Looking at 1-Methyl-3-Ethylimidazolium Tetrafluoroborate: More Than Just a Solvent
Historical Development: Tracing the Roots of Ionic Liquids
Chemists didn’t always have an answer for the challenges of volatile organic solvents. Early lab notebooks from the mid-to-late 20th century started mentioning compounds like 1-Methyl-3-Ethylimidazolium Tetrafluoroborate — often nicknamed [EMIM][BF4] among researchers — whenever greener chemistry hit the agenda. Unlike traditional solvents, these new liquids had almost no vapor pressure, meaning much less environmental release. The push for non-flammable, stable substances grew as more process chemists dealt with fires, spills, and regulatory headaches. Despite being overlooked in the early years, ionic liquids — this one included — ended up in various labs simply because scientists needed reliable, non-volatile options. At first, researchers struggled with cost and purity, but those who tried them saw the potential. Slowly, companies began to scale production and the volume of published research picked up steam, often echoing stories of stubborn solvents swapped out for this innovative option.
Product Overview: Not Your Everyday Salt
1-Methyl-3-Ethylimidazolium Tetrafluoroborate doesn’t fit the mold we expect from salts. Rather than powdery or crystalline, it forms a clear, sometimes slightly yellowish liquid at room temperature. I remember the first time I handled it: the distinct viscosity made pouring more like handling syrup than water. Its main job as a solvent skips the line in applications that punish traditional organics — think electrochemistry and catalysis. The ability to dissolve both organic and inorganic compounds convinced plenty of researchers to try it for odd-ball mixtures that usually defy blending.
Physical & Chemical Properties: What Sets It Apart
1-Methyl-3-Ethylimidazolium Tetrafluoroborate usually presents itself as a viscous, colorless to pale yellow liquid. Its melting point falls well below most common table salts, and it boasts remarkable thermal stability. Without a significant vapor pressure, you don’t catch a whiff of it in the lab, which proves a relief after years of inhaling solvents that linger in the air. The ionic nature brings an unusually high electrical conductivity — something that grabbed the attention of engineers searching for new electrolytes. The chemical balance between the imidazolium core and the tetrafluoroborate anion confers a broad electrochemical window, a crucial reason for its adoption in battery and capacitor research. It also resists typical hydrolysis, which means once made, it stays intact even with some water around or under mild acidic conditions.
Technical Specifications & Labeling: Reading Between the Lines
Lab labels for 1-Methyl-3-Ethylimidazolium Tetrafluoroborate usually list purity over 99 percent for advanced applications, especially in electrochemical setups. Spec sheets reveal details about residual water and halides, since even a trace of these impurities can throw sensitive experiments off track. Safety labels call out the low vapor pressure and relative non-flammability; that offers a major practical advantage during handling compared to acetone or ether. Even so, meticulous handling remains universal, since any ionic liquid can cause issues upon skin or eye exposure.
Preparation Method: How It’s Made
Modern manufacturing often begins with 1-methylimidazole, a staple in many organic syntheses, and an electrophile like ethyl bromide or ethyl chloride. The resulting 1-methyl-3-ethylimidazolium bromide or chloride trades its halide for tetrafluoroborate through a straightforward metathesis reaction, often using sodium tetrafluoroborate. After filtration and washing, the final product gets dried under vacuum. Labs invested in rigorous research typically go through repeated purification cycles, often washing with ethyl acetate and drying with molecular sieves, to knock out every last trace of moisture.
Chemical Reactions & Modifications: More Than a Bystander
Far from acting as a simple solvent, 1-Methyl-3-Ethylimidazolium Tetrafluoroborate gets involved in chemical reactions, sometimes as a phase transfer catalyst, sometimes as an actual reactant in ionic liquid-promoted synthesis. Some research follows the path of modifying the imidazolium ring itself, tweaking the side chains to tune solubility and reactivity, though the tetrafluoroborate version frequently serves best as-is for electrochemistry. Its stability has driven chemists to use it in organometallic catalysis, biphasic extractions, and even in polymerization reactions.
Synonyms & Product Names: Decoding the Labels
This compound often goes by 1-Methyl-3-Ethylimidazolium Tetrafluoroborate in catalogs, but those who spend time browsing chemistry journals recognize abbreviations like [EMIM][BF4]. Older literature sometimes lists it as methyl-ethylimidazolium tetrafluoroborate or MEIM BF4. These variations spring from the sometimes wild-west history of chemical nomenclature, though major vendors now help clear up confusion by following systematic names whenever possible.
