Looking Beneath the Label: Understanding 2-(2-Aminoethylamino)Ethanol
The Roots: How Science Built Up to 2-(2-Aminoethylamino)Ethanol
Stepping into a chemical lab, even in the modern era, you come across glass bottles with names stretching across the label, sometimes more tongue-twisting than the molecules inside. 2-(2-Aminoethylamino)ethanol falls precisely into that category. This molecule didn’t come out of thin air; it represents the culmination of years of growing understanding about amines, ethanolamines, and the push by industrial chemists to modify simple molecules for more targeted uses. People unfamiliar with chemical history may glance past the importance of semi-structured molecules such as this one, whose backbone comes from putting amine and alcohol groups together. Ask a seasoned chemist about chemical building blocks in their toolkit, and they’ll mention that basic amine-ethanol combos—like monoethanolamine—helped spark a world of research into surfactants, corrosion inhibitors, and stabilizers. 2-(2-Aminoethylamino)Ethanol traces its story through labs that patiently tweaked chain-lengths and polarity, all in search of better performance in industrial and biomedical settings.
The Substance — Product Overview and What It Means in Practice
Let’s ignore the stock photos of test tubes for a second, and focus on what you actually get with 2-(2-Aminoethylamino)ethanol. It’s a colorless to slightly yellow liquid that introduces itself with a faint ammonia-like smell. This compound has an unmistakable duality: there’s the ethanol group offering some water-loving properties, and two amine groups that drive its ability to grab onto other molecules through hydrogen bonding. The trick is in that ethylene bridge connecting both amines, which changes how it interacts with metals and organic compounds. The compound’s structure often plays a part in making selective binders and tailored surfactants. In regular lab speak, people use it to boost solubility and tweak reaction profiles—think of it as a chemical tool for those who like adjusting dials more precisely than usual.
Physical and Chemical Properties: More Than Meets the Eye
Properties matter because they dictate where and how you use a chemical. Here, the liquid state at room temperature, relatively high boiling point, and good solubility in water spell out its strengths. 2-(2-Aminoethylamino)ethanol doesn’t evaporate at the drop of a hat and can get along well with both hydrophilic and hydrophobic compounds. The dual amine groups increase its basicity compared to regular ethanolamine, which often means it acts as a better buffer or chelating agent. Its pKa values tell anyone who’s used it in the lab that you can count on stable behavior in common pH ranges for industrial, cleaning, or biological prep work. Its ability to form salts with acids is handy if you need to shuttle it into water-based processes. It tends to resist oxidation, but will grab up carbon dioxide and acids if left exposed.
Technical Specs and How Labels Shape Understanding
Most bottles you’ll see in the supply cabinet carry labels specifying purity percentages—usually above 98% for research, but lower grades exist for more rugged applications. Because trace impurities like water or aldehydes can affect reactions, labeling often demands strict clarity not just on concentration, but also on storage conditions and shelf life. The technical sheets spell out recommended handling—usually with gloves, not because it’s immediately lethal, but because strong bases can irritate skin and eyes. In my own research, a clear label means less interruptions from Quality Assurance down the line. Accurate specs give confidence in batch-to-batch consistency, which matters most for people running multi-ton syntheses rather than quick student experiments.
How It Comes to Be: The Preparation Method
Synthesizing 2-(2-Aminoethylamino)ethanol doesn’t require mysterious procedures, just a solid understanding of basic organic reactions. Most processes begin with aminoethylethanolamine or by ethoxylating ethylenediamine. In the large reactors, careful addition of reactants and tight control over temperature slows side reactions, which can otherwise produce unwanted byproducts. The odor noticed in many labs generally lingers after synthesis, but careful distillation and scrubbing shed most off-notes. Even though yields are appreciable, thorough purification takes effort. There’s a reason reputable chemical suppliers invest in decent filtration and drying steps—skipping those, and your final product drags along enough impurities to ruin specialty applications, especially in pharmaceuticals or electronics.
