Unpacking 1-Methyl-3-Hexylimidazolium Chloride: Real Talk on an Ionic Liquid
Historical Development
Curiosity about 1-Methyl-3-hexylimidazolium chloride, commonly abbreviated as [C6mim]Cl, sparks up as soon as you glance at its name. It sits inside the larger family of imidazolium-based ionic liquids, a class of compounds that has taken the chemical world by storm over the past three decades. Back in the 1990s, scientists were hunting for cleaner alternatives to volatile organic solvents. This was about the same time environmental awareness really began picking up steam, and industries started feeling the heat over emissions and chemical waste. Early research often circled around simple imidazolium ions with short chains, but tweaking those chains for better solubility, melting point, and stability led to the birth of derivatives like 1-Methyl-3-hexylimidazolium chloride. This compound caught attention for its ability to dissolve a range of organic and inorganic compounds and for standing up to heat and stress, something traditional solvents just couldn’t do as reliably.
Product Overview
Peering into the beaker, 1-Methyl-3-hexylimidazolium chloride might look unassuming, usually presenting as a colorless to pale yellow liquid or solid, depending on room temperature and humidity. What goes unnoticed at first glance is the unique blend of properties this substance offers. Users often reach for [C6mim]Cl when regular solvents quit the job early—its stability and ionic nature have earned it a coveted spot on the workbenches of academic labs and chemical plants. Unlike many chlorinated solvents with a notorious reputation for toxicity and volatility, [C6mim]Cl doesn’t evaporate and stink up a room, nor does it set off alarm bells when used in small amounts with the right precautions.
Physical & Chemical Properties
Physical profiles drive much of the discussion around ionic liquids. [C6mim]Cl brings in a melting point that can land close to room temperature, depending largely on water content and impurities. Its hexyl group not only adds some bulk to the molecule but also lowers the melting point compared to its shorter-chained cousins, making it more accessible for liquid-phase processes. Chemically, this compound keeps a steady hand under both acidic and basic conditions, and its solubility in water and certain organic solvents brings practical versatility. The substance doesn’t freeze like water or boil off after a short stint on a hot plate, which translates to longer reaction times and less fiddling with environmental controls during experiments.
Technical Specifications & Labeling
People who deal with chemicals know label details aren’t just for show. For [C6mim]Cl, technical data covers purity, water content, and any trace contaminants that could affect performance. It typically gets bottled in airtight, light-resistant containers to keep degradation and uptake of water in check. You don’t want unexpected reactivity because of sloppy handling or ambiguous labeling. Professional operators check batch numbers, purity percentages, and safety instructions—a sensible approach when you consider trace water can tip delicate reactions off-balance or impact the truly low viscosity prized in some syntheses.
Preparation Method
If you ever tried to whip up [C6mim]Cl in the lab, you’d notice it isn’t rocket science, but it still needs finesse. The process usually starts with a classic alkylation—methylimidazole reacts with hexyl chloride, yielding the target imidazolium compound and a byproduct—typically handled under an inert atmosphere to keep impurities out. After the reaction, purification steps like washing with organic solvents and recrystallizing come in, ridding the batch of any unreacted starting materials. Factory-scale processes don’t stray far from this script, just scaled up with better equipment to manage heat and maximize yield. There’s an art to getting a crystalline, water-free product, and the best chemists pay attention to every step.
Chemical Reactions & Modifications
Possibilities open wide when modifying [C6mim]Cl or reacting it with other chemicals. The chloride anion sitting beside the imidazolium cation is more than just a spectator. It can be exchanged out for other anions through metathesis reactions, swapping chloride for something like tetrafluoroborate or hexafluorophosphate. This uncomplicated swap transforms not only the solubility but also the toxicity and reactivity, opening doors to new research routes. The imidazolium core can act as a phase-transfer catalyst or a template for coordination chemistry with transition metals, which anchors its spot in the field of catalytic green chemistry and material sciences.
Synonyms & Product Names
Beyond its formal IUPAC name, this compound pops up in literature with aliases like [C6mim]Cl, BMHexImCl, or 1-methyl-3-hexylimidazolium chloride. Researchers lean on shorthand because it’s a mouthful to say every time, but accuracy matters to keep confusion at bay, especially with so many similar-sounding imidazolium variants floating in the ether. Product names usually track the structure closely, adding another layer of confidence for those tracking chemicals across suppliers and journal papers.
