1-Methoxy-2-Propyl Acetate: Science, Safety, and Shifting Opportunities
Historical Development
Learning about 1-Methoxy-2-Propyl Acetate, or what chemists often call Propylene Glycol Monomethyl Ether Acetate (PMA), drags us back through a century of modern solvent science. When industries demanded safer, less volatile substitutes for older, more hazardous chemicals, companies started exploring glycol ethers. Once introduced to coatings and inks, PMA didn't take long to catch on because processors valued its convenient evaporation rate and milder odor. Growth in manufacturing, especially in electronics, printed materials, and automotive paints, saw a steady uptick in use of this solvent. Newer environmental regulations certainly nudged that along, pressuring the paint and coating sectors to move away from highly toxic ones like xylene and toluene. You can see how a technical breakthrough remakes entire product lines without most of the public ever knowing the backstory.
Product Overview
Out on the work floor or in a laboratory, 1-Methoxy-2-Propyl Acetate serves as a powerful solvent. It works especially well in dissolving resins, dyes, and greases that can puzzle other liquids. I've seen product engineers opt for it when they wanted better flow and fewer streaks in waterborne coating formulas. Factory managers appreciate that drying times with PMA behave predictably, helping them keep batches moving without constant fuss. Efforts to boost energy efficiency in industrial settings favored such solvents. While PMA doesn't hog the spotlight, it delivers in contexts requiring something tough yet less harsh or flammable.
Physical and Chemical Properties
The clear, colorless appearance gives away little about what this solvent handles behind the scenes. It reads as a middleweight on volatility, not so fast as to flash off before it does its job, yet not sluggish enough to cause problems with residue. Its mild odor is less likely to create headaches on a busy line compared to some pungent alternatives. Solubility stretches from water-based to oil-based systems, making it flexible for process engineers. Chemical stability in PMA matters: it won’t decompose easily under normal conditions, so it fits industrial settings that need reliability more than surprises.
Technical Specifications and Labeling
Producers package PMA under tight standards, following global and domestic chemical labeling guidelines. Labels underline its flammable nature and stress the need for ventilation. Regulatory bodies in the United States and Europe require hazard pictograms and clear wording about risks. European REACH rules and the U.S. OSHA guidelines both weigh in, seeking to limit harmful exposures and insist on rigorous workplace safety measures. Chemical supply managers keep track of purity, moisture content, and acidity, because real-world usage hinges on transparent documentation and honest disclosure—something I’ve learned never to take for granted.
Preparation Method
Manufacturers synthesize PMA by esterifying propylene glycol monomethyl ether with acetic acid. This process takes careful temperature control and strong attention to catalyst selection. The aim is to maximize output while reducing byproducts like free acids, which affect performance and shelf life. Getting this step right saves on expensive downstream purification and helps meet the expectations of demanding industries. Big plants make it at scale, but even boutique operations have found their stride producing consistent batches by refining their reaction conditions over years of trial and error.
Chemical Reactions and Modifications
PMA’s acetyl group leaves it open for hydrolysis in the presence of water and acids, forming methoxypropyl alcohol and acetic acid. In well-run operations, technicians keep a watchful eye, maintaining low water activity to avoid breakdown. In the world of advanced coatings, chemists look at further modification, like reaction with isocyanates or epoxy resins, seeking next-generation film properties for automotive and electronics applications. The adaptability lowers technical barriers for innovation where other solvents fall short.
Synonyms and Product Names
Walk through any technical catalog, and you’ll find PMA sold as 1-Methoxy-2-Propyl Acetate, Propylene Glycol Monomethyl Ether Acetate, or often “PMA” for short. Some traditions call it PGMEA, not to be confused with close relatives like PM or PnB. The world of chemical trade codes and acronyms brings confusion, so process engineers and quality managers always cross-check using CAS numbers to avoid costly mix-ups—a lesson anyone in logistics or compliance learns sooner rather than later.
