An Editorial Look at 1-Methyl-3-Ethylimidazolium Bromide: Beyond the Beaker
Historical Development: Roots in Discovery and Progress
There aren’t many corners of chemistry where you find such a blend of old-fashioned curiosity and cutting-edge solutions as in the story behind 1-Methyl-3-Ethylimidazolium Bromide. Chemists first started turning their attention toward ionic liquids back in the late twentieth century, especially as traditional solvents began to show more downsides than anyone would like to admit. Long before sustainability became a buzzword, researchers wanted alternatives to volatile, polluting substances. The imidazolium family hit the lab bench with a dramatic flair, taking cues from foundational work on molten salts and ionic compounds from the previous era. After pioneers realized these substances could stay liquid far below the temperatures associated with standard salts, focus shifted toward tweaking the alkyl groups bound to the imidazole ring. Adjustments led to hundreds of new salts, all with unique profiles, and among them, 1-Methyl-3-Ethylimidazolium Bromide carved out its own space due to attractive handling properties, decent stability, and versatile uses.
Product Overview: What Sets It Apart
There’s a simplicity and robustness that comes with 1-Methyl-3-Ethylimidazolium Bromide. The structure, composed of a methyl and an ethyl group flanking the imidazole ring, plus a bromide anion, looks unassuming on paper. In practice, it proves a practical choice for scientists who can’t always waste time with substances that break down or mix unpredictably. Its reliable liquid form under normal room temperatures scores points where many powders or viscous gels fall short. After working with it in several processes, it becomes clear that this compound bridges the gap between the favorable ionic character needed for electrochemical work and the chemical resilience researchers demand when pushing boundaries.
Physical and Chemical Properties: The Why Behind its Versatility
Describing this ionic liquid gets easier once you’ve held a sample or monitored its behavior. You quickly see why researchers rely on it for so many purposes. 1-Methyl-3-Ethylimidazolium Bromide appears as a clear, nearly colorless viscous liquid—a subtlety that hides real power. It boasts a high thermal stability with melting points typically hovering near room temperature, making it workable throughout a range of climates and lab setups. Its high ionic conductivity and low volatility are no accident; these features mean less risk of inhalation and lower loss to evaporation, issues every researcher dreads. Throw in its fairly wide electrochemical window, and you’ve got a strong candidate for battery research, catalysis, and organic synthesis. When I handled materials with noxious odors or unsafe vapor pressures, I always reached for alternatives like this one—less risk and more reliability go a long way.
Technical Specifications & Labeling: Clarity for Users
Every chemical bottle should come with labeling that means something beyond regulatory feel-good statements, and this one doesn’t disappoint. 1-Methyl-3-Ethylimidazolium Bromide must arrive to the bench with full disclosure: its identification as an ionic liquid, its purity (often 98% or above for most research-grade applications), and its CAS number. The detailed molecular weight, melting and boiling points, and bromide content cut down on errors in analytical runs. This level of transparency isn’t about ticking boxes; it’s about ensuring researchers work with what they think they are getting. I’ve seen how impurities or vague specs derail projects and exposures, so this degree of openness reassures anyone relying on consistent properties during repeated experiments.
Preparation Method: Science in the Making
Making 1-Methyl-3-Ethylimidazolium Bromide tends to follow a pretty direct route. Most syntheses start by quaternizing 1-ethylimidazole with methyl bromide, resulting in the target cation paired with bromide. It sounds simple in theory, but controlling the atmosphere, handling reagents with care, and managing temperature swings take skill and experience. What stands out isn’t the novelty of this process, but the predictability—a benefit for anyone scaling up without introducing surprises. During my time in graduate school, running reactions like this drilled in the importance of purification steps: repeated crystallization or extraction helps remove side products, and the practical know-how passed from lab veterans to newcomers shapes both safety and quality.