Safety & Operational Standards: Not Completely Harmless
Those who treat ionic liquids as risk-free quickly discover otherwise. While its vapor pressure stays low and it resists ignition, 1-Methyl-3-Ethylimidazolium Tetrafluoroborate brings its own set of challenges. The compound can irritate the skin and eyes, and it stays persistent in the environment if spilled. Cleanup takes work, and you can’t just flush it down the sink. I’ve learned the value of gloves and safety glasses the hard way: splash a drop on an uncovered wrist, and the mild burning sensation follows soon after. Proper handling practices remain essential, alongside rigorous waste management that fits with local and global environmental standards.
Application Area: Where It Excels
Batteries and supercapacitors now count on this ionic liquid as an electrolyte, thanks to its stability and high conductivity. Electroplating and metal finishing labs value its ability to keep metal ions dissolved and evenly spread without the hazards of nitric acid or cyanide solutions. The solvent power covers everything from herbal extractions to high-performance liquid chromatography. Green chemistry circles have championed it as an alternative to volatile solvents, especially in catalysis and biphasic reactions that need a clear separation between phases. Academic laboratories still pick it up for materials science, synthetic organic chemistry, and separation science.
Research & Development: Working Past the Hurdles
Research never stands still. A decade or two ago, price and limited supply made this compound available mostly for small-scale work. Now, broader access has encouraged new experiments in fuel cells, solar energy, and ionic-based lubricants. Green chemists keep refining the synthesis route to cut waste and energy use. Some groups work to swap out the tetrafluoroborate anion for something less likely to persist in the environment, aiming to combine the practical benefits with less long-term environmental impact.
Toxicity Research: Reality Check
It’s tempting to assume “non-volatile” means “safe,” but years of careful study paint a mixed picture. As much as the low vapor pressure and lack of fire risk help, that doesn’t exempt this ionic liquid from toxicity concerns. Long-term exposure or improper disposal can hurt aquatic life, and recent studies have found certain ionic liquids stagnate in soil or water, making their way into food chains under some conditions. The nuanced toxicological data tells researchers to approach these substances with caution. More work is underway to pin down their breakdown products, chronic effects, and environmental fate, with many labs pushing for third-party and long-term studies that go well beyond the initial safety testing.
Future Prospects: Challenges and Innovation Around the Corner
More labs push for energy storage and environmental-friendly chemical processes, so everything from power grids to specialty syntheses points toward ionic liquids like this one. Yet, wider adoption faces a stubborn truth: cost still runs higher than commodity solvents, and questions linger about life-cycle impacts. Engineers work at finding recycling or regeneration routes, aiming to close the loop and bring down costs by cutting waste. Synthetic chemists tinker with both the imidazolium ring and the anion, looking for versions with better performance or lower ecological footprint. The push for cleaner energy, safer labs, and faster automation means 1-Methyl-3-Ethylimidazolium Tetrafluoroborate likely stays on the frontlines of research and industry. If the field nails scalable, less toxic versions and closes environmental loops, the next decade could see this “liquid salt” grab even more headlines.
What are the main applications of 1-Methyl-3-Ethylimidazolium Tetrafluoroborate?
The Story Behind This Ionic Liquid
Ionic liquids turn a few heads these days, and 1-Methyl-3-Ethylimidazolium Tetrafluoroborate—sometimes shortened as [EMIm][BF4]—definitely pulls its own weight. Coming from a chemistry background, I remember spotting this salt in research labs for its sheer resilience; it doesn’t evaporate easily, shrugs off decomposition up to high temps, and doesn’t catch fire like common solvents. That makes a huge difference, especially if you’re concerned about workplace safety or green chemistry.
Electrochemistry: A Cleaner Path to Energy Storage
Walk through any modern battery research center, and you’ll spot scientists using [EMIm][BF4] for supercapacitors and advanced batteries. The salt stands out because it stays liquid across a wide temperature window, offering a safer route than flammable organic solvents. In real-world batteries, electrolytes with this ionic liquid give more stability, slowing degradation and widening the voltage range. Research out of Asia and Europe shows much higher performance levels and cycle longevity, both critical for scaling up energy grids or building better electric vehicles.
Green Solvent in Chemical Processing
If you’ve ever seen the mess solvents make in a chemical plant, you’d understand the hunt for cleaner options. [EMIm][BF4] works as a green solvent, ditching the environmental baggage from many volatile organic chemicals. Factories chasing more efficient catalyst systems, recyclable solvents, and reduced air pollution often turn to this salt. In cellulose processing, for instance—breaking down tough natural fibers without creating a toxic cloud—this salt lets engineers push for sustainable processes. Academic journals keep highlighting this switch as a genuine step toward friendlier manufacturing.