Safety & Operational Standards
Safety isn’t just about donning lab coats and goggles—it’s also about a real understanding of risk. While [C6mim]Cl doesn’t carry the volatile stench or acute toxicity of legacy solvents, careful handling still makes sense. Direct skin or eye contact brings irritation risks, and without gloves, you might pay the price with rashes or discomfort. No one should assume an ionic liquid is green just because it resists vaporizing. Spent compounds demand responsible disposal, since incomplete data on long-term environmental breakdown persists. Smart labs focus on containment, good ventilation, and proper spill response—a heavy lesson after seeing what happens when containment fails.
Application Area
Ionic liquids such as [C6mim]Cl don’t spend their lives stuck in test tubes—real work happens in industry. In my experience, chemists find [C6mim]Cl especially handy for extracting bioactive compounds, dissolving cellulose, and running non-aqueous electrochemistry. Its stability at elevated temperatures turns it into a go-to for high-temperature catalysis and advanced separation processes. Pharmaceutical research teams crank up yields by loading this solvent into custom synthesis of active ingredients. More engineers look to ionic liquids while separating precious metals from waste streams, since traditional water-based methods don’t always succeed or come with greater hazards. The story keeps turning as biofuel researchers keep testing it for breaking down rigid plant fibers more efficiently than water or caustic soda ever could.
Research & Development
Watching the shift in R&D projects over time really brings perspective. Early on, research centered around describing basic properties of [C6mim]Cl, testing how different substitutions tweak melting points or alter miscibility. In recent years, research targets ways to recycle and reuse the compound or adjust its structure for biodegradability. Academics team up with industry trying to design custom-tailored solvents, optimizing every aspect for their unique applications. There’s also a push to integrate it into next-generation batteries, thanks to its resistance to degradation and broad electrochemical windows. These advances aren’t just academic footnotes—they shorten timelines in chemical manufacturing, shrink environmental footprints, and lower the overall risk of process upsets.
Toxicity Research
Trust in any new solvent boils down to how it behaves in the real world, especially when it comes to health and environmental safety. Research so far says [C6mim]Cl shows relatively low acute toxicity, but chronic effects remain less clear. The longer alkyl chain compared to shorter imidazolium relatives may hike up toxicity, especially to aquatic life. Studies show potential for slow environmental breakdown, raising eyebrows about its persistence if spilled or released in large volumes. Regulatory agencies and scientists urge caution—thorough risk assessments, in-depth studies focused on degradability, and possible transformation products if exposed to sunlight or microbes. Putting restrictions on discharge, along with investing in better recycling and waste treatment, can go a long way toward keeping risk levels low without slowing innovation.
Future Prospects
[ C6mim]Cl stands at an interesting crossroads. As regulatory pressure on legacy solvents ramps up, chemists gravitate toward ionic liquids for their lower volatility and higher selectivity. Emerging trends suggest tweaking the molecule for faster breakdown or incorporating it into benign matrix systems for next-gen polymer processing. Energy storage, sustainable extraction, and advanced catalysis could all see breakthroughs driven by careful engineering of these compounds. Getting there means doubling down on toxicity research and long-term ecological studies—something that’s tough, but absolutely crucial if ionic liquids will become a mainstay rather than a fleeting trend. Real progress in process integration, cost reduction, and greener synthesis can tip the balance, making this compound and its relatives an everyday tool for tomorrow’s chemists and engineers.
What is 1-Methyl-3-Hexylimidazolium Chloride used for?
A Modern Tool in Chemistry
1-Methyl-3-hexylimidazolium chloride, often shortened to [HMIM]Cl, steps into the spotlight when scientists and engineers want something reliable for dissolving, separating, or catalyzing chemical reactions. This compound doesn’t look flashy in a bottle, but chemistry labs, battery manufacturers, and some green technology startups see a lot of promise in this colorless liquid salt.
Why Industry Looks at Ionic Liquids
Ionic liquids—liquids made wholly of ions—don't just evaporate in open air like water or alcohol. That means less risk for air pollution, less stink, and less worry about fires. When I first encountered [HMIM]Cl, I saw a chemical that handles high temperatures, doesn’t burn your nose, and refuses to boil away quickly. That caught my attention in a college research lab, where cleaning up solvents meant headaches at the end of the day.
Think about the jobs liquids do in industry. They dissolve stubborn solids, carry electricity in batteries, move heat from one part of a machine to another, help break down plants and even plastics. Most alcohols or hydrocarbons create their own set of problems—flammability being a big one. [HMIM]Cl shifts the equation toward safety in many of these roles.
