1-Ethylpyridinium Bromide: Chemistry’s Reliable Cornerstone
Historical Development
Long before ionic liquids started filtering into labs across the globe, researchers spent years searching for manageable, tunable compounds that didn’t bring the hazards of old-school solvents. 1-Ethylpyridinium bromide came onto the chemical scene through patient organic synthesis work, and the shift from pyridine derivatives to more useful pyridinium salts took dedicated effort from academics and industrial chemists alike. Pyridinium-based salts turned up in papers as far back as the early and mid-20th century, but back then they stayed largely in the world of theoretical work or niche syntheses. Interest spiked again as chemists sought solvents that could handle extreme conditions without breaking down, or materials that facilitated safer, more eco-friendly chemistry. The growth of “green chemistry” forced a relaunch for compounds like 1-ethylpyridinium bromide, which now shows up in research that spans electrolytes for batteries, organocatalysis, and electrochemistry. This backstory matters, since it shows how chemistry keeps circles running—a once-specialized laboratory compound can find new life as the push for sustainability demands better tools.
Product Overview
Anyone who has ever unpacked a bottle of 1-ethylpyridinium bromide knows it doesn’t call attention to itself. It’s a salt in the literal sense: typically solid, sometimes a white crystalline powder, occasionally a clumpy off-white block if storage conditions lapse. The molecule combines the recognizable pyridine ring with an ethyl side chain, paired with a bromide counterion. On paper, the structure looks simple. In practice, this ionic nature opens doors in both academic and applied chemistry, turning what seems basic into something quite useful. Having handled similar pyridinium salts, I find their physical consistency practical—easy to weigh, easy to store, and rarely prone to sudden degradation, at least when kept away from excess moisture. The lack of odor makes lab life easier than with some amine relatives, which helps when working in cramped shared spaces.
Physical & Chemical Properties
As a primarily crystalline solid, 1-ethylpyridinium bromide dissolves well in polar solvents, especially water and certain alcohols, which makes it a flexible partner for formulating electrolyte solutions or working up chemical reactions. Its melting point lands higher than room temperature, so it doesn’t turn to liquid unless forced; this trait holds steady compared to shorter- or longer-chain relatives. Thermal stability puts it ahead of many quaternary ammonium salts. The salt carries the characteristic chemical resistance of pyridinium systems—strong against both acids and bases under ordinary conditions. Unlike reactive organohalides, this compound doesn’t dash off to start unwanted side reactions. My own time working with pyridinium salts taught me that their ion-pairing can influence solubility and electrochemical behavior in ways you only appreciate after handling a few batches. For researchers blending solutions or examining conductivity, these details matter.
Technical Specifications & Labeling
Every bottle I’ve received—whether for research or teaching purposes—comes clearly labeled with molecular formula (C7H10BrN), molecular weight (about 188.07 g/mol), and hazard statements. Purity tends to stay above 97% when sourced from reputable suppliers, as even a 2–3% impurity can skew data in sensitive electrochemical measurements or catalytic testing. Any technical document worth its salt includes melting point, solubility data, and safe handling notes. I value how regulatory bodies push for these clear statements, especially as more labs encourage students and early-career chemists to work with ionic species. While technical specs alone don’t tell the full story, they’re a checkpoint by which you compare batches, monitor shelf life, and satisfy both safety audits and funding agencies demanding traceability. Anyone who’s sat through an inspection knows the value of transparency and accurate documentation.
Preparation Method
The route to 1-ethylpyridinium bromide isn’t shrouded in mystery. Most labs start with pyridine, reacting it with ethyl bromide under controlled conditions, either as a simple alkylation or through solvent-free approaches when researchers want to avoid waste. The setup needs good fume extraction, since ethyl bromide can sting both nose and eyes, and pyridine is notorious for its sharp, fishy odor. In my own experience, patience matters—a rushed reaction leads to byproducts and lost yield. Purification usually involves crystallization from ethanol or another compatible solvent. These synthetic steps seem straightforward, yet controlling moisture and temperature determines success or lack thereof. I’ve seen batches fail when humidity crept into the reaction or purification. It serves as a reminder: chemistry at the bench isn’t always by-the-book, but small procedural tweaks make all the difference.
