Understanding 2,2,3-Trimethylpentane: A Deep Dive into Its Past, Present, and Future
Historical Development
During the early twentieth century, gasoline engines demanded more and more from their fuels. Knocking — the telltale rattle that every engine builder dreads — pushed chemists and engineers to tinker with hydrocarbon structures. Enter 2,2,3-Trimethylpentane, a molecule emerging from the surge of interest in branched alkanes. As the petroleum industry chased higher octane ratings, it wasn’t enough to just refine more—there was a push to create fuel components that could meet the appetite of increasingly sophisticated engines. Research led to new branching patterns, and this compound popped up as a key player, especially after World War II. The search for performance standards and cleaner burning fuels drew attention to how molecules like 2,2,3-Trimethylpentane could be produced at scale—we’re talking catalytic reforming, isomerization improvements, and much more. Industry and academia both got behind this, creating a legacy of collaboration and pushing past "business-as-usual" refinement.
Product Overview
If you pick apart a can of gasoline, you won’t find one recipe scrawled on a label—it’s an orchestra of hydrocarbons, each helping engines resist pre-ignition and squeeze out power efficiently. 2,2,3-Trimethylpentane sits in this cast as an octane booster, lending its resilience against knocking. The compound not only helps interior combustion engines run smoother—its stable structure also appealed to those shaping racing fuels and specialty blends. In labs, researchers measure octane with test blends, and this hydrocarbon helps benchmark fuel behaviors. The world doesn’t often see it on the shelf by name, but it plays a behind-the-scenes role where performance matters.
Physical & Chemical Properties
Let’s get down to brass tacks: 2,2,3-Trimethylpentane is a clear liquid at room temperature. Don’t count on it dissolving in water—like its fellow alkanes, it’s hydrophobic through and through. As temperatures climb, its volatility stands out, which forces attention to handling protocols in warm climates or industrial settings. The molecule’s non-polar nature makes it blend smoothly with other similar hydrocarbons, forming stable mixtures without fuss. Its boiling point hovers comfortably in the range typical for medium-chain branched alkanes, adding a measure of predictability for refineries and labs. No strong odors, no wild coloration—just a reliable component for those who know where to look.
Technical Specifications & Labeling
In the drum or the barrel, industry keeps track of purity, density, and hydride content for this compound. Standards exist, remembering that what shows up in fuel blends must not trip up emissions controls or ignite too soon. Some chemists track infrared spectra or run chromatograms to check batches. Those juggling product safety never lose sight of flammability warnings, spill protocols, and clear labeling mandates. Handling guidelines put safety goggles and gloves on the shortlist. Storage calls for tight seals, away from heat or open flame, a familiar refrain for those working with volatile organics.
Preparation Method
Start with petroleum. Refineries process crude through catalytic reformers, breaking and reshaping molecules under heat and pressure, helped along by specific catalysts. These steps promote branching at strategic points, producing 2,2,3-Trimethylpentane among other isomers. Sometimes, isomerization routes or alkylation with specific catalysts gain the upper hand for making particular branched octanes, adjusting reactor conditions to favor the output. The complexity of separating this compound relies on distillation, using boiling points and fractionation columns, not brute force. The industry keeps innovating, always looking for routes that save energy, cut emissions, and boost yield ratios.
Chemical Reactions & Modifications
2,2,3-Trimethylpentane doesn’t just sit quietly on a shelf. In atmospheres rich in oxygen and under extreme conditions, it breaks down into smaller, sometimes highly reactive fragments. Combustion in engine cylinders turns it into water and carbon dioxide, releasing the energy drivers crave. Under the microscope, you’ll see it resist many routes of chemical attack due to its saturated nature, yet lab chemists have coaxed it into various oxidation reactions and halogenations under the right push. As a hydrocarbon, it dodges most acids and bases, showing its stubborn side. Its structure isn’t up for easy rearrangement without significant intervention—something that sets branched alkanes apart from their straight-chained cousins.
Synonyms & Product Names
Step into a refinery or chemistry textbook, and the same chemical shows up under a few monikers. The IUPAC name, 2,2,3-Trimethylpentane, spells out its structure—three methyl groups decorating a pentane backbone. Literature sometimes refers to it simply as a trimethylpentane, or by shorthand as TMP. For engine testers, it lands under “isoparaffins” or “high-octane isomer.” These names matter, especially when cross-referencing data sheets or regulatory filings, though true insiders rarely confuse this molecule’s aliases with others in the family.