Key Uses Across Industries
ElectrochemistryRechargeable batteries and fuel cells benefit from [HMIM]Cl and other similar ionic liquids. Regular batteries heat up and wear out faster. [HMIM]Cl keeps its cool for longer and lets ions travel more easily. The battery industry chases longer life and higher safety standards; this compound can help deliver both.
Catalysis and Chemical SynthesisLabs and factories turn to [HMIM]Cl as a solvent or co-solvent for reactions that traditional solvents struggle with. Copper-catalyzed click reactions or processes that pull value from agricultural leftovers run smoother with an ionic liquid in the mix. I’ve watched researchers replace volatile solvents in favor of [HMIM]Cl, eager to pitch their process as “greener” and often less toxic, both to workers and to the world outside a factory’s fence.
Biomass ProcessingAnyone looking at cellulose or lignin from plants faces a challenge—standard solvents barely touch these tough materials. [HMIM]Cl dissolves cellulose, helping turn wood, crop waste, or paper sludge into sugars, fuels, or even specialty chemicals. This prospect excites scientists championing a circular economy since it opens new doors to sustainable sources for plastics, textiles, or fuels.
Environmental and Safety Considerations
[HMIM]Cl refuses to ignite or explode. It doesn’t easily vaporize into thin air, so workers face less danger breathing in fumes. On the other side, the real world rarely matches a safe lab. Disposal matters. We still need better data on long-term toxicity and what happens when this compound leaks outside the plant. Some studies question how “green” any ionic liquid really is, especially if it lingers in soil or water. Big progress in safety can give way to new questions on persistence and biodegradability.
Room for Improvement
The chemistry world, like most fields, doesn’t sit still. Chemists push for even safer, faster, and cheaper ways to run reactions. [HMIM]Cl solves some headaches, introduces others. Its cost sits higher than basic alcohols, so big projects look for ways to recycle and reuse it. Research teams work on new versions of ionic liquids showing quicker breakdown in the environment while keeping the properties that work in labs and factories.
In the end, [HMIM]Cl gives us a sharp tool—a liquid salt fit for a modern lab or energy startup. How we use it depends on how far we chase greener chemistry and safer workplaces. As with any clever tool, the challenge isn’t just in its abilities, but also in using it wisely for the future.
What are the chemical properties of 1-Methyl-3-Hexylimidazolium Chloride?
Getting to Know the Molecule
1-Methyl-3-hexylimidazolium chloride might sound like a mouthful, but it’s just one member of a fascinating group called ionic liquids. I’ve seen this molecule pop up in research labs and industry experimentations, mostly because it can do things regular solvents can’t. The “imidazolium” core—a stable ring with nitrogen atoms—anchors the molecule, while a methyl and a hexyl group swing off the ring, shaping its physical and chemical personality. The chloride ion tags along, bringing the salt’s ionic nature to life.
Solubility and Stability in Practice
This ionic liquid behaves very differently from the solvents under the kitchen sink. It doesn’t evaporate like water, and its vapor pressure barely registers at room temperature. Picture a salt that looks and acts like a thick, clear oil. I remember during my research days, handling this kind of compound felt almost slippery, a tactile reminder of its low volatility. Its melting point sits well below that of table salt, so you don’t see it forming crystals at room temperature. It mixes well with water and many organic solvents, letting chemists fine-tune reaction conditions far beyond the reach of classic salts.
Chemical Reactivity and Interactions
One feature that sets it apart is its chemical resilience. Most acids and bases barely faze it, though strong bases like sodium hydroxide can start breaking it down. The imidazolium ring holds firm against gentle heating, surviving temperatures that send most organic liquids running for cover. I’ve read about it standing strong up to 200°C—sometimes even higher—depending on its environment. Chloride, the counterion, brings ionic conductivity into play. This makes the liquid handy for electrochemical experiments, where moving charges matter more than taste or smell.
Environmental and Safety Notes
Here’s something researchers keep in mind: not every “green” solvent truly helps the planet. It won’t burst into flames or explode, but that doesn’t mean it’s harmless. Many ionic liquids, including this one, resist breaking down in nature. If someone dumps it down a drain, microbes can struggle to digest it, raising long-term environmental questions. If handling spills in the lab, it’s sticky and persistent, clinging to surfaces. Wearing gloves isn’t just for show—skin contact brings risks, and inhaling any dust or vapor (if heated) gets frowned upon in safety training.
