1-Nitrobutane: A Straightforward Look at an Understated Chemical
Historical Development
Long before chemists started digging into the details of big pharma molecules and green solvents, research into nitroalkanes caught plenty of attention in the labs of the late 19th and early 20th centuries. 1-Nitrobutane hit the scene as part of that wave. Its synthesis tied closely to early work on nitro compounds, which saw an explosion of interest once nitration reactions of alkanes became reliable. Folks working with nitromethane and nitroethane naturally took the step to larger chains like nitrobutane. Literature from the middle of the last century marks its steady emergence in organic synthesis and energetic materials circles, with its value growing in line with the demand for simple building blocks in chemistry. Older texts don’t obsess over 1-nitrobutane the way they do the smaller nitroalkanes, but the paper trail shows it steadily showing up on chemical inventories, quietly supporting bigger innovations.
Product Overview and Synonyms
Now, you won't find 1-nitrobutane on grocery store shelves, but it earns a spot in chemical supply catalogs around the globe. Also called nitro-n-butane, nitrobutane, or n-butyl nitro, it slides into the family of nitroalkanes—those are organic molecules with one or more nitro groups (-NO2) attached to a hydrocarbon backbone. People sometimes lump it in with its isomer, 2-nitrobutane, though their properties and uses aren't identical. The chemical’s identity hinges on its simple structure and single nitro group, and those characteristics keep its market steady for research and specialty applications rather than mass consumer use.
Physical & Chemical Properties
Take a look at 1-nitrobutane on the bench, you see a colorless to pale yellow liquid, with a faint odor that's not unlike the smell of other light nitroalkanes—sharp, chemical, easily noticeable if a bottle isn’t capped tight. It has a density that's a bit higher than water but lighter than a lot of other nitro compounds. The boiling point sits above room temperature but below the point of easy distillation hazards, with a flash point that means careful storage away from sparks is wise. In water, it doesn’t dissolve much, which often makes cleanup simpler but can challenge analytical work. What sticks with me working in labs is the chemical stubbornness of these small nitroalkanes: they resist easy breakdown, stand up to mild acids or bases, and require strong reducing conditions to budge that nitro group.
Technical Specifications & Labeling
Bottles of 1-nitrobutane usually show common warning icons because of its flammable and toxic nature. Precise purity levels depend on how it was made, but most labs call for material above 98 percent. You’re likely to spot UN numbers for shipping and handling, alongside pictograms for acute toxicity. Regulatory requirements jump in at certain storage volumes, so companies set up their handling stations well-ventilated, with spark-free tools close by. Precise labeling keeps people alert—mislabeling even a single container could bring big headaches down the line.
Preparation Method
Most commercial 1-nitrobutane comes from the direct nitration of butane or by reactions involving n-butyl halides with silver nitrite. The direct approach demands exacting control of temperature and concentration, as over-nitration or side products can spoil the yield. In research settings, batch reactions with careful distillation and washing give a clean product, but large-scale production might use continuous flow setups for safer and more efficient handling. Over the years, as environmental scrutiny sharpened, producers looked for cleaner byproduct management, trying catalytic or phase-transfer methods to push yields and keep waste down. This isn’t just academic; safer, higher-yield processes drive cost lower and reduce risk for both workers and the environment.
Chemical Reactions & Modifications
1-Nitrobutane’s biggest value in the lab comes from its chemical versatility. That nitro group can transform in several directions—reduction leads to amines, often in steps with iron and acid or catalytic hydrogenation. The molecule works for carbon–carbon bond formation in Michael additions or Henry (nitroaldol) reactions, building up complexity from small pieces. Heat or strong bases can strip the nitro group off entirely or shift it, generating a mix of products that advanced synthetic chemists know how to separate and use. Over time, people found that by tweaking catalytic systems or playing with reaction partners, even this small molecule could ink a path toward totally new chemical products.
Safety & Operational Standards
Safety matters a lot with 1-nitrobutane—explosive incidents in poorly ventilated rooms have injured otherwise cautious workers who underestimated its fire hazard. Labs store it in explosion-proof refrigerators, well away from acids, oxidizers, or open flames. Goggles and gloves stay on throughout transfer or sampling, since the chemical can irritate skin and eyes on contact. Spills demand quick cleanup with proper absorbents; some shops opt for storage in secondary containment trays to catch drips. Modern SOPs go beyond government regulation, leaning on hard-won experience to keep labs safe. Emergency response training drills, fire blankets, and spill kits stand ready—all signs that people take these lessons seriously.
