1-Methyl-3-Ethylimidazolium Hexafluorophosphate: An Editorial Review
Historical Development
1-Methyl-3-ethylimidazolium hexafluorophosphate, often known in the lab as [EMIm][PF6], belongs to a group of chemicals called ionic liquids. The early days of ionic liquids started over a century ago when people first noticed that some salts stay liquid below the boiling point of water. It wasn’t until the 1990s, with an accelerating push for green chemistry, that these compounds caught real attention. Many researchers wanted clean reactions without relying on volatile organic solvents; environmental regulation pressure also played a part. [EMIm][PF6] came along as one of the heavy hitters here, providing a relatively stable, air-resistant, nonflammable liquid. Its reputation grew as a replacement for hazardous solvents in both laboratory and industrial settings. The fact that it didn’t burst into flames and performed reliably in batteries and other tech applications made it a favorite. Soon, its use fed a wave of curiosity that shaped the direction of modern electrochemistry and chemical engineering.
Product Overview
Calling [EMIm][PF6] just another chemical doesn’t cut it. This salt, staying liquid near room temperature, stands out because it doesn’t act like a traditional solvent. Unlike acetone or ethanol, it hardly evaporates and doesn’t have a strong smell wafting through the air. In the lab, folks handle it with care, respecting its water-reactive nature and its capacity to dissolve an impressively wide range of organic, inorganic, and metal-based compounds. Many in industry find its stability appealing—it won’t catch fire, and its decomposition point sits higher than solvents found under the kitchen sink. The go-to applications often rely on its unique mix of chemical inertness and ionic conductivity, especially in research focusing on electrochemistry, catalysis, and advanced separation processes.
Physical & Chemical Properties
[EMIm][PF6] doesn’t look particularly exciting on the bench: clear, slightly viscous, and heavier than common household liquids. Its melting point floats just under room temperature, which explains why it pours easily yet almost feels oily. No one wants an unpredictable substance in their research, and its chemical structure provides significant resistance to oxidation and heat. The combination of a bulky imidazolium cation with a robust hexafluorophosphate anion leads to a liquid with low volatility and strong solvent capabilities for both polar and nonpolar solutes. Unlike common lab solvents, you won’t catch its fumes out in the open—its vapor pressure is practically undetectable. Handling it reminds you that not every transparent liquid is water: mix it with water, and it gets cloudy fast, marking out separate phases. Its density means a small spill can trick you—always best to clean up immediately.
Technical Specifications & Labeling
Professional labs demand a clear picture when using new substances. With [EMIm][PF6], purity ranks at the top of the list, usually above 99%. Even tiny impurities can mess up experimental outcomes, especially in electrochemical cell studies. Labels from reputable suppliers spell out concentration, moisture content, recommended storage temperatures, and warnings against exposure to strong acids or bases. Storage follows suit—keep it in airtight containers, away from light and humidity. Direct contact calls for gloves and eye protection, since accidental exposure can touch off sensitization or irritation. Accurate labeling ensures every researcher working with this liquid gets the real deal, not a watered-down or contaminated variant that could crash a semester’s worth of data in a single afternoon.
Preparation Method
Synthesizing [EMIm][PF6] might sound simple, but cutting corners brings trouble. Typically, chemists start by mixing 1-methylimidazole with ethyl halides to form the precursor, 1-methyl-3-ethylimidazolium halide. This salt then meets up with potassium hexafluorophosphate in a controlled reaction, often in anhydrous acetone. The “exchange” reaction swaps out the halide for hexafluorophosphate. Anyone who’s tried this reaction knows patience pays off. If you skimp on purification steps, such as repeated washes with organic solvents and drying over molecular sieves, ionic impurities and leftover halides can stick around, spoiling conductivity or catalytic properties. Moisture creates headaches—water can quickly hydrolyze [PF6]- and degrade your product. The prepared ionic liquid needs careful bottling and quick transfer to sealed storage.