Chemical Reactions and Modifications: A Foundation for Innovation
The real value of 1-Methyl-3-Ethylimidazolium Bromide turns up during downstream chemistry. It doesn’t just play the role of solvent; it gets involved in phase-transfer catalysis, organometallic reactions, and even serves as a template for polymerization. The imidazolium core stays chemically robust, and swapping out the bromide for other anions opens up new properties: hydrophobic versus hydrophilic behavior, for instance. I watched researchers fine-tune the anion to match specific reaction conditions, turning a single substance into a whole catalogue of related materials. This adaptability allows chemists to build on a known foundation, speeding up experimental cycles and leaning into targeted applications.
Synonyms and Product Names: Avoiding Confusion
In chemical literature, names can confuse even seasoned professionals. 1-Methyl-3-Ethylimidazolium Bromide regularly appears under abbreviations like [EMIM]Br or 1-ethyl-3-methylimidazolium bromide. Each label nods to the simple logic of its structure; EMIM reflects ethyl and methyl groups attached to imidazole. Regulators and suppliers standardize labeling to cut down on miscommunications. Sorting through samples and papers with inconsistent names drove home the value of sticking to authorized nomenclature—minor differences in language trip up searches, lead to accidental mixing of chemicals, and complicate data analysis. In a world where every mishap costs time, adhering to recognizable names saves more than just paperwork.
Safety and Operational Standards: Protecting People and Process
Too often, discussion of chemical safety turns into a recitation of standard phrases and pictograms. Real protection comes from building habits anchored in science and respect for every compound. 1-Methyl-3-Ethylimidazolium Bromide stands out for its low volatility, minimizing inhalation risks, but that doesn’t excuse lapses in personal protective equipment, ventilation, or waste management. Overexposure through skin contact or accidental ingestion represents a bigger danger than many realize, especially when handling ionic liquids over long shifts. Training staff to understand the potential for irritation or harm, along with upfront discussions of disposal methods, reinforces a culture where safety isn’t an afterthought. In my experience, labs that treat even “milder” chemicals with deliberate caution foster an atmosphere where students and professionals feel empowered to speak up and watch out for each other.
Application Area: Pushing the Boundaries of Science
Whenever academia or industry rolls out advances in electrochemistry, catalysis, or even green energy, odds are good that 1-Methyl-3-Ethylimidazolium Bromide helped smooth the path. Its use in batteries, especially as a non-flammable, conductive medium, keeps showing up in the pursuit of safer, longer-lasting power sources. The compound also holds sway in organic synthesis as both a solvent and a catalyst, steering reactions down more selective pathways and reducing side waste. Polymers and plastics, pharmaceuticals, and even waste treatment draw on its stability and tuneable character. On a personal note, working alongside groups blending this ionic liquid into their research, I regularly saw setbacks turn into progress as teams swapped in 1-Methyl-3-Ethylimidazolium Bromide and suddenly gained tighter control over their systems. It’s no small feat to help drive fields as different as clean energy storage and drug manufacturing at the same time.
Research and Development: Where Curiosity Meets Commercial Push
Every new wave of material science seems to return to ionic liquids for answers, and 1-Methyl-3-Ethylimidazolium Bromide sits firmly within researchers’ toolkits. R&D teams don’t just look at its basic properties—they dissect how adjustments to its structure open up new doors. Current work often centers around designing greener, less toxic versions or incorporating bio-based starting materials. Responsive systems that shift their behavior in the presence of other chemicals, or that transport ions and molecules more efficiently, keep drawing chemists back to this versatile core. Meetings with startups and tech hubs reveal ongoing projects where traditional solvents lose ground in the face of improved safety and efficiency. Investment in better characterization and upscaling methods promises a future where lab-scale discoveries actually reach commercial applications without compromise.
Toxicity Research: Facing Reality with Data, Not Hype
No discussion feels complete without confronting the health and environmental questions facing any new material. Data on the toxicity of 1-Methyl-3-Ethylimidazolium Bromide continues to grow, reflecting both hope and caution. While many tests show low acute toxicity compared to conventional solvents, chronic effects remain under study, and questions about bioaccumulation or breakdown products still loom. Direct skin and eye contact with even low-volatility liquids won’t always end in a hospital visit, but irritation and long-term exposure risks shouldn’t be minimized. Real progress comes from transparency—sharing both negative and positive findings in open journals, conducting third-party audits, and keeping environmental release to a minimum. Past mistakes with “harmless” industrial solvents taught the importance of ongoing vigilance, not just marketing a chemical as inherently green without thorough, independent study.