Chemical Reactions and Modifications
Chemists gravitate to molecules that offer both flexibility and reliability, and this compound’s dual-functionality enables multiple downstream reactions. 2-(2-Aminoethylamino)ethanol acts like a chemical ‘middleman,’ eager to bind with acids in salt formation or participate in condensation reactions to produce everything ranging from detergents to corrosion inhibitors. The molecule’s primary and secondary amines allow for custom-tailored N-alkylations or acylations. In metalworking, its affinity for copper and iron ions underscores its popularity as a corrosion inhibitor when blended with lubricants or coolants. The alcohol group tempts chemists into further modification—some, for instance, convert it into urethanes or couple it to polymer chains to create emulsifiers with unique performance profiles.
The Many Names It Answers To
Names in chemistry add as much confusion as they do clarity. Look up this compound, and besides 2-(2-Aminoethylamino)ethanol, you’ll run into synonyms such as N-(2-Hydroxyethyl)ethylenediamine, or more cryptically, AEEA. Some people, tired of the mouthful, simply refer to it as 'bis(2-aminoethyl) ethanol.' While labels differ from catalog to catalog, the substance inside the bottle holds true. Knowing these synonyms becomes more than trivia—industry contracts or scientific publications might list only one, and not knowing all of them can stall a project or trigger a regulatory headache.
Laying Down Safety and Operations
Walk through any site using 2-(2-Aminoethylamino)ethanol at scale, and you pick up on safety routines adopted because of hard-learned lessons. Direct contact brings irritation; fumes, if concentrated, sting eyes and lungs. It doesn’t belong near open flames due to its combustibility. Reliable respirators and eye wash stations aren’t just set dressing—they prevent medical emergencies. Storage rules matter: keep the container tightly closed, away from acids and oxidizers. Regulations, in my experience, frequently update as more data turns up on long-term exposure or possible decomposition products. Proper ventilation cuts down on worker risk, and handling protocols, enforced with regular training, keep incident rates lower than they were a few decades back.
Where It Finds a Purpose: Application Areas
2-(2-Aminoethylamino)ethanol proves its worth most clearly where chemical stability, binding power, and buffering meet. In industrial water treatment, it stands out as a corrosion inhibitor, safeguarding cooling systems and boilers against metal loss. Its buffering capacity and solubility place it in specialty detergents or cleaning agents, where standard amines fall short. Look at epoxies or urethanes, and chemists rely on it as a curing agent, locking in polymers in a way that improves strength and extends service life. In making lubricants, it blocks rust and forms stable films on metal surfaces. Some niche pharmaceutical and biotech applications harness its biocompatibility to customize drug delivery systems, but these uses demand higher purity and assurance against trace toxic byproducts. Its role in textile processing, printing inks, or even photography chemicals hints at an adaptability not every amine-alcohol demonstrates.
Research, Development, and What Scientists Are Chasing
Lab notebooks from the past decade highlight an ongoing hunt for safer, greener alternatives in every chemical sector. The unique backbone of 2-(2-Aminoethylamino)ethanol inspires attempts at better chelators for environmental cleanup, and more robust surfactants for tough cleaning jobs. Pharmaceutical researchers remain curious about tweaking this molecule to ferry more complex drug cargos. In my own experience, every research proposal about improved amine-ethanol derivatives involves finding ways to reduce toxicity without losing the desired chemical backbone. Emerging work focuses on blending it with biodegradable polymers to make new coatings, or revisiting its role in copper corrosion for modern electronics—a nod to the balancing act between raw performance and sustainable life cycles.
Turning Over Stones: Toxicity and Long-Term Safety Research
Safety research doesn’t just mean running a checklist; scientists study repeat exposure, accumulative dosing, and rare breakdown byproducts. 2-(2-Aminoethylamino)ethanol’s basic nature demands respect—prolonged skin or respiratory exposure can sensitize workers, and ingestion carries recognized toxicity risks. Environmental impact, particularly aquatic toxicity, drives regulatory scrutiny. Studies over the past twenty years document that, at moderate concentrations, aquatic organisms register adverse effects, underscoring the need for careful wastewater management. Chronic exposure data on humans remains less thorough, which is typical for many industrial chemicals whose usage volumes dwarf pharmaceutical analogs. Lessons from occupational health push for smarter engineering controls and robust spill containment, especially at scales above laboratory synthesis.