Chemical Reactions & Modifications
[EMIm][PF6] shows up as a silent partner in many lab syntheses. Its real power lies in its ability to stabilize both anions and cations during tough reactions. Organic chemists favor it for tasks like alkylation, Diels-Alder reactions, and organometallic catalysis, thanks to its low nucleophilicity and high ionic strength. While it usually keeps to itself, strong acids, strong bases, or elevated temperatures can encourage it to break down, sometimes making hydrofluoric acid—a real safety worry. Folks also take interest in tweaking the [EMIm] cation or swapping out [PF6]- for less toxic or more hydrophobic alternatives. Modifying the structure allows researchers to target specific solubility, viscosity, and conductivity profiles, tailoring the ionic liquid to applications ranging from battery electrolytes to pharmaceutical extraction fluids. Each tweak brings both promise and fresh technical hurdles.
Synonyms & Product Names
Search around, and you’ll find [EMIm][PF6] listed under names like 1-methyl-3-ethylimidazolium hexafluorophosphate, methyl-ethyl imidazolium PF6, and EMIM PF6. Some catalogs abbreviate it to [C2mim][PF6]. Academic literature sometimes even opts for its CAS number to avoid confusion. You’ll spot these names across research papers, patent filings, and material safety data sheets, reflecting the many disciplines drawing on its unusual blend of properties.
Safety & Operational Standards
There’s a tendency to call ionic liquids “green” or “benign,” but the reality is more complicated. [EMIm][PF6] doesn’t evaporate into the air, and it won’t light up without provocation, which spares researchers the hazards of flammable solvent bottles. That being said, running a reaction with this stuff takes preparation. Direct skin exposure isn’t as harmless as pure water, and it can bite back, causing skin and eye irritation after prolonged contact. Accidental spills and leaks into drains present another headache—the hexafluorophosphate anion can break down, releasing noxious byproducts like PF6 gas or hydrofluoric acid. Good practice means handling it with chemical-resistant gloves in well-ventilated hoods, sealing waste for proper collection, and avoiding heating it past its decomposition threshold. Every researcher picking up this bottle needs proper training, an up-to-date safety data sheet, and reliable gloves; skipping these basics in the pursuit of speed only invites trouble. Industrial setups now lean on containment systems, tool-specific waste protocols, and constant monitoring for leaks, especially around large-scale operations.
Application Area
The reach of [EMIm][PF6] keeps expanding. In the battery world, it works well as an electrolyte with both high ionic conductivity and remarkable thermal stability, fueling demand in energy storage for electric vehicles and grid systems. Electrochemists draw on its ability to cycle ions smoothly between electrodes, paving the way for safer, longer-lasting devices. The pharmaceutical industry values its solvation properties, using it for organic extraction and purification processes without risk of flammable vapor buildup. Anyone working on advanced catalysis will probably stumble across it at some stage, leveraging its inert qualities to boost reaction rates. Wastewater treatment, carbon capture, and even cellulose processing have started tapping into the unique abilities of ionic liquids like [EMIm][PF6], seeking environmental gains and less reliance on fossil-derived solvents. I’ve seen teams use it to dissolve stubborn biopolymers, release rare earth metals from ores, or keep precious metals stable during refinement. Each breakthrough makes clear how a single chemical can open doors across industries—sometimes at the risk of shifting hazards from the air to soil and water, which regulators and researchers alike need to keep tracking.
Research & Development
Academic and industrial labs around the world dig into [EMIm][PF6] for new ideas. Much curiosity centers on improving battery tech or replacing toxic organic solvents, but there’s also a strong push toward greener syntheses. Researchers dive into adjusting the cation or anion structure for better recyclability, lower toxicity, or greater specificity for particular reactions. Teams experiment with recycling protocols, thermal stability, and ways to lower production costs for broader commercialization. Some try to pair it with renewable energy, aiming to boost the storage capacity and longevity of solar or wind power systems. When breakthroughs happen—either in the purity of the ionic liquid, the development of entirely new ionic frameworks, or easier production—it sends ripples across chemistry conferences. It becomes clear that the conversation never stays technical for long; ethical debates, cost-benefit analyses, and environmental impact discussions always find their way into the mix.