Safety & Operational Standards
Nobody pours 2,2,3-Trimethylpentane casually, least of all in a crowded facility. It qualifies as a flammable liquid, demanding respect. Spillage means vapors can spread quickly, and ignition sources turn a minor slip into a major incident. Facilities handling it stick to grounding, vapor management, and secure container storage. Staff know to shuttle it in well-ventilated areas, avoid direct skin contact, and never use compromised containers. Regulatory bodies keep a watchful eye on reporting, emissions, and any potential environmental releases, especially near waterways or ground supplies.
Application Area
Automotive fuels make up the number one arena for 2,2,3-Trimethylpentane. High-compression performance engines depend on knock resistance, a property where branched alkanes excel. Racing fuels and specialty aviation blends sometimes call for a touch of this hydrocarbon to sharpen octane profiles. From my own visits to engine testing labs, the molecule shows up again and again in calibration blends, useful for evaluating detector sensitivities or baseline octane behaviors. Blending engineers and formulation chemists see it as an essential puzzle piece to hit octane targets without propping up toxic side products.
Research & Development
Labs across the globe dig into ways to synthesize branched alkanes more efficiently. Clean energy drives this research, not just profits. Recovery methods aim to reduce greenhouse output and waste. Catalytic design, reactor optimization, and smart separation technologies attract top minds interested in both chemistry and environmental stewardship. Computational chemistry now leans heavily on 2,2,3-Trimethylpentane in model systems, leveraging its known behaviors to validate simulation packages. Data-sharing through open-access projects makes each incremental advance ripple through academic and industrial spheres faster than ever.
Toxicity Research
Toxicology keeps the public and workforce safe by digging for risks, and 2,2,3-Trimethylpentane doesn’t get a free pass. Inhalation stands out as a primary exposure path—workers who handle it without proper ventilation risk headaches, dizziness, or worse if things go sideways. Regulatory studies place it among the lower-toxicity alkanes, shy of persistent or bioaccumulative behavior, but not without concern for chronic exposure or accidental ingestion. Aquatic risk studies flag it for low solubility but note that spills can harm surface-dwelling organisms. Every risk assessment underscores the importance of proper storage, quick spill response, and rigorous air quality monitoring—no shortcuts, especially with volatile organics.
Future Prospects
The world chases cleaner transportation, and attention shifts from just making burning smoother to rethinking entire fuel cycles. 2,2,3-Trimethylpentane remains important as a reference chemical, benchmark fuel, or blend component wherever internal combustion engines remain in play. Whether that’s for classic cars, backup generators, or transitional aviation solutions, performance testing relies on molecules with known, stable properties. Synthetic biology and green chemistry aim to create alkanes in bioreactors or from recycled wastes, promising new ways to source high-octane hydrocarbons. If hydrogen and electric drives grab the market, demand for this molecule could shift toward more niche and experimental territories. For now, it earns its spot on industry and research shelves, a reminder that even simple hydrocarbons can spark decades of innovation and challenge.
What is 2,2,3-Trimethylpentane used for?
The Story of An Everyday Gasoline Ingredient
Most people would never recognize the name 2,2,3-Trimethylpentane. Still, anyone filling up their car's tank benefits from this compound’s practical value. Known to chemists as one of the isomers of octane, this clear liquid matters a lot for engines running on gasoline. Years ago, car performance hinged less on computers and more on the very molecules poured into the tank. In that world, 2,2,3-Trimethylpentane grew famous for its resistance to knocking, that annoying rattle in old engines that made drivers wince at green lights.
A Key Ingredient in High-Octane Gasoline
Refineries don’t just squeeze crude oil into gasoline and pump it into cars. Each batch needs careful blending, and choosing the right set of chemicals makes all the difference between a noisy, polluting engine and a quiet, cleaner-running one. 2,2,3-Trimethylpentane’s claim to fame lies in its high-octane rating. “Octane” here doesn’t mean the energy in fuel; it marks the ability of gasoline to withstand premature ignition. Fuels with more of this ingredient let engines run faster and hotter, boosting efficiency while trimming down wear.
