Rethinking Chemical Progress: A Commentary on 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate
Historical Development
Life in the chemical world never slows down, and ionic liquids like 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate (some chemists like to call it [MBPyrr][OTf] for short) tell a story of shifting attitudes toward safer solvents and greener processes. Before the rise of these compounds, researchers often stuck with volatile organics—think chloroform, benzene, or toluene. The shift began around the nineties when labs and industry alike started pushing for alternatives less likely to catch fire, less likely to poison someone, and a lot friendlier to the environment. Academic chemists first saw pyrrolidinium-based salts and their triflate anion pairings as a new toolkit for non-aqueous chemistry, though it didn’t take long before people saw promise far beyond the beakers and glassware. In a career spent both on the bench and in the classroom, I’ve watched these ionic liquids move from textbook curiosities to real players in battery electrolytes and catalytic processes—and every time, it comes back to safety and tunability.
Product Overview
Mention 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate and you’re stepping into the world of ionic liquids, which pretty much defy the old-school expectations of what a liquid should look or behave like. It’s a salt, sure, but you’ll find it as a room-temperature liquid, clear to faintly yellow, with a viscosity that doesn’t scream “water” but doesn’t act like syrup either. The cation, 1-methyl-1-butylpyrrolidinium, links up with the triflate anion—trifluoromethanesulfonate—not just for stability but to build a liquid that can take some heat, resist breaking down in air, and avoid vaporizing into the lab. Anyone who’s cracked open a bottle of the stuff knows there’s almost never that chemical sting to the nose you’d find in old-fashioned solvents. For industrial or research labs, that quality alone signals progress.
Physical and Chemical Properties
This ionic liquid won’t give you stories of dangerous vapors or low boiling points. Its melting point sits well below room temperature, so it remains fluid across a wide range of conditions most syntheses require. Non-flammability is another big plus—a lab mishap never escalates to a fireball just from a small spill. Polarity and low volatility stand out among its most-discussed features, and so does ionic conductivity. People working in electrochemistry value these numbers; energy researchers pay close attention because it blends the best of a salt’s charge-carrying abilities with a solvent’s practicality. This unique blend holds together across everything from inert gas environments up to crowded, open-beaker chemical reactions. Solubility swings widely depending on which organic or inorganic mates you pair it with, which often opens unexpected doors for creative mixing or phase-separating tricks in separation or extraction work.
Technical Specifications and Labeling
Accuracy in handling and tracking matters a lot—nobody wants the wrong liquid dropped in the wrong experiment. Batches of this ionic liquid carry purity grades tailored for distinct goals: some go into highly sensitive battery research, others head for the demands of reaction catalysis. Chemists look at water content, since moisture can mess with both reactivity and material longevity. Stability under temperature and exposure to light shows up on the radar for shipping and storage, not just during use. For the last few years, the rise of green chemistry has led synopses and technical papers to track not just what’s in the bottle but what it keeps out of the air or waste stream—something that builds trust with both regulators and health watchdogs.
Preparation Method
Making 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate skips elaborate engineering and instead relies on careful organic synthesis. Typical routes start with N-methylpyrrolidine, which then meets up with 1-butyl bromide. This nucleophilic substitution gives the core pyrrolidinium cation. A metathesis with sodium trifluoromethanesulfonate yields the final ionic pair. Purification means more than just a quick filter—residues like water, reaction byproducts, and unreacted starting material can trip up later applications and corrode confidence in research outcomes. These steps sometimes raise headaches for scaling up: solvents need reclaiming, energy use climbs, and process waste demands smart disposal to dodge new chemical headaches.