Future Prospects: More than Just Hope
The future for 1-Methyl-3-Ethylimidazolium Bromide doesn’t rest on hype or isolated breakthroughs, but rather on persistent, careful development. Researchers look for ways to push beyond current limits in smart materials, recycling, and safe, energy-efficient chemical processes. Demand for renewable energy storage—where safety, longevity, and environmental responsibility matter—aligns squarely with the substance’s potential. Work on pairing it with renewable feedstocks or biodegradable ionic liquid systems opens up another avenue for green chemistry. At every conference and publication I see, the conversation turns toward better documentation of life cycle impacts, improved understanding of its ecological destiny, and broader adoption in established industries. Responsibility for this future lands on everyone in the field—chemists, engineers, decision-makers—to use both their expertise and hard-won lessons from past chemical innovations.
What is 1-Methyl-3-Ethylimidazolium Bromide used for?
Everyday Chemistry, Modern Needs
If you spend any time in a chemistry lab, ionic liquids show up often. One compound, 1-Methyl-3-Ethylimidazolium Bromide, comes up in research and industry alike. Its long name hides some practical roles. This salt holds a unique set of properties: it remains liquid at room temperature, it dissolves a wide range of compounds, and it holds a steady chemical structure even in challenging environments. These traits open doors in fields like green chemistry, energy, and even pharmaceuticals.
Making Solvents Safer
Solvents used to mean harsh chemicals and stinging fumes. In the shift to cleaner methods, 1-Methyl-3-Ethylimidazolium Bromide appeals to chemists who want safer options. Its ability to replace volatile organic solvents means less harmful emissions in several processes. In tasks like cellulose processing and dye removal, labs choose this salt to cut down on toxic waste. I’ve seen friends in research move to these alternatives, chasing a smaller environmental footprint without giving up performance.
Boosting Catalysis and Extraction
Many reactions speed up in the presence of the right catalyst. Some catalysts struggle to work efficiently in water or oil. Here’s where 1-Methyl-3-Ethylimidazolium Bromide steps in. Chemists use it to dissolve both organic and inorganic compounds, mixing things that typically don’t play well together. It serves well in extraction, letting researchers pull out target compounds—natural products, metal ions—with greater selectivity and less fuss. It’s used for separating rare earth metals and recycling electronics waste, helping stretch out dwindling resources.
Energy Storage and Battery Research
New approaches to batteries often depend on finding better electrolytes. Ionic liquids like this bromide salt provide stable, non-volatile options for carrying ions between battery electrodes. Engineers add it to lithium-ion or flow batteries, chasing longer lifespans and improved safety. The compound’s broad thermal window comes into play for batteries that need to survive stress, cold, or heat. Its use in capacitors and solar cells shows how chemistry supports renewable energy, even if you rarely hear about these behind-the-scenes advances.
Improving Pharmaceutical Processes
Drug development needs precise tools. From dissolving stubborn drug molecules to helping purify delicate peptides, 1-Methyl-3-Ethylimidazolium Bromide makes pharmaceutical synthesis smoother. Even in formulation, its unique ability to mix different chemical worlds helps ensure better bioavailability. That means medication works as intended, getting where it needs to go in the body.
Challenges and Room for Growth
Just because something seems cleaner doesn’t mean it always is. Ionic liquids sometimes hang around in the environment longer than expected, and toxicity varies. It’s not perfect—scientists need more data on long-term safety and impact. Right now, the push is on for recycling these salts and designing versions that break down completely and safely. Every time the chemistry world comes up with a solution, another puzzle turns up.