Safety and Operational Standards
In day-to-day work, attention to PMA safety isn’t optional. People on shop floors wear gloves, goggles, and sometimes respirators when handling this solvent, because even though toxicity sits lower than with some alternatives, it can still irritate skin, eyes, and airways. Safety Data Sheets (SDS) go out with every shipment, outlining fire prevention routines and what to do in a spill. Ventilation systems and explosion-proof storage became standard in factories using PMA in significant volumes. The short-term exposure limits set by agencies like OSHA reflect decades of research on what workers can handle. Firms audited for environmental compliance stay strictly within disposal guidelines thanks to these standards, limiting PMA in waste streams to minimize ecosystem harm.
Application Area
If you’ve ever seen a flawless automotive finish, intricate printed circuit board, or vivid magazine cover, chances are good PMA took part. Paint and coating manufacturers profit from its deft balance of solvency and controlled evaporation. In electronics, it cleans photoresist layers without destroying delicate etch patterns. Printing shops gravitate to PMA because it lets inks set crisply, even for high-speed jobs. These uses show how progress in chemistry touches everything from cars to daily mail without much fanfare, quietly improving durability, appearance, and longevity.
Research and Development
Universities and industry labs don’t stop at using PMA the same way year after year. Researchers probe for greener synthesis, aiming to reduce waste and energy use during manufacture. People in coatings science investigate additives and blends that can tune PMA’s drying or solvency for specialized films. Lab groups monitor performance changes under different temperatures, humidity, and light, ensuring reliability through the lifespan of finished products. Some groups turn to molecular modeling to predict reactivity and ultimately design safer, more effective alternatives, if any can surpass PMA in versatility.
Toxicity Research
Toxicologists know PMA as a less-aggressive solvent, especially compared to ancestors like benzene, but they don’t let caution slide. Studies over decades track effects from short-term inhalation, skin exposure, and accidental ingestion. Most findings show low chronic risk, but high doses can lead to central nervous system symptoms—headache, dizziness, and breathing trouble. Regulatory scientists continue animal testing and epidemiological reviews, seeking gaps in existing data. The challenge remains to balance industrial needs with safety, especially since safer alternatives often run less effective in key technical roles; manufacturers weigh these trade-offs as part of responsible supply chains.
Future Prospects
Demand for PMA looks steady, with global trends in electronics, green coatings, and advanced materials pushing continued use. Environmental advocacy groups keep watch, asking producers to cut emissions and invent biodegradable substitutes where possible. The best shot at next-generation solvents comes from targeted research: finding molecules as effective as PMA, yet with faster natural breakdown and lower workplace exposure risks. Until chemists strike that balance, manufacturers will stick with what works while upgrading safety engineering and collection of exposure data for better, more informed decision making down the road.
What is 1-Methoxy-2-Propyl Acetate used for?
A Silent Workhorse in Modern Industry
Whenever I hear about manufacturing or painting, I think of the sharp, sometimes sweet smell that lingers in workshops and studios. That’s the signal that solvents are hard at work. One chemical that doesn’t get much attention outside the factory or laboratory floor is 1-Methoxy-2-Propyl Acetate, often called PMA or propylene glycol monomethyl ether acetate. It's not a topic for dinner conversation, yet it quietly shapes the world—especially in how products look and last.
How It Touches Everyday Life
This colorless liquid acts as a solvent in countless products. For folks in painting, automotive refinishing, and electronics, PMA is a familiar companion. Paint doesn’t glide easily over metal and plastic without something to help other ingredients dissolve, mix, and flow. That’s where PMA steps up: it dissolves resins and pigments, keeps mixtures stable, and makes sure everything dries at the right speed.
A close friend of mine works in automotive repair. He told me about spray booths where this chemical gives paint the ability to lay flat on car panels, without runs or splotches. And inside those glossy smartphones in our pockets? The printed circuit boards relied on PMA during manufacturing to get their protective coatings just right.
Fact-Based Insight Into Real-World Use
Numbers tell the story, too. According to industry reports, global demand for PMA has risen over the years, driven by the electronics and automotive sectors’ hunger for precise coating results. PMA helps adjust drying times for paints and coatings. Without that control, assembly lines would need to slow down or endure quality issues. In electronics assembly, PMA’s presence lets workers clean delicate parts without damaging sensitive circuits.
