tight-sealing glass or metal bottles, marked specifically for chemical storage. Never try to stretch the life of old containers, and skip plastics that can soften or crack from the solvent. I’ve run into plenty of labs still using scuffed plastic jugs from years back, only to find puddles under them later. Permanent labels showing not just the name, but the date received and initials of the user help others spot issues early. If you’re in a shared space with rotating students or staff, tracking who last handled each bottle proves worth the extra effort.
Ventilation and Workspace Practices
Fume hoods don’t just keep you safe while pouring—they remove stray vapors that collect even from supposedly closed bottles. Labs working without good ventilation often notice the smell increasing around storage spots. Storing 2-Butanol on high shelves may seem like a space-saver, but in a spill, the liquid trickles down and makes cleanup tougher. Eye-level storage, with unobstructed doors, lets you spot condensation or leaks right away.
Controlling Quantity and Access
Only stock what you’ll use in a reasonable window. Hoarding big drums looks efficient on paper, but increases the fallout if a leak starts. Even veteran lab workers sometimes get overconfident, pulling extra bottles to their bench and leaving them open for hours. Limiting access to staff with proper training, and storing surplus material in locked cabinets, adds a layer of safety that pays off when new people start handling chemicals.
Preparing for Accidents
Easy-to-find spill cleanup kits—sand, absorbent pads, and disposal bags—should live close to flammable cabinets. Keeping a powder or carbon dioxide extinguisher in arm’s reach, checked monthly, reduces panic if flames break out. Emergency contacts and chemical data sheets taped inside cabinet doors make sure everyone knows what action to take fast, without hunting for instructions.
Learning from Real Mistakes
In my early days, I saw storage rules ignored in the rush of late-night experiment runs. Labels peeled off, bottles jammed in wherever they fit, and warnings about flammability chalked up to over-cautious managers. After a scare involving a corroded bottle and a call to the fire department, everyone got a lot more serious. Just following these basics makes work safer—and allows research (or industry) to keep moving forward without a hitch.
Is 2-Butanol soluble in water?
Real Chemistry Meets Real Life
People in schools and labs often ask if 2-butanol mixes with water. On paper, the answer is yes—2-butanol dissolves in water. You get this from organic chemistry textbooks, but that’s only half the story. In practical use, solubility always comes back to what you want out of the mixture. If you pour 2-butanol into a beaker of water, you won’t get a full-on solution like sugar in your coffee, but the two will blend well enough for most uses.
Understanding Solubility in Everyday Terms
Let’s strip things down to what matters. In water, polar molecules move and mingle well, and alcohols usually mix pretty decently. 2-butanol raises more questions because it’s got both a polar side (thanks to the –OH group) and a nonpolar tail (the rest of the molecule). If you’ve ever spilled vodka and noticed how easily it blended with ice water, you’ve already seen alcohol’s talent for dissolving in water. But as the alcohol molecule gets bigger, mixing gets a bit more complicated.
Why 2-Butanol Actually Mixes with Water
I’ve worked in kitchens, labs, and DIY setups—everywhere liquids get dumped and problems need solving. 2-butanol dissolves in water, but not as much as smaller alcohols like ethanol. Water’s hydrogen bonds pull at the –OH group of 2-butanol, letting some of it sneak between water’s molecules. Scientific data say you can get up to 29 grams of 2-butanol into 100 grams of water at room temperature. Not as much as with methanol or ethanol, but plenty for making tinctures, solvents, or reaction mixtures.
Try mixing more than that, and you’ll see layers start to separate—similar to what happens with oil and water, just less dramatic. Anyone prepping chemical solutions or running a distillation setup knows you always need to check these numbers if you want things to go smoothly.
Why Solubility Matters Beyond the Classroom
This blend of chemistry finds its way into daily life. Solubility data shape everything from how you clean a countertop to how medicines get absorbed by the body. In the pharmaceutical industry, solubility tells scientists whether a pill gets swallowed by the body or just passes through. For industry, knowing just how much 2-butanol goes into a solution means better outcomes from glue to fuel additives. Users in labs focus on dosages and concentrations, because too much can shift a solution into a messy mixture.
What Can Be Done With This Knowledge?
If you’re mixing 2-butanol with water at home or in a professional lab, the important thing is to know why you want them together. For better results, always check temperature—hotter water holds more 2-butanol. For green chemistry and safety fans, this also brings up handling: even though it blends in, 2-butanol can still release vapors or irritate skin. Gloves, good ventilation, and careful measurements help you use it safely and efficiently.
Science works better in the real world when you balance hard facts with hands-on experience. Checking solubility tables gives you a start, but pouring, observing, and adjusting finishes the job. Mixing 2-butanol and water is less about textbook trivia and more about getting the right solution for what you want to accomplish.
| Names | |
| Preferred IUPAC name | butan-2-ol |
| Other names |
sec-Butanol sec-Butyl alcohol 2-Butyl alcohol methyl ethyl carbinol |
| Pronunciation | /ˈtuːˈbjuː.tə.nɒl/ |
| Identifiers | |
| CAS Number | 78-92-2 |
| Beilstein Reference | 1713886 |
| ChEBI | CHEBI:28398 |
| ChEMBL | CHEMBL1359 |
| ChemSpider | 6269 |
| DrugBank | DB03374 |
| ECHA InfoCard | 100.004.882 |
| EC Number | 200-751-6 |
| Gmelin Reference | 82813 |
| KEGG | C00474 |
| MeSH | D001943 |
| PubChem CID | 6560 |
| RTECS number | EL5425000 |
| UNII | LFQ7SNO3N7 |
| UN number | UN1120 |
| Properties | |
| Chemical formula | C4H10O |
| Molar mass | 74.12 g/mol |
| Appearance | Colorless liquid with a characteristic odor |
| Odor | sweetish |
| Density | 0.806 g/cm³ |
| Solubility in water | Miscible |
| log P | 0.61 |
| Vapor pressure | 2.7 kPa (at 20 °C) |
| Acidity (pKa) | 16.1 |
| Basicity (pKb) | pKb = 15.20 |
| Magnetic susceptibility (χ) | -53.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.399 |
| Viscosity | 2.98 mPa·s (at 20 °C) |
| Dipole moment | 2.79 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 157.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –318.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2674 kJ/mol |
| Pharmacology | |
| ATC code | R02AD03 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Precautionary statements | P210, P261, P280, P301+P312, P304+P340, P337+P313, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 24 °C |
| Autoignition temperature | 400 °C (752 °F; 673 K) |
| Explosive limits | 2–11.8% |
| Lethal dose or concentration | LD50 Oral Rat 2190 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2190 mg/kg (oral, rat) |
| NIOSH | NIOSH: SE9100000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 2-Butanol: 150 ppm (450 mg/m³) |
| REL (Recommended) | 300 ppm |
| IDLH (Immediate danger) | 3400 ppm |