Chemical Reactions and Modifications
Researchers tinker with both the cation and anion to tune properties like viscosity, conductivity, or chemical stability. Methyl and butyl groups on the pyrrolidinium ring can swap places for other alkyls or even functional groups, each substitution offering a slight nudge toward better solubility or stronger chemical resistance. On the anion side, switching out trifluoromethanesulfonate for, say, bis(trifluoromethylsulfonyl)imide, tweaks solubility in water and electrochemical window. Among the most ambitious tricks: ionic liquids like this sometimes anchor onto solid supports to create hybrid catalysts or extraction media. These efforts rely on years of expertise and detailed trial-and-error—something that big libraries of academic references and the informal sharing of “what worked” between research groups quietly support.
Synonyms and Product Names
Anyone who’s spent time reading technical papers or digging through catalogs knows that names can multiply and confuse. [MBPyrr][OTf] sticks as a typical shorthand in journals, though labels switch to longer forms like 1-Methyl-1-Butylpyrrolidinium Triflate, N-Butyl-N-Methylpyrrolidinium Trifluoromethanesulfonate, or similar mouthfuls. Keeping track of synonyms matters, especially for those searching safety records, patent filings, or regulatory databases—getting lost between names means missing crucial safety or technical facts. Chemical Abstracts Service or EC numbers often show up side-by-side with trade names; most researchers now check these before placing orders or stacking bottles on their shelf.
Safety and Operational Standards
Nobody walks into the lab expecting trouble, but it pays to respect new chemicals, even those promoted for safety. 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate does away with volatility and flammability, yet mishandling still raises risks. Skin contact or accidental ingestion doesn’t typically result in severe effects, but chronic exposure stories haven’t played out long enough for a full verdict. Gloves, goggles, and ventilation come as second nature to anyone working with chemical innovation—insurance against uncertainty, not just box-checking for compliance. Waste streams, especially those containing fluorinated components, draw attention from environmental agencies and local regulators. Over time, tighter labeling standards and batch tracking become industry norms, tracing each bottle from synthesis to final use in research or production lines.
Application Area
People started using this ionic liquid mostly in niche research projects, but its reach has spread into commercial areas thanks to how well it gets along with both organic and inorganic materials. In energy storage, especially lithium batteries and supercapacitors, manufacturers look for solvents that don’t dry out, don’t catch fire, and keep ions moving at a steady clip. Chemical processing values stability under heat, where legacy solvents might degrade or evaporate. Extraction and separation science likes the ionic liquid’s knack for dissolving or carrying compounds that dodge both water and regular oils. Even in pharmaceuticals or polymer syntheses, labs use it to push reactions into cleaner, higher-yielding territory. Green chemistry, a field where I’ve seen more progress in the last decade than almost anywhere else, often relies on tricks learned from handling ionic liquids—a step away from the hazards and emissions associated with traditional organic solvents.
Research and Development
Companies and academic labs keep pushing the boundaries of what [MBPyrr][OTf] can do. Newer projects explore its use in CO₂ capture, waste-water treatment, and advanced material processing. Analytical chemistry research tests its performance in chromatography and electrochemical sensors, with the focus shifting to miniaturized or field-portable devices. I’ve watched research pivot from proof-of-concept papers and lab demonstrations to side-by-side comparisons of ionic liquids and industry standard solvents. Direct collaborations with battery companies, environmental engineers, and university research teams help move the compound from isolated curiosity to valuable staple. As more data rolls in, and as performance benchmarks improve, proprietary blends and purification methods keep appearing, each outfitted for targeted end-use scenarios.
Toxicity Research
Toxicity hasn’t disappeared just because volatility is low. Recent toxicity studies on pyrrolidinium-based ionic liquids note limited acute effects but warn about long-term exposures and environmental buildup. Early work on aquatic organisms points to some bioaccumulative tendencies, particularly for ionic liquids with persistent fluorinated anions like triflate. Some studies flag possible cytotoxic effects at higher concentrations, underscoring the need for careful waste management and monitoring. Regulators and green chemistry advocates push for lifecycle analysis of waste streams, not just product handling. In my experience, the conversation never ends with “it’s less toxic”—responsible labs keep data on downstream effects, and regulatory shifts can follow as toxicologists gather new insights.