As car engines have grown more advanced, the need for high-octane blends increased. Factories produce 2,2,3-Trimethylpentane in large quantities through chemical reactions starting with petroleum feedstocks. Then, refiners mix it into gasoline to reach the blend that works best with current engine technology. My experience at a busy service station showed me drivers usually care about price, yet those little labels about octane levels point right back to chemicals like this one making a real-world difference.
Cleaner Air and Better Performance
The world cares more each year about air quality. Governments keep tightening rules on what comes out of exhaust pipes, and car makers work hard to squeeze out every possible mile per gallon. Here, 2,2,3-Trimethylpentane stands out again. Adding this molecule helps gasoline burn more completely and cleanly, giving fewer emissions of pollutants like carbon monoxide and unburned hydrocarbons. Better gasoline doesn’t just help engines last longer—it also keeps the air clearer for everyone.
Moving Forward: Green Alternatives and Blending Challenges
Petroleum refineries depend on tried-and-true chemistry, but cleaner alternatives are pushing everyone to reconsider how gasoline gets made. Adding ethanol has become common, yet it brings its own set of headaches—water absorption, fuel stability, lower energy content. Some researchers are hunting for ways to make molecules like 2,2,3-Trimethylpentane from renewable feedstocks instead of fossil fuels. This sort of innovation takes time, investment, and a willingness on the part of industry to shift away from what’s familiar.
As the world moves toward electric mobility, traditional gasoline ingredients may seem less relevant to city dwellers who see charging stations everywhere. But many regions still run on internal combustion—trucks rolling down highways, tractors in fields. If fuels keep improving, air gets cleaner even before every car runs silently. From my perspective, the chemistry behind those incremental improvements shapes daily life in ways even the most dedicated motorists rarely stop to notice.
Making Every Drop Count
2,2,3-Trimethylpentane reminds us that small changes at the molecular level can mean smoother engines, cleaner streets, and even saved money. Each time scientists, engineers, or drivers think about what flows into a fuel tank, they’re dealing with a legacy of chemistry that quietly shapes the modern world. Adapting this knowledge to fit new needs will keep giving society reliable, practical energy—at least as long as engines need fuel to run.
What is the chemical formula of 2,2,3-Trimethylpentane?
Seeing the Big Picture in Chemical Nomenclature
It’s easy to look at a name like 2,2,3-Trimethylpentane and feel lost. During my chemistry studies, chemical names with numbers stuffed in the middle seemed complicated. It gets easier once you decode how those numbers fit the molecules together. This isn’t just a mental exercise—it makes an impact in industries, fuels, and risk management.
Breaking Down the Structure
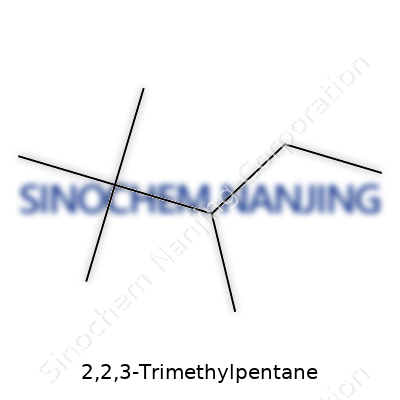
2,2,3-Trimethylpentane uses the backbone of pentane—an alkane with five carbon atoms. The name flags extra pieces: methyl groups sitting on the second and third carbon. Each “methyl” means another CH3 group. So, at carbon 2, two methyls attach. Carbon 3 takes one more methyl. The key isn’t memorizing lists; it’s visualizing how each carbon fits those pieces together, where the branches sit, and how the skeleton builds up.
The Actual Formula: C8H18
Pulling it all together, 2,2,3-Trimethylpentane adds up to eight carbons and eighteen hydrogens—a chemical formula of C8H18. Getting from name to formula isn’t as abstract as it seems. Sketching structures on scratch paper helped me avoid mistakes. I’d count every carbon and hydrogen, double-check every branch. Mixing up a methyl or losing one hydrogen can wreck entire lab batches, especially during synthesis.
Why Does 2,2,3-Trimethylpentane Matter?
This molecule isn’t just a textbook problem. The real world needed better gasoline right after the widespread adoption of the automobile. Engine knocking slammed early drivers. Researchers tried a mix of compounds, but some molecules stood out for resisting knock. Iso-octane is the common name for 2,2,3-Trimethylpentane. Its structure smothers premature ignition, banishing knock, and sets the 100 point on the octane rating scale—an essential factor for performance and safety.
