Understanding 1-Nitropropane: Legacy, Challenges, and Future Horizons
Historical Perspective
Chemicals like 1-nitropropane didn’t just pop up overnight. They carry layered stories, looping back to a time when organic chemistry started shifting from curiosity-driven work to labs full of hardwood benches, glassware, and real industrial ambition. As a writer and observer of science, I often see how lesser-known substances ride the tides of chemical history. 1-nitropropane found its way onto the scene as researchers hunted for better solvents and tried to squeeze more out of nitro chemistry. Exploring the legacy of nitroalkanes helps us understand not only how science moves forward but how past generations made tough choices, usually balancing risk and utility with what they knew back then.
What Sets 1-Nitropropane Apart
1-nitropropane isn’t just another entry in an endless catalog of specialty chemicals; it’s an example of how small changes in molecular structure can change a substance’s fate. It takes a propyl backbone and slaps on a nitro group, instantly shifting behavior and giving rise to new possibilities. Most folks outside of chemical circles don’t realize that nitroalkanes like this are not household names, but inside research and industry, they make things tick—especially as solvents, intermediates for other compounds, and tools in tricky organic reactions. To those who work with it, 1-nitropropane is a helper, sometimes a hurdle, and always something to handle with a mix of respect and real-world caution.
Physical and Chemical Personality
Open a container, and you’ll notice 1-nitropropane arrives as a pale liquid, sometimes colorless, with a sharp smell that lets you know it means business. Its relatively low boiling point means it moves between gas and liquid at temperatures where some chemists sweat, and it doesn’t vanish so easily; this persistence is handy in procedures calling for steady evaporation rates and dependable solvent behavior. On a chemical level, its structure offers a single nitro group on the end, which gives it a distinctive thumbprint compared to relatives like 2-nitropropane. Such subtle differences don’t just sit in textbooks—they shape reactivity, toxicity, and where the compound fits best.
Making Sense of Technical Specs and Labels
Anyone handling industrial chemicals learns fast that you have to trust—and double-check—technical specifications. Purity typically comes rated in the high nineties, with water content and other nitroalkane isomers noted because contamination changes everything. Regulatory labeling demands clarity. Flammability, toxicity, and environmental impacts show up up front. Through years following such details in labs and factories, I know missing one detail on a label can upend a project or blindside a safety audit. Transparency and clarity with specifications anchor not only compliance but daily trust between producers and end users.
The Art and Science of Preparation
Behind every bottle of 1-nitropropane rests expertise you don’t see on the shelf. Preparation usually draws from classic organic reactions: the vapor-phase nitration of propane, where mixing nitric acid with the hydrocarbon at elevated temperatures in presence of a catalyst yields several nitroalkanes. Tweaks in temperature, pressure, and proportions shift yields toward 1-nitropropane or relatives like 2-nitropropane or nitromethane. The real-life process isn’t as neat as academic papers suggest. Engineers wrestle with byproducts, reactor fouling, and the headaches of separating chemical cousins. For every desired kilo of 1-nitropropane, there’s a lesson in patience and process discipline—skills any hands-on chemist can vouch for.
Chemical Reactions and Modifications
Anyone who has run a reaction with 1-nitropropane soon finds that nitro groups act like chemical magnets. Under basic conditions, it undergoes condensation, forming carbon-carbon bonds into new chains—think Henry reactions, which broaden synthetic routes to valuable intermediates like nitroalkenes. Reductions turn the nitro group into amines, opening doors to medicinal chemists and polymer formulators. Heating or hitting it with acids can send it off into side reactions, chewing up yields or introducing complicated mixtures. After a decade seeing lab trials and reading failure reports, I’ve found success with 1-nitropropane hinges on understanding these tricks and traps up front.
The Many Faces: Synonyms and Names
Someone scanning databases or talking with old-school chemists will hear 1-nitropropane called by a few different names. N-propylnitro, nitro-n-propane—each tags the same molecular entity, but confusion rises fast if suppliers or users don’t align their terminology. I’ve seen cases in research and procurement where a misread synonym led to the wrong compound arriving, wasting time and causing paperwork headaches. Consistent naming under IUPAC makes sense, yet the world of commerce and regulation stubbornly sticks with variant names. Getting this part right underpins traceability and safety, and it often requires slow, careful cross-checking.