Toxicity Research
Many early adopters of ionic liquids hoped they’d solve the toxicity problems of classic solvents. That optimism never quite matched reality. Studies on [EMIm][PF6] indicate it doesn’t evaporate, but it doesn’t just disappear—leaked material and accidental spills can persist in soil and water. Some toxicity assessments show moderate risks for aquatic environments and a need to avoid careless disposal. Hydrolysis in water or under acidic conditions can form corrosive byproducts, ramping up the hazard level. Researchers continue digging into chronic effects, aiming to understand exactly how long-term exposure influences lab technicians, industrial workers, and ecosystems. Many call for stricter guidelines not just for personal protective equipment and storage but also for end-of-life treatment and environmental cleanup. As new ionic liquids reach the market, the same rigorous evaluation process proves essential to avoid swapping one pollutant for another.
Future Prospects
The future of [EMIm][PF6] and ionic liquids overall remains tied to both their promise and their drawbacks. There’s excitement around their potential to revolutionize everything from clean energy systems to rare metal recovery, yet the real challenge hits at scale—cost, lifecycle, and actual environmental impact. Researchers keep pursuing less hazardous alternatives and better recycling schemes, ensuring these liquids don’t just shift the pollution problem out of sight. Faster, greener syntheses, tailored physical properties, and hybrid ionic liquids all promise to unlock new applications in areas like fuel cells, chemical separations, and smart materials. Future success relies on practical training, solid regulatory oversight, and a willingness to weigh both the benefits and hidden costs. As with many breakthroughs, the genuine value comes not just from what’s possible in the lab but from what proves safe, affordable, and truly sustainable outside it.
What are the primary applications of 1-Methyl-3-Ethylimidazolium Hexafluorophosphate?
Changing How Chemists Approach Solvents
For decades, traditional solvents ruled the lab. They’re volatile, they pollute, and we keep searching for cleaner ways to separate and analyze chemicals. That’s where 1-Methyl-3-Ethylimidazolium Hexafluorophosphate, often abbreviated as [EMIM][PF6], steps in and changes the way we look at liquid solutions in chemistry. As an ionic liquid, this compound brings some major advantages — it barely evaporates at room temperature and stands up to chemical punishment.
In my experience, replacing harsh organic solvents with [EMIM][PF6] made the job safer and less smelly, and it slashed the risk of fire. Labs using ionic liquids suddenly started recycling more of their materials, which meant less waste and lower costs. The move toward these green solvents supports ethical research and lines up with safety protocols that have grown stricter in recent years.
Electrochemistry and Battery Research
Battery research leans on electrolytes tough enough to survive high currents and potential spikes. Traditional liquid electrolytes break down, short out, or dry up over time. [EMIM][PF6] gives batteries and supercapacitors a way to push energy density higher with fewer risks to performance. Researchers point toward lower volatility and high thermal stability — both key for safer energy storage. The compound’s ability to dissolve salts, especially lithium salts, gives scientists a toolkit for building advanced lithium-ion and sodium-ion cells that live longer and weigh less.
I’ve watched university teams experiment with [EMIM][PF6] in battery prototypes that could handle abuse and remain steady. This flexibility pushes new ideas forward in electric vehicles and grid storage solutions. The big carmakers and energy companies take notice when a replacement electrolyte doesn’t burst into flames or evaporate after long use.
Separating and Recycling Chemicals
Anyone who has worked in process industries knows the challenge: getting high-purity materials often means multiple steps, lots of heating, and a parade of solvents. [EMIM][PF6] steps up in liquid-liquid extractions and gas separations, making life easier for chemical engineers. Take gas absorption — industries use it to scrub harmful gases from exhaust. Ionic liquids like [EMIM][PF6] trap carbon dioxide more efficiently than many amine solutions. With the world watching carbon emissions, every bit of progress helps.
This compound appeared in solvent extraction setups, especially for rare earth elements — the ones crucial for smartphones and renewable energy tech. Instead of nasty acids, teams lean on ionic liquids, which can be recycled and reused across cycles. Having seen plants try to hit environmental targets, the switch has saved headaches and regulatory fines.
Catalysis and Green Synthesis
Chemical manufacturing depends on complex reactions, some requiring conditions tough on traditional solvents. [EMIM][PF6] supports catalytic processes by dissolving both organic and inorganic compounds. Reports show higher yields and cleaner products, especially for pharmaceuticals and specialty chemicals. From my lab time, using ionic liquids sped up tricky organic reactions and sidestepped by-products that would have required extensive cleanup.