Creating Opportunity From Innovation
Innovation rarely comes from a single breakthrough. Usually, it’s subtle changes in practice that shape safer industries and greener technology. 1-Methyl-3-Ethylimidazolium Bromide matters because it marks progress away from outdated, polluting methods. It shows how science adapts—using creativity and good data—toward something more sustainable and efficient.
What is the chemical structure of 1-Methyl-3-Ethylimidazolium Bromide?
Understanding the Makeup
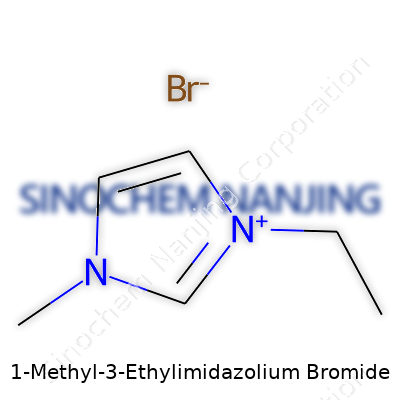
Science runs on clear details, not guesswork. The chemical structure of 1-Methyl-3-Ethylimidazolium Bromide tells a straightforward story: a ring made of three carbon and two nitrogen atoms, called an imidazole ring. A methyl group attaches to one nitrogen, while an ethyl group joins the other nitrogen. The positive charge sticks with the ring—making it a cation. Partnered with that sits bromide, a negative counterion. Chemists sketch it as C6H11N2Br. The molecule’s shape matters—not just for textbook drawings, but for how it mixes into solutions and interacts with everything around it.
Why This Structure Matters for Real Applications
Ionic liquids, like 1-Methyl-3-Ethylimidazolium Bromide, don’t act like everyday salt. They melt below 100°C, sometimes even at room temperature. You won’t find them in your kitchen, but labs and factories count on them. The imidazolium cation, made stable by the arrangement of methyl and ethyl groups, creates a liquid that resists evaporation. It also stands up well to heat. The bromide anion sits there and keeps the whole thing electrically neutral. Chemistry researchers see a lot of potential in these salt-like liquids.
Environmental and Industrial Impact
Most folks want to hear that a chemical can do something good, or at least avoid harm. Traditional solvents—think of acetone or toluene—release plenty of fumes. Ionic liquids can step in as a greener choice in many settings, because low volatility means less going up in the air. For example, 1-Methyl-3-Ethylimidazolium Bromide carries out many types of reactions without sending strong smells into the room. Workers breathe easier, the air stays cleaner, and explosions become less likely.
In my own experience, swapping to ionic liquids like this one let me set up reactions indoors without breaking sweat over fire risk. The low vapor pressure brings peace of mind, especially when someone turns up the heat.
Challenges Scientists Face
Nobody gets a free ride, though. The imidazolium part, with its easy-to-tune side groups, means millions of possible ionic liquids are out there. Not all of them break down easily in the environment. Some stick around in water or soil. Studies in the past decade have shown that certain cations and anions hurt aquatic life if they get out of the lab. The bromide in this case, while less toxic than some, can still pose problems if handled carelessly.
Researchers stay busy testing how these ionic liquids behave outside flasks and beakers. Biodegradability matters—some labs have turned attention to swapping bromide for greener anions. Imidazolium salts also come under scrutiny, especially when released into wastewater.
Improving Safety and Sustainability
Better choices start with clear policies and strong training. Labs with solid disposal plans keep ionic liquids from escaping down the drain. Most teams now check the toxicity of both cation and anion before scaling up production. On the industry side, engineers can lean on closed-loop systems to recover and reuse these chemicals, instead of treating them like single-use solvents.
Making greener versions, perhaps swapping bromide for biodegradable ions, already shapes the research agenda. Groups like the European Chemicals Agency recommend pre-market testing to avoid repeating mistakes from the past. Facts still drive the work, but respect for both safety and sustainability grows year by year.
How should 1-Methyl-3-Ethylimidazolium Bromide be stored?