Chemical Reactions & Modifications
Once in hand, 1-ethylpyridinium bromide offers a platform for further chemical adventure. The pyridinium core acts as both electron sink and reactive scaffold; with the right substitutions, chemists generate a range of ionic liquids or fine-tune reactivity to fit the latest electrochemical project. This salt can play a direct role as a phase-transfer catalyst, or support synthetic steps such as nucleophilic substitution and organic coupling reactions. Swapping out the anion, modifying the alkyl chain, or adding functional groups to the ring can tweak solubility, thermal stability, or even antimicrobial properties. This kind of modular chemistry attracted me during my graduate studies because being able to steer both physical and chemical profiles by adjusting small features means fewer dead ends in the research phase. Flexibility on the molecular level often saves resources in the long term.
Synonyms & Product Names
Literature and catalogs don’t always agree on naming. It goes by 1-ethylpyridinium bromide and N-ethylpyridinium bromide, and on rare occasions, the IUPAC “ethylpyridinium bromide” without the locant. For researchers jumping between papers, checking these synonyms prevents costly mix-ups—it’s easy to run into a paper reporting one name, only to order the wrong salt from a supplier using a different one. Older publications occasionally use alternative descriptors or abbreviations (like EPyBr), especially when discussing broader pyridinium-based systems or ionic liquids. My experience reading primary literature and catalog listings over the years has made me triple-check chemical names—one slip-up can set a project back by weeks.
Safety & Operational Standards
Working with pyridinium salts, including 1-ethylpyridinium bromide, means taking real steps to stay safe. The salt itself doesn’t pack the toxic punch of smaller alkylating agents, but improper handling exposes risk through inhalation, ingestion, or skin contact. Ethyl bromide and pyridine residue from incomplete reactions can amp up hazard levels, so diligent ventilation and PPE are non-negotiable. Good practice starts with gloves, eye protection, and clean workspace habits. Disposal follows local rules for halogenated organics—never down the drain. I once saw a student develop a rash from casual handling, so I teach that “benign” does not mean “harmless.” Documented toxicity remains moderate, yet ongoing research into its chronic effects and environmental fate makes proper respect crucial. The growth in undergraduate research puts an even larger burden on supervisors to enforce protocols, and technical regulations have only grown tighter in labs keen on chemical safety.
Application Area
1-Ethylpyridinium bromide’s calling card shows up in new energy materials, catalysis, and organic synthesis. Chemists prize it as both model compound and functional ingredient in ionic liquids, where the pyridinium framework enables robust, low-volatility systems that make battery research and green solvent science both safer and more efficient. Electrochemists benefit from its stable salt profile in electrolyte formulations, which influence ionic conductivity and lifecycle in flow batteries, lithium-ion cells, and emerging capacitors. In synthetic organic chemistry, it figures in quaternization reactions, as well as in phase-transfer catalysis, where shifting reactants between phases becomes more controllable. I once relied on it to simplify a cumbersome N-alkylation step, streamlining a process that chewed up hours beforehand. Its low toxicity and strong solubility parameters also lead to biomedical research, especially in drug delivery or as a foundation for novel antimicrobial materials. These diverse fields thrive on adaptability—and here, practical, reliable compounds form the backbone of innovation.