The history of gasoline grades uses iso-octane as a benchmark. Gasoline grades reflect the concentration of high-octane, branched alkanes like 2,2,3-Trimethylpentane. Using this molecule in fuel blends allows engines to use higher compression ratios, generating more power and better fuel economy.
Looking Through the Lens of Environmental and Health Impacts
Not everything about branched hydrocarbons is positive. Blending for cleaner combustion cuts down dangerous emissions, but the same efficiency can mean more complicated refinery processes and tougher environmental controls. Leaks and spills put hydrocarbons into soil and water. Health risks—ranging from headaches to respiratory irritation—appear for those exposed to high vapor concentrations. A push for tighter controls—improved containment and real-time detection—helps avoid environmental contamination and public health risks.
Making Real Progress Beyond Paper Chemistry
Solving these challenges means better ways of producing and handling hydrocarbons. Upgrading refinery tech and setting stricter environmental standards protect communities. In my own work, practical safety training and using up-to-date protective gear made the difference for me and my coworkers. Small habits—ventilating labs, double-sealing containers, taking vapor exposures seriously—save more trouble than any rule on paper.
Science does more than fill shelves with formulas. Each chemical has a story that shapes industries, environmental policy, and public health. Diving deep into names like 2,2,3-Trimethylpentane shows how much there is beneath the surface and the value of staying thoughtful and careful—both on the page and in the field.
Is 2,2,3-Trimethylpentane hazardous or toxic?
Getting to Know 2,2,3-Trimethylpentane
2,2,3-Trimethylpentane mostly shows up in the context of fuels, especially as a part of gasoline blends. Some chemistry folks call it iso-octane. Refineries rely on it to boost octane ratings and prevent a car engine from knocking. The compound itself boils at a lower temperature than water and smells like gasoline. It’s clear, colorless, and doesn’t dissolve well in water. You touch it, you instantly know you’re handling something flammable. Anyone who has worked in a laboratory or a refinery recognizes the importance of handling such liquids with care.
Health Hazards: Not for Casual Handling
Reading a material safety data sheet on 2,2,3-trimethylpentane, the health warnings jump out. Breathing in the vapors or getting it on your skin starts trouble — headaches, dizziness, and irritation are common complaints. Skin contact often leads to dryness and redness. People who have worked around volatile fuels know how fast solvent fumes can sneak up on you. Toxicity data does not rank it as a super-poison like some solvents. Still, given enough exposure, it harms. Chronic exposure may affect the nervous system, causing symptoms like tiredness or trouble focusing. No parental figure needs to explain why splashing this chemical near your mouth or eyes is a bad idea; the burning sensation says enough.
Environmental Risks: Not Just a Lab Problem
Leaks can cause headaches beyond human health. Dropped into soil or water, 2,2,3-trimethylpentane persists longer than you’d hope. Microbes have a hard time breaking it down, which means it hangs around — and once in the environment, it can slip into the food chain. Fish exposed to hydrocarbon spills face death or mutations. Growing up near an oil refinery, odd odors hung in the air after rainstorms, hinting that not all chemicals villainize only humans. Gasoline runoff affected neighborhood gardens, leaving old-timers comparing tomato crops before and after refinery expansions. The consequences rarely stop at the fence line.
Facts and Figures: Why Smart Handling Matters
The U.S. Agency for Toxic Substances and Disease Registry keeps public records on compounds like this one. Short-term exposure above 300 parts per million often leads to headaches or nausea. Getting it in eyes or inhaling large amounts can send someone to the doctor. The substance is also flammable — a single spark near an open container can ignite a fire or explosion, filling a space with black smoke and poisonous fumes. You only need to watch a single safety demonstration or see a gasoline fire to respect that danger. OSHA sets workplace exposure limits, and for a good reason. Regulators do not want people treating fuel chemicals like harmless household cleaning products.
Taking Practical Steps
Reducing risk calls for more than rules on a poster. The real answer involves proper ventilation, gloves, and eye protection. Keeping storage areas away from heat matters. Prompt cleanup of spills keeps accidents from turning into news headlines. Everyone, down to the new kid at the refinery, benefits from regular safety drills. Awareness campaigns remind users about hidden risks, while research looks for safer additives for tomorrow’s fuels. Talking with old mechanics or refinery workers always turns up stories of folks underestimating what a can of volatile liquid can do. A little knowledge and respect for these chemicals go a long way toward keeping everyone around them safe and healthy.
