2,5-Hexanedione: A Closer Look at an Industrial Chemical’s Journey and Future
Historical Development
The story of 2,5-Hexanedione traces back to the gradual growth of organic chemistry and its role in the modern economy. Early research into aliphatic diketones shed light on this compound, as scientists chased new intermediates for synthetic processes. By the late twentieth century, people in chemical industries started taking a keen interest in this molecule, largely due to incidents involving toxic workplace exposures. Before regulations brought workplace safety into sharper focus, 2,5-Hexanedione often popped up in places like paint shops, glue factories, and laboratories. The compound earned a certain notoriety, not for its productivity, but because it raised difficult questions about safety and responsibility in industry.
Product Overview
This transparent, slightly oily liquid comes with a distinct sweet, acetone-like smell. Chemists classify it as a diketone, key for unlocking a range of transformations in chemical laboratories and production lines. Most users see this as both a valuable tool and a potential risk. It shows up in solvent mixtures, paints, inorganic chemical syntheses, and even in the manufacture of some plastics. It quickly dissolves in water and most organic solvents, which makes it easy to use but also easy to spill or leak.
Physical & Chemical Properties
You can spot 2,5-Hexanedione by its faintly yellow hue in concentrated form, though it’s mostly colorless in solutions. The liquid boils at around 197°C and flows with moderate viscosity. Under normal atmospheric conditions, it evaporates at a rate similar to that of ethylene glycol, so the risk of inhalation lingers unless ventilation runs well. Structurally, it stands as a straight chain with two carbonyl groups at the 2 and 5 positions, which strongly influence its reactivity and interactions with other molecules.
Technical Specifications & Labeling
With every bottle, producers typically provide a label detailing its purity, molecular weight (114.14 g/mol), and hazardous designations under systems like GHS. For chemistry labs, a purity higher than 98% is standard, largely because technical impurities can trigger side reactions or interfere with experiments. I remember more than one instance in graduate school where a less-than-pure reagent ruined days of work, showing that an eye for technical details saves time and headaches. For bulk industrial orders, batch consistency becomes more critical than even trace purity, showing how end-use shapes what “quality” really means.
Preparation Method
Industrial routes produce 2,5-Hexanedione mostly by oxidizing n-hexane or related alkanes, but small-scale syntheses often start from adipic acid or its esters. Oxidative cleavage using strong acids or peroxides turns up as a popular laboratory method, especially for researchers looking to make only a few grams. Large chemical companies prefer methods that generate fewer toxic byproducts, not just for cost but due to mounting regulatory scrutiny. There’s no getting around the fact that anyone working with this compound needs gear that withstands strong acids, oxidizing agents, and sometimes high heat.
Chemical Reactions & Modifications
2,5-Hexanedione acts as both a reagent and a synthetic intermediate, thanks to those active carbonyl groups. Organic chemists appreciate its usefulness in forming heterocycles, including pyrroles and other nitrogen-containing rings. In the industry, it often serves as a building block for advanced adhesives and certain pharmaceuticals. Researchers can use simple condensation reactions to turn it into more complex molecules. Its reactivity brings up its downside too: it doesn’t just react in the flask; it also interacts with proteins in the body, which ties directly into its safety profile.
Synonyms & Product Names
In scientific papers, you’ll see people mention 2,5-Dioxohexane, Hexane-2,5-dione, or even Pyrodione. In supply catalogs or regulatory lists, the simplest name typically surfaces, to avoid confusion during shipment or customs inspections. Over the years, some European safety agencies have flagged it under older terms, illustrating how chemical naming conventions shift with the language and legal guidelines of different countries.
Safety & Operational Standards
Anyone spending time around this chemical learns quickly that safety isn’t optional. Even at low doses, long-term exposure has left workers with severe nerve damage, a fact documented in case studies throughout the 1970s and 80s. That history laid the groundwork for tough controls: air monitoring, personal protective equipment, and restricted access in most reputable facilities. Chronic exposure in some jobs started the conversation about occupational disease registers and exposed the loopholes of earlier regulatory systems. Today’s standards reflect a history of harsh lessons, including real suffering. Most countries set strict short-term and long-term exposure limits, enforced with on-the-spot workplace inspections and rigorous documentation of handling procedures.