Health and safety authorities, including the European Chemicals Agency and the US Environmental Protection Agency, watch PMA use closely. The chemical’s low odor, moderate evaporation rate, and small molecular size make it safer and more versatile than some harsher solvents of the past, such as toluene or xylene. This softer profile does not mean skipping protective gloves and proper ventilation, because even less aggressive solvents can cause headaches and skin irritation with frequent exposure.
Why Responsible Choices Matter
Standing in hardware store aisles, I often glance at the warning labels on cans of paint stripper or spray paint. PMA sometimes appears there. The choice to include it isn’t casual. Paint makers look for solvents that do the job using less product and create less air pollution. Regulatory guidelines encourage the switch to options like PMA where possible, since it produces lower levels of volatile organic compounds (VOCs) compared to older alternatives.
Anyone who spends their workday around paints and solvents has stories about chemical headaches, smelly workshops, and stubborn stains. Switching to safer solvents like PMA can mean fewer health complaints, a cleaner job site, and cleaner air for all of us. Yet, as anyone who has spilled even a small bottle knows, safe use must go hand-in-hand with training, ventilation, and care during disposal.
A Path Forward
The industries that rely on PMA keep finding ways to use less of it, contain fumes, and recycle leftover chemicals. I’ve seen small tech manufacturers partner with chemical companies to recover solvents from their processes, saving costs and waste. These efforts support cleaner production, healthier workers, and longer-lasting products for everyone. True progress means using smart chemistry and practical habits together—and that’s a job for all of us, not just the scientists.
Is 1-Methoxy-2-Propyl Acetate hazardous to health?
A Common Solvent, Not Without Danger
Anyone who’s spent time in paint shops or printing rooms probably knows the harsh tang of solvents in the air. 1-Methoxy-2-Propyl Acetate often makes an appearance on safety sheets and container labels. Also known as propylene glycol methyl ether acetate (PMA), this clear liquid helps dissolve pigments, resins, and various chemicals. Factories, warehouses, and even small workshops rely on it for fast drying and consistent results in coatings. Still, amongst all the buzz of production lines and finished batches, health questions keep cropping up.
Getting Exposed
Most folks who handle 1-Methoxy-2-Propyl Acetate meet it through skin contact, breathing fumes, or sometimes the unlucky mistake of splashing it into eyes. Short bursts of exposure can cause headaches, nausea, nose or throat irritation, and dizziness. Eyes sting. Hands dry out and crack. Even if a job only takes an hour or two, the sticky smell sticks with you. NIOSH warns that extended or heavy breathing of its vapors affects the central nervous system in unpredictable ways and long-term exposure may harm the liver and kidneys. The evidence isn’t as publicized as it is for substances like toluene, but the risks haven’t gone away.
Scientific Consensus and Evidence
Scientists have run studies using rats, finding tissue damage and shifts in blood chemistry after long-term exposure to high levels. Overdose cases in people are rare, but industrial records document symptoms much like solvent poisoning: forgetfulness, coordination trouble, and chronic migraines. The European Chemicals Agency lists it as an irritant, and OSHA tells employers to fit their workers with gloves, goggles, and fume hoods. Manufacturers themselves publish safety data showing limits: 50 ppm in the air stands as the threshold for workers, and going beyond brings consequences that can last months.
Addressing the Issue in Workplaces
People shouldn’t have to accept health problems as the cost of doing a job. Even small steps make a difference. Factories that switched to local exhaust fans and closed mixing systems report better air and fewer sick days. Workers who wear nitrile gloves and splash-proof eyewear sidestep the worst daily symptoms. Management support matters: when the boss checks air monitors, stocks proper gear, and schedules health training, you see fewer incidents. The “ounce of prevention” saying fits—the up-front cost of proper ventilation and regular checks pays off in both safety and fewer lawsuits or worker replacement costs.
Better Choices for the Future
Alternatives exist, from water-based solvents to “green” variants claiming reduced toxicity. Switching isn’t always simple, but companies that started using safer solvents haven’t looked back: less turnover, fewer doctor’s visits, and real peace of mind. Regulatory bodies in the US and Europe keep updating exposure limits as new research appears, pushing companies toward safer processes every year. For consumers, knowing what’s inside that can of paint or bottle of ink gives some power—safer products often cost a bit more, but the payoff shows up in long-term health.