Research & Development
Recent years brought a surge in interest for tailoring ionic liquids to control viscosity, electrochemical window, and environmental compatibility. 1-Ethylpyridinium bromide stands out as a go-to “building block” for ionic liquid synthesis, giving chemists an entry point to adjust anions or further functionalize the pyridine ring. Research groups investigate how small changes in the alkyl or aryl substitutions shift properties like toxicity, hydrophobicity, and biodegradability. Applications spill into nanotechnology and biotechnology, where ionic liquids bridge gaps between traditional solvent science and materials engineering. In collaborative projects I’ve followed, 1-ethylpyridinium bromide gets used to check benchmarks against new, more complex systems; its reliability and cost-effectiveness make it a staple for method validation and process optimization. Outside the lab, startups exploring battery and sensor technology keep the compound on their inventory lists, focusing on scaling its synthesis and minimizing environmental impact during production.
Toxicity Research
Toxicological data on 1-ethylpyridinium bromide doesn’t command the spotlight the way more infamous chemicals do, but steady work has mapped its acute and chronic effects. The salt collects a moderate-to-low hazard classification, with some irritation risk at elevated exposures. Animal studies evaluate its routes of elimination and long-term biocompatibility, with research teams drilling down on biodegradation and aquatic toxicity as its use spreads in energy storage and pharmaceutical applications. Regulatory reviews reflect growing pressure on chemical manufacturers to track persistence in the environment and potential impacts on microbial life—especially now that the chemical pops up beyond purely academic use. My take, shaped by daily lab routines, puts heightened emphasis on residue management and responsible disposal. Every step toward fully mapping its toxicological profile reinforces safe-by-design approaches in new chemical development.
Future Prospects
With the explosion of interest in performance chemicals for sustainable technologies, 1-ethylpyridinium bromide has a future as a reference material and as a workhorse in greener solvent systems. As battery R&D pivots toward ionic liquids that balance eco-compatibility with high efficiency, expect more projects harnessing the pyridinium framework for safer electrolytes and advanced storage devices. Neuroscience, pharmaceutical pre-formulation, and advanced separations offer new outlets for exploration. Environmentalists and regulatory bodies will push transparency, forcing chemists to adapt production and recovery processes to tighten up lifecycle and cradle-to-grave management. Based on my time mentoring the next wave of chemists, I see the willingness to adapt safer, well-characterized compounds like this one—rather than jump straight to the most exotic structures—as a sign of growing scientific maturity. Chemistry communities need practical, trustworthy “standards” if they want to transform ideas into real-world progress.
What is the chemical structure of 1-Ethylpyridinium Bromide?
Understanding the Makeup
1-Ethylpyridinium bromide comes from a world of ringed molecules and charged particles. The backbone is pyridine, a six-membered ring almost like benzene, except one carbon in the ring swaps out for a nitrogen atom. On that ring, an ethyl group – two carbon atoms chained together – hangs off the nitrogen, making the molecule positively charged.
What keeps that positive charge in check is a bromide ion, a simple bromine atom holding a single negative charge. This salt-like relationship stabilizes 1-ethylpyridinium. The chemical formula displays this partnership as C7H10NBr, telling you at a glance every atom present.
Seeing the Pieces Fit Together
From years in the laboratory, I’ve learned that the real magic in molecules is in their shape. Imagine fastening an ethyl group to the nitrogen of a pyridine ring. That positive charge prefers balance, so bromide always tags along. The nitrogen, now attached to three things and carrying the charge, gives the compound fresh chemistry compared to plain pyridine.
The ethyl group bulks up the structure, adding flexibility and altering how this molecule interacts with others. In a classroom setting, I watched students draw the ring in hexagonal fashion, then stick the ethyl group straight off the nitrogen. The bromide shuttles around, separate from the main structure, but never far away.
Practical Use and Impact
I’ve seen 1-ethylpyridinium bromide appear in research searching for greener solvents and in experiments that look for new ways to control electrical charge. The ionic liquid field values structures like this, because the ions break apart easily in the right context yet stick together enough to create stable mixtures.
One big advantage: the molecule breaks free from old-school solvent rules. It can form liquids at much lower temperatures than you’d expect for a salt. Walk into a lab using ionic liquids and you see less reliance on volatile, flammable chemicals.