Application Areas
Even though 1-nitrobutane doesn’t pull headlines like other chemicals, several industries and research fields count on its reliability. In organic synthesis, it serves as a precursor for pharmaceuticals, agrochemicals, and dyes. Energetic materials research taps its ready nitro group, studying decomposition and reactivity for propellants or explosives. Analytical chemists use it as a standard compound for certain instrument calibrations. Specialty applications tap it for electronic materials or as a probe molecule in gas-phase studies. Each use relies on that mix of volatility, reactivity, and manageable toxicity—qualities that make 1-nitrobutane useful, but keep it off the mass market.
Research & Development
Researchers often look for ways to handle 1-nitrobutane more sustainably and safely. Recent work chases higher-yield synthesis with less waste, aiming to curb energy use and solvent output in the process. Chemists hunt for new catalytic systems that let the molecule slip into more efficient or selective bond-forming reactions, which would open new doors in pharmaceutical or materials chemistry. Studies on environmental fate track how it breaks down in soils or waterways, with a focus on understanding and minimizing its ecological impact. Better toxicity models and exposure guidelines come out of this push, helping both scientists and regulators manage risk with more certainty.
Toxicity Research
There’s no sugarcoating the hazards: 1-nitrobutane can harm both people and wildlife if mishandled. Its vapors irritate mucous membranes, with acute exposure sometimes leading to headaches, dizziness, or in severe cases, central nervous system depression. Long-term toxicity hasn’t seen the exhaustive study given to bigger-volume industrial solvents, but caution rules the day until more robust data shows otherwise. Environmental scientists worry about runoff or accidental spills, as breakdown can produce secondary pollutants. Ongoing animal studies and cell assays work toward clearer answers on thresholds, chronic effects, and safe exposure limits, putting science out ahead of any future regulatory crackdown.
Future Prospects
Looking down the road, 1-nitrobutane sits at the intersection of tradition and innovation in organic chemistry. As synthetic pathways call for ever more tailored building blocks, even modest molecules gain new value. Green chemistry initiatives push producers and researchers to find cleaner routes to nitroalkanes, and stricter safety rules nudge storage and handling standards higher. If new catalysts or reaction systems make it cheaper and safer to use, demand could inch upward in specialty areas. What stands out is the steady curiosity of chemists—always asking where a molecule like this could take the next idea, so even chemicals in the shadows of bigger names keep earning their spot on the shelves.
What is 1-Nitrobutane used for?
The Honest Role of 1-Nitrobutane
Walk into any industrial lab or chemical plant and you’ll spot stacks of unfamiliar compounds with difficult names. One of them, 1-nitrobutane, flies under the radar even though it shows up in some places people don’t always expect. If you have ever worked in a facility that makes plasticizers or solvents, you might catch the sharp, fruity odor of this colorless liquid. Its main job isn’t about making headlines—it's more about making other chemicals possible.
How 1-Nitrobutane is Used on the Ground
Most chemists reach for 1-nitrobutane as a building block. Synthesis labs and specialty chemical manufacturers need it to start reactions that hand off nitrogen, nitro, or butyl groups into new molecules. In practice, this means the compound helps produce pharmaceuticals, dyes, and pesticides. 1-nitrobutane is a key piece in the toolbox for making chemicals like n-butylamine, which shows up in everything from rubber accelerators to drugs that treat anxiety or depression. It’s not glamor work, but without 1-nitrobutane, the chain breaks.
The Health Factor—Why Vigilance Counts
No one’s bottling 1-nitrobutane for home use, and there’s a reason. Handling it in an industrial setting, I’ve seen the safety team suit up with thick gloves and proper ventilation. Brief skin contact stings; breathing vapors could set off headaches or dizziness. The U.S. National Library of Medicine lists it as hazardous—workers need training and equipment that actually work. Companies with strong safety cultures take this seriously, though lapses still happen. Federal agencies like OSHA and the EPA set exposure limits and demand clear storage and accident protocols, but the follow-through at the ground level varies from place to place. Investment in real training—not just posters on a wall—cuts risk. I learned that lesson the tough way early in my career, after seeing how fast a chemical burn can escalate if ignored.