Application Area
People use 2,5-Hexanedione where its carbonyl power makes a difference: custom resins, special adhesives, and the lab-scale synthesis of bioactive compounds. It factors into environmental analysis too, especially as a marker for certain industrial emissions. In my years working alongside analytical chemists, I’ve seen it turn up as a background contaminant, posing extra burdens for cleanroom processes. Some investigative toxicology labs maintain samples as references to monitor for workplace exposure, underscoring its role in both industry and health sciences. Its value to organic synthesis keeps it on the research bench, even as its risks lead some companies to search hard for alternatives.
Research & Development
Research into 2,5-Hexanedione breaks down into two camps: new syntheses and health science. In laboratories, chemists keep pushing boundaries, using this diketone as a lynchpin in building up molecules with more practical benefits, such as flame-resistant coatings or pharmaceutical candidates. From a toxicological angle, research digs into the nuts and bolts of how exposure leads to nerve injury, and how to predict or block those effects. In recent years, advances in non-toxic synthetic pathways have started to appear, reflecting mounting environmental regulation and pressure from advocacy groups for safer workplaces.
Toxicity Research
Much of the notoriety attached to 2,5-Hexanedione comes from its grim track record with nerve toxicity. Researchers learned decades ago that this chemical builds up in nerve tissue and causes progressive, sometimes irreversible neuropathy. Studies in lab animals echo findings in human workers: long exposure eats away at nerve fibers, causing loss of coordination and muscle weakness. That knowledge sits at the base of workplace engineering controls. For scientists and workers, current research investigates the exact metabolic pathways that convert 2,5-Hexanedione’s structure into nerve poisons, with the hope of finding interventions or treatments. Medical monitoring, once unheard of for many industrial chemicals, now runs as standard practice in facilities with known exposures.
Future Prospects
Looking ahead, two paths run alongside each other. Chemical industries keep tweaking their methods to either minimize or fully phase out 2,5-Hexanedione in places where workers come into contact with it. At the same time, the compound sticks around in highly specialized synthesis and advanced-materials research, where no easy substitute exists. Ongoing push from occupational safety advocates and regulatory bodies guarantees that research into less-toxic analogs and alternative processes won’t slow down any time soon. In the best scenarios, we see smarter workflows, better medical surveillance, and safer chemicals. The wider conversation shows how a legacy of harm and innovation can sit side by side, shaping the next decades of chemical safety and creativity.
What is 2,5-Hexanedione used for?
Behind the Scenes in Industry
Factories often keep 2,5-hexanedione out of sight and out of mind, but it makes a deep mark on chemical manufacturing. This colorless liquid pops up most along the winding route to nylon and certain plastics. It steps in as an intermediate, helping to build larger molecules needed for fibers and engineering materials. Nylon production drives its use; manufacturers rely on it to stitch together molecules that will turn into the tough, flexible threads found in everything from carpets to airbags.
The Human Side of Chemical Exposure
People who work in these plants, and folks living in nearby communities, can end up facing unexpected hazards. Medical journals flagged 2,5-hexanedione’s neurotoxic side after workers developed nerve problems. Researchers saw that long-term exposure, even if it wasn’t noticeable day to day, led to muscle weakness and numbness, mostly in the hands and feet. This isn’t just another chemical quirk—it has forced companies to rethink safety. Even now, some countries check workplace air for traces and require personal protective gear.
Why Workers Stay Vigilant
My grandfather, a plant mechanic in the 1970s, shared stories of coworkers who learned the hard way about the risks of solvent fumes. They didn’t get clear warnings until symptoms crept up. Today, science backs up these experiences—2,5-hexanedione messes with the way nerves work by attacking the axons. In practical terms, regular check-ups, better ventilation, and using less risky substitutes make more difference than just paper rules on safety.