Adoption isn’t perfect. The price tag of [EMIM][PF6] stands higher than bulk solvents. Cleanup isn’t a total breeze, either. But the shift to greener, safer processes keeps gaining ground. Making climate-friendly choices in a research or industrial setting matters, and ionic liquids like [EMIM][PF6] earn their place by ticking important boxes for safety, reusability, and performance.
Possible Solutions for Broader Adoption
Scaling up production often brings costs down. Researchers continue to search for easier synthesis methods and more ways to recycle these ionic liquids after use. Partnerships between industry and academic labs often open doors to better handling, waste management, and recovery. If public policy keeps nudging industry toward greener materials, expect to see more [EMIM][PF6] in everything from labs to big factories.
What is the chemical stability and storage requirement for this compound?
Getting Real About Stability
Many of us who handle chemicals know that a lot hangs on how steady a compound remains before it ever reaches the lab bench or assembly line. In chemistry classes, you only hear about breaking bonds under heat or light, but the real test starts when a compound sits on a shelf for months, exposed to air, fluctuating temperatures, and the hundreds of small handling mistakes humans inevitably make. Stability isn’t static. Sometimes moisture from humid weather sneaks past a screw cap and starts slowly wrecking everything inside. Peroxides form, odors develop, results shift — and nobody wants to discover this halfway through an experiment or production run.
Certain chemicals hold up better than others, and that comes from their underlying structure. For instance, sodium chloride, or table salt, shrugs off most dry and wet conditions, but put a bottle of sodium metal near an open window and any stray drop of water could spell disaster. The trouble always comes from reactivity: with air, water, acids, or even light. Take silver nitrate. It’s colorless at first, but after too much exposure to daylight, the solution turns brown or black as it breaks down into silver metal. The same lesson pops up with vitamins, certain pharmaceuticals, and biological reagents—waste kicks in fast if the climate and container aren’t right.
Storage: Practical Wisdom from Years on the Job
Most chemical labels tell folks to “store in a cool, dry place,” but the phrase barely scratches the surface. My first job in a research lab drove this home after a refrigerator power outage ruined two years’ worth of expensive enzymes. Water trickled down unnoticed into the freezer, ice melted and refroze, and the enzymes—unstable by nature—lost their punch. Some compounds want total darkness (think riboflavin or retinol), while others demand an inert gas blanket, not just a tight screw cap. Labs with the best reputations invest in humidity sensors, explosion-proof fridges, and regular drills to remind people what’s actually inside those amber bottles.
Even small details matter. Solid powders spill less often than liquids, but a streak of carelessness with a hydroscopic salt guarantees clumping and time wasted trying to dry what was once easy to measure. In some cases, folks get creative: storing containers inside desiccators, wrapping reagents in foil, tucking acid-sensitive chemicals away from metal shelves. Lapses in storage rarely cause problems instantly, but the damage builds up. If safety data sheets sound dry, it’s because the stakes are rarely as obvious as a laboratory fire. Quality slips, reproducibility drops, and everyone downstream feels it—from researchers to end users who rely on those compounds to work as advertised.
Aiming for Solutions That Actually Work
Experience teaches that checklists and reminders go further than trust alone. In shared workplaces, a clear log beside every dangerous or shelf-sensitive compound makes a difference. Chemical companies could start including expiry dates that really mean something for each batch. Getting people to actually read and follow storage precautions matters just as much as buying the right freezer.
No one wins when a lab’s supply of an expensive reagent fails from something as pointless as a sweaty storage room or a cracked rubber stopper. Companies serious about quality control set up regular audits: not just at delivery, but before use. For professionals on the front line, swapping stories and sharing near-misses sometimes saves more lives and money than a dusty manual. Training that dives into why certain rules exist sticks with folks longer than a sign nagging “Keep refrigerated.”
For anyone relying on chemicals—whether in research, healthcare, or manufacturing—treating stability and storage like part of the day’s real work, not afterthoughts, pushes outcomes in the right direction. That’s knowledge drawn not just from the books but from the decades of small successes and failures that come from handling real stuff in a real world.
Is 1-Methyl-3-Ethylimidazolium Hexafluorophosphate toxic or hazardous to handle?