Future Prospects
This isn’t just another flash-in-the-pan laboratory chemical. The rise of 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate follows bigger trends in industrial chemistry—less risk, tighter controls, and new markets for cleaner technologies. I see opportunities ahead in battery storage, niche chemical syntheses, and even large-scale environmental cleanup. What comes next involves both chemistry and collaboration, as specialists in materials science, energy, and environmental engineering look for new ways to stretch what this ionic liquid can achieve. At the same time, growing minds in sustainability and toxicity testing keep pressure on manufacturers to document impacts and reduce risks. Ultimately, the future of this ionic liquid depends on a combined push for innovation and responsibility, a hallmark that people—not just the molecules—shape the path forward.
What are the main applications of 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate?
A Closer Look at This Unusual Chemical
Some chemicals end up known only to a handful of researchers. 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate doesn't show up on supermarket shelves, but its impact trickles down into everyday life. My work in university labs and later, collaborating with battery startups, gave me a front-row seat to what this salt delivers. Let's pull back the curtain on how industry leans on this ionic liquid to keep progress moving.
Powering Modern Batteries
Ask anyone building next-generation batteries what holds back progress, and “safety” lands near the top. The flammability of older battery solvents slows down bigger leaps in energy storage. This is where 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate steps up. With a solid track record for chemical and thermal stability, its use in lithium-ion and sodium-ion battery electrolytes has spread. These batteries, found in everything from smartwatches to grid-scale storage, need electrolytes that don’t break down or catch fire. By blending in this pyrrolidinium salt, chemists create safer, longer-lasting batteries. It delivers wide electrochemical windows and stays stable under tough operating conditions, reducing the risk of circuit failure and costly breakdowns.
Electroplating and Metal Processing
Shiny gadgets do not come from nowhere. Behind every polished smartphone bezel sits a process called electroplating, where surfaces get a metal coating for durability or appeal. Traditional methods depend on harsh, sometimes hazardous chemicals. Over the past decade, the push to swap toxic solvents with “greener” ones led scientists to try ionic liquids. This compound, with its robust ionic conductivity and resistance to decomposition, became a favorite in research labs exploring low-waste electroplating methods. Jewelry makers and electronics manufacturers benefit from more precise, controlled metal layers—and fewer environmental headaches.
Solving Separation Challenges in Industry
After spending a long week troubleshooting separation columns in a pilot plant, I learned the value of a stable solvent. Chemical manufacturing often calls for separating or purifying tough mixtures. Ionic liquids thrive in these environments. 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate stands out for its ability to dissolve both polar and nonpolar compounds. Petrochemical companies and fine chemical producers use it to help isolate valuable ingredients, streamline recycling of rare metals, or remove sulfur from fuels. Not by theory, but by practice in scaled-up processes.
Pushing Research Forward
Researchers often look for solvents that stay inert during high-stakes experiments. This compound resists water, stands up to heat, and survives strong oxidizing conditions, making it a backbone in studies on catalysis and new material synthesis. Its non-volatility means labs avoid the hazards of unexpected fumes, supporting safer research environments. With the rise in “green chemistry” policies and the sharp eye of regulatory agencies, the need for stable, task-specific ionic liquids like this one grows. Early skepticism is fading as the evidence piles up—both in peer-reviewed literature and in industrial performance reviews.
Looking Ahead
Chemical manufacturers, battery engineers, and research groups now treat 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate as a building block for progress. That’s not marketing—it’s the result of years of lab tests, patent filings, and production runs. To move more applications into real-world markets, scientists work to cut costs and fine-tune purity so average consumers and small companies can benefit. Sharing best practices, supporting scalable production, and maintaining strict safety data will ensure that the real promise of this ionic liquid reaches industries that rarely get the spotlight.
Is 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate safe to handle and what precautions should be taken?