Environmental Footprint
Spills or air emissions from 1-nitrobutane don’t just vanish. In wastewater, it can persist if it isn’t properly treated. It breaks down with sunlight and certain bacteria, though not quickly enough if dumped recklessly. This is especially tough near chemical plants that border water supplies. Oversight and honest reporting lower the risk. Some facilities push to recover and recycle nitrobutanes, which not only saves money but also keeps the compound out of rivers and air.
Better Paths Forward
Workers in the chemical industry often push for more transparent monitoring, frequent health screenings, and smarter process controls to catch vapor leaks. It helps to foster a workplace where nobody ignores even a small spill. Environmental groups urge more investment in greener alternatives, nudging companies to explore cleaner substitute chemicals or additional filtration for wastewater. Real change depends on listening to frontline staff and investing in engineering upgrades—not settling for the same old routines. As chemists and safety specialists keep tightening up rules, 1-nitrobutane might move out of the shadows, making the case for stronger safety and environmental stewardship right where it counts most—on the factory floor and in nearby towns.
What are the safety precautions for handling 1-Nitrobutane?
Understanding the Risks
1-Nitrobutane looks harmless enough in a labeled bottle, but anyone who's poured from that container knows the risks are real. Breathing in nitroalkanes can irritate the nose and lungs, and the liquid itself brings a fire risk. Beyond the warning labels, it’s about working with a chemical that belongs on the “handle with respect” list. Before unsealing that bottle, it's important to know where the danger lies.
Protecting Yourself
Gloves aren’t optional here. Nitrile or butyl rubber stand up well to 1-Nitrobutane’s tendency to pass straight through weaker materials. My own mistake using vinyl gloves years ago taught me how easily liquid chemicals can seep through. Eyes need protection too. A good pair of chemical splash goggles, not your daily safety glasses, stops splashes dead. Lab coats keep the liquid off clothes and skin, and long sleeves close the gaps.
Air Quality Matters
Working near an open container of 1-Nitrobutane without ventilation feels like rolling the dice. Fume hoods turn that risk way down. Labs that cut corners on air flow end up exposing everyone to fumes. A recent review in "Occupational Medicine" found nitroalkane vapors trigger headaches and dizziness fast. Some folks even report nausea after short exposure. Watching out for your own health and your coworkers’ just makes sense. Keep the airflow strong, and keep the containers closed tight between uses.
Spills and Fires
Once, during a night shift, a coworker knocked over a flask of 1-Nitrobutane. The smell hit fast and sharp, but the bigger problem was fire. This stuff lights up at fairly low temperatures. Keeping sources of ignition far away isn’t overkill—it’s just smart. Static sparks, open flames, and even certain hotplates can spell trouble. Spills need a plan: absorb with sand or a specialized spill kit and bag it up for safe disposal. Waste bins should be metal with tight lids, never left open in the lab. I never walk past a spill kit station without checking it’s stocked for this reason.
Storing It Right
Ever see a shelf stacked with chemicals all jammed together? That’s a recipe for trouble, especially with something as touchy as 1-Nitrobutane. Store it in tightly sealed containers, in cool and well-ventilated cabinets, far from acids, oxidizers, or even sunlight. Keeping incompatible chemicals separated is basic, but it saves you from a lot of problems down the line. If your lab fridge looks more like a janitor’s closet, it's time to start organizing.
Training and Culture
Training isn't just a box to check. Newer lab techs learn good habits from steady hands. Watching someone else stop mid-pour to fix a glove, or step back from a container to turn on the vent, sticks with you. Labs with a strong safety culture keep accident rates lower. In my experience, people who ask questions and review the chemical’s safety data find themselves ready when something unexpected happens.
Disposing of Waste
Pouring leftover 1-Nitrobutane down the drain sounds fast, but it lands you in trouble with both the law and your conscience. This chemical counts as hazardous waste and goes in marked containers for licensed disposal. Local rules spell out the details, but the principle is simple—keep it separate, label it clearly, and call for collection when storage fills up.