Handling Chemicals Beyond the Lab Bench
Ask anyone who has worked long hours in a research lab or at an industrial site, and stories will come out: cracked gloves, mystery spills, chemical odor sticking to clothes. Certain chemicals call for extra attention, and 1-methyl-3-ethylimidazolium hexafluorophosphate (EMIM-PF6) fits that category. A mouthful of a name, but the real challenge comes with handling and safety, not pronunciation.
What's Inside This Ionic Liquid?
EMIM-PF6 belongs to the family of ionic liquids. These compounds usually stay liquid at room temperature, making them handy for chemists who need non-volatile solvents. That may sound reassuring, but the smooth surface hides a risky core.The structure includes a fluorinated anion: hexafluorophosphate (PF6-). It can break apart and release PF5 or hydrogen fluoride if heated, handled with water, or mixed with strong acid. Anyone who’s smelled a faint, sharp sour note near these chemicals will recognize trouble: hydrogen fluoride can burn through organics, concrete, and bone.
Skin, Eyes, and Lungs: Points of Contact
If you’ve ever splashed an unknown liquid on unprotected skin, you know the initial worry. From the data, EMIM-PF6 can cause significant irritation. Safety sheets and studies both point the same direction: redness, pain, sometimes blisters. Eyes are at an even higher risk, with exposure leading to burns or long-term damage. It vaporizes less than traditional solvents, but that doesn't eliminate hazard. Some people underestimate vapor risk with low-volatility chemicals, but if EMIM-PF6 gets heated or reacts, you might breathe in dust or vapors from decomposition. More than a few chemists have let their guard down, only to end up coughing or with a burning sensation in their throat after a fume hood mishap.
Decomposition: Where the Risks Compound
Simple handling can turn hazardous—water spills, chemical residues, or elevated temperatures start chemical breakdown. Inhaled hydrogen fluoride doesn't just irritate—it attacks lung tissue. Chronic exposure to breakdown products raises risks of tooth decay, bone issues, and kidney problems. This isn’t theory; safety organizations have logged real-world accidents in university and factory settings.
Environmental and Waste Issues
Labs often generate waste streams containing EMIM-PF6. Pouring it down the drain or dumping in regular trash can pose risks to wastewater staff and downstream wildlife. Some ionic liquids break down slowly, sticking around in the natural environment. As someone who’s sorted chemical waste containers at the end of a long day, it’s easy to feel tempted to skip safety steps, but environmental consequences don’t stay in the lab—they become a community problem.
Safer Handling and Alternatives
Gloves, goggles, and working behind a fume hood are non-negotiable with EMIM-PF6. Cursory glances at labels don’t cut it—it’s all about preparation and having neutralizing agents on hand, such as calcium gluconate gel for fluoride exposure. Training every new face at the bench, discussing the risks openly, and keeping up with Material Safety Data Sheets can prevent accidents.Some researchers choose alternatives. Newer ionic liquids and water-based solvents get developed for the same reasons: reducing toxicity and environmental persistence. Cost and performance matter, but many groups now weigh hazards alongside effectiveness.
Why This Matters
Professional pride grows from taking care of each other and the environment. Accidents don’t care about experience level—mistakes happen to veteran chemists and newcomers alike. Knowing the facts and respecting the risks with EMIM-PF6 means the next generation of chemists inherits some wisdom and a safer working space.
How should this product be disposed of after use?
Everyday Decisions After Use
Many people finish using a product and drop it straight in the trash, thinking their part is done. The story hardly ends there. What happens next might impact a neighbor's health, a child’s creek, or even your tap water. I’ve tossed batteries before, only to learn what leaches out in a landfill can stick around for decades. Trash doesn’t just vanish. Choices made at home echo far from the kitchen bin.
Sorting Fact from Habit
Too many of us stare at packaging and guess. Food wraps, shampoo bottles, worn-out sneakers—each brings a little confusion about proper disposal. Some products shout, “Recycle me!” with bold arrows. Others hide tiny codes or offer no clue. Once I learned what the numbers tucked under a plastic cup actually mean, recycling felt less like shooting in the dark. For example, plastics #1 and #2 see high curbside recycling rates in many U.S. cities, but #6 and #7 rarely make it through, ending up in landfill. Labels matter, but clear info would make a bigger difference.