On the Horizon — Future Prospects for 2-(2-Aminoethylamino)ethanol
Peering forward, the chemical landscape around this compound thrums with possibility and caution. On the plus side, moves toward circular chemistry—recycling, bio-derivation, or reprocessing—align with the molecule’s core strengths, placing it in contention for greener solvents, better corrosion control, and adaptive surfactants. As industries squeeze every bit of efficiency from materials, the adaptability of dual-functional molecules stands in stark contrast to off-the-shelf single-use reagents. On the flip side, shifting regulatory landscapes and demand for lower toxicity alternatives urge companies to pour more resources into toxicological testing, process improvements, and closed-loop applications that seal potential exposures. In fields like energy storage, textiles, and pharmaceuticals, innovators continue to tinker with molecular analogues, hoping to unlock better performance and safer profiles while protecting worker and environmental health. My experience tells me that the story for 2-(2-Aminoethylamino)ethanol is far from finished—every successful application uncovers new questions about balance, safety, and the next generation of purposeful molecules.
What is 2-(2-Aminoethylamino)Ethanol used for?
Getting to Know 2-(2-Aminoethylamino)Ethanol
Science classrooms have a way of sticking strange names to things that barely show up on anyone’s radar. 2-(2-Aminoethylamino)ethanol, sometimes called AEEA, hides behind a mouthful of syllables, and yet does plenty in the background. I first saw the chemical on a faded drum at an industrial site while consulting on wastewater concerns. Most folks wouldn’t think twice about some off-white liquid labeled with a number as its name, but dig a little and this stuff connects to plenty of practical stories.
Life as a Chemical Building Block
AEEA acts as a handy middleman in a lot of chemistry. Companies producing detergents or fabric softeners rely on modifiers that make surfactants work better or stay more stable. AEEA fills that role, stepping in, boosting properties like foaming or cleaning, and helping companies dial in a product’s cost and performance. It isn’t glamorous, but soap and detergent aisles would look emptier without ingredients like this working quietly.
Changing the Game for Water Treatment
Factories and city treatment plants spend real money to keep water free from the sort of build-up that clogs pipes or coolers. In these operations, 2-(2-Aminoethylamino)ethanol pops up in specialty chemicals that fight scale and corrosion. I once watched a maintenance team switch to a treatment solution made with AEEA and, over a few weeks, that sticky mess in the filter system slowly vanished. Less breakdown means less downtime and repair cost, which any facility manager will tell you makes a noticeable difference in the annual budget.
Supporting Other Chemical Processes
Besides cleaning up water and boosting detergents, AEEA steps into the lab as a raw ingredient in creating bigger chemical structures. Epoxies and resins for adhesives, coatings, and paints often need secondary amines to help cure or give the final product a certain flexibility or weather resistance. AEEA checks those boxes without bringing along harsh byproducts. Some developers searching for specialized metalworking fluids rely on this molecule, blending it for lubricants that cut down tool wear and even give a little extra rust defense on finished parts.
Health and Handling
Not everything about AEEA is rosy. Handling chemicals with strong amine groups always brings up skin and eye irritation. During my time visiting manufacturing floors, safety managers kept a strict log on who handled it, and insisted on gloves and goggles each time. On top of that, everyone had the safety sheets handy—the sort that explains what to do if there’s a splash or smell in the air. Strict regulations cover storage and disposal, and that careful chain of oversight cuts down injuries and environmental headaches.
Steps Toward Safer and Cleaner Chemistry
Plenty of experts search for greener alternatives to chemicals with challenging safety profiles. Some research teams look at plant-based amines for surfactant production, but AEEA’s balance of availability, cost, and performance keeps it on the roster for now. For those aiming to reduce risks, investing in upgraded ventilation, constant training, and clear hazard labeling are good first moves. Companies who listen to shop floor feedback often find practical ways to improve safety around these kinds of ingredients.