Tools for Modern Chemistry
This salt’s chemical properties open doors in chemical synthesis and energy storage. Chemists use it to dissolve tough substances, coaxing reluctant molecules to react. It can carry electricity in batteries or pull toxins out of polluted water. Not every application works smoothly—cost and recycling challenges get in the way—but teams keep tweaking the formula to cut waste and keep toxicity in check. The structure allows designers to swap in longer or shorter chains, tweak the ions, and push for less-harmful, more recyclable alternatives.
Looking for Solutions
Solving issues with persistence and toxicity means rethinking design from the ground up. I’ve seen university teams working with biodegradable tweaks to the imidazolium core, aiming for salts that break down faster in soil or water. Companies invest in collecting and reusing these liquids, much like metal catalysts. Regulation moves slowly, yet better policies encourage safer choices and responsible disposal. Anyone working with these salts ends up part of a bigger conversation about balancing performance with responsibility—putting both science and conscience to work in the lab.
Is 1-Methyl-3-Hexylimidazolium Chloride hazardous or toxic?
Understanding the Chemical
Plenty of folks working in research labs have noticed the rise of ionic liquids, including 1-Methyl-3-Hexylimidazolium Chloride. Some call these compounds “designer solvents,” and chemists love them for tasks ranging from catalysis to extraction. Still, a chemical with a long name can pack some risks, so it makes sense to ask if this one’s safe to use day in and day out.
Looking Over the Hazards
Regulatory data on 1-Methyl-3-Hexylimidazolium Chloride doesn’t exactly fill library shelves, but what’s out there tends to raise an eyebrow or two. Studies show this ionic liquid carries moderate toxicity both for humans and for aquatic organisms. Its imidazolium ring is known to disrupt cell membranes. Some researchers have described issues such as skin and eye irritation after brief exposure. If you’re unlucky or working without gloves, extended contact can lead to more severe burns or inflammation. When I worked with similar chemicals, safety data sheets recommended thorough ventilation and double-checking the fit of safety goggles and gloves.
Exposure risks don’t stop with skin contact. Inhaling the dust or vapors could irritate the respiratory system, and there’s not enough long-term data to know much about possible chronic effects. We do know imidazolium ionic liquids have shown genotoxic properties in some in vitro tests. Swallowing isn’t likely in a typical lab, but even accidental ingestion can cause gastrointestinal distress. Putting facts in perspective, these risks aren’t just hypothetical; there’ve been enough published accounts to encourage careful handling across research and industrial settings.
Environmental Concerns
People often assume lab chemicals break down quickly after use, but not all do. 1-Methyl-3-Hexylimidazolium Chloride degrades slowly in nature and lingers in soil and water. This can threaten aquatic life, as ionic liquids tend to accumulate in fish and other organisms up the food chain. Research shows even low concentrations can hurt the growth of plants and microorganisms essential for clean water and healthy soil. The persistence means one careless spill could affect an eco-system for years. During my graduate work, we spent days developing waste protocols specifically because these types of salts don’t just “wash away” like some old solvents might.
What Safety Looks Like in Practice
To stay safe around 1-Methyl-3-Hexylimidazolium Chloride, standard lab protocol won’t cut corners. That means sealed containers, well-ventilated workspaces, and gloves that actually resist ionic liquids — not just the standard latex. Anyone working near this chemical should use splash goggles and lab coats. Local exhaust ventilation matters because fumes may irritate lungs, especially over a full workday. Waste needs to head straight to a designated collection site. I saw whole labs in university research buildings get written up for pouring dilute solutions down drains without thinking about where those chemicals end up.
Better Practices and the Future
It’s possible to enjoy the benefits of these chemicals without trading away personal safety or environmental responsibility. Better training, stricter safety policies, and clear labeling help. Reading the latest peer-reviewed studies can help teams recognize updated hazards or smarter disposal options. Chemical manufacturers now include clearer hazard statements and toxicology summaries thanks to more outspoken regulatory agencies. As new green chemistry options grow, a shift to less persistent solvents might spare workers and waterways alike from harm. My advice: know what your team handles, and never make assumptions about a compound’s safety — that knowledge keeps people and the planet healthier in the long run.
How should 1-Methyl-3-Hexylimidazolium Chloride be stored?
Looking Out for Longevity and Safety
It’s easy to ignore the quiet dangers chemicals bring, especially in places where work gets busy and time for best practices slips away. With a chemical like 1-Methyl-3-Hexylimidazolium Chloride, a little care goes a long way. Stories out of academic labs stress this: minor spills turn into real problems, and leftover containers sometimes leak or form deposits when left out in humid corners.