The solvent game keeps changing. Safety practices, clear labeling, and a willingness to invest a little extra in systems and protective gear create cleaner air and healthier workers. It’s not just about checking the box on a compliance form. It’s about making sure you go home feeling as sharp as you arrived.
What is the proper storage condition for 1-Methoxy-2-Propyl Acetate?
Straight Talk on 1-Methoxy-2-Propyl Acetate
1-Methoxy-2-Propyl Acetate pops up in places you might not expect, from paints to cleaning products. Most people don't spend their day thinking about how these chemicals are stored, but a mistake can spell trouble. I learned early in my lab days that a missed detail in storage can shut down a project, or worse.
The real deal with 1-Methoxy-2-Propyl Acetate is that it’s flammable and it evaporates easily—stuff anyone working with solvents has to take seriously. I once saw a careless seal on a jug lead to headaches in the storeroom and a scolding from the safety officer. This isn't just about following a rulebook; it's about making sure everyone goes home the same way they arrived.
Conditions Matter: Heat and Air Are No Friends
Don't park 1-Methoxy-2-Propyl Acetate near a heat source. I’ve watched solvents swell and hiss just from being too close to a sunny window. Keeping it cool protects product quality and reduces vapor buildup that can linger in the air. A temperature under 30°C (86°F) keeps things in check for most common workplaces, though a dedicated flammable cabinet does even better.
Always seal those containers tightly. Each crack in a cap means more fumes. If you’re in a place with poor ventilation, that vapor can build up and trip alarms or worse—cause a burn. Good airflow repels those fumes. I saw one workshop turn off their fume hood and instantly the harsh smell grew. It’s not just unpleasant, it ramps up risk.
Separate, Shield, and Label
Solvents and acids or oxidizers never mix well—sometimes literally. In my experience, a busy shelf gets cluttered fast. Grouping chemicals by hazard class keeps things straight. Use clear labels with purchase and opening dates. It sounds fussy, but in one emergency spill in a warehouse, real pain came from not knowing what was on the floor.
Choose containers built for chemicals, ideally the one the supplier provided. That factory seal means no surprise reactions with the plastic or metal. My team once used a generic plastic jug in a pinch; it slowly got brittle and cracked, leaking before lunchtime.
Handling Spills and Disposals
It’s tempting to shrug off a small spill, but even a few drops can make trouble. I saw a coworker lose a week’s worth of data for cleaning up a splash with the wrong gloves. Absorbent spill pads in easy reach save time and skin. Never toss unused solvent down the drain. Most cities treat this as hazardous waste. Local guidelines usually spell out the process, but any reputable disposal company can walk you through it.
Why This Matters
I’ve seen my fair share of safety audits. Inspectors don’t want to play “gotcha”; they are there to keep people safe. Flammable vapor, spoiled material, or accidental mixing with reactive chemicals all cause headaches and dangers. Good storage practices protect your health, your coworkers, and your company’s reputation. If something feels off—a weird smell, a swollen container—stop and check. It doesn’t take long, and it keeps small problems from turning into headlines.
Solid habits in chemical storage come down to paying attention and not skipping steps. Flammable solvents demand respect. Locking in the right temperature, air flow, and clear labels can turn a risky stockroom into just another safe corner of your workspace.
How should 1-Methoxy-2-Propyl Acetate be handled safely?
Understanding the Real Risks
1-Methoxy-2-propyl acetate, often called PMA, turns up often in paints, inks, and cleaners. The chemical helps dissolve sticky stuff and lets products dry evenly without streaks. Anyone who has spent hours in a workshop knows how useful a good solvent is—it does the heavy lifting, but the cost comes with the fumes, and the way it soaks through gloves that aren't made for chemicals.
It’s easy to forget how sneaky some chemicals can be. PMA comes with a sweet smell, almost pleasant, which tricks people into lowering their guard. Breathing in a little over a few hours can make your eyes itch, your throat burn, and even bring on headaches or dizziness. In a closed, unventilated space, things can get riskier, especially after long exposure. Over time, throwing safety rules out the window brings real health headaches—literally and figuratively.