The Bottom Line
2-(2-Aminoethylamino)ethanol rarely gets a headline, but plays a role that touches everything from clean water to shiny cars to streak-free laundry. Those of us with an eye on chemical use know these small pieces keep factories and daily life running, as long as we keep a close watch on health, cost, and the next wave of innovation.
What are the safety precautions when handling 2-(2-Aminoethylamino)Ethanol?
Respect the Chemical, Not Just the Label
If you spend much time in a lab, you start treating each bottle for what it can do—good, bad, and everything between. 2-(2-Aminoethylamino)Ethanol doesn’t light up headlines, but overconfidence or carelessness around this compound quickly teaches a hard lesson. Skin, eyes, and lungs can all take a hit from casual handling. Years working with amines drive home this truth. One quick mistake, and you’ll remember the sting or burn for days.
Personal Protective Equipment is a Starting Line
Nitrile gloves become a second skin for anyone who values their own. Standard cotton offers nothing against corrosives. Safety goggles matter—splashes don’t send warnings. Even a small drop in the eye can put your sight at risk, turning an ordinary day sideways fast. Lab coats and long sleeves stop spills from finding bare arms, and it later saves worry about delayed burns. I’ve seen new technicians think these steps slow them down; scars show up much quicker than safety habits.
Ventilation Outshines Good Intentions
Handling volatile substances in a closed-in space punishes those who ignore airflow. 2-(2-Aminoethylamino)Ethanol gives off vapors that irritate throats and lungs, sometimes before you smell anything wrong. Fume hoods actually do their job—pushing air and chemicals away from your nose. I’ve worked afternoons smelling amine after someone skipped the hood. Headaches and sore sinuses don’t lie. Proper airflow solves the problem before it starts.
Don’t Play Catch-Up with Spills
Spills don’t follow schedules. You plan and they still show up. Fast clean-up depends on simple steps being ready before anything leaves a bottle. Absorbent pads and neutralizing agents belong within arm’s reach. My earliest spill taught me that hunting for supplies soaks precious seconds you never get back. Prompt clean-up stops both injuries and ugly messes.
Label What You Use and Store It Right
People ignore labels until confusion leaves them scrambling. Mixing up clear liquids produces more chaos than chemistry. Sharp, clear writing on every bottle stops mistakes. Dedicated cabinets keep hazardous chemicals far from everyday supplies. I’ve seen bottles hidden behind snacks or paper towels—it’s an accident waiting for a headline. Secure storage gives peace of mind long after the workday ends.
Take Training Seriously—It’s Not Just for Newcomers
Anyone who spends time around chemicals benefits from refreshers. I picked up things after years in the field that formal training missed. Chemical hazards evolve, so do safe handling strategies. Sharing near-misses with coworkers helps everyone adapt. Nobody is too experienced to stop learning, and the risks don’t shrink just because you know the material by heart.
Better Safety Builds Lasting Confidence
Workplaces grow safer when everyone expects the best from each other and holds each other accountable. This isn’t about managing fear; it’s about making safety feel automatic. From PPE choices to storage, each step done right today makes it easier for people tomorrow. The value of that lesson never fades, and neither does the respect it brings for what you’re handling.
Looking Forward: Keeping Hazards in Check
Although perfection doesn’t exist, steady habits, proper equipment, and real respect for chemical potential go further than any label ever can. 2-(2-Aminoethylamino)Ethanol remains a tool, not a trap, if you treat it with care every single time.
What is the chemical formula and molecular weight of 2-(2-Aminoethylamino)Ethanol?