Keeping People and Places Safe
I’d wager few things keep chemical professionals up at night more than safety, especially with volatile, toxic, or environmentally tricky chemicals, and 1-nitropropane ticks several boxes. Flammable vapors, toxic effects, and potential groundwater impacts all demand proper training, good ventilation, and smart storage solutions. Wanting to cut corners never ends well. Experienced hands know picking the right gloves, goggles, and spill measures cannot be a half-step. Industry has pushed harder lately for best-practices, with updated Material Safety Data Sheets, better labeling, and real-life scenario training. The cost of skepticism—thinking nothing will go wrong—shows up not only in OSHA logs but in lost years of trust.
Application Areas: From Paints to Pharmaceuticals
For years, 1-nitropropane found its niche as a solvent, making tough-to-dissolve resins manageable in certain coatings and lacquers. Folks in paints and sealants know its reputation: good solvency, decent evaporation rate, and compatibility with other nitroalkanes in bespoke mixtures. Some custom syntheses in pharma and agrochemicals dip into this toolbox, pulling from its reactivity. I’ve talked with formulators who swear by its unique edge, while others steer clear for reasons of safety, regulation, or greener alternatives. Each application comes with trade-offs—solvency weighed against health or environmental questions—and innovation often means thinking two steps ahead, both in chemistry and public trust.
Research, Innovation, and Evolving Roles
Scientists and engineers keep pushing the envelope, even with familiar chemicals. New research keeps exploring efficient nitration routes, separation methods, and catalytic systems that might reduce waste streams when making nitroalkanes. In academia, 1-nitropropane’s role in exploratory synthesis hasn’t faded; it still pops up in medicinal and materials research, mainly as a versatile carbon and nitrogen source. Industrial labs pursue process improvements hoping for productivity gains and smaller footprints. My experience echoes plenty of others: progress here requires teamwork and open sharing, as proprietary silos and inertia slow industry more than technical hurdles do.
Toxicity and Health Realities
Reality checks in chemistry hit hardest in toxicity research. Early on, nitroalkanes like 1-nitropropane were handled with few restrictions; now, the body of research points to acute and chronic toxicity. Inhalation, skin exposure, and accidental ingestion can bring on headaches, depression of the central nervous system, and longer-term organ effects. Chronic exposure links to potential liver and kidney damage, highlighting the need for consistent, modern risk assessments—especially as lab protocols and workplace exposures continue evolving. Controls should never become box-ticking routines. Real deployment means clean air systems, robust training, and policies that reflect the most recent findings, not faded standards from decades past.
Looking Down the Road: Future Prospects
What comes next for 1-nitropropane depends on how global industry, science, and regulation adapt. Stricter environmental rules push for lower emissions, safer facilities, and alternatives with lower toxicity. For fields wedded to its properties, such changes echo louder: finding substitutes is tough, and revamping processes costs both time and money. Some forward-looking chemists are targeting bio-based production, greener syntheses, and molecules that mimic the benefits without the baggage. I’ve watched trends in specialized chemicals move quickly from acceptance to scrutiny, often over just a few years. If 1-nitropropane holds a place going forward, success will rest on informed use, continued innovation, and an honest reckoning with both its strengths and its risks.
What is 1-Nitropropane used for?
The Role of 1-Nitropropane in Industry
Factories across the globe rely on specialized chemicals to get tough jobs done, and 1-nitropropane seems to keep showing up on this list. As someone who’s spent years watching manufacturing change shape, I’ve seen that no plant wants to waste money or time. Solvents have to work hard and work fast. Reality is, 1-nitropropane makes the cut because it dissolves things that other solvents just can’t touch. Consider paint and varnish. Stripping down an old wooden staircase or prepping steel beams before a new coat—clean-up has to be tough, but not brutal on the material underneath. This chemical steps up, handling stubborn stains or residue that water or weaker mixes leave behind. It is common in heavy-duty paint removers and parts cleaning formulations.