Alternatives: The Push for Safer Choices
Driven by mounting evidence about health risks, some companies seek out less toxic chemicals. Even though 2,5-hexanedione fits its job, businesses that make a habit of reviewing their processes—and swapping out hazardous ingredients—tend to avoid surprise lawsuits and lost workdays. Industry groups push for data sharing, so others know which chemicals can do the same job with less harm. As a journalist, I’ve seen real improvements once plants face public pressure or work with unions to flag unsafe material.
Sustainable Chemistry Steps Forward
Beyond immediate health, the demand for cleaner chemistry lines up with bigger environmental goals. Making nylon with greener ingredients trims down waste and shrinks the carbon footprint. Academic labs invest time in designing new routes that cut out 2,5-hexanedione or break it down safely after use. Some European regulations push producers to limit or report their releases into water and air. This whole process doesn’t just help the planet—it keeps local communities safer and builds trust in brands.
Looking Ahead: Shared Responsibility
Anyone working around 2,5-hexanedione deserves honest information and a voice in safety plans. It pays to read the small print on chemical labels and ask questions about substitutes. Real change comes when governments, companies, and everyday folks agree to draw a line, favoring health and safety over quick fixes. In my reporting, I’ve seen workers who spoke up spark company-wide overhauls. For new chemicals and old standbys alike, respect for people goes hand in hand with solid science.
What are the health hazards of 2,5-Hexanedione?
Toxic Impact on Nerves
2,5-Hexanedione catches most folks off-guard because the risks do not come with a strong smell or obvious warning sign. This chemical drifts through industrial sites and labs, showing up in places tied to making adhesives, coatings, and plastics. A lot of people find out about its dangers only after symptoms kick in. The biggest concern centers on its effect on the body’s nerves. Workers exposed through breathing or skin contact can start to feel tingling, numbness, or weakness, especially in their hands and feet. Over time, these nerve effects stretch up arms and legs. Long-term exposure links clearly to a nerve disorder called peripheral neuropathy.
Unlike sudden poisoning, the nerve damage creeps in slowly. A person might keep working even as symptoms worsen, not connecting small signs like clumsiness or muscle pain to a chemical at work. Years ago, shoe and glue workers often developed what they called “drop foot” or lost the ability to pinch small objects. Medical scans now confirm that 2,5-Hexanedione attacks nerve fibers’ axons, blocking signals that control movement and feeling.
How it Gets Into the Body
Some folks think toxic chemicals mostly enter through swallowing, but 2,5-Hexanedione breaks that notion. The chemical moves in through breathing dust or vapor, and it slips through the skin as well. Areas with poor ventilation or workers not using gloves raise the risk. In smaller workshops, personal protective equipment gets skipped because the chemical does not cause fast, painful effects. Chronic exposure from small, repeated contact can cause just as much harm as a one-time large spill.
Other Health Effects
2,5-Hexanedione causes more than just nerve problems. Animal research connects higher exposures to liver and kidney injury, meaning this chemical can tax organs responsible for clearing poisons from blood. Some evidence ties it to reproductive effects, including lower sperm counts. While these studies come from labs using high doses, the signal cannot just be ignored, especially for people mixing or handling the chemical every day at work.
Workplace Safety and What Works
No one stays safe by guessing. The only way to control 2,5-Hexanedione’s risks uses a few old-fashioned principles. Good ventilation pulls harmful air away from people. Gloves and protective clothing block it from sinking into skin. Air testing in workplaces pinpoints problem areas before workers get sick. Strict government exposure limits mean something only if people measure the air and act on the results. Education goes a long way too. Workers who have seen firsthand what chronic exposure does tend to double-check that their gloves are on and their masks fit.
Regulation by agencies like OSHA helps by setting exposure limits, but supervisors and safety leaders must take those limits seriously. Management builds trust by sharing risks openly. I have watched people skip safety steps until someone close gets sick, then demand stronger rules and more testing. Bringing in outside safety trainers or medical experts helps keep the conversation honest. In every workplace, prevention proves cheaper and kinder than treating chronic nerve damage.