Chemical Formula and Molecular Weight
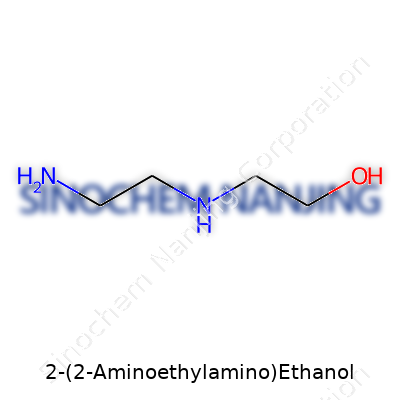
2-(2-Aminoethylamino)ethanol—often called AEEA in the lab—packs a punch when it comes to practical chemistry. Its chemical formula is C4H12N2O. Every molecule brings together four carbons, twelve hydrogens, two nitrogens, and a single oxygen atom. The molecular weight lands at 104.15 g/mol, based on the calculation of atomic weights for each element. It looks like a simple string of atoms, but its uses stretch far beyond basic stats.
Why Structure Matters in Real-World Chemistry
AEEA isn’t just another name on a list of obscure chemicals. I spent summers in college working with amine compounds for water treatment. The structure of this molecule—two amine groups and one ethanol group—delivers real advantages. It snatches up stray acids and traps carbon dioxide with impressive efficiency. This kind of hands-on reactivity makes it a choice pick in both gas sweetening operations and specialty surfactant production.
Pulling apart its structure tells a story. Those two amine groups can play both offense and defense in reactions. They catch acid gases, work in buffers, and even help in polymer modifications for custom resins. That ethanol chain tacked on the side brings a touch of solubility, helping AEEA blend smoothly in water. In my experience, these traits shave off headaches when dosing solutions or fine-tuning blends for system compatibility.
Applications and Practical Concerns
In gas treatment, refineries hook up systems that depend on amines like AEEA to scrub out harmful components. Efficiency in this game means lower energy bills and cleaner air. I’ve seen operators swap formulations to include more of these efficient “ethanolamines” and slash carbonation costs in the process. Laboratories turn to this molecule for its handy reactivity and low toxicity compared to harsher amines. In textile work, tweaks to AEEA’s formula lead to dyes that grab onto fibers and hold their color through rough washes.
Still, some challenges follow chemicals wherever they go. Managing exposure matters most for those handling the raw material. Direct contact can irritate skin and eyes, and nobody enjoys a strong amine odor in confined spaces. Wearing gloves, keeping the workspace well-ventilated, and labeling containers sharply makes life easier and safer. Small steps like these cut down on emergency visits and product contamination—we learned this the hard way after one summer intern splashed a sample during a demo.
Building Safer and Smarter Chemical Practices
Folks in education and industry take safety training seriously for a reason. Handling any amine compound involves investment in good ventilation, clear communication, and shared responsibility across the team. Data from OSHA show that simple controls and consistent labeling reduce accidents by over half, which matches what I’ve seen in plants with strong safety cultures. Sharing those basic lessons early and often changes outcomes.
Better stewardship starts at the small-batch scale. Track each transfer, give every container a clear label, and keep close tabs on inventory. This prevents mix-ups—especially with chemicals as similar as AEEA and its cousins. When supply chain disruptions hit, having a trained eye on compatibility and substitution helps avoid costly downtime.
Understanding the nuts and bolts of 2-(2-Aminoethylamino)ethanol means safer labs, smarter production, and fewer costly mistakes.How should 2-(2-Aminoethylamino)Ethanol be stored?
Why Safe Storage Matters
2-(2-Aminoethylamino)ethanol can push curious people to look beyond its tongue-twisting name. Most folks don’t think about chemical storage too often, but anyone who works in a laboratory or industrial space learns pretty fast that detail makes all the difference. Container choice, temperature, and labeling sometimes seem like minor things. They’re not minor for this one. I know from years spent in research labs that cutting corners here usually ends up biting you later. Unsafe storage could expose workers to irritation, chemical burns, or even fires.
Solid Steps Keep Chemical Drama at Bay
Over the years, I’ve learned it’s vital to start with the right container. This chemical reacts with acids and oxidizers, so metal drums or old jugs don’t make the cut. Stick with tightly sealed plastic or glass bottles that can handle corrosive materials. Clear labeling turns into your best friend after a week or two, because half-remembered mysteries on the shelf never end well.