Driven By Demand in Inks and Coatings
Most of us don’t think much about what goes into printer ink. I’ve watched commercial printing, and the art isn’t just about color—it’s about consistency and drying time. 1-nitropropane ends up blended into the manufacturing of inks and coatings because its properties help inks spread evenly and dry fast without ruining the paper or clogging expensive machines. Commercial packaging needs to last months on shelves—labels must resist smudging and flaking. This single chemical quietly holds a lot of responsibility behind the scenes.
The Impact on Chemical Synthesis
Every supply chain relies on building blocks, the starting points for bigger, more complex chemicals. Chemists pick 1-nitropropane for knocking together other molecules that end up in crop sprays, pharmaceuticals, or plastic additives. It works well for creating nitroalkanes—necessary intermediates for larger scale production. For example, if you walk through a fertilizer production plant, you realize that each batch needs precise control over what goes in, and 1-nitropropane supports consistency process after process.
Health, Safety, and Environmental Challenges
Handling chemicals like this calls for more than a pair of gloves and a well-ventilated room. Inhaling the vapors over a shift, or getting it on your skin, carries risks long-term. There’s no mystery here—workplace safety gear and clear protocols matter. I remember chatting with a factory foreman about training new hires. It’s not enough to post rules on a break room corkboard; regular drills and real conversations about hazards mean people are ready if something spills.
The environment can’t shrug off chemicals either. Waste streams from paint stripping or cleaning processes can find their way into the ground and water if factories cut corners. Wastewater treatment and chemical recovery systems have to become part of the conversation early. Cleaner alternatives might exist, but replacing 1-nitropropane without sacrificing performance takes research and investment.
Room for Improvement and Practical Steps
Future routines in factories and print shops may look different. Exploring less harmful solvents or safer blends could ease the pressure on workers and the planet. Some companies already use closed-loop systems to recycle solvents, shrinking both costs and waste. Regulations push industries to rethink their choices, not just to follow rules, but to keep workers healthy and protect communities nearby. It’s worth companies spending time on in-depth review of their chemical lists and bringing in experts to spot smarter substitutions.
It’s true—there’s no silver bullet in chemical safety or environmental health. But taking each solvent, like 1-nitropropane, and breaking down what it does, why it matters, and how risks stack up, makes the whole industry stronger. This kind of work sits at the core of trust between producers, workers, and neighbors.
Is 1-Nitropropane hazardous to health?
A Close Look at the Chemical
Most people haven’t heard of 1-nitropropane before stepping into a factory or chemistry lab. It’s a clear liquid, smells strong, and usually shows up in paint thinners, coatings, inks, and sometimes as part of adhesives. In my years working with industrial chemists, I’ve noticed that this chemical rarely makes headlines, but quietly sits on shelves, trusted to do a job. That’s exactly why a casual attitude toward its risks creeps in so often. One can’t dismiss what’s in play just because it isn’t a household name.
Short-Term Dangers in the Air
Plenty of workers report headaches, dizziness, and irritation after exposure. These are the common first flags: lightheadedness, breathing trouble, and sore throats. Safety Data Sheets published by chemical suppliers spell this out in plain language, but it helps to hear from folks who’ve spent years on paint shop floors. A painter I know shared stories of sudden nausea after working too fast without enough ventilation. Whenever these effects show up, it’s clear the body is sounding a warning.
Long-Term Exposure Carries Weight
Short bursts of 1-nitropropane in the air irritate quickly and fade once people get outside. Longer stretches are another story. Health researchers from OSHA and NIOSH found that repeated exposure can set off more lasting harm. Liver and kidney damage start to look real after months in poorly ventilated shops. Animal studies give us a peek at liver changes and changes in blood chemistry, and while you can’t draw perfect comparisons, it’s reason enough to pay attention.
Eye and skin contact add another layer of risk. I’ve seen techs run into rashes and red eyes, especially when they get careless with gloves or goggles. Risks ramp up fast if skin is left unprotected, or if splashes get ignored. Nobody in the industry shrugs off a chemical burn for long.