Understanding What You’re Working With
From my own experience working in chemical research, anything with a complicated name like 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate tends to bring along a healthy dose of caution. This compound falls under the family of ionic liquids, and although researchers love using it in batteries or as a solvent, there’s no such thing as “just another liquid” in the lab. Any substance that mixes organic cations and sulfonate anions deserves full respect for both its utility and its risk.
Direct Contact: Not Worth the Gamble
I’ve watched talented colleagues get a little too casual around new chemicals, only to find themselves with rashes or headaches a few hours later. This chemical comes with risks common to organic liquids—direct skin or eye contact causes irritation. No one wants to explain a mysterious hand rash or eye burn to the safety officer. Safety goggles and nitrile gloves—these are the bread and butter here. Even a small splash stings or lingers, so make a habit, not an exception, of covering up.
Inhalation and Ventilation
Heating ionic liquids might seem harmless because they don’t boil quickly. Still, trace vapors and small particulate releases can sneak up if the fume hood is running below standard. I’ve had sensitive friends develop headaches around these substances, so I always double-check the hood’s airflow before cracking open a fresh bottle. Never get lazy about airflow—breathing in unknown fumes isn’t a cheap lesson.
Gloves and Beyond: Personal Protective Equipment (PPE)
Not all gloves handle every chemical the same way. Nitrile generally blocks out trifluoromethanesulfonate-based compounds better than latex, which can degrade. Wash hands after handling, not before eating or answering your phone. Lab coats protect your arms and keep spills off your clothes. Closed-toe shoes have saved many a toe from caustic drips—including my own.
Spill Management: Act Fast and Smart
Spills rarely happen on purpose, but in real-life labs, they’re always possible. Absorbent pads and a bottle of neutralizing solution live close to my workbench. If a spill hits—contain it, clean it, dispose of the waste in hazardous bins. Normal paper towels won’t cut it; these compounds don’t play nice with everyday cleaning tools. Alerting a supervisor can save a lot of trouble for others down the line.
Know Your Chemistry and Training
Material safety data sheets only do so much on their own. Before even opening a container, read the section on toxicity, stability, and reactivity. No one expects to memorize the MSDS, but you ought to know whether the chemical reacts with water or decomposes when heated. Ask experienced colleagues, as “tried and true” beats a guessing game every time.
Smart Waste Disposal
Pouring ionic liquids down the drain just creates problems for everyone. Chemical waste needs handling via specialty disposal containers suited for organics and fluorinated substances. I once sat through a painful sustainability seminar where the presenter documented the unchecked flow of lab waste—fines aside, the environmental toll sticks.
Conclusion: Practical Safety is Everyday Responsibility
Chemistry’s value comes from curiosity and careful work, not shortcuts. Every step, from glove choice to disposal, reflects personal integrity and respect for health. Follow the precautions and stay alert—handling 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate becomes safer for everyone, not just you.
What is the chemical stability of 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate under various conditions?
Understanding Stability Beyond the Lab
1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate—many call it a mouthful, but in the world of ionic liquids, it’s gotten solid attention. This compound brings flexibility to battery technology, catalysis, and solvents designed for tough jobs. Real questions on its chemical stability, though, make the difference between moving from research to daily use.
Heat, Humidity, and Workplace Realities
From personal experience working in university labs, I’ve seen reactions stall or fail because researchers put too much faith in a chemical’s reputation and too little in actual storage conditions. This ionic liquid keeps pretty steady under ambient temperatures, making it attractive for standard storage. Raised temperatures, though, start shifting things. Stability under heat gets tested above 100°C. Studies show it doesn’t decompose until it approaches around 250°C. Pushing it near this range doesn't just trim shelf life; it risks unwanted byproducts that could ruin precision in synthesis. Folks at Lawrence Berkeley National Lab report that the trifluoromethanesulfonate anion stands up to quite a bit of stress, but moisture is always an enemy. Hygroscopic tendencies mean careless capping pulls in water—and water content in an ionic liquid isn’t just a nuisance. It can lower conductivity, tweak viscosity, and in some cases set off slow degradation.