Solutions for Safer Handling
Switching to less volatile alternatives makes the lab safer, though it isn’t always practical. Automation can help reduce direct contact. Regular drills on spill response sharpen reactions. Most of all, an open-door culture where people feel safe pointing out unsafe storage or outdated gloves lifts everyone’s game. Safety isn’t about checklists. It's about habits, vigilance, and pride in protecting yourself and your team at every step.
What is the chemical structure of 1-Nitrobutane?
Building Blocks of 1-Nitrobutane
1-Nitrobutane stands as a reminder of how simple tweaks in chemical bonding can switch up the behavior of a molecule. Picture a straight chain of four carbon atoms, each with a respectable load of hydrogen hanging on—this is butane’s backbone. Swap out a hydrogen on the first carbon with a nitro group (–NO2), and the chemistry shifts. What you get is 1-Nitrobutane, written out in shorthand as CH3CH2CH2CH2NO2. Under the microscope, the first carbon bonds not just to three hydrogens and the next carbon—one position steps aside for the nitro group, known for its electronegativity and reactivity.
Why This Structure Matters
The nitro group does more than take up space; it flips the script on reactivity. Experience in the lab taught me that nitro compounds make bold choices—they bring explosive tendencies, act as solvents and intermediates, and play by different chemical rules than their non-nitrated relatives. 1-Nitrobutane’s structure puts it in a family of chemicals used for organic synthesis, fuel additives, and sometimes in propellant chemistry. That NO2 group pumps up the molecule’s ability to react, opening doors in synthetic applications or making safety a bigger concern.
Supporting Evidence from Research
Chemists count on knowledge about structure to predict how chemicals behave. The nitro group’s arrangement—one nitrogen doubly bonded to an oxygen atom and singly bonded to another oxygen which latches onto the butane—spells strong electron withdrawal. Peer-reviewed literature like Ullmann’s Encyclopedia of Industrial Chemistry lays out how nitroalkanes get used to build pharmaceuticals and custom polymers. The industrial sector watches these molecules for their unique ability to spark chain reactions or add stability in the right mixture. I’ve come across safety data that points out enhanced volatility and toxicity, common risks for nitroalkanes, and public health documents like the CDC’s profiles on nitro compounds back up those concerns.
Facing the Safety Challenge
As soon as a nitro group comes into play, the stakes rise. I remember one practical exam during graduate school—handling nitroalkanes always meant double-checking ventilation and eye protection. 1-Nitrobutane doesn’t explode with a careless spark like nitroglycerin, still, it deserves respect, both for fire safety and for its effects if mishandled. There’s research pointing to its potential for environmental persistence if accidents occur in industry settings. This speaks to the need for tighter storage protocols and rapid response plans at plants that use or produce it, so local communities aren’t put at risk.
Pathways to Responsible Use
Recognizing structure lets both workers and regulators address hazards at the source. Engineering controls, airtight storage, and routine monitoring guard against most chemical mishaps. Updated protocols based on real-world incidents help the whole sector learn from past oversights. Where possible, green chemists look to substitute nitroalkanes with less hazardous alternatives or build reaction schemes that reuse these compounds instead of dumping them. Funding for waste treatment upgrades and public transparency about chemical processes can also tamp down the long-term risks tied to these molecules.
How should 1-Nitrobutane be stored?
Packing Away Hazard: Respecting 1-Nitrobutane’s Risks
Keeping 1-nitrobutane safe is no backroom detail for science teams and suppliers. Anyone who’s spent time handling volatile chemicals knows accidents often begin with ignored basics. This compound isn’t one that loves sunlight, heat, moisture, or open air. From my time in smaller chemistry labs to walking through large-scale supply rooms, I’ve found the same principle plays out: discipline around storage keeps people healthy and experiments reliable.
What 1-Nitrobutane Demands
Leaving a sealed drum of 1-nitrobutane in a hot storeroom turns it from manageable to menacing fast. Vapor pressure spikes, and what feels solidly closed can push out toxic fumes or kick off a fire risk. This chemical wants a cool, dry, and well-ventilated spot. I learned early on that swinging doors, poor labeling, and cluttered flammables shelves are a recipe for exposures nobody wants to clean up. A simple sign that lays out the real dangers reminds people that safety isn’t decoration; it keeps the future open for everyone in the room.