CO2 Capture and Separation
A lot of people talk about carbon capture, but the engineering reality gets dicey fast. [EMIm][BF4] has carved out a niche in separating carbon dioxide from flue gases. Power plants and industrial facilities need robust methods that soak up greenhouse gases without churning out more problems. The ionic liquid absorbs CO2 better than traditional amines in some cases, which translates into less energy wasted and fewer byproducts. The research community in Germany and the US keeps running real-world tests, hoping this route eases the chokehold of industrial emissions.
Electroplating and Metal Processing
Anyone in electronics manufacturing has seen the headaches traditional plating baths bring: toxic spills, harsh acids, strict handling. [EMIm][BF4] pops up in plating precious metals like silver and gold, giving finer control over deposits while cutting waste. Reports from small-scale plating shops to industrial engineering conferences all point to this: smoother coatings, better resistance, and a safer workspace. That lines up with the growing push for RoHS and other green certifications around the globe.
Tackling the Challenges
Innovation takes more than lab breakthroughs; cost and large-scale supply must follow suit. At the moment, [EMIm][BF4] still costs more than traditional chemicals in some regions. This limits rollout to niche applications or well-funded R&D projects. If chemical companies invest in larger reactors and recycle spent liquid efficiently, cost comes down. More open data on long-term performance, health effects, and end-of-life disposal will also help reassure regulators and businesses.
Real Potential, Right Tools
From my stint in chemical industry consulting to late nights in grad school labs, I’ve seen this ionic liquid start small and grab more attention each year. Fact-driven policies, plus industry partnerships, can smooth out bumps in adoption. By focusing on these key areas—energy storage, cleaner solvents, carbon management, plating—1-Methyl-3-Ethylimidazolium Tetrafluoroborate shows how advanced chemistry fits into better, cleaner industry.
What is the chemical stability and storage condition of 1-Methyl-3-Ethylimidazolium Tetrafluoroborate?
Looking at Stability from the Lab Bench
Anyone handling chemicals day in and day out comes to know the signs of a "sensitive" compound. Some change color over a weekend, others give off odd smells at the slightest rise in room temperature. 1-Methyl-3-ethylimidazolium tetrafluoroborate does not belong in either of those categories. It stands out for its toughness among ionic liquids, showing little trouble under normal lab conditions.
This compound has a low vapor pressure. No one catches a whiff during distillation or transfer. A tightly capped container keeps its original state, even after months on the shelf. The crystal-clear liquid shows almost no sign of decomposition unless someone brings in high heat or damp air. Simple common sense—keeping it sealed, avoiding sunlight, working with clean hands and tools—prevents small problems from becoming big headaches.
The Role of Water and Air
Tetrafluoroborate salts have one quirk that matters in practice: water loves the tetrafluoroborate anion. Even a brief swap from a dry glovebox to a humid fume hood can draw enough moisture to cause slow hydrolysis. As a result, one ends up with small—but bothersome—amounts of hydrofluoric acid after a while. I remember seeing etching on glassware after careless storage.
While 1-methyl-3-ethylimidazolium cation holds together, the BF4- part does not support exposure to open air in a damp climate. Flushing headspace with dry nitrogen or argon after opening makes a difference. After sitting open over a long lunch, minor breakdown becomes visible through changes in conductivity and hints of cloudiness.
Getting Storage Right
Proper storage plays a key part in keeping this ionic liquid in top shape. A cool, dry chemical fridge does more good than leaving it out at room temperature, especially in seasons with high humidity. Suppliers pack it in amber glass to cut down on UV effects—direct sunlight triggers slow changes over months, and I have seen liquid darken from poor storage near a sunny window.
If someone needs to work with the compound daily, splitting the supply into smaller vials cuts down on contamination risk. Less air exposure, less water getting in, and less chance for change. For long-term storage—project delays can stretch—keeping it under dry inert gas works best. I often plug vials with septa for needle transfers, avoiding air contact as much as possible, inspired by good glovebox practice from my own research group.
Potential Solutions for Common Issues
No two labs get the same level of environmental control. Lab managers in humid cities may run into trouble with hydrolysis more often, while others in arid climates rarely notice it. Regular checks help. Traces of hydrolysis products show up with NMR or ion chromatography—catching them early beats discovering ruined product during a synthesis or electrochemistry run.