Why Chemical Storage Matters
Stuffing a new bottle of 1-Methyl-3-Ethylimidazolium Bromide into a random cabinet never feels right. I learned early on that sloppy storage can wreck both the chemical and a project budget. Even common lab salts turn risky if ignored, but ionic liquids bring extra quirks. That’s why experience teaches that storage isn’t just a box to tick; it’s a real risk-control measure.
Factors That Should Guide Storage
Let’s get direct—1-Methyl-3-Ethylimidazolium Bromide absorbs water easily. Anyone who’s handled these imidazolium-based solvents knows a sticky clump means moisture got in. Contamination affects purity and reactions will run off-course. You don’t want to troubleshoot a failed project only to find the culprit was improper storage.
Keep Moisture Out
Every time I see a new grad open a reagent with sweaty gloves or leave lids ajar, I cringe. This compound drags water vapor right out of the air. Dry storage turns into a necessity, not a luxury. Tightly sealed bottles with a PTFE-lined cap get the job done. If you can open a nitrogen or argon atmosphere cabinet for long-term batches, protective atmosphere storage pulls ahead for peace of mind.
Temperature Isn’t an Afterthought
Most ionic liquids don’t burn or decompose right away at summer heat, but the story changes with time. Heat accelerates breakdown and fosters impurities. My habit is simple: store at room temperature, somewhere the sun doesn’t reach. Forgetting vials on a windowsill led to a sticky, discolored mess—the kind you can’t fix. Refrigerators can help for high-purity samples, as long as the bottle is dry and closed up tight.
Keep It Clean, Keep It Labeled
Sloppiness invites trouble. Dust, old labels, or unidentified clear liquids (it happens in crowded labs) cause accidents or even legal fines. Clear, chemical-resistant labels survive years and tell everyone what’s inside. If you ever had to rummage through cabinets for the right substance, you learn to over-communicate with labels: date received, date opened, even the last person who used it. Once, a near-miss with a similarly labeled acid bottle burned the lesson into my brain.
Factoring In Safety
Every material safety data sheet points out: avoid eyes, wash off spills, and breathe as little as possible. Spillage of this salt on benches or gloves spreads contamination. Old habits of wearing goggles and gloves, plus keeping absorbent pads nearby, protect both people and the chemical. A clean, cool, dry spot near a fume hood has proved the best choice. I never store it where food, coffee, or personal items sit nearby—cross-contamination is too easy and not worth the risk.
Better Solutions for Shared Spaces
A busy communal lab means mistakes multiply. Centralizing rare or hazardous chemicals in a lockable cabinet helps keep everyone safe and the material uncontaminated. If only one person checks supplies monthly, missed spills or mislabeling can escape notice. Assigning routine checks and keeping a shared digital logbook takes five minutes but has saved hours, especially during safety inspections or sudden audits.
Final Thoughts on Diligence
Simple habits—dry, cool storage, solid labeling, and basic vigilance—outperform any fancy storage tech. Most losses in my experience come from shortcuts: mislabeled jars, unsealed caps, or hurried returns to the shelf. Consistency protects both the sample and the experiment. Over years in the lab, small habits around storage have prevented big problems and preserved more than just chemical purity.
Is 1-Methyl-3-Ethylimidazolium Bromide hazardous or toxic?
Getting to Know This Chemical
1-Methyl-3-Ethylimidazolium Bromide crops up in many research labs and industries focused on advanced materials, batteries, and chemical processing. It belongs to a family called ionic liquids—a group that’s earned praise for low volatility and usefulness as “green” solvents. Nothing about this compound looks wild on the surface, but questions about danger or toxicity deserve more than casual curiosity.
What Science Tells Us
People might feel reassured when they hear about “green solvents,” but being green doesn’t always mean harmless. Studies do exist on the imidazolium class (this substance fits right in), and a few common findings keep turning up. On skin, direct contact tends to bring irritation. If the chemical splashes in the eyes, stinging and redness often follow. Breathing in dust or mist can trigger coughing or a scratchy feeling, especially for those working in closed or poorly ventilated areas.