Keep the storage area cool and well-ventilated. This stuff reacts fast if the room heats up too much, and that's a risk nobody wants. I once worked in a lab where a chemical cabinet shared space with a sunny window, and we wound up scrapping half our supplies after a spike in temperature. Direct sunlight doesn’t just mess with stability here — it speeds up decomposition, releasing fumes you definitely don’t want hanging in the air.
Moisture is another problem. Humidity and open bottles set the stage for unwanted reactions, so store the container in a dry spot. Floors can get wet in a hurry if someone trips or during cleaning, so seal all containers and place them above floor level. Pouring anything back into the original bottle pretty much guarantees contamination down the line. Fresh containers slow that risk.
The Real Risk of Mixing and Mishandling
One small mistake—like stashing this on the wrong shelf—can lead to cross-contamination with acids or oxidizers, and that puts property and people at risk. I’ve seen close calls where leftover acid on gloves turned a routine task into a medical emergency. Workers deserve supplies and signage that get checked regularly. Emergency eyewash stations and showers should be easy to reach. This kind of preparation keeps everyday accidents from turning ugly.
Regulation is Not Optional
OSHA and local safety rules aren’t just red tape. They reflect lessons learned from accidents others have faced. Ignoring rules around temperature control, signage, and personal protective equipment leaves real scars. Training sessions might seem like a drag, especially for experienced staff, but staying sharp on protocols is how teams steer clear of injuries and fines.
Making Storage Work for Everyone
A shared commitment to smart storage often starts with leadership. 2-(2-Aminoethylamino)ethanol storage works best in locked cabinets with restricted access. Check inventories often, use fresh containers with each new shipment, and document every transfer. Good lighting makes it easier to spot leaks or labeling problems. My own habit involves a checklist before and after using any stock, and it’s saved headaches more than once.
Storage can’t become an afterthought just because a chemical “usually behaves.” Respect the routine, and keep asking questions. Teams build healthier habits and nobody has to wonder where that sharp stinging smell is coming from. Reliable storage protects people, buildings, and the entire workflow, and that’s worth real effort every day.
Is 2-(2-Aminoethylamino)Ethanol hazardous or toxic?
Looking at Hazards in Chemical Handling
2-(2-Aminoethylamino)ethanol sounds technical, but its story isn’t unique in a world filled with complex chemicals. This compound plays a role in industry—often showing up as an ingredient in specialty surfactants or chemicals used for water treatment. It pops up where strong cleaning or reactive systems are needed. People working in labs or factories sometimes see it on the ingredient list or a shipping drum. Reading the safety sheet is part of the job, and there’s good reason for that.
Human Health Risks Before Safety Gear Goes On
When workers open a drum, they get a whiff—sharp, ammonia-like, enough to make most people step back. Coming into direct contact with it can cause skin irritation or burning, and it doesn’t spare the eyes. Over a long day, even a single careless moment—rubbing your eye after handling a valve, skipping gloves once—can lead to a trip to first aid. Breathing its vapors doesn’t do lungs any favors. Coughing, throat irritation, headaches show up in those who underestimate its power. Swallowing even a small amount sends someone to the emergency room with possible internal burns.
Science Backs Up Our Caution
Technical documents, along with the Globally Harmonized System (GHS), put 2-(2-aminoethylamino)ethanol in a category of serious irritants. The National Institute for Occupational Safety and Health (NIOSH) treats it with respect. Data shows skin absorption can lead to more than just a rash—it can move on to systemic effects, especially if used daily or spilled over large areas of skin. Workers report headaches, fatigue, and even confusion after heavy exposure. Lab animals developed liver and kidney problems after chronic exposure, so the concern isn’t made up or exaggerated.