I’ve witnessed a misplaced bottle lead to minor headaches—literally and bureaucratically—resulting in a scramble for new MSDS sheets, repeat audits, and even interrupted research. Flammable liquid cabinets graded for proper venting offer the right level of containment. Standard fireproof lockers with visible latches signal that safety isn’t getting shortchanged. This culture matters as much as the science itself, especially if students or new hires rotate through shifts on different projects.
Fighting Ignition: The Heat and Spark Problem
Anyone working with 1-nitrobutane can’t rely on luck. Even small leaks or forgotten caps turn regular rooms into potential accident sites. Electrical equipment nearby should pass inspection for spark-proofing because one loose wire or outdated fixture could set everything off. I’ve had old timers in the industry roll their eyes at “too many rules,” but slip-ups don’t discriminate between veterans and rookies. Static charges build up naturally. Grounding the storage containers—actually running a copper wire, not just talking about it—makes a real difference in lowering ignition risk.
Air and Water Don’t Mix Here
Keeping this chemical sealed stops any unwanted reactions. Humidity finds its way into poorly capped bottles, breaking down the product and sometimes creating new hazards unplanned and unwanted. The best setups feature desiccators or sealed secondary containment. I remember one instance years ago when a roof leak brought moisture onto a shelf; it took a week to sort and repack: wasted hours that could have been spent on work that mattered. Catching small issues early pays off multiplied by the traffic any storage room sees in a month.
Training and Oversight Make the Difference
Labels, training seminars, and posted emergency contacts aren’t red tape. They turn unknowns into routine. Even with perfect shelving and monitoring, an untrained tech can undo careful planning. Refresher courses once a year give people muscle memory. In my experience, sharing close calls in staff meetings—without blame—raises everyone’s alertness. Walking through storage checks with new staff rather than handing them a checklist builds confidence and habit.
Safer Storage Leads to Better Work
Poor storage means lost time, lost samples, and in rare but real cases, lost lives. 1-nitrobutane, handled with respect from purchase to disposal, turns from a threat into a tool for progress. Managing hazards with real-world attention isn’t optional; it’s the quiet backbone behind what good labs produce every day.
Is 1-Nitrobutane hazardous to health or the environment?
Looking at 1-Nitrobutane Through a Practical Lens
I’ve seen plenty of chemicals treated either as everyday tools or ticking time bombs, depending on who’s handling them and how. 1-Nitrobutane falls into the former category for many folks working in labs or factories—often used as a solvent or intermediate. Yet, setting aside the technical lingo, let’s dig into what matters to anyone near the stuff: Is it hazardous to your health or the environment?
Health Concerns—Straight from Experience
1-Nitrobutane doesn’t top lists of notorious toxins, but don’t mistake low profile for low risk. Breathing in its vapors can leave you dizzy or with a burning throat. I remember stepping into a storage room without enough ventilation and feeling my head start to spin—later learned the culprit was a nitro compound vapor that had slipped out. Nausea and headaches are real possibilities if the room isn’t aired out properly.
Long-term exposure brings even bigger problems. Animal studies tell us repeat exposure affects the liver, kidneys, and nervous system. Workers who handle chemicals day in and day out face real risks of organ damage unless they suit up, work in hoods, and respect time limits. If it gets on your skin, irritation flares up fast, which happened to me once—gloves with a tear, a little spill, and a red patch that took a day to calm down.
Environmental Impact—What Happens After Disposal
Pouring anything nitro-based down the drain isn’t a harmless act. Once 1-nitrobutane escapes into water or soil, it sticks around longer than you’d like. It doesn’t break down quickly, so it finds ways into groundwater and slowly inched its way through nature. Fish and microorganisms can pay the price, since toxicity doesn’t just vanish once the chemical leaves human hands.
Air emissions can be an issue if 1-nitrobutane evaporates. Combustion creates even nastier byproducts—nitrogen oxides and other pollutants that harm lungs and aggravate asthma. A small release near a storm drain spreads farther than people think, and the ripple effect ends up being much bigger than a stained floor.