To stop glassware etching, use high-density polyethylene bottles instead of glass for storage, or coat sensitive areas of glassware. Store stocks in small, dry ampoules for teaching labs or places with inconsistent handling. With over ten years working around ionic liquids—including countless experiments with 1-methyl-3-ethylimidazolium tetrafluoroborate—small steps like these have paid off, keeping research moving forward and minimizing waste.
Summary
With its robust stability, 1-methyl-3-ethylimidazolium tetrafluoroborate handles everyday lab life well. Paying attention to air and moisture, keeping bottles sealed, and using the right storage techniques protects its quality for months on end. This approach supports safe, reliable results—something any scientist or educator values.
Is 1-Methyl-3-Ethylimidazolium Tetrafluoroborate hazardous or toxic?
The Real Risks in Chemistry
Most folks outside the lab probably don’t know much about 1-methyl-3-ethylimidazolium tetrafluoroborate, unless they’re neck-deep in chemistry or battery research. To give some context: it’s an ionic liquid—one of those compounds folks dream up in research circles that turns out to work where other solvents or salts won’t. It pops up in electrochemistry, catalysis, and more, mostly because of its stability and low volatility. But as with any specialty chemical, scratching below the surface to see if it brings risk or toxicity makes sense.
Understanding The Chemistry—Not Just a Label
The big question is whether working with this stuff brings danger beyond the usual chemical lab hustle. Many ionic liquids gained a reputation for being “green solvents.” People saw the lack of vapor and figured they were safer. From what I’ve seen, that turns out to be half the story. 1-methyl-3-ethylimidazolium tetrafluoroborate doesn’t boil off into the air, so you’re not breathing it day-in, day-out. But the bigger issue rarely shows up in the air—the real problem lands on your skin, or worse, in waterways after improper disposal.
Direct Contact and Exposure
Let’s be practical. Splashing a solution like this on your skin or getting it in your eyes causes irritation. Lab workers run glove-to-glove with ionic liquids because repeated exposure can lead to dermatitis. There’s some early evidence that certain imidazolium compounds have cytotoxic effects, which means cells don’t like them much once exposure gets high enough. So, while you won’t pass out from a whiff, it’s no hand sanitizer. And, crucially, chronic low-level exposure hasn’t yet been mapped—dark territory for people using these materials daily.
Spills and Waterways
Environmentally, ionic liquids offer a false sense of security. They don’t easily evaporate, so people tend to overlook drainage and disposal. The tetrafluoroborate part breaks down into boron and fluoride—elements that, in large concentrations, upset aquatic life and can be tough to snatch back once they slip into soil or water. Tossing spent liquid down the drain might seem insignificant, but over years and labs worldwide, the impact can add up.
Looking at Real-World Handling
In a busy electrochemistry setup, stricter controls become a must, not an afterthought. I’ve sat through safety seminars where ionic liquids seem glossed over since they don't stink up the lab like volatile organics. That's shortsighted. Disposable gloves aren’t just a suggestion, and proper eyewear never becomes optional. Chemical waste management plans can’t just pull old protocols off the shelf; they need to match the unique breakdown chemistry of each compound, tracking “secret green risks” that show up outside the fume hood.
What Works Better
It comes down to habits. I’ve worked with chemists who log every milliliter, manage controlled waste streams, and avoid skin splashes like the plague—almost always the same folks whose labs have spotless safety records. Institutions benefit from regular updates in safety protocols and better funding for disposal resources. Manufacturers should publish in-depth data sheets that cover not just acute hazards, but also chronic toxicity and environmental fate.
The Takeaway for Everyday Labs
Treating 1-methyl-3-ethylimidazolium tetrafluoroborate like any cutting-edge chemical: with respect. Assume new solvents could carry unseen risks, even those billed as “safe.” Personal protective equipment isn’t an option, it’s baked into daily routine. Waste needs careful handling. If the field keeps growing, ongoing research on long-term health and environmental impact is non-negotiable. In science—especially chemistry—out of sight often doesn’t mean out of harm’s reach.
What is the purity and available packaging sizes for this product?
Purity Drives Performance
Pureness isn't just a number on a label—it can shape everything about an ingredient or compound. Higher purity means fewer surprises down the line. In pharmaceuticals, tiny traces of other substances could mean big differences in quality, safety, or how a medicine acts. Food manufacturing also draws a sharp line: even a little contamination can break public trust or trigger recalls.