Acidic and Basic Challenges
Strong acids and strong bases strip away stability. In the presence of strong acid, the pyrrolidinium ring may hold, but the cation-anion partnership starts shaking. If you toss it with a strong base for long periods, the methylpyrrolidinium group loses integrity fast. Decomposition under these conditions doesn’t always explode, but subtle changes—color shift, odor—act as early warnings.
Impurities and Contamination in Real-World Settings
Synthetic chemicals rarely show up completely pure. In pilot plant settings or scale-up environments, trace metals or other ionic species sneak in. These can either serve as unplanned catalysts or unpredictable degradation triggers. I recall one project where an unnoticed trace of iron jump-started decomposition earlier than expected. Even certified “high purity” stocks need regular testing. It’s tempting to skip this, but quality control involves more than following a checklist. Lab teams track NMR or MS data over months, looking for any hints of instability.
Industry Use: Balancing Safety and Innovation
Battery makers and green chemistry firms chase after ionic liquids like this one because of that promise of non-flammability and low volatility. Yet, long-term exposure to light, air, or industrial mixing can stress even the best of them. If a storage drum sits uncapped or exposed, even short spells in humidity cut into performance. Health and safety regulations demand strict labeling and airtight storage. Some teams in pharmaceutical synthesis add desiccant chambers and set up monthly QC on their stock to head off surprises.
Moving Toward Reliable Use
Stability boils down to vigilance. Choose storage away from heat and moisture, run periodic checks, and never trust a container to stay perfect on its own. For engineers, chemists, or factory workers, knowing these pitfalls saves both budgets and safety records. Research always runs into unknowns, but sticking to solid handling and real-time quality checks puts you ahead of the curve. Reliable chemical products need more than good numbers on paper—they thrive only with respect for their limits.
How should 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate be stored and what is its shelf life?
Why Stability Means Everything for Chemicals
Chemicals in the lab can be like that forgotten carton of milk in the fridge—ignore them for too long and they turn into a surprise nobody wants. Keeping 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate (let’s call it BMPyrr OTf) stable is less about fancy gadgets and more about understanding how everyday choices in storage mean the difference between a reliable reagent and an expensive mistake.
Watch the Temperature—and the Humidity
BMPyrr OTf likes cool, dry places, just like most ionic liquids. Extremes in temperature or humidity chip away at its reliability. Anything over 30°C speeds up possible degradation, while moisture in the air starts to encourage hydrolysis. Leaving it unsealed on a shelf invites in problems. I’ve seen labs where someone ‘just put it there for a minute’ and next day the bottle held a sticky mess with odd smells—that’s cross-contamination in action. Nobody needs that.
Store the bottle in a tightly sealed container. Glass jars or bottles with chemical-resistant caps (like PTFE-lined lids) block unwanted air or water vapor from sneaking in. Toss in a few silica gel packs, and you’ve built a cheap, effective insurance policy against moisture. A desiccator also works great for ionic liquids—shelves in ambient lab air just lead to trouble.
Keep Away from Light, Acids, and Bases
BMPyrr OTf doesn’t throw a fit in sunlight right away, but prolonged exposure to UV or strong visible light can upset delicate chemical structures. A brown glass bottle keeps anything light-sensitive in better shape. Storing it near acids or alkaline chemicals—either in the cabinet or at the workbench—can trigger slow side reactions. These aren’t always obvious overnight, but over a month or six, the difference can show up as weird color changes and inconsistent results.
Why Dates Matter: Shelf Life Basics
BMPyrr OTf doesn’t have the short shelf life of old-school solvents, but it’s not immortal. Unopened, sealed in the right bottle, and sitting in a cool, dark spot, this ionic liquid typically lasts about two years. Open the bottle, and that clock speeds up. Residual air from each opening and closing acts like a slow, invisible thief—stealing purity each time. After six months from opening, most reputable labs test samples for quality or just order fresh stock.