Instead of waiting for new medical treatments, everyone involved—company leaders, workers, doctors, and regulators—owes it to themselves to push for safer handling, better equipment, and frank information about the costs of ignoring 2,5-Hexanedione’s hazards. Early action keeps hands steady and feet strong, letting workers return home healthy at the end of the day.
How should 2,5-Hexanedione be stored?
Understanding the Dangers
2,5-Hexanedione doesn’t get much attention outside industry, yet the dangers deserve real respect. I’ve spent years in spaces where this colorless liquid shows up—often as a by-product or an industrial cleaner. Even seasoned teams can get too comfortable, letting procedures slide. Trust me, nobody wants to learn about chemical neurotoxicity the hard way. Inhalation and skin contact can produce long-term nerve problems. There’s a reason why the storage room for this stuff always catches the eye on safety inspections.
Practical Lessons on Safe Storage
Simple label warnings don’t stop accidents. From experience, the difference between a dusty corner and a dedicated flammables cabinet decides whether someone goes home healthy. 2,5-Hexanedione gives off vapors that both ignite and sicken. Locked cabinets built for fire resistance make a huge difference. I’ve watched coworkers roll their eyes, but one spill next to a space heater can do more than ruin a shift.
Strong ventilation in storage areas cuts down vapor buildup. Most places have an OSHA poster in the hallway, but few invest in actual airflow. Money spent on proper fans will always beat the cost of dealing with a health investigation. I once saw a lazy workaround—a chemical drum next to an open window. Wind direction changed, and a passing technician landed in the ER. The right solution never involves shortcuts.
Choosing the Right Containers
Not every bottle is tough enough. Polyethylene and glass work well with 2,5-Hexanedione. Metal and some plastics corrode, creating leaks that go unnoticed until a strong odor or a funny-tasting lunch breaks the news. Tightly sealed, purpose-built containers stop those vapors from escaping. Every chemist I know checks container seals before and after each transfer, no matter how many times they’ve done it.
Labels and Segregation—Not Just “Red Tape”
Mix-ups and unmarked containers turn minor incidents into medical emergencies. I’ve helped sweep up broken glass where some joker used an unlabeled bottle, and it always leads to panic. Universal, chemical-resistant labels, updated with the date and initials of the person responsible, save more than paperwork. Never place 2,5-Hexanedione next to acids or oxidizing agents; these combinations lead straight to disaster. Store this liquid in areas built for organic solvents, away from anything that could trigger unwanted reactions.
Training: The Missing Piece
Safety gear gets flashy posters, but real safety starts with knowledge. Most labs and workshops do the bare minimum, handing out a sheet and hoping nobody asks questions. I remember my first thorough training—walking through what to do in a spill, where the eyewash sat, and how to recognize vapor exposure early. After that, sloppy habits disappeared. Annual, hands-on refreshers work better than any lecture or online quiz. Encourage questions, share near-miss stories, and give everyone a stake in keeping the place safe.
Looking Beyond the Store Room
Safe storage of 2,5-Hexanedione means creating a culture that refuses shortcuts. Serious accidents often come from small mistakes—a cracked lid, an outdated label, a forgotten procedure. Respect for proper storage grows from real understanding of what this liquid can cost in health and reputation. If your facility uses or stores 2,5-Hexanedione, keep a sharp eye on every step, and never accept “close enough” as good enough. Investing in simple, consistent practice pays for itself every single day.
What is the chemical structure of 2,5-Hexanedione?
Understanding the Basics
2,5-Hexanedione takes a straightforward approach compared to some unwieldy organic compounds. You find six carbon atoms lined up in a row. At the second and fifth spots along that chain, ketone groups—the ones that put the “-one” in hexanedione—stick out. In chemical shorthand, the structure maps as CH3COCH2CH2COCH3. Staring at that skeleton in a textbook catches attention because of its symmetry: two carbonyl groups facing each other, three carbons apart, nothing fancy hung from the sides.
Why Structure Matters More Than a Drawing
Getting this layout into your head goes way past just memorizing a formula for an exam. The real-world impact pops up in unexpected places, from the workshop to research labs. 2,5-Hexanedione walks straight out of textbooks and right into industrial life, especially as a solvent. This skeleton lets the molecule interact with a wild range of organic materials. Think of it as a versatile handshake in the world of solvents—good at dissolving both polar and some nonpolar compounds because of the way those carbonyl groups tug electrons.