Safety also stands out. Traditional solvents have a reputation for fumes and fire hazards. Ionic liquids like this cut back on these problems. Less volatility means fewer accidents, and if your work keeps you near a fume hood all day, this shift in practice reduces both stress and harm.
The Broader Backstory
Diving into scientific literature, you see that pyridinium compounds – especially ones with simple alkyl chains like ethyl – play key roles in pharmaceutical research, catalysis, and new battery chemistries. They act as building blocks for more complex ionic liquids or help shepherd reactions toward cleaner, more predictable outcomes.
It isn’t only about what the molecule does in one reaction. Its structure – including the positive charge anchored on the nitrogen, the easy-to-swap bromide, and the stable ring – creates options for scientists looking for safer chemicals. It’s small changes in structure that often unlock benefits like lower toxicity, higher efficiency, or better recyclability.
Looking at Next Steps
From my own challenges with waste streams, I believe that understanding and harnessing molecules like 1-ethylpyridinium bromide opens doors to industry-scale improvements. By leaning on these stable, tunable structures, chemists can lower their environmental footprint, design easier recycling processes, and reduce exposure to hazardous compounds.
More education about how ions work and why their pairing matters could give the next group of chemists a stronger foundation. Any effort to replace risky solvents with custom ionic liquids needs both technical skills and an understanding of how small tweaks in molecular structure ripple all the way through workplace safety and environmental outcomes.
What are the typical uses of 1-Ethylpyridinium Bromide?
An Everyday Look at a Less-Than-Everyday Chemical
People working in labs don’t always talk about what goes on behind the bottles and jars they reach for—unless you ask about something like 1-ethylpyridinium bromide. This molecule rarely pops up in daily conversation, though you’ll hear a buzz about it in research groups digging into ionic liquids and modern chemical synthesis.
I first ran into this compound back in grad school. The task: find a stable ionic liquid for carrying out a tricky organic reaction. My advisor insisted we test a few pyridinium salts. 1-ethylpyridinium bromide made the cut, blending into the collection of chemicals that helped get cleaner results than old-fashioned solvents. Its unique mix of a positively charged ring and a bromide counterion offered enough thermal and chemical stability for us to push reactions a little harder or keep things running for longer periods.
Why Do Chemists Reach for It?
One of the first things seasoned chemists learn: solvents shape a reaction. 1-ethylpyridinium bromide often shows up as a building block for making ionic liquids. These liquids break some rules. Instead of acting just as solvents, they can help shuttle electricity or even serve as both solvent and reactant. They don’t evaporate away like water or ethanol, making lab life safer and greener—no strong smells filling the air.
A growing area of research uses these compounds in electrochemistry. At some university labs, you’ll see researchers testing ionic liquids with 1-ethylpyridinium cations in batteries and capacitors. The low volatility cuts down on leaks and increases safety for rechargeable batteries, which has always been a sticking point for consumer electronics. An MIT group reported better thermal stability for certain device prototypes when they swapped in these pyridinium-based ions.
Getting Creative with Synthesis
Synthesis teams have found other reasons to keep a vial of this compound around. Working with rare or unstable catalysts, sometimes traditional sodium or potassium salts gum up the works. 1-ethylpyridinium bromide dissolves quickly in both water and organic solvents, which gives reactions a helpful push. Some pharmaceutical researchers have used it to fine-tune how much active ingredient crystallizes out of a mixture, which can help with drug purity.
Material science teams in Europe publish results using this compound for shaping unconventional polymers. Special coatings that resist water and oils gain a boost in production when they use this salt instead of more famous alternatives.
Looking at the Future—and Challenges
This class of chemicals offers promise, yet the industry keeps a close eye on safety. Research journals and government agencies flag the need to better understand long-term effects on health and the environment. Even if a salt like this one helps make greener solvents, production and waste management must follow strict guidelines. People working on safer batteries or electronics seek recyclable or low-toxicity versions of these ionic liquids.