What are the physical properties of 2,2,3-Trimethylpentane?
Fuel Science on the Lab Bench
2,2,3-Trimethylpentane shows up in every conversation about gasoline quality. Its clear, colorless liquid form tells us right away it fits right in with other hydrocarbons that we blend into fuels. Take a sample in the lab and you’ll note it stays liquid far below freezing, with a melting point around -107°C. In gas tanks and pipelines through a Minnesota winter or a freezer-grade test facility, this behavior makes things reliable and predictable.
Boiling Point and Handling
The boiling point stands close to 122°C. It doesn’t flash off at room temperature like lighter alkanes, which helps with safe storage and transport. In my early years working with fuel batches, nobody wanted a compound that vanished into thin air if a barrel stood open too long. This respectable boiling point gives us a manageable evaporation rate.
Density Matters at the Pump
At 20°C, the density hovers near 0.695 grams per cubic centimeter. This figure means real things for blending gasoline — the energy packed per gallon and the volume versus mass calculations that go into every refinery ledger. Lighter fuels shift shipping weights, storage demands, and burn rates. Every tenth of a point on density translates to dollars and efficiency for producers and drivers.
Viscosity and Flow
2,2,3-Trimethylpentane pours like water but with just enough thickness to handle well in pumps and pipelines. Its viscosity makes it simple to blend with other hydrocarbons without clogging or separation. If you’ve ever dealt with stubborn or gunky fuel mixtures, you get why this property matters.
Ignition and Safety
Flash point data puts it just below -12°C. There’s always fire risk in fuel work, and such a low flash point requires respect, but given proper storage, normal safety procedure manages it. Its vapor pressure at room temperature stands at about 7 kPa. That says something to anyone storing or hauling fuel across rough distances or temperature swings. Good fuel handling isn’t just about clean-burning engines — it keeps tanks from swelling or venting unexpectedly, which protects worksites and communities.
Octane and Application
The structure of 2,2,3-Trimethylpentane gives it a high octane rating. We actually use it as a reference point in octane testing. It can resist knocking in engines with high compression ratios. Real-world drivers care about that because it means smoother acceleration, better performance, and less engine damage. Blending this compound into gasoline helps automakers build better engines that use less fuel and pump out fewer emissions per mile.
Moving Forward with Safety and Sustainability
Improved valves, vapor recovery systems, and stricter tank insulation all come into play when working with chemicals of this volatility. Practical safety is part checkbox, part culture. I’ve seen companies train workers to not only follow the rules but understand why low flash points matter and how even a cup of spilled hydrocarbon can change local air quality. Longer term, the way we use trimethylpentanes feeds into questions about greener chemistry. Maybe new processes can lower emissions during synthesis or encourage recycling from petrochemical streams. Strong regulation, smart engineering, and honest reporting keep these powerful fuels safe for everyone who depends on them — which, at the end of the day, means all of us.
How should 2,2,3-Trimethylpentane be stored and handled?
Get to Know What You’re Handling
2,2,3-Trimethylpentane doesn’t turn heads in everyday conversation, but it earns attention in labs and workplaces. Many know it as isooctane, a big player in making high-octane fuel for cars. I’ve seen people underestimate chemicals like this. They look clear, sometimes smell sweet, and get shrugged off as “just another liquid.” That’s not a good plan. Without a real respect for its risks, things can go sideways fast.
Why the Way You Store Matters
Letting 2,2,3-Trimethylpentane sit in any old spot isn’t an option. This chemical snaps up vapor pressure, meaning it turns to vapor easily at room temperature. Those vapors catch fire even from small sparks. Every storage mishap story I’ve heard—whether it was an unlabeled bottle or a sunny shelf—ends with regret and sometimes the fire department. Solid sources like the National Fire Protection Association warn against keeping this stuff near any heat, flames, or exposed wiring.
You want metal containers with tight lids. Glass works for lab amounts but, in bulk, go with drums that stay grounded. I’ve worked in places where someone stored it in a plastic bottle picked from the break room. That bottle softened and collapsed by week’s end, leaking this volatile liquid everywhere. Stick with containers rated for flammable liquids and keep them in a well-ventilated, cool spot.