Why This Matters for Workplaces
A lot of small businesses cut corners hoping to save time or money, especially when profit margins shrink. There’s always a temptation to open up a drum and “get this done quick.” Still, the cost of a hospital visit or a lawsuit can crush a small outfit. It doesn’t take a catastrophic spill for real trouble to start. A vent fan, fresh gloves, and regular training go further than any quick fix once someone starts feeling sick.
Safer Habits and Solutions
The best safeguard stays simple: respect for what’s on hand and clarity around what’s risky. Plenty of safety professionals I’ve met keep workplace posters up and check protective gear stock weekly. For high-use areas, upgrading to local exhaust systems and air monitoring tools can turn a guessing game into certainty. Substitution offers another road. Some companies have started switching to less toxic solvents, trading off a bit of price or performance for peace of mind.
Getting serious about chemical safety doesn’t always look dramatic. Sometimes it shows up in small moves—a supervisor checking lids, someone swapping out cracked goggles, or a crew taking their break outside after a messy job. 1-nitropropane isn’t the most infamous name on a hazard sheet. The real risk comes from ignoring it.
What are the storage requirements for 1-Nitropropane?
Keeping Safety in Focus
Most folks who work with chemicals know the importance of respecting what’s in the drum. 1-Nitropropane isn’t just another liquid—its low flash point means fire can become a real risk. This chemical catches fire faster than many would expect, turning a regular storage room into a danger zone if handled carelessly. Trying to stash it next to an open flame or even in a sun-soaked shed can end with a scare—or worse.
Every container matters. Metal drums with tight-fitting lids do a much better job at locking in fumes and holding out unwanted moisture. Never ignore leaks, since the smell may be mild but vapor can still gather where you least expect it. Fact is, those fumes aren’t just flammable, they’re heavier than air. I’ve seen vapor drift along floors, slipping into places far from their source, so keeping good ventilation going is more than a box to tick—it's a real-world safeguard.
Conditions Matter More Than You Think
Not every warehouse works for 1-Nitropropane. Leaving it in direct sunlight or where it gets hot offers trouble an open door. Heat ramps up pressure inside the drum, making bursts and leaks more likely. It’s not just about accidents, either. With enough warmth, vapor concentrations rise and the risk climbs alongside them. Areas with steady temps—think cool, shaded spaces—make a better home for this chemical.
Humidity brings its own headaches. Many forget that nitro compounds don’t play well with water. Dampness invites corrosion, and once a storage drum starts to rust, you’re trading long-term safety for short-term convenience. Concrete floors with a spill tray underneath, or at least some non-sparking mats, provide a stronger safety net against what I’d call the “Oops” moment.
Segregation Saves Lives
Anyone who mixes nitroparaffins and oxidizers in storage is tempting fate. 1-Nitropropane should stay far from strong acids, alkalis, and especially oxidizing agents. Chemicals like peroxides or bleach can kick off dangerous reactions faster than most imagine. Working in industry, you see these “shortcut” storage solutions more often than you’d like, but they’re just accidents biding their time.
Even the best-trained teams need reminders. Clear signage, updated inventory sheets, and restricted access all trim down risk. No one benefits from surprises around chemicals. I once walked into a storage room that smelled faintly sweet—just enough to notice that someone hadn’t closed a drum tightly. Fixing small mistakes early keeps faith with safety culture.
Planning for the Worst
It’s easy to overlook emergency gear. Spill kits packed with absorbent pads, fire extinguishers rated for chemical fires, and nearby eyewash stations set the tone for a prepared workplace. Where 1-Nitropropane gets stored, emergency exits need to stay clear, and everyone should know the route in their sleep. It’s not just about getting checked off during audits—it’s about coming home safe every day.
One last thing—regular inspections matter. Not every problem gives warning signs you notice right away. Spotting drum damage or a missing label can mean the difference between order and chaos. In the end, storing 1-Nitropropane demands respect. Easy shortcuts fade quickly when safety stands on the line.
What is the chemical formula of 1-Nitropropane?