Safety Culture and the Right to Know
Years ago, many people in industry learned safety protocols the hard way. Back then, warning labels weren’t always clear, and new workers found out why caution matters through burns and blisters. Now, safety data sheets are more available, and hazard pictograms warn people to suit up. Standard gear includes goggles, gloves made of nitrile or neoprene, rubber aprons, and sometimes full-face shields. This didn’t come about because people wanted to be dramatic—injuries and hospitalizations forced those changes. Every incident and exposure gets logged, and over time, companies learn what really works to prevent them.
Training and Accountability Make the Difference
Some places put all the right PPE in the storeroom, but training makes the real difference. Walking new workers through the safety process—showing how to store chemicals, how to clean up spills, why double gloving is more than just overkill—protects not just individuals but whole teams. Regular refresher courses, hands-on practice, and having visible emergency showers and eyewash stations help everyone feel less rattled if something does go wrong. The process builds trust and lowers accident rates.
Seeking Safer Alternatives and Smart Practice
Some manufacturers look for replacements with milder properties, especially as regulations get stricter and as buyers push for green chemistry. The lesson isn’t to avoid all chemicals, but to use knowledge and vigilance to keep people safe. Proper ventilation, environmental controls, and responsible waste disposal all add another layer of defense. Nobody enjoys reading safety sheets, and wearing goggles all day makes the job less comfortable, but the facts about 2-(2-aminoethylamino)ethanol remind us why health can’t wait for an accident before it becomes a priority.
| Names | |
| Preferred IUPAC name | 2-(2-aminoethylamino)ethan-1-ol |
| Other names |
2-(2-Aminoethylamino)ethanol N-(2-Hydroxyethyl)ethylenediamine 2-[(2-Aminoethyl)amino]ethanol N-(beta-Hydroxyethyl)ethylenediamine N-Ethanol-2,2′-diamine 2-Amino-N-(2-hydroxyethyl)ethanamine |
| Pronunciation | /tuː tuː əˈmiːnoʊˌɛθəlˌəˈmiːnoʊ ˈɛθənɒl/ |
| Identifiers | |
| CAS Number | 111-41-1 |
| Beilstein Reference | 1701121 |
| ChEBI | CHEBI:40561 |
| ChEMBL | CHEMBL12782 |
| ChemSpider | 12708 |
| DrugBank | DB04292 |
| ECHA InfoCard | 100.026.660 |
| EC Number | 205-793-9 |
| Gmelin Reference | 80140 |
| KEGG | C01602 |
| MeSH | D019335 |
| PubChem CID | 16398 |
| RTECS number | KL9275000 |
| UNII | 2M83AO56D5 |
| UN number | UN3336 |
| CompTox Dashboard (EPA) | DTXSID3054673 |
| Properties | |
| Chemical formula | C4H12N2O |
| Molar mass | 106.16 g/mol |
| Appearance | Colorless to light yellow transparent liquid |
| Odor | Ammonia-like |
| Density | 1.027 g/mL at 25 °C(lit.) |
| Solubility in water | Soluble |
| log P | -2.02 |
| Vapor pressure | 0.01 mmHg (25°C) |
| Acidity (pKa) | 9.62 |
| Basicity (pKb) | 5.02 |
| Magnetic susceptibility (χ) | -62.7e-6 cm³/mol |
| Refractive index (nD) | 1.482 |
| Viscosity | 17 cP (25°C) |
| Dipole moment | 3.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -223.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3785 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | C04AX21 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, causes skin irritation |
| GHS labelling | GHS07,GHS05 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335 |
| Precautionary statements | P261, P264, P280, P305+P351+P338, P337+P313, P337, P314 |
| NFPA 704 (fire diamond) | 1-3-0 |
| Flash point | 100°C |
| Autoignition temperature | 410°C |
| Lethal dose or concentration | LD50 Oral Rat 3000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 605 mg/kg |
| NIOSH | PC2625000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | REL (Recommended Exposure Limit): 3 ppm (8-hour TWA) |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
Ethanolamine Diethanolamine Triethanolamine 2-(2-Aminoethylamino)acetic acid N-(2-Hydroxyethyl)ethylenediamine |