Some forward-thinking teams also talk about scaling up production. Basic chemistry in the lab is just one step. Factories looking to churn out tons of custom materials need cost-effective synthesis and responsible ways to handle waste. Students who learn about ionic liquids in university now could drive much safer and more sustainable processes in the future.
What is the recommended storage condition for 1-Ethylpyridinium Bromide?
Conditions That Make Sense for Chemical Longevity
Most chemists recognize the importance of taking care of chemicals, both for safety and for research quality. With 1-Ethylpyridinium Bromide, the key lies in stability. Small things make big differences. Put this compound in a tightly sealed container — not a battered old jar. Glass or HDPE works fine, but make sure the cap fits well, keeping humidity and air out where they belong. This keeps the powder from clumping or turning odd in color or performance.
Moisture: The Hidden Enemy
This salt doesn’t love water. Moist air can set off slow changes you probably won’t spot by eye, but over time, they register in lab results. The melting point starts drifting, or some reactions don’t go the way you expect. Dumping desiccant packets in your chem cupboard can sound tedious, but silica gel beats the headache of spoiled batches, repeat experiments, or contaminants in your data.
Keep the Heat Down
Every summer, old buildings turn labs into ovens. Heat speeds up chemical breakdown. Room temperature often means under 25°C. If work runs long hours or weekends, a dedicated chemical fridge keeps conditions more predictable. It’s easy to let this slide, especially since 1-Ethylpyridinium Bromide looks stable and unassuming, but heat sneaks up as a slow thief.
Keep Light Out
Clear glass bottles look sharp on a shelf. Sunlight and fluorescent bulbs, though, don’t do any favors for this compound. Over time, UV rays can eat away at chemical purity. An amber bottle, or just a dark corner of a cupboard, blocks the worst of the energy. From some frustrated afternoons with photo-sensitive reagents, I’ve seen low-tech solutions save the day.
Label Honestly and Clearly
No one likes digging through mystery bottles. Give the label a clear name and, most importantly, a date. Don’t skip safety information or hazard pictograms if applicable. If you re-bottle, jot down the transfer date. This helps anyone who inherits your workspace trust the stock.
Health and Safety: Science Protects Itself and Us
1-Ethylpyridinium Bromide doesn’t try to hurt you, but like any lab chemical, skin contact, inhalation, or eating lunch next to it isn’t wise. This makes strong storage choices even more relevant. Spills or crumbling containers can mean exposure, but with regular checks and sturdy storage, risk drops. I’ve seen old storerooms where lazy storage led to wasted material and avoidable accidents.
Take Home Wisdom from Experience
Reliable research starts in storage, not at the benchtop. Good storage conditions keep costs down and make science more trustworthy. There’s no grand secret. Dry, cool, and dark are the golden rules for 1-Ethylpyridinium Bromide. Any corner store carries silica gel, weather apps warn about heat, and every lab has space out of the sun. Attention to these details keeps projects on track, budgets under control, and researchers safe.
Better Supplies Serve Better Science
If any lab struggles with a stuffy storage room or wilting climate control, talk it over as a team. Even basic upgrades—a small fridge, airtight jars, or extra storage racks—make a night-and-day difference in chemical quality. There’s pride in a well-run stockroom, and one wrong turn on storage can waste weeks of research. Smart, careful storage isn’t about fussiness. It’s about setting everyone up for honest, reliable results.
Is 1-Ethylpyridinium Bromide hazardous or does it require special handling?
Looking Beyond the Lab Bench
Walking into a chemical storeroom, glass bottles stacked like chess pieces, every chemist faces the question: Does this one demand extra care? 1-Ethylpyridinium bromide crops up in research involving ionic liquids and organic synthesis, and anyone handling it wonders what kind of respect it commands.
Hazard Profile: Getting the Details Straight
This compound isn’t a household name, but the way it behaves deserves a closer look. According to the Globally Harmonized System (GHS), 1-ethylpyridinium bromide falls into the “irritant” category—think itchy skin, burning eyes, maybe coughing if you breathe in its dust. Government databases, including the European Chemicals Agency (ECHA), tag it for causing both skin and eye irritation. Accidentally eating or inhaling some could trigger nausea, redness, and headaches.