Chemists spot the significance of that 1,4-diketone motif. That arrangement isn’t just theoretical — it means increased reactivity. In actual reactions, those carbonyl groups can start talking to each other through the chain, setting up possible cyclizations and enolizations. For synthetic chemists, these features unlock creative pathways to build bigger, more complex molecules.
Safety Stories from the Laboratory
It’s impossible to talk about 2,5-Hexanedione without touching on what happens if it strays from a controlled environment. Industry and academia have both documented its neurotoxic properties. Too much exposure puts nerves at risk. Workers in old-school workshops using glue or paint thinners found out the hard way: exposure resulted in nerve damage and in some cases, persistent motor issues in hands and feet.
A molecule can look unassuming on paper but pack a punch in the real world. The symmetry in 2,5-Hexanedione contributes to its metabolic fate. Human and animal studies connect the dots from this structure to harmful byproducts in the body. The dione motif turns into a reactive intermediate, gamma-diketone, which crosslinks with essential proteins in nerve tissue. Recognizing the hazard, employers and chemists turned to personal protective equipment, ventilation, and strict exposure monitoring practices. Simple habits, like always labeling workspaces and storing chemicals with purpose, make a big difference. Over the years, tough regulations on solvent emissions and routine nerve testing for workers proved themselves as must-haves.
Searching for a Better Way Forward
Industry continues to chase safer substitutes without sacrificing performance. Researchers revisit the basic structure, looking for tweaks that quietly reduce toxicity and keep useful solvent properties. Training programs for safe chemical handling don’t just flash warnings—they put practical, regular routines in the spotlight. Today’s best practices didn’t appear out of nowhere but grew from listening to everyone: chemists, health and safety officers, and workers who saw issues up close.
2,5-Hexanedione tells its own story. Its structural simplicity offers impressive versatility in chemical synthesis and manufacturing. Yet that same structure also highlights human risks lurking when safety is skipped. Real progress shows up when chemistry knowledge shares the floor with practical experience and genuine care for the people using these compounds every day.
Is 2,5-Hexanedione regulated or restricted in any countries?
Why 2,5-Hexanedione Draws Attention
Many industrial solvents come with a list of hazards, but few raise eyebrows among workplace safety experts like 2,5-hexanedione. Its use in making adhesives, inks, or rocket propellants might sound distant to most people, but the health effects tie directly to everyday job safety. Exposure to this compound can cause severe nerve damage, known as peripheral neuropathy—a slow, sometimes irreversible breakdown in the body’s ability to sense and move. Workers in facilities that handle such chemicals sit closest to risk. Their health records show what extended contact with 2,5-hexanedione can do, and their stories have shaped the regulatory debate worldwide.
What the Rules Look Like Around the World
Some countries waste little time raising alarms. In the United States, the Occupational Safety and Health Administration (OSHA) adds 2,5-hexanedione to its air contaminants list. Factories must keep workplace levels below strict limits—usually around 0.5 ppm (parts per million) as an 8-hour time weighted average. Nobody wants a chemical linked to nerve disorders floating in the air, especially not near workers spending years in those rooms. States and local health departments echo these standards. Not following them opens doors to lawsuits, investigations, and in the worst cases, serious medical crises.
Across the Atlantic, the European Union brings even more scrutiny. Laws under REACH (Registration, Evaluation, Authorisation and Restriction of Chemicals) require companies to report how and where they use 2,5-hexanedione if quantities reach certain thresholds. This means more paperwork, but also more transparency—and a chance for safety managers and regulators to spot dangerous trends before a tragedy happens. Germany and France, known for their tough labor protections, monitor the chemical closely. If science reveals new dangers, expect stricter rules to follow.
Asian regulators start with guidelines from the World Health Organization, then build their own. Japan includes 2,5-hexanedione in its list of substances prohibited or controlled in workplace air. South Korea reviews workplace exposure records yearly, offering health monitoring for workers exposed to neurotoxic solvents. Factories must install ventilation and use protective equipment, not just post safety warnings.