Why Small Steps Make a Big Difference
Years ago, I worked in an old printshop where nobody gave much thought to ventilation. Windows stayed closed, exhaust fans collected dust, and gloves weren't the chemical-resistant kind. Colleagues ended up with skin rashes and coughing fits by the end of a shift. These days, safety teams remind folks about gloves and goggles, but putting rules into practice beats reading long memos any day.
Gloves and Protection:Nitrile gloves hold up against PMA. A neighbor working as a lab tech swears by them for all handling tasks—her hands stay safe, even after hours of cleaning up spills. Standard latex gloves, on the other hand, break down quickly. For eyes, a pair of well-fitting safety goggles is a must, not a suggestion, since the stuff splashes easily and burns the skin on contact.
Keep the Air Moving:Leaving a door cracked or switching on an extractor fan means the fumes don’t linger. Industrial settings install local exhaust vents right above workbenches for a reason—it pulls the worst of it away before it settles in your lungs. Masking up with a respirator rated for organic vapors fills in the gap when you can’t trust the air.
Storage Habits:Few people enjoy taking out the hazardous waste drum or triple-checking the lids on every bottle. Still, the habits keep fumes from spreading or, even worse, bottles from leaking. Metal cans with tight seals and clear labels give a small but real layer of security nobody appreciates until a spill happens. Keeping PMA far from sparks or open flames makes sense, given its flash point; a single stray spark makes for a bad afternoon.
Steps Toward Safer Workspaces
OSHA lists PMA as a hazardous chemical for a reason. Reviewing safety data sheets before the workday only takes a few minutes. If someone on the team has questions, bringing in a safety rep or checking local guidelines beats guessing. Keeping eyewash stations and showers nearby pays off in emergencies. Spills happen fast, so absorbent pads within reach save a lot of cleanup.
At the end of the day, working safely with PMA comes down to habits. Wearing the right gear, airing out the space, and labeling every bottle matter much more than the fine print on technical sheets. Over time, these small steps add up to a smoother, safer workshop—where the air stays clear and hands avoid burns or rashes.
What are the environmental impacts of 1-Methoxy-2-Propyl Acetate?
Looking Beneath the Surface
Most folks rarely give a second thought to what goes into their paint or cleaning products, as long as they do the job. The story shifts for those like me who grew up watching marshes and creeks change color after a nearby warehouse hosed out its floors. Chemicals get into places that regulations and paperwork often miss. 1-Methoxy-2-Propyl Acetate, a solvent found in coatings, inks, and cleaners, slips through the cracks much the same way. Its track record with the environment matters because communities—especially those close to industrial centers—sit on the receiving end.
Where It Lands and How Long It Stays
Once washed down the drain or sprayed on a surface, this chemical rarely stays put. It evaporates into the air, seeps into soil, and sometimes hits the water table. As a volatile organic compound (VOC), it contributes to the formation of ground-level ozone. Ozone in the upper atmosphere shields us; near the ground, it means respiratory problems, especially for kids and older folks. People in cities with lots of manufacturing already struggle to catch their breath during the summer—this solvent only makes things worse.
The Fallout for Water and Wildlife
Many times I’ve heard anglers complain about fish kills or frogs disappearing like they never existed. Runoff from factories, whether in paint shops or print houses, drags 1-Methoxy-2-Propyl Acetate along for the ride. Aquatic life handles these chemicals worse than we do. Research from the European Chemicals Agency flags the solvent for its toxicity at higher doses, especially for aquatic invertebrates and fish eggs. Growth rates tumble, and some species lose their ability to reproduce at concentrations far lower than what kills outright.
Human Health at the Receiving End
People who work with paints and solvents smell this chemical every shift. It stings eyes, gives headaches, and after long exposure, liver problems aren’t out of the question. All it takes is one leaky drum at an industrial site or a poorly ventilated warehouse. Once these stories break, the focus lands on EPA guidelines and air quality reports, but families are left dealing with the results much longer. According to the U.S. National Library of Medicine, repeated exposure over time can compound the risk, especially indoors where air doesn't refresh quickly.