There’s hardly any data showing it’s a mutagen or carcinogen, so it doesn’t get lumped with the nastier chemicals, but it still doesn’t earn a “harmless” badge. If no one’s studied something at length, it’s safer to assume more risks might exist that haven’t popped up yet. Plenty of researchers, myself included, handle lesser-known salts like this one with the same respect they’d give more notorious chemicals. Even without spectacular dangers, basic precautions separate veterans from the careless.
Working Safely: The Basics Matter
I remember an old professor repeating, “Let the lab coat, gloves, and goggles do the worrying.” Working with 1-ethylpyridinium bromide follows this rule. Good sense says: don’t touch it without gloves. Splash-proof eyewear is non-negotiable because that white, crystalline powder stings like soap in the eye, only worse. Have it open under a fume hood, not the bench, since dust likes to take flight and travel right into the airways.
If it hits the skin, rinse with water the way you would for most laboratory chemicals. Eye exposure? Forget pride—dash for the eyewash station, and let water flow for a full quarter hour. Most labs keep safety showers for a reason, and this salt justifies keeping those on standby. Clean up spills with an eye toward dust suppression: damp cloths work better than dry paper towels. A closed, labeled waste container does more than keep the lab neat; it keeps others out of harm’s way.
From Storage to Disposal: Practical Approaches
Keep the container sealed tight, stored in a dry spot, and clearly labeled. Stick it somewhere away from acids or bases—mixing unknown pairs can bite you. Disposal lines up with your standard hazardous waste protocol. In my experience, no one ever regretted overlabeling their waste bottle. Local regulations might tell you to separate halogenated and non-halogenated waste, so double-check before tossing anything.
What Drives Caution
The lesson: a lack of horror stories doesn’t mean free rein. Chemical safety depends on anticipating the unexpected, not just following what’s already written on a safety sheet. Not every hazard screams with skulls and crossbones; many chemicals hide their dangers behind plain packaging.
Years in the lab have taught me shortcuts compound risk. For 1-ethylpyridinium bromide, the routine—gloves, goggles, fume hood—answers the question. Hazards don’t disappear with familiarity; if anything, experience means not getting sloppy.
What is the purity or grade specification of 1-Ethylpyridinium Bromide sold?
Chemical Grade Isn’t Just a Detail
Purity speaks louder than marketing tags in chemistry labs. Pick up a bottle of 1-ethylpyridinium bromide and the first thing to check isn’t the fancy label, but the fine print where purity sits. In practice, most suppliers ship this compound with a purity between 97% and 99%. Lab techs expect nothing less, especially when each decimal can tilt the outcome of a reaction.
I remember working on a synthesis project, thinking purity was a minor point. The supplier listed 98% for the batch. Turns out, that last 2% came with enough moisture and colored residues that results grew inconsistent and separation needed extra time. Those “small” impurities stick around—water, pyridine, or even leftover reactants. Few chemists get excited about spending twice as long washing and drying their product. Once, we had to toss three days’ worth of effort because a lower grade let side products slip past. Nobody wants to burn money and time like that.
Spotting the Gaps in Purity
Some folks depend on certificates of analysis as gospel, but not every supplier checks the same things. Good ones give a detailed breakdown: water content, chloride, color, and that crucial heavy metals line. Anything less, and you run blind. If high-resolution NMR and HPLC data don’t come with, that’s a red flag. A “white to off-white powder” sounds reassuring until those off-white bits creep into your results.
Not all buyers need 99.9%. Some industrial tasks, like handling large ionic liquid batches, can tolerate slightly less pure lots, as long as the impurities don’t interfere. Still, nobody wants surprise elements. For anyone scaling up, those small fractions of bromide or leftover solvents build up and muck with catalyst performance, lower yields, or cause headaches in downstream steps. In research, chasing high purity pays off—murky data or ambiguous peaks in spectroscopy burn away confidence in every result. Reliable research thrives on clean reagents.