Understanding 1-Nitropropane: An Everyday Chemical
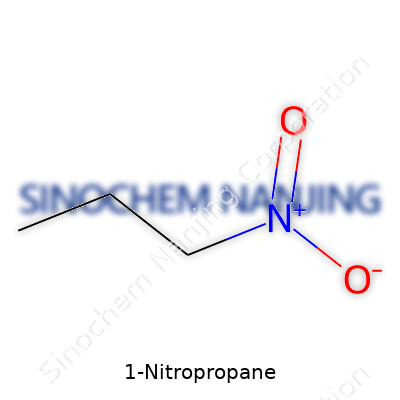
It’s pretty easy to overlook the things that make up the backbone of chemistry labs and industrial formulas, but real learning starts by breaking molecules down to their nuts and bolts. 1-Nitropropane stands as a good example. It’s not one of those chemicals you hear about at the grocery store, but it affects paints, coatings, and even fuel additives that show up in day-to-day life. Chemically, 1-Nitropropane comes from the simple joining of a three-carbon propane chain with a nitro group. Its formula, C3H7NO2, tells a story many folks gloss over. There’s a lot more to unpack than memorizing a string of characters.
Looking Into the Details: What That Formula Means
C3H7NO2 means three carbons, seven hydrogens, a nitrogen, and two oxygens team up to build a small molecule, but that still doesn’t show what separates this substance from a bunch of other nitro compounds. The “1” in the name tells you the nitro group hangs off the first carbon. This small tweak changes everything: the boiling point, reactivity with other chemicals, and even its risks to health.
Folks might wonder why it matters. At school, I saw classmates memorize formulas for quizzes without asking what they could do. Later, working with solvents, I found that knowledge of structure made the difference between a safe day and a trip to the health center. That small nitro group isn’t shy about releasing toxic fumes or causing irritation if spilled.
Value for Industry — And Why Regulations Matter
In the world of industry, C3H7NO2 offers value thanks to its solvency and ability to mix with certain organic compounds. It pops up in the manufacture of paints, varnishes, and lacquers, giving products the right finish or enabling certain properties in coatings. Demand for it runs parallel with the growth in building, furniture-making, and even some specialty adhesives. In cases like mine, where factory staff worked in tight quarters, poor ventilation showed just how important chemical literacy could be when you’re exposed every shift.
Health agencies, both local and global, draw limits on what industries can release into the air. 1-Nitropropane features in the European Chemicals Agency’s database with careful warnings. It’s flagged for its harmful effects on lungs and skin if safety rules get ignored. One neighbor of mine handled it at a local plant, and stories of headaches and routine air checks were normal. That experience taught me that without proper labels, training, and monitoring, a seemingly simple chemical formula turns into a long-term hazard.
Potential for Safer Use and Better Education
Solutions stick closer to diligence and transparency than big innovations. Companies investing in training, using closed systems, and enforcing regular workplace checks cut down incidents year after year. In classrooms, teachers who walk students through each atom’s role go beyond test prep—they build the foundation for safer workplaces and smarter policies. Some newer green solvents promise less toxicity, but adoption hinges on cost and performance.
Knowing why the formula for 1-Nitropropane matters and how it fits in regulations and industry keeps the focus rooted on people and safety. It’s not about chemistry in a vacuum but chemistry right where lives, livelihoods, and lungs are on the line.
How should I handle spills or leaks of 1-Nitropropane?
Understanding What You're Dealing With
1-Nitropropane often turns up in industrial workplaces and labs. It’s a yellowish liquid with a smell similar to ether or chemicals you might have noticed in an auto shop. Coming in contact with it or breathing its vapor sometimes causes headaches, skin or lung irritation, and—in large enough doses—serious health effects. Besides, 1-Nitropropane burns fast and throws off toxic smoke. That’s a recipe for trouble if treated lightly.
Immediate Steps After a Spill
Spills can cause panic. Someone might grab the nearest rag or bucket, but cleaning up 1-Nitropropane means more than just mopping the floor. I’ve seen inexperienced coworkers try to improvise, and trust me, shortcuts pose risks to everyone nearby.
Ventilating the area has to come first. Opening windows and activating fume hoods can help prevent vapor buildup. Don’t reach for fans that could spark—electric motors may ignite the fumes. Evacuate anyone not involved in clean-up to reduce exposure, because even a minor slip-up can have costly consequences.