Label Everything
Clear labels cut confusion. I’ve walked into storerooms, seen rows of unlabeled containers, and felt that knot in my stomach. All it takes is mixing chemicals by accident or not knowing there’s a fire risk right under your nose. Every container gets the name, the date, and a “flammable” warning in big letters. If someone plans to move it, make sure they know exactly what’s sloshing around in that jug.
Take Handling Seriously
Pouring 2,2,3-Trimethylpentane without gloves or goggles is asking for trouble. Even the pros slip up. I watched a colleague, someone with a chemistry Ph.D., fail to tighten a lid. The next person walked in, caught the fumes, and had to sit down because of the headache. Vapors don’t just risk fire—they can hit your health. Splashing it onto skin or breathing fumes isn’t just unpleasant, it can cause real problems like dizziness or irritation.
Good ventilation keeps the room safe. Fume hoods, vents, open windows—anything that clears the air. Keeping the room clean and tidy also makes a difference. I like to keep absorbent pads or spill kits within arm’s reach because leaks don’t wait for your calendar.
Trained Eyes, Calm Hands
Training every person who handles 2,2,3-Trimethylpentane should never feel optional. People learn best from stories, not just manuals. Sharing true mishaps and practicing emergency drills beats reading the same dry warnings year after year. Knowing how to use fire extinguishers and spill kits makes the difference between a near miss and an incident report.
Better Habits, Safer Workplaces
The lessons from 2,2,3-Trimethylpentane stretch far beyond one chemical. Every person who walks into a storage room or lab comes away with responsibility. You can’t guess or coast through safety. Clear labels, the right containers, trained hands, and constant attention keep risk under control. Stories of well-handled chemicals rarely make headlines, but they keep people safe, gear working, and workplaces running smooth. Responsible storage isn’t busywork; it’s the difference between a routine shift and a disaster.

| Names | |
| Preferred IUPAC name | 2,2,3-Trimethylpentane |
| Other names |
Isooctane Iso-Octane |
| Pronunciation | /ˌtraɪˌmɛθəlˈpɛnˌteɪn/ |
| Identifiers | |
| CAS Number | 564-02-3 |
| Beilstein Reference | 1721217 |
| ChEBI | CHEBI:8836 |
| ChEMBL | CHEMBL43070 |
| ChemSpider | 20238 |
| DrugBank | DB01975 |
| ECHA InfoCard | ECHA InfoCard: 100.006.258 |
| EC Number | 203-604-4 |
| Gmelin Reference | 78677 |
| KEGG | C06738 |
| MeSH | D015229 |
| PubChem CID | 11231 |
| RTECS number | RH6900000 |
| UNII | RU4S6TU4J6 |
| UN number | UN1262 |
| CompTox Dashboard (EPA) | DTXSID0033913 |
| Properties | |
| Chemical formula | C8H18 |
| Molar mass | 114.23 g/mol |
| Appearance | Colorless liquid |
| Odor | sweet odor |
| Density | 0.690 g/cm³ |
| Solubility in water | insoluble |
| log P | 2.93 |
| Vapor pressure | 3.3 mmHg (20°C) |
| Acidity (pKa) | 16.0 |
| Magnetic susceptibility (χ) | -8.63 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.391 |
| Viscosity | 0.696 mPa·s (at 20 °C) |
| Dipole moment | 0.13 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -248.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | –5195.0 kJ/mol |
| Pharmacology | |
| ATC code | C04Ax19 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H304, H315, H336, H411 |
| Precautionary statements | P210, P243, P261, P271, P273, P301+P310, P303+P361+P353, P304+P340, P331, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-4-0 |
| Flash point | 40 °C (104 °F; 313 K) |
| Autoignition temperature | 416 °C |
| Explosive limits | 1.0 - 6.0% (V) |
| Lethal dose or concentration | LD50 oral rat 12,003 mg/kg |
| LD50 (median dose) | LD50 (median dose) = 5000 mg/kg (rat, oral) |
| NIOSH | RN91075 |
| PEL (Permissible) | 300 ppm (US OSHA) |
| REL (Recommended) | 350 mg/m3 |
| IDLH (Immediate danger) | 500 ppm |
| Related compounds | |
| Related compounds |
Isobutane Neopentane Isooctane 2,2,4-Trimethylpentane 3,3-Dimethylpentane |