What Makes Purity Worth the Price?
Paying more up front for a clear certificate often saves in other ways. Higher purity batches trim time spent on column cleanups and re-crystallizations. Techs don’t have to waste precious solvent trying to fish out extra water. Waste disposal costs drop. Fewer failed reactions mean the wallet breathes easier. For companies, recalls or failed production runs cost more than a premium price tag on a tightly controlled compound.
Check where your 1-ethylpyridinium bromide comes from. Suppliers in the US, Europe, and reputable Asian labs follow strict quality programs and update documentation. Some list not only the purity, but also batch-to-batch variation, keeping surprises rare. Buying from less established sellers for a bargain might end with more questions than answers.
Raising the Bar for Quality
Rely on transparent companies that lay out raw data from every batch. Push for tighter specs on water, halides, and metallic traces. Building a good working relationship with your chemical provider keeps supply chains honest. Request samples accompanied by full characterization. Good suppliers don’t shy away from third-party testing. Labs that set higher standards for purity square away risks from the start.
1-Ethylpyridinium bromide isn’t cheap, but nothing eats money like wasted reactions caused by subpar grade. Investing in true 98-99% material—and demanding proof—delivers steady results. Clean chemistry isn’t fancy: it’s just honest, reliable, and built on details that pay off in the long run.
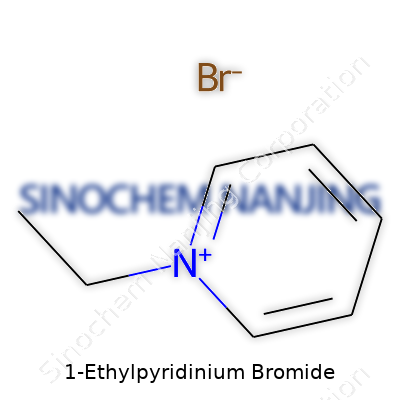
| Names | |
| Preferred IUPAC name | 1-ethylpyridin-1-ium bromide |
| Other names |
1-Ethylpyridinium bromide N-Ethylpyridinium bromide Ethylpyridinium bromide |
| Pronunciation | /waɪˌɛθ.ɪl.pɪˈrɪd.i.ə.ni.əm ˈbroʊ.maɪd/ |
| Identifiers | |
| CAS Number | 1124-11-4 |
| Beilstein Reference | 63619 |
| ChEBI | CHEBI:132578 |
| ChEMBL | CHEMBL1478505 |
| ChemSpider | 22988349 |
| DrugBank | DB04258 |
| ECHA InfoCard | 100.047.864 |
| EC Number | 216-528-8 |
| Gmelin Reference | 61383 |
| KEGG | C02713 |
| MeSH | D017870 |
| PubChem CID | 11770644 |
| RTECS number | UX8775000 |
| UNII | ZZR98BTE8F |
| UN number | UN2811 |
| CompTox Dashboard (EPA) | DTXSID7044158 |
| Properties | |
| Chemical formula | C7H10BrN |
| Molar mass | 186.07 g/mol |
| Appearance | white to off-white solid |
| Odor | Odorless |
| Density | 1.37 g/cm³ |
| Solubility in water | Very soluble |
| log P | -2.1 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.2 |
| Basicity (pKb) | 4.86 |
| Magnetic susceptibility (χ) | -62.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.546 |
| Viscosity | 84 cP (25 °C) |
| Dipole moment | 4.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 247.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -85.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P270, P273, P301+P312, P330, P501 |
| NFPA 704 (fire diamond) | 2-0-0 |
| Flash point | 129°C |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
1-Ethylpyridinium chloride 1-Ethylpyridinium iodide 1-Butylpyridinium bromide 1-Methylpyridinium bromide |