Environmental Impact
Tossing away electronics creates a chain of headaches. E-waste dumps can leak lead into soils and water, hurting kids who play nearby. Paint rinsed down the sink clogs treatment plants and sends toxins toward rivers and lakes. Studies from the EPA show Americans send over 120 million phones to landfills every year. That’s a mountain of metals and plastics with nowhere safe to rest. Having watched rivers in my hometown turn murky after heavy rain, I trace some of that pollution back to discarded household goods. Nothing just disappears; everything heads somewhere.
Solutions Within Reach
Better product labeling stands out as a fix. Clarity works—no one benefits from recycling symbols that require a decoder ring. Local governments could require easier instructions, with text large enough to spot. Mobile apps and barcode scanners help bridge the knowledge gap. I tried an app that tells me where each item belongs, cutting my curbside confusion to almost zero. Store take-back programs also help, especially for electronics and batteries. Retailers already run battery collection in many places; expanding these efforts keeps fewer toxics in the community trash stream.
Some towns reward households for sorting waste properly. I joined a pilot program that tagged bins for accuracy, and neighbors started swapping tips and competing to see who could get the best marks. Incentives can shift habits faster than lectures or fines. Companies who design single-use items might also change the game by switching to genuinely compostable or widely recyclable materials. I've seen compostable cups turned into garden mulch at a local facility—it only happened because the cup’s labeling lined up with the region’s compost rules.
The Personal Stakes
I learned early that out-of-sight, out-of-mind leads to problems for someone else. Asking “How should this product be disposed of?” isn’t about guilt. It answers, “How do I want to leave the place I live?” Every careful step—reading a label, checking with the city website, dropping off at a collection event—chips away at bigger public health costs. What goes in the bin might shape the air, water, and soil for years ahead.
What are the available purity grades and packaging sizes for this product?
Why Purity Matters
Standing in the warehouse surrounded by sacks and drums, I’ve often noticed just how wide the range of chemical purity can be. Choice doesn’t come down to preference, but to safety, quality, and compliance. In the lab, one small impurity can throw off results or even ruin equipment. In manufacturing, impure materials cost companies money and lead to frustrating troubleshooting down the line.
The decision on purity always begins with specific project demands. Perfume development in a cosmetic lab usually calls for reagent-grade materials, which must meet tight standards. These grades generally run between 99% and 99.9% pure, ensuring that trace contaminants are either very low or documented. On the other hand, most routine cleaning agents or industrial processes can use technical-grade products with purities in the 90–98% range. Lesser purity brings down the price, but contamination risks go up.
For my work in food processing, using food-grade chemicals isn’t a luxury — it’s often a legal mandate. These follow government regulations on things like heavy metals, solvents, and handling. Skipping that step opens the door to recalls or consumer illness.
How Packaging Influences Results
After looking at the shelf in a university storeroom, it’s obvious that packaging size shapes workflow. Small-scale research or quality testing involves bottles or ampules starting from 10 grams or 100 milliliters. These are manageable, reduce waste, and hold up well on the shelf. They almost always feature tamper-evident seals. On the other end, commercial operations want drums, bags, or carboys — 25 kilograms, 50 kilograms, even 200-liter barrels. Freight costs drop when buying in bulk, yet handling safety factors rise.
During a project for water treatment, our team found that choosing larger packaging cut costs, but introduced new risks. Improper storage, humidity exposure, and improper resealing lead to caked material and safety hassles. Keeping bulk material fresh often means investing in dedicated storage rooms or dry boxes, which might not be worth it for small-batch work.
Evaluating the Choices
Not all suppliers list every possible option, but the common grades on the market include technical, laboratory, analytical (ACS or AR), food, and pharmaceutical. Analytical-grade products fetch higher prices but remove the risk of contamination that can derail research or legal approvals. Large manufacturers, though, might accept technical grade in some settings, banking on robust filtration or downstream quality control to remove contaminants.
Suppliers will ship as little as a vial or as much as a pallet. For most of my smaller customers, storage space and frequency of use matter more than price per kilogram. Paying a premium for small sizes means less hassle with handling and inventory. For government contracts or multinational factories, larger drum or tote options win out due to cost savings and streamlined logistics.