Why These Rules Matter to People and Business
Long-term, the cost of cutting corners with 2,5-hexanedione never works out. Neurological injuries mean hospital bills, lost work, and legal disputes that reach much further than the factory gates. Factory owners might win in the short term by skipping controls, but the risk to people’s health and the company’s reputation isn’t worth it. In places where regulations lag behind, unions and advocacy groups fill the gap by demanding local governments act. Recent studies show lasting nerve damage among workers not protected by strict standards, which brings fresh pressure on lawmakers to update old rules.
Solutions often come from the ground up. Workers speak up, and doctors share research showing early signs of nerve damage. Companies that take safety seriously invest in air monitoring and personal protective gear, which often costs less than dealing with years of medical claims. Sometimes manufacturers reformulate products, switching to less toxic alternatives and promoting these changes to customers who value social responsibility.
Where Action Makes the Difference
Clear rules and tough enforcement offer the best shot at preventing suffering. Chemical safety demands vigilance, not just compliance with old standards. People’s health takes priority. Policymakers and leaders look beyond minimum requirements, listening to those who have lived with the consequences of weak controls. That’s where real change starts—and where the damage stops.
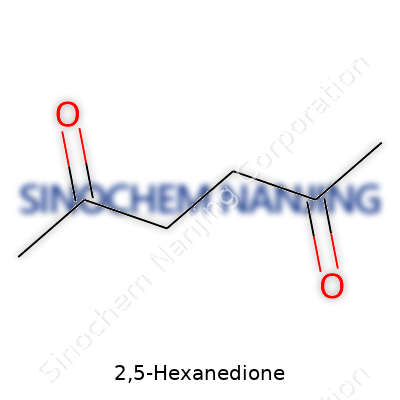
| Names | |
| Preferred IUPAC name | hexane-2,5-dione |
| Other names |
2,5-Dioxohexane Acetonylacetone Hexane-2,5-dione |
| Pronunciation | /ˌhɛk.səˈniː.di.oʊn/ |
| Identifiers | |
| CAS Number | 110-13-4 |
| Beilstein Reference | 1208733 |
| ChEBI | CHEBI:16476 |
| ChEMBL | CHEMBL16221 |
| ChemSpider | 68264 |
| DrugBank | DB02141 |
| ECHA InfoCard | ECHA InfoCard: 100.003.852 |
| EC Number | 211-128-3 |
| Gmelin Reference | 87255 |
| KEGG | C00742 |
| MeSH | D006534 |
| PubChem CID | 7658 |
| RTECS number | MN0875000 |
| UNII | 99P7427X5D |
| UN number | UN 2528 |
| CompTox Dashboard (EPA) | DTXSID0022077 |
| Properties | |
| Chemical formula | C6H10O2 |
| Molar mass | 114.15 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Fruity |
| Density | 0.842 g/mL at 25 °C (lit.) |
| Solubility in water | miscible |
| log P | -0.37 |
| Vapor pressure | 0.67 mmHg (20 °C) |
| Acidity (pKa) | 9.61 |
| Basicity (pKb) | pKb = 13.41 |
| Magnetic susceptibility (χ) | -54.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 1.019 cP (25°C) |
| Dipole moment | 2.72 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 234.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -321.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3153.0 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07,GHS08 |
| Signal word | Danger |
| Hazard statements | H226, H319, H336, H373 |
| Precautionary statements | P210, P261, P280, P304+P340, P312, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 57 °C |
| Autoignition temperature | 190 °C |
| Explosive limits | 1.1–9.4% |
| Lethal dose or concentration | LD50 oral rat 1.48 g/kg |
| LD50 (median dose) | 1.56 g/kg (rat, oral) |
| NIOSH | NIOSH: MN0875000 |
| PEL (Permissible) | 1 ppm (parts per million) |
| REL (Recommended) | 0.4 mg/m³ |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
Acetylacetone 2,3-Butanedione Hexane Glutaraldehyde |