Chloride-based ionic liquids such as this compound don’t react violently in most room-temperature settings. Even so, they grab water from the air and can slowly degrade if the moisture builds up. After years working near chemical stockrooms, I’ve learned how quick humidity changes can ruin supplies. Caps left off for just an hour sometimes lead to crystals or cloudy solutions, and that’s a mess that nobody wants to clean up.
Controlling Temperature and Air
A room running too warm or an open window on a muggy day can turn a secure shelf into an accident waiting to happen. Heat tends to increase the risk of decomposition. This substance prefers living in a cool, dry space — think refrigerators made for chemicals, not kitchen fridges filled with food. Routine checks with digital thermometers make sure settings stay within range. Once, a broken seal on a fridge let condensation seep into glass bottles, causing them to corrode at the neck. In that case, the chemical itself clung to every drop, creating unexpected sticky patches across the shelf.
Dust and outside vapors act as silent saboteurs. Small particles collect inside containers if lids don’t fit tightly or bottles crack. Investing in airtight glass containers saves everyone headaches down the line. After a few rounds of inventory spills, our lab staff moved toward amber bottles. Not only do amber bottles block sunlight, but they also take the edge off unnecessary exposure and slow down photo-degradation. Light exposure can shift colors or alter behavior in the ionic liquid, sometimes permanently.
The Importance of Labels and Logs
Mislabeling turns routine storage into a guessing game no one wins. It’s a rule in any reliable lab or industrial facility: always write the exact date, substance name, and any hazards right on the container. My own experience tells me that busy people neglect this step, leading to confusion when the next shift tries to identify a mystery vial. Teams do better with shared digital inventory logs tracked alongside shelf labels.
Practical Solutions for Safer Storage
Good habits start with training. New staff should see demonstrations on decanting, wiping spills, and the safest way to handle even small containers. Double containment goes a long way; keeping the main bottle inside a shatterproof secondary dish helps catch leaks. In our lab, one chemical-resistant tray beneath every row already caught two minor leaks last year.
Disposal matters as much as storage. Old or unusable stocks belong in chemical waste, never down the sink or in regular trash. Local and federal rules spell out these steps clearly — any place that ignores them invites fire marshals or health officials, not to mention putting colleagues at risk.
Looking around a well-kept chemical storeroom, you’ll find dry shelves, tightly sealed and clearly labeled bottles, temperature logs, and secondary containment everywhere. Sticking to those basics means cleaner work, fewer spills, and a safer space for everyone who sets foot in the stockroom.
What is the typical purity of commercially available 1-Methyl-3-Hexylimidazolium Chloride?
Why Purity Matters in Practice
Back in my university days, I remember a colleague struggling all semester because the ionic liquid she ordered showed wild results due to a low-grade product. It’s easy to overlook purity, especially when a catalog lists a shiny number that sounds professional, but that number makes or breaks everything in both lab work and industry. 1-Methyl-3-Hexylimidazolium Chloride often lands on that dividing line between research success and frustrating setbacks.
Typical Purity Ranges You See on the Market
Commercial suppliers like Sigma-Aldrich, IoLiTec, and Strem list their 1-Methyl-3-Hexylimidazolium Chloride at purities ranging from 95% up to 99%. The 98% mark seems to dominate most listings. If you’re sourcing from Asia, you might spot offerings at 95% and “ultra-pure” batches closer to 99.5%. The lower end sometimes slips in trace contaminants—starting with leftover solvents, halides, or even unreacted starting materials. These numbers sound small, but a contaminant at one percent can spark trouble. In catalysis, a trace of sodium chloride can tank the yield, and in electrochemistry, nothing ruins a run faster than unexpected chloride signals.
Behind the Number: What Does 'Purity' Actually Mean?
That stated percentage rarely covers every impurity that counts. Most sellers post specifications on water content (Karl Fischer checks might say <0.5%), chloride sources, or color—sometimes even UV-Vis data if they're catering to a fussy crowd. True, routine NMR or IR scans help flag off-the-wall contaminants, but the average researcher will notice only if a batch comes with yellowish hints or a musty odor. Unless you’re willing to splurge on the priciest bottles from niche producers, it helps to expect some variability—even within the same stated purity.
Pitfalls from Cutting Corners on Purity
My first failed synthesis using a budget ionic liquid still haunts me. Instead of a clear reaction, I ended up with a solid mess that wouldn’t dissolve. Anyone who thinks “a couple of percent won’t matter” probably hasn’t spent hours trying to clean a stubborn product or troubleshoot a dead sensor. In areas like fuel cells or green chemistry, slipping past quality checks means entire efforts can go right down the drain. It’s not just about cleaner results; higher-purity materials cut costs by saving time, materials, and reputation.