No amount of careful storage can undo repeated temperature swings, contamination, or constant exposure to lab air. I’ve seen entire batches tossed when someone skipped simple labeling or delayed reordering, betting the bottle could last ‘just a little longer.’
Labels Continue the Legacy
Every bottle deserves a label counting the arrival month and the opening date. Lab managers who train their teams to log these dates run fewer risks and avoid finger-pointing during troubleshooting. Some tests—like NMR or mass spec—will catch bigger problems, but those cost time and money versus keeping track from day one.
Practical Path Forward
BMPyrr OTf keeps its promise with a little care: cool, dry, dark, sealed tight, and tracked by date. Invest in a small desiccator, brown glass bottles, and good labels. Teach everyone in the lab to treat incoming chemicals with the same care as breakable glassware. These steps pay off in reliable results and fewer surprises the next time someone reaches for the bottle.
What is the purity and typical physical properties of 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate provided by suppliers?
Why Purity Matters
Diving into ionic liquids, people working in labs count on getting exactly what’s printed on the bottle label. If you’ve ever handled 1-methyl-1-butylpyrrolidinium trifluoromethanesulfonate (also called [BMPy][OTf]), you know that purity isn’t just a number for an MSDS sheet—it shapes experimental results from start to finish. Suppliers usually offer this salt with purity not dipping below 98%. The remaining 2% can sound small, until that contaminant blocks a catalyst or interferes with a battery’s performance. Anyone setting up a reaction or studying electrochemical windows can run into data headaches if that last fraction of impurity refuses to cooperate. Labs with strict protocols usually look for a suppliers’ NMR and elemental analysis data, not just the certificate of analysis.
Peeking at Physical Properties
The [BMPy][OTf] salt sets itself apart with a melting point well above room temperature, usually resting between 50°C and 85°C. Once the vial sits in a heat block, the once-chunky solid turns into a clear liquid showing low volatility. That low vapor pressure helps a lot in glovebox work or high-voltage applications. For my own part, walking into a cramped electrochemistry lab, a low-odor, low-volatility salt is more than convenience—no one wants to lose reagent to random evaporation or worry about breathing odd vapors all day.
Viscosity turns out to be one of those deal-breaker properties the first time you try moving this liquid with a pipette tip or try charging it into a microfluidic channel. Close to room temperature, the liquid gets syrupy, pushing above 100 centipoise, so warming the sample makes life easier. That high viscosity helps as an electrochemical medium, limiting leakage or cross-contamination, but might slow things down if you want rapid ion transport.
Density hovers near 1.3 g/cm³, easy enough to measure, and usually doesn’t throw a wrench into weighing out portions. Water solubility feels more pressing to me. This salt dissolves well in water, ethanol, and some polar organic solvents. If you’ve ever watched your ionic liquid pull moisture from humid lab air, you’ll realize that hygroscopicity isn’t an academic detail. Extra water shifts conductivity, electrochemical window, and can ruin the salt for certain applications. Storage under inert gas or in a desiccator avoids nasty surprises.
Trust—Not Just Specs on a Sheet
Every supplier, whether in Europe or Asia, prints product specs in their catalogs. But I’ve seen a surprising range day-to-day between sources. A bottle labeled “99%” purity from two different companies can show clear color or brown tint, mild odor or none, and vastly different NMR spectra. For people working on batteries, supercapacitors, or catalysis, confirmation from independent lab tests beats faith in supplier assurance.
Several respected producers include chromatograms and ion chromatography data. Still, the best approach involves sending a sample for third-party testing. Verification cuts down wasted experiments and builds trust in the supplier’s processes. Synthetic chemists or energy storage scientists can’t afford to ignore solvent remnants or trace halides.