Handling Risks and Finding Smarter Paths
Safer handling starts at the source. In my lab days, switching to a closed-system transfer cut down exposure instantly—no more pouring from open drums. Spill kits kept chemicals out of drains, and weekly drills drove home quick response habits. Signs and labels seem minor until a visitor grabs the wrong bottle, so I favored clear, bold labels on every shelf.
Disposal companies, not municipal trash bins, tackle waste solvents best. I once saw a facility store full drums outside with only a tarp for “protection,” and one downpour later, the nearby creek foamed and smelled for days. It takes commitment and regular inspections to keep storage safe and compliant.
Substitution isn’t always possible, but reviewing alternatives forms part of regular safety meetings. Certain green chemistry replacements can step in for nitro compounds in many jobs. Regulations exist for a reason, yet it’s up to frontline workers, supervisors, and regulators to hold one another to a higher standard if health and local rivers matter at all.
Knowing how hazardous a compound like 1-nitrobutane can be means thinking beyond routine chemical lists. It calls for vigilance, a healthy dose of respect, and solid habits that outlast any single bottle or drum. Costs of shortcuts or carelessness stick around for a long time—sometimes in the water, sometimes as a cough that never fully goes away.
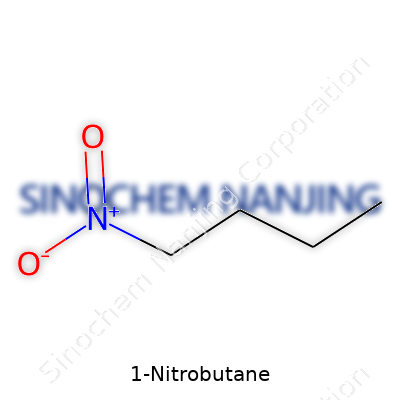
| Names | |
| Preferred IUPAC name | 1-nitrobutane |
| Other names |
1-Nitrobutane n-Butyl nitrite Nitrobutane Nitrobutan-1 |
| Pronunciation | /ˈwʌnˈnaɪ.trəʊ.bjuː.teɪn/ |
| Identifiers | |
| CAS Number | 626-21-7 |
| Beilstein Reference | 1209223 |
| ChEBI | CHEBI:35809 |
| ChEMBL | CHEMBL3181914 |
| ChemSpider | 6194 |
| DrugBank | DB14112 |
| ECHA InfoCard | ECHA InfoCard: 100.007.857 |
| EC Number | 209-703-4 |
| Gmelin Reference | 69601 |
| KEGG | C19136 |
| MeSH | D009642 |
| PubChem CID | 12154 |
| RTECS number | EO1400000 |
| UNII | X8339M5P38 |
| UN number | UN2608 |
| Properties | |
| Chemical formula | C4H9NO2 |
| Molar mass | 89.11 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet odor |
| Density | 0.991 g/cm³ |
| Solubility in water | 12 g/L |
| log P | 0.97 |
| Vapor pressure | 2.9 mmHg (20 °C) |
| Acidity (pKa) | pKa = 9.7 |
| Basicity (pKb) | 10.86 |
| Magnetic susceptibility (χ) | -7.35×10⁻⁶ |
| Refractive index (nD) | 1.398 |
| Viscosity | 1.276 mPa·s (20 °C) |
| Dipole moment | 4.09 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -120.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2775 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H315, H319, H335, H351 |
| Precautionary statements | P210, P261, P280, P304+P340, P312, P370+P378 |
| NFPA 704 (fire diamond) | 2-3-1 |
| Flash point | 43 °C (109 °F; 316 K) |
| Autoignition temperature | 285 °C |
| Explosive limits | 1.8–8.3% |
| Lethal dose or concentration | LD50 oral rat 840 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 1300 mg/kg |
| NIOSH | NA9070000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1-Nitrobutane: 100 ppm (parts per million) |
| REL (Recommended) | REL: 100 ppm (410 mg/m3) |
| IDLH (Immediate danger) | 140 ppm |
| Related compounds | |
| Related compounds |
2-Nitrobutane Nitromethane Nitroethane 1-Nitropropane 2-Nitropropane |