This goes beyond rules set by organizations like the United States Pharmacopeia or the European Pharmacopoeia. Lab teams test purity with methods like chromatography and infrared spectroscopy. These tests root out contamination, giving buyers peace of mind. Over the years, I've watched engineers and purchase managers inspect certificates of analysis like detectives hunting for clues—because a small spike in impurity sets off a row of headaches, from lost batches to compliance fines.
Packing Up for Different Needs
Packaging sizes can feel like a detail, but mismatched formats bring waste or slowdowns. Many suppliers divide their product ranges into packages like 500-gram bottles, 1-kilo tubs, and 25-kilo sacks. In research or specialty production, a small glass bottle protects expensive contents and lets users measure out just what they need. Bulk orders for factories show up in drums or flexible sacks, stacked on pallets for forklifts to clear paths through busy warehouses.
Working in labs and on loading docks, the importance of size came down to more than convenience. Opening a huge bag for a small project raised the risks—exposure, spills, moisture creeping into sensitive powders. On the other hand, too many tiny bottles meant heaps of plastic to dispose of, hurting both budgets and sustainability goals. Teams learned to look for vendors able to offer just enough choice—no one wants to pay for ten kilos when two will do, but equally, no engineer wants to halt production waiting for the next shipment to arrive.
Why Transparency Matters
People expect suppliers to be upfront with specifications: they want to see clear purity grades, packaging formats, and shelf-life right away. Over the years, I've fielded calls where missing details tripped up entire projects. A few grams of extra impurity or a box in the wrong size can derail weeks of planning, burn extra cash, and cause trust to take a hit. Reputable brands display certificates and batch numbers, trackable anytime. This focus on transparency, recommended by groups like the FDA or GFSI, protects both sides—brand reputation and user safety.
Building Better Solutions
What can companies do? Start with routinely audited labs, detailed test reports, and secure packaging. Listening to feedback leads to practical extras: resealable bags for powders that clump with moisture; bottles with built-in dispensers for easier handling. Some producers partner with recycling programs to ease the disposal of tough packaging, tackling environmental issues head-on. I’ve seen packaging innovations reduce breakage during shipping and help even small labs cut down on waste.
For anyone choosing products that matter—chemicals, food ingredients, or specialty compounds—the clearest path involves demanding information. Pick suppliers who stand by their quality, list their sizes without hedging, and give proof when needed. These small choices make a world of difference, not just on a spreadsheet but in safer products, smoother workflows, and lasting trust.
Can 1-Methyl-3-Ethylimidazolium Tetrafluoroborate be used in electrochemical or ionic liquid applications?
Real-World Experience With Ionic Liquids
Once you get into research labs or industries working on energy storage, solvent technology, or specialty chemistry, you realize ionic liquids always spark debate. Folks like me who have fiddled with batteries or tried to coax new behavior out of electrolytes end up valuing hands-on results. 1-Methyl-3-ethylimidazolium tetrafluoroborate, sometimes abbreviated as [EMIM][BF4], shows up on supply shelves for good reason. It combines a certain practicality with a hint of risk—enough to keep everyone awake and honest.
Chemistry Backed Up By Facts
Chemically, its imidazolium backbone mixed with the tetrafluoroborate gives a stable salt that's liquid at room temperature. That opens applications where regular solvents break down. Researchers hit big milestones using this compound in supercapacitors and lithium battery prototypes. Articles in the Journal of Power Sources or Electrochimica Acta point out measurable improvements in energy density and temperature range compared to classic electrolytes. I’ve seen bench prototypes hit cycle counts unheard of with old salt or carbonate blends—minimal vapor pressure, solid conductivity, less fuss over water pickup.
Why Focus on This Compound?
Here’s why its value stands out. In a real lab, water contamination ruins data. With [EMIM][BF4], the low volatility means fewer hours fighting with vacuum ovens or gloveboxes. You notice fewer fires, less stench, and less scrambling to find PPE when pouring solutions. The low hazard profile boosts morale and safety, especially for junior staff. It means projects move faster: less stoppage for cleaning up accidental spills or tracking down strange odors.
Performance and Drawbacks in Electrochemical Systems
Every compound has a tradeoff. Cycle your system too aggressively or apply too much voltage, and the imidazolium cation starts to degrade, especially at the edges of electrochemical windows. Some studies, like the ones at Argonne National Lab, showed that trace breakdown can lead to fouling of electrodes. The cost of ionic liquids also rarely escapes anyone’s attention. Even bulk orders run higher than classic organic solvents. In specialty batteries or sensitive sensors, the payoff can justify the price. Scale up to grid storage or commodity devices, and those numbers pinch.