Accidents can happen, and anyone who’s handled fine powders in a hurry knows how quickly a cloud forms. 1-Methyl-3-Ethylimidazolium Bromide isn’t the worst offender compared to heavy metals or volatile solvents, but that doesn’t give a free pass. The Global Harmonized System (GHS) rates it as an irritant; long-term effects haven’t shown up in big studies, but toxicology work—often done on rodents and aquatic life—suggests nervous system impacts at higher doses. Fish and algae exposed in testing sometimes stop reproducing as easily, or their growth takes a hit. The blunt truth: life forms struggle in water with too much imidazolium compound.
What This Means for the Lab and Beyond
Many of us have stood at a cluttered lab bench, gloves on, distracted by deadlines. I’ve handled similar ionic liquids, and there’s a temptation to downplay risk when the name sounds technical or when no sharp odor warns the senses. A single drop on skin feels greasy, leaving a reminder to clean up—yet people sometimes slack on washing up or keep the same gloves for too long. Mistakes add up. Even careful use inside a fume hood doesn’t make spills or splashes impossible.
Smaller labs sometimes cut corners due to tight budgets, skipping eye protection or relying on old ventilation. Over time, these habits chip away at safety. Waste disposal also poses challenges. Pouring leftover solutions down a drain might look easy, but local regulations frown on sending ionic liquids into public water, since sewage treatment won’t break them down. I’ve seen facilities scramble after inspectors flagged improper disposal.
Smart Handling Keeps Risk Down
A practical approach works best. Use gloves, eye shields, and closed shoes. Good ventilation keeps vapors down and limits accidental breathing exposure. After handling, wash up right away. Clean spills with absorbent pads and treat the mess as hazardous waste. Those working with this compound every day should take extra steps—like using designated containers, recording spills, and making sure disposal happens through licensed services.
Public databases, including European Chemicals Agency (ECHA) and U.S. EPA records, keep expanding on what’s known about long-term toxicity. Right now, there aren’t big stories of severe chronic illnesses tied directly to 1-Methyl-3-Ethylimidazolium Bromide. Still, I’ve learned in work settings that chemicals without strong acute effects can surprise you after years of routine exposure.
Treating 1-Methyl-3-Ethylimidazolium Bromide with a little extra respect, building smarter habits, and keeping up with changing rules help protect not just workers, but the environment too.
What is the purity and molecular weight of 1-Methyl-3-Ethylimidazolium Bromide?
Understanding What’s in the Bottle
Peering into the world of laboratory chemicals, the numbers on the label tell a story. Look at 1-methyl-3-ethylimidazolium bromide — most folks call it [EMIM]Br, for short. This isn’t just another white powder in a glass bottle. Its purity and molecular weight make all the difference for anyone working in synthesis, electrochemistry, or materials research.
The Weight Behind Every Molecule
Start with the numbers: 1-methyl-3-ethylimidazolium bromide’s molecular formula is C6H11N2Br. Punch this into any chemistry calculator, and you get a molecular weight of 207.08 g/mol. Manufacturers and chemists reference this value to build accurate, predictable reactions. Slip up on this number, and measurements lose their meaning—results stop lining up, no matter how clean the gear might be. I remember planning an ionic liquid solvent mix, only to realize that using the wrong molecular mass threw our concentrations off. Mistakes like that eat up time and materials.
Purity’s Real-World Impact
At the bench, purity tells you how much of your bottle is what you’re actually after, with contaminants kept to a strict minimum. The industry standard often lands at over 98%. Sure, there are purer samples, often at a much higher cost. Most suppliers take pride in pushing that last percentage point, since a trace of impurity can sink sensitive experiments. I’ve run extractions where low-purity ionic liquid leached out color or left unexpected salts. Those moments teach you to dig deeper than a glossy product sheet.
Purity influences everything from conductivity to thermal stability, especially for researchers probing physical properties of ionic liquids. Even in battery research, a pinch of a leftover starting material can mess up cycle testing. I’ve watched a colleague chase a mysterious performance drop in a supercapacitor, only to find trace chloride in their EMIM-Bromide. Labs spend real hours and dollars analyzing this, not just out of habit, but because good data depends on it.