PPE: No Place for Duct Tape Solutions
Standard work gloves don’t cut it against a chemical like 1-Nitropropane. Nitrile gloves, chemical resistant boots, and goggles or a face shield protect skin and eyes. I always recommend a respirator designed for organic vapors in case the fumes get thick. In my experience, regular dust masks leave you wide open to trouble, so don’t rely on them here.
Containing and Cleaning the Spill
Speed matters—the longer 1-Nitropropane sits, the further vapors travel. Absorb the liquid with sand or a commercial spill kit meant for flammables. Skip sawdust, because it can trigger combustion. Once absorbed, scoop up the material into a container that seals tightly. Placing a label with the date and contents helps avoid any mistakes during disposal.
I’ve seen some people tempted to wash down spills with water. That's risky. Adding water may not dilute the risk—it could spread contamination or carry flammable material into the drains. Companies like OSHA recommend keeping the clean-up dry unless a chemical expert gives the green light.
Safe Disposal—Out of Sight Isn’t Out of Mind
Hazardous material regulations exist for a reason. Tossing waste in regular trash exposes sanitation workers and can lead to hefty fines. Reach out to a certified hazardous waste provider to handle both the contaminated absorbent and any ruined protective gear.
Never store the collected material near sources of heat or open flames. If space is tight, a flammable cabinet keeps everything contained until picked up for disposal. One misstep can give fire the opening it needs.
Long-Term Thinking—Training Makes the Difference
After a spill, take the chance to review what happened. Running regular safety drills and making sure everyone has quick access to spill kits and contact info for emergency help saves lives and property. I’ve seen shops run smoother once each worker knows who to call and which steps to follow—no second-guessing or scrambling.
Safety with chemicals like 1-Nitropropane isn’t just about reacting in the moment—it’s about setting up systems and culture that don’t let little mistakes become big disasters.
| Names | |
| Preferred IUPAC name | 1-nitropropane |
| Other names |
1-Nitropropane 1-NP n-Nitropropane Nitropropane |
| Pronunciation | /ˌwʌn‿ˌnaɪtrəʊˈprəʊpeɪn/ |
| Identifiers | |
| CAS Number | 108-03-2 |
| Beilstein Reference | 1640226 |
| ChEBI | CHEBI:35806 |
| ChEMBL | CHEMBL152797 |
| ChemSpider | 8912 |
| DrugBank | DB01907 |
| ECHA InfoCard | ECHA InfoCard: 100.003.189 |
| EC Number | 211-740-3 |
| Gmelin Reference | Gmelin Reference: **8002** |
| KEGG | C01775 |
| MeSH | D009617 |
| PubChem CID | 7922 |
| RTECS number | **TZ9625000** |
| UNII | 5M67C7NP8T |
| UN number | UN2608 |
| Properties | |
| Chemical formula | C3H7NO2 |
| Molar mass | **75.07 g/mol** |
| Appearance | Colorless liquid |
| Odor | sweetish odor |
| Density | 0.991 g/mL at 25 °C |
| Solubility in water | 8.75 g/100 mL (20 °C) |
| log P | 0.94 |
| Vapor pressure | 3.3 kPa (at 20 °C) |
| Acidity (pKa) | 10.22 |
| Basicity (pKb) | 4.56 |
| Magnetic susceptibility (χ) | -9.6e-6 cm³/mol |
| Refractive index (nD) | 1.393 |
| Viscosity | 2.17 mPa·s (20 °C) |
| Dipole moment | 4.23 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 180.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -120.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2054.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H315, H319, H335 |
| Precautionary statements | P210, P243, P261, P280, P305+P351+P338, P304+P340, P308+P311, P370+P378, P403+P233 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | Flash point: 41°C |
| Autoignition temperature | 410°C |
| Explosive limits | 1.8 – 11.0% |
| Lethal dose or concentration | LD50 oral rat 954 mg/kg |
| LD50 (median dose) | LD50 (median dose) of 1-Nitropropane: **950 mg/kg (oral, rat)** |
| NIOSH | # NL1400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Nitropropane is "25 ppm (90 mg/m3) TWA |
| REL (Recommended) | 0.1 |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Nitroethane 2-Nitropropane Nitromethane Isopropyl nitrate Propyl nitrate |