Better Choices and Smarter Practices
A lot of companies now test water and air for VOCs, but spot checks don’t make a real dent when solvents keep getting dumped or vented. Closed-loop systems—where vapors get captured and scrubbed—drop emissions. Less toxic alternatives in paints, such as water-based formulas, cut VOCs dramatically. Users play a role, too: proper storage, clear disposal instructions, and local collection points keep smaller spills from turning into big problems. Industries can’t operate behind closed doors, and regular reporting with real community oversight brings neighbors into the conversation.
Lessons from the Ground
Growing up near an industrial area taught me the value of clean water and air. That experience underscores why sustainable practices matter more than the bottom line. 1-Methoxy-2-Propyl Acetate isn’t just a line in a safety data sheet—it’s part of the air we breathe and the rivers where kids fish. Every choice, from manufacturers to everyday users, has ripples that reach far beyond the factory gate. Reducing use, improving containment, and choosing safer substitutes chip away at the damage. That’s a future both workers and neighbors deserve.
| Names | |
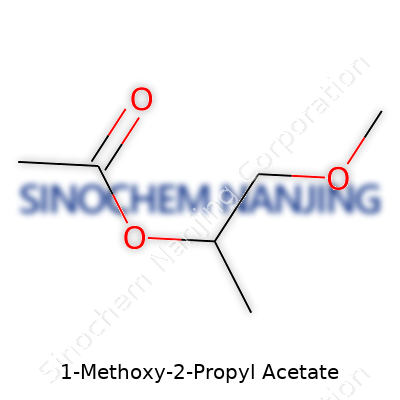
| Preferred IUPAC name | 1-methoxypropan-2-yl acetate |
| Other names |
Propylene glycol monomethyl ether acetate PGMEA 1-Methoxy-2-acetoxypropane 2-Methoxy-1-acetoxypropane Methoxypropyl acetate |
| Pronunciation | /ˈwʌn ˈmɛθ.ɒk.si tuː ˈprəʊ.pɪl əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | 108-65-6 |
| Beilstein Reference | 878088 |
| ChEBI | CHEBI:8966 |
| ChEMBL | CHEMBL1544949 |
| ChemSpider | 7585 |
| DrugBank | DB14159 |
| ECHA InfoCard | ECHA InfoCard: 100.023.876 |
| EC Number | 203-603-9 |
| Gmelin Reference | 1330861 |
| KEGG | C19699 |
| MeSH | D052438 |
| PubChem CID | 7923 |
| RTECS number | AI8925000 |
| UNII | K7O76887AP |
| UN number | UN3272 |
| Properties | |
| Chemical formula | C6H12O3 |
| Molar mass | 132.16 g/mol |
| Appearance | Colorless liquid |
| Odor | Mild, pleasant odor |
| Density | 0.966 g/cm³ |
| Solubility in water | soluble |
| log P | 1.2 |
| Vapor pressure | 0.49 hPa (20 °C) |
| Acidity (pKa) | 8.1 |
| Basicity (pKb) | pKb: 15.3 |
| Magnetic susceptibility (χ) | -8.36×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.406 |
| Viscosity | 0.95 mPa·s (at 20 °C) |
| Dipole moment | 3.98 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 222.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -576.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3557.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H336 |
| Precautionary statements | P210, P243, P261, P271, P280, P303+P361+P353, P304+P340, P312, P337+P313, P403+P233, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | 46°C |
| Autoignition temperature | 340 °C |
| Explosive limits | 1.5% - 7.0% |
| Lethal dose or concentration | LD50 oral rat 8532 mg/kg |
| LD50 (median dose) | LD50 (Rat oral): 8532 mg/kg |
| NIOSH | WN4725000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1-Methoxy-2-Propyl Acetate is "100 ppm (540 mg/m³)". |
| REL (Recommended) | 50 ppm (TWA) |
| IDLH (Immediate danger) | 150 ppm |
| Related compounds | |
| Related compounds |
Propylene glycol methyl ether Propylene glycol Isopropyl acetate Ethylene glycol monomethyl ether acetate Propylene glycol n-butyl ether Butyl acetate |