One challenge that comes up is the “one size fits all” mentality. Labs adopting packaging that’s too large often face spoilage after breaking a seal. Similarly, small bottles cost more in the long run for any consistent process. Discussing specific use cases with vendors, or even touring a supplier’s facility, can reveal packaging or purity options not listed in standard catalogues.
Navigating the Marketplace
Finding the best fit always means considering project scale, regulatory burden, and customer needs. Highly regulated industries demand both grade documentation and lot traceability. Someone working in jewelry cleaning cares about brightness, not about testing each drum for heavy metals. By focusing on the required outcome, teams make better choices and save time on reordering or waste disposal.
Asking tough questions about how a product was handled, where it came from, and which certifications back it up can reveal weak points or new opportunities for savings. Through years of trial and error, it’s clear that purity and packaging aren’t just specs — they impact quality, cost, and peace of mind.
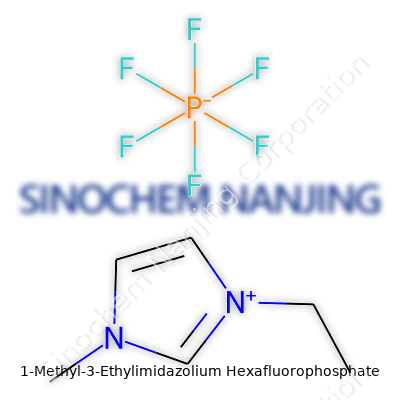
| Names | |
| Preferred IUPAC name | 1-ethyl-3-methyl-1H-imidazol-3-ium hexafluorophosphate |
| Other names |
1-Methyl-3-ethylimidazolium hexafluorophosphate EMIM PF6 1-Ethyl-3-methylimidazolium hexafluorophosphate EMIPF6 EMIM+ PF6– |
| Pronunciation | /waɪ ˈmɛθ.əl θri ˌɛθ.ɪl ɪˌmɪd.əˈzoʊ.li.əm ˌhɛk.səˌflʊə.roʊ.fɒsˈfeɪt/ |
| Identifiers | |
| CAS Number | 331717-63-6 |
| Beilstein Reference | 3921590 |
| ChEBI | CHEBI:39298 |
| ChEMBL | CHEMBL1231850 |
| ChemSpider | 290924 |
| DrugBank | DB11172 |
| ECHA InfoCard | ECHA InfoCard: 100.131.654 |
| EC Number | EC 700-390-8 |
| Gmelin Reference | Gmellin Reference: **104272** |
| KEGG | C18196739 |
| MeSH | D000068449 |
| PubChem CID | 2734161 |
| RTECS number | VP2300000 |
| UNII | 2Z7M8S1MWP |
| UN number | UN3488 |
| Properties | |
| Chemical formula | C6H11N2PF6 |
| Molar mass | 284.19 g/mol |
| Appearance | White to off-white solid |
| Odor | odorless |
| Density | 1.36 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.59 |
| Vapor pressure | 0.0000163 mmHg at 25 °C |
| Acidity (pKa) | -0.6 |
| Basicity (pKb) | pKb = 9.4 |
| Magnetic susceptibility (χ) | -82.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.415 |
| Viscosity | 87 cP (25 °C) |
| Dipole moment | 4.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | **570.8 J·mol⁻¹·K⁻¹** |
| Std enthalpy of formation (ΔfH⦵298) | -1472.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5048.6 kJ/mol |
| Pharmacology | |
| ATC code | No ATC code |
| Hazards | |
| Main hazards | Harmful if swallowed, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H226, H302, H315, H319, H373, H400 |
| Precautionary statements | P261, P264, P271, P273, P280, P301+P312, P305+P351+P338, P337+P313, P330, P391, P501 |
| NFPA 704 (fire diamond) | 1-1-0-W |
| Flash point | 184 °C |
| Lethal dose or concentration | LD50 (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose), Oral, Rat: >2000 mg/kg |
| NIOSH | MH6515000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.05 ppm |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
1-Butyl-3-methylimidazolium hexafluorophosphate 1-Ethyl-3-methylimidazolium hexafluorophosphate 1-Hexyl-3-methylimidazolium hexafluorophosphate 1-Methyl-3-propylimidazolium hexafluorophosphate |