How to Make Purity Work for You
Instead of playing purity roulette, always read past the bold numbers. Request a detailed datasheet. If you can, grab a tiny sample and run your own checks: NMR, melting point, water content. For essential applications—a pharma synthesis, a breakthrough sensor—don’t hesitate to invest in the top-grade version, even if it hits the budget. Some colleagues swear by in-house purification, like charcoal treatment or gentle distillation, to bring purity past what the label guarantees.
It’s tempting to cut costs but, from experience and what the literature makes clear, contamination piles up costs and confusion. Ask smart questions, push your suppliers for specifics, and keep your own quality bar high. Chemistry rewards those who prepare, not those who gamble on “good enough.”
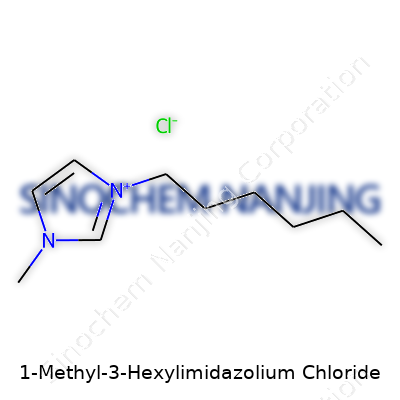
| Names | |
| Preferred IUPAC name | 1-hexyl-3-methyl-1H-imidazol-3-ium chloride |
| Other names |
HMIMCl 1-Methyl-3-hexylimidazolium chloride 1-Hexyl-3-methylimidazolium chloride |
| Pronunciation | /ˈwʌn ˈmɛθ.əl θri ˈhɛk.sɪl ɪˌmɪd.əˈzoʊ.li.əm ˈklɔː.raɪd/ |
| Identifiers | |
| CAS Number | 324025-38-1 |
| 3D model (JSmol) | `[1H-imidazol-3-ium, 1-hexyl-2-methyl-, chloride]` JSmol 3D Model String: ``` [H][C+]1=C(N=CN1CCCCC)C.[Cl-] ``` This is the SMILES string you would use for the JSmol 3D model. |
| Beilstein Reference | 3920395 |
| ChEBI | CHEBI:140220 |
| ChEMBL | CHEMBL2029457 |
| ChemSpider | 21871392 |
| DrugBank | DB11197 |
| ECHA InfoCard | 100.247.565 |
| EC Number | 613-404-9 |
| Gmelin Reference | Gm:2167607 |
| KEGG | C221073 |
| MeSH | D000068280 |
| PubChem CID | 11505722 |
| RTECS number | GV0805000 |
| UNII | Z4C0T0G0SJ |
| UN number | Not assigned |
| Properties | |
| Chemical formula | C10H21ClN2 |
| Molar mass | 237.80 g/mol |
| Appearance | white to off-white solid |
| Odor | Odorless |
| Density | 1.06 g/cm³ |
| Solubility in water | soluble |
| log P | -1.27 |
| Vapor pressure | Vapor pressure: <0.01 mm Hg (20 °C) |
| Acidity (pKa) | 14.2 |
| Basicity (pKb) | 1.9 |
| Magnetic susceptibility (χ) | -81.5×10^-6 cm^3/mol |
| Refractive index (nD) | 1.484 |
| Viscosity | 180 cP (25 °C) |
| Dipole moment | 7.28 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 193.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -520.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -711.3 kJ/mol |
| Pharmacology | |
| ATC code | This product does not have an ATC code. |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. |
| GHS labelling | GHS07, Warning, H315, H319, H335 |
| Pictograms | GHS05, GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | Precautionary statements: P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Lethal dose or concentration | LD50 oral rat > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): >2000 mg/kg (oral, rat) |
| PEL (Permissible) | PEL (Permissible exposure limit) for 1-Methyl-3-Hexylimidazolium Chloride is not specifically established by OSHA, NIOSH, or ACGIH. |
| REL (Recommended) | 0.02 mg/m³ |
| IDLH (Immediate danger) | NIOSH: Not established |
| Related compounds | |
| Related compounds |
1-Butyl-3-methylimidazolium chloride 1-Ethyl-3-methylimidazolium chloride 1-Octyl-3-methylimidazolium chloride 1-Methyl-3-propylimidazolium chloride 1-Methyl-3-hexylimidazolium bromide 1-Methyl-3-hexylimidazolium tetrafluoroborate |