Trust but Verify: Documentation and Testing
Reliable suppliers will show off detailed certificates—like NMR, elemental analysis, or water content by Karl Fischer titration. If someone hesitates to share data, I get suspicious. A trustworthy product comes with a file thick enough to tell any curious scientist what’s lurking inside. One lab I worked with routinely ran their own purity checks, just to sleep better at night. Sometimes, the extra cost for “high-purity” isn’t just mark-up; it keeps reproducibility intact and gives peace of mind in high-stakes research.
Supporting Progress Through Standards
Standards from organizations like ISO or ASTM serve as the backbone for chemical suppliers. By sticking to these, they give us all a common language. If there’s room for improvement, it’s in making detailed documentation a non-negotiable part of every purchase. More transparency levels the playing field, letting every lab—no matter the funding—get reliable, honest product data. I’ve had projects stalled by vague specs and had progress jump once a supplier shared everything, warts and all.
Making the Most of Every Gram
1-methyl-3-ethylimidazolium bromide offers more than a string of atoms. The numbers—207.08 for molecular weight and at least 98% for purity—set the stage. They open doors for precise results, fewer surprises, and real breakthroughs. Whether you’re scaling up a process or running a cutting-edge experiment, knowing exactly what’s in the bottle isn’t just good sense—it’s what keeps science honest and moving forward.
| Names | |
| Preferred IUPAC name | 1-ethyl-3-methyl-1H-imidazol-3-ium bromide |
| Other names |
1-Methyl-3-ethylimidazolium bromide EMIMBr EMI Br 1-Methyl-3-ethylimidazole bromide |
| Pronunciation | /waɪ ˈmɛθ.əl θriː ˌɪθ.ɪl ɪˌmɪd.əˈzoʊ.li.əm ˈbroʊ.maɪd/ |
| Identifiers | |
| CAS Number | 64697-40-1 |
| Beilstein Reference | 8532057 |
| ChEBI | CHEBI:72856 |
| ChEMBL | CHEMBL1429699 |
| ChemSpider | 22891576 |
| DrugBank | DB04314 |
| ECHA InfoCard | 19d220ac-cf63-46fd-befb-5cc80c1c1e4e |
| Gmelin Reference | 75585 |
| KEGG | C18761 |
| MeSH | D000070243 |
| PubChem CID | 2735121 |
| UNII | 47P86I35F7 |
| UN number | Not assigned |
| CompTox Dashboard (EPA) | DTXSID30889870 |
| Properties | |
| Chemical formula | C6H11BrN2 |
| Molar mass | 207.09 g/mol |
| Appearance | white to off-white solid |
| Odor | Odorless |
| Density | 1.432 g/cm³ |
| Solubility in water | soluble |
| log P | -2.15 |
| Vapor pressure | Vapor pressure: <0.01 mm Hg (20 °C) |
| Acidity (pKa) | pKa ≈ 7.7 |
| Basicity (pKb) | pKb = 10.2 |
| Magnetic susceptibility (χ) | -86.5 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.545 |
| Viscosity | 265 cP (25 °C) |
| Dipole moment | 6.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 181.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -92.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -526.7 kJ/mol |
| Hazards | |
| Main hazards | Causes serious eye irritation. Causes skin irritation. May cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319 |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| Lethal dose or concentration | LD₅₀ (oral, rat) > 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose) (oral, rat): >2000 mg/kg |
| NIOSH | Not established |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.02 ppm |
| IDLH (Immediate danger) | NIOSH has not established an IDLH value for 1-Methyl-3-Ethylimidazolium Bromide. |
| Related compounds | |
| Related compounds |
1-Methyl-3-ethylimidazolium chloride 1-Methyl-3-ethylimidazolium iodide 1-Methyl-3-propylimidazolium bromide 1-Butyl-3-methylimidazolium bromide 1-Ethyl-3-methylimidazolium bromide 1-Methylimidazolium bromide 3-Ethylimidazolium bromide |