Finding Solutions to Compatibility and Cost
Some groups are addressing the stability questions by blending [EMIM][BF4] with additives like lithium salts or bis(trifluoromethanesulfonyl)imide anions. These blends sometimes hold up better under cycling, based on data coming from DOE labs and university centers. On the cost front, recycled ionic liquids are starting to show promise. Reclaiming from spent electrolytes with dialysis or distillation, a few pilot plants have managed to drop lifecycle costs by at least 20%. Policy incentives for “green chemistry” could help, too, if leadership wants to push for ionic liquids over hazardous organic solvents.
Why This Matters
Ionic liquids like 1-methyl-3-ethylimidazolium tetrafluoroborate build a bridge between safety and performance. The ability to push battery, capacitor, or sensor development without fighting constant chemical hazards is no small thing; it leads to fresher ideas and more creative researchers. If future supply chains bring costs down and more long-term cycling data covers the stability side, this compound will keep making its mark on energy and sensor science. No magic bullets exist in chemistry, but some tools beat others by letting you focus on progress rather than firefighting avoidable problems.
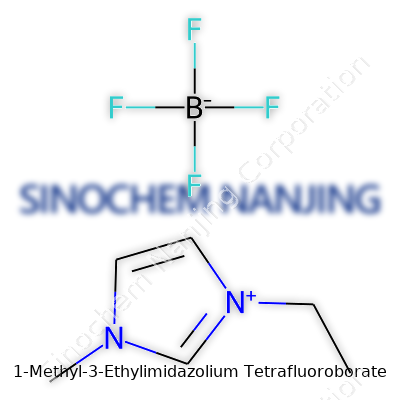
| Names | |
| Preferred IUPAC name | 1-ethyl-3-methyl-1H-imidazol-3-ium tetrafluoroborate |
| Other names |
1-Ethyl-3-methylimidazolium tetrafluoroborate EMIM BF4 1-Methyl-3-ethylimidazolium tetrafluoroborate EMI BF4 |
| Pronunciation | /ˈwʌnˈmɛθɪlˈθriːˈɛθɪlɪˌmɪd.əˈzoʊliəm ˌtɛtrəˌflʊə.rəˈbɔːreɪt/ |
| Identifiers | |
| CAS Number | 138079-76-6 |
| Beilstein Reference | 173876 |
| ChEBI | CHEBI:139482 |
| ChEMBL | CHEMBL4296592 |
| ChemSpider | 122733 |
| DrugBank | DB11104 |
| ECHA InfoCard | 100.224.293 |
| Gmelin Reference | 108215 |
| KEGG | C21504 |
| MeSH | D000068367 |
| PubChem CID | 10490493 |
| RTECS number | RNAMG1 |
| UNII | 1XW054IW10 |
| UN number | UN3464 |
| CompTox Dashboard (EPA) | DTXSID5022236 |
| Properties | |
| Chemical formula | C6H11BF4N2 |
| Molar mass | 192.03 g/mol |
| Appearance | Colourless to pale yellow liquid |
| Odor | Odorless |
| Density | 1.242 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | -0.43 |
| Vapor pressure | 0.156 mmHg at 25 °C |
| Acidity (pKa) | -0.14 |
| Basicity (pKb) | pKb = -3.8 |
| Magnetic susceptibility (χ) | -5.88 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.409 |
| Viscosity | 72 cP (25 °C) |
| Dipole moment | 5.93 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 349.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -484 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4791.7 kJ/mol |
| Pharmacology | |
| ATC code | NO ATC |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02,GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | Hazard statements: H302, H319 |
| Precautionary statements | P261, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | ''Greater than 100 °C (212 °F; 373 K)'' |
| Autoignition temperature | > 362 °C |
| Lethal dose or concentration | LD50 Oral Rat > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): >2000 mg/kg (rat, oral) |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) of 1-Methyl-3-Ethylimidazolium Tetrafluoroborate: "Not established |
| Related compounds | |
| Related compounds |
1-Butyl-3-methylimidazolium tetrafluoroborate 1-Ethyl-3-methylimidazolium tetrafluoroborate 1-Methyl-3-propylimidazolium tetrafluoroborate 1-Methyl-3-ethylimidazolium hexafluorophosphate 1-Methyl-3-ethylimidazolium chloride |