Improving Reliability
Greater transparency between suppliers and labs builds scientific credibility and better results. More companies began including detailed batch analysis and trace impurity breakdowns, not just broad purity percentages. I’d like to see more products accompanied by a QR code linking to live batch test data, instead of relying on PDFs updated months apart.
For anyone using 1-methyl-1-butylpyrrolidinium trifluoromethanesulfonate regularly, smart handling—testing for water content and verifying purity by NMR—cuts unpleasant surprises. As demand grows across analytical chemistry and electrochemical fields, communication between suppliers and users becomes part of the scientific process, not just good customer service.
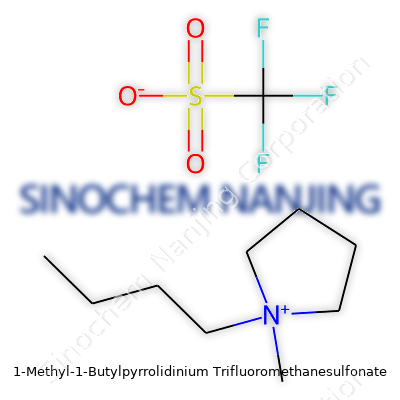
| Names | |
| Preferred IUPAC name | 1-methyl-1-butylpyrrolidin-1-ium trifluoromethanesulfonate |
| Other names |
N-Butyl-N-methylpyrrolidinium trifluoromethanesulfonate BMPTfO 1-Butyl-1-methylpyrrolidinium triflate 1-Butyl-1-methylpyrrolidinium trifluoromethanesulfonate BMPTf N-Butyl-N-methylpyrrolidinium triflate |
| Pronunciation | /wanˈmɛθ.əl.wənˈbjuː.təl.paɪˌrɒl.ɪˈdɪn.i.əm ˌtraɪ.flʊə.rəʊˌmiː.ˈθeɪn.sʌlˌfəʊ.neɪt/ |
| Identifiers | |
| CAS Number | 616476-31-6 |
| Beilstein Reference | 1361081 |
| ChEBI | CHEBI:189481 |
| ChEMBL | CHEMBL2150783 |
| ChemSpider | 16023372 |
| DrugBank | DB11213 |
| ECHA InfoCard | 01baf5e6-e4cf-4b7e-87a0-8e02def7a0e8 |
| Gmelin Reference | 563341 |
| KEGG | C22106811 |
| MeSH | D000077308 |
| PubChem CID | 16066573 |
| RTECS number | UJ9780000 |
| UNII | KT9Z48L8X3 |
| UN number | UN3488 |
| CompTox Dashboard (EPA) | DTXSID1062388 |
| Properties | |
| Chemical formula | C10H20F3NO3S |
| Molar mass | 284.33 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | Odorless |
| Density | 1.34 g/cm³ |
| Solubility in water | Soluble |
| log P | 0.458 |
| Vapor pressure | 4.3E-5 mmHg at 25 °C |
| Acidity (pKa) | 13.5 |
| Basicity (pKb) | pKb = -3.6 |
| Magnetic susceptibility (χ) | -68.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.430 |
| Viscosity | 84.2 cP (25 °C) |
| Dipole moment | 5.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 523.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1077.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4752.7 kJ mol-1 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P233, P240, P241, P242, P243, P280, P303+P361+P353, P305+P351+P338, P370+P378 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 98 °C |
| Autoignition temperature | 230 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat) >2000 mg/kg |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Methyl-1-Butylpyrrolidinium Trifluoromethanesulfonate is not established. |
| IDLH (Immediate danger) | Not listed/Not established |
| Related compounds | |
| Related compounds |
1-Butyl-1-methylpyrrolidinium chloride 1-Butyl-1-methylpyrrolidinium bromide 1-Butyl-1-methylpyrrolidinium tetrafluoroborate 1-Butyl-1-methylpyrrolidinium hexafluorophosphate 1-Butyl-1-methylpyrrolidinium bis(trifluoromethylsulfonyl)imide |

