1-Chloro-2-Propanol: A Closer Look at a Versatile Chemical
Historical Development
Chemistry often finds its way into everyday corners of life, even if many people have never heard of 1-Chloro-2-Propanol outside a laboratory. Back in the mid-20th century, the push in industrial chemistry to create new intermediates for everything from pharmaceuticals to plastics led researchers to explore halogenated alcohols. 1-Chloro-2-Propanol didn’t just pop up by chance; it showed up as part of the run to improve reaction routes and create building blocks for new compounds with unique properties. Researchers working in the 1950s and 60s were hungry for new synthetic ways to alter simple alcohols, and chlorination was a natural route. Documentation from older journals hints at its adoption in fields that wanted reliable, reactive intermediates without the bulk production limitations associated with larger or more complex molecules.
Product Overview
1-Chloro-2-Propanol belongs to the category of halohydrins, which means it contains both a halogen and a hydroxyl group on adjacent carbons. Its structure gives it more than one trick up its sleeve: the chlorine provides interesting reactivity for substitution, while the alcohol end makes it compatible with a range of catalytic and synthetic processes. It doesn’t take much for chemists to realize the potential of combining these moieties in a single molecule, especially when aiming to synthesize epoxides or to serve as intermediates for other specialty chemicals.
Physical & Chemical Properties
In the beaker, 1-Chloro-2-Propanol usually appears as a clear or slightly yellow liquid. Its solubility in water and organic solvents like ethers gives it practical value for those mixing it into multi-phase reactions. Molecular weight falls around 94.5 g/mol, and its boiling point, somewhere above 100°C, makes it stable enough to withstand moderate heating. The presence of the chlorine atom changes its volatility and introduces a layer of reactivity you won’t get with plain propanols. This classification as a halohydrin also means that it can serve as a handy precursor to epoxides by simple intramolecular reactions under basic conditions.
Technical Specifications & Labeling
While labeling standards differ by region, chemical labeling always needs to keep users informed about hazards and handling. Standard labels usually include its chemical name, formula (C3H7ClO), and safety symbols signaling irritant or harmful effects. Specifications for lab or industrial supply often call for minimum purity, most often above 98 percent, to ensure reliability in research and manufacturing. Suppliers flag any trace contaminants, because slight impurities could throw off catalyst systems or poison sensitive syntheses.
Preparation Method
One of the classic approaches involves the reaction of propylene oxide with hydrochloric acid, which yields 1-Chloro-2-Propanol through a ring-opening process. This method became popular due to its relatively straightforward nature and high conversion rates. Process engineers watch temperature and acid strength to push the reaction toward mono-chlorination rather than getting a mix of chlorohydrin isomers. Some labs prefer chlorination of 2-propanol under controlled conditions, but side products and cleanup hassles usually tip the odds toward the propylene oxide route, which sets up better yields and cleaner separations.
Chemical Reactions & Modifications
For synthetic chemists, this molecule offers a fun playground. The alcohol end opens up oxidation, esterification, and etherification avenues, while the chlorinated site suits nucleophilic substitution by all sorts of agents—ranging from amines to alkoxides. A favorite trick involves treating it with a base like sodium hydroxide to kick off an epoxidation, yielding propylene oxide, which itself is big business in the plastics and coatings world. In other cases, you might see chemists swap the chlorine out for a range of functional groups, crafting new intermediates for custom syntheses, dyes, or specialty pharmaceuticals.
Synonyms & Product Names
Scientists and industry use a handful of alternate names so it pays to recognize them on paperwork and safety documents. You’ll see 1-Chloropropan-2-ol, 2-Hydroxy-1-chloropropane, Isopropanol chloride, or simply Chlorohydrin. Each name highlights a different feature or nomenclatural system, and cross-referencing them becomes important, especially for those handling international shipments or regulatory paperwork where naming quirks can cause delays or miscommunication.
Safety & Operational Standards
Extended contact with chlorinated alcohols rarely bodes well for skin or lungs. Like a lot of halogenated solvents, 1-Chloro-2-Propanol carries the risk of irritation and toxic effects, so the standard kit of gloves, goggles, and well-ventilated spaces comes in handy—without it, headaches and skin burns can follow. Industrial plants install scrubbers and tight controls on waste disposal because spills in soil or water can stick around, causing lingering environmental impact. Proper chemical hygiene—like tight seals and good labeling—protects everyone from accidental ingestion or inhalation. Groups like OSHA and the European Chemicals Agency offer compliance roadmaps, but in busy labs and warehouses, a culture of vigilance often proves more effective than checklists alone.
Application Area
This chemical finds most of its industrial strength in serving as an intermediate. In making propylene oxide—a key ingredient in polyurethanes and other plastics—1-Chloro-2-Propanol stands as one of the most effective precursors. Polymer and resin manufacturers run large-scale processes using this intermediate, allowing for efficient production of flexible foams or rigid coatings. Beyond that, smaller-scale uses, such as starting material for synthesizing custom pharmaceuticals or fine chemicals, add to its profile. Its versatility in organic reactions means research teams always keep it around as a go-to reagent for exploring new molecular scaffolds or reaction pathways.
Research & Development
Research labs still dig into new uses and reaction mechanisms for 1-Chloro-2-Propanol. Academic publications continue to map out the best ways to utilize its reactive centers—some studies explore asymmetric syntheses to generate enantiomerically pure derivatives, which could unlock new drug candidates with fewer side effects. Collaborative projects between universities and industry focus on cleaner, safer chlorination steps, trying to squeeze down waste or hazardous byproducts. Researchers also examine alternate catalysts for its substitution reactions, aiming to lower the reaction temperatures or swap out problematic solvents. The chemical’s flexibility keeps it on the radar for folks working in green chemistry and process optimization.
Toxicity Research
Toxicologists have placed chlorohydrins like 1-Chloro-2-Propanol under the microscope for decades. Acute exposure studies show it irritates the eyes, skin, and mucous membranes, and chronic exposure can affect central nervous function. Reports document cases of accidental poisoning with symptoms like dizziness, nausea, and even kidney or liver effects at higher doses. Animal studies indicate possible mutagenic effects, prompting regulatory bodies to keep a close eye on occupational exposure. Researchers continue to test breakdown products and metabolic pathways in mammals, checking for long-term carcinogenic risks. Routine medical screening and air monitoring offer front-line defenses, catching problems before they escalate.
Future Prospects
Looking ahead, industry and academia both have their eyes on process safety and green chemistry innovations for 1-Chloro-2-Propanol. With regulatory pressure pushing for products and production methods with smaller ecological footprints, manufacturers chase cleaner synthetic routes and seek out better containment systems. Advances in automation, sensor networks, and waste minimization equipment could help shrink the risk footprint for workers and the environment. Research teams might uncover new derivatives or applications, especially in pharmaceutical synthesis, biocatalysis, or even as reactants for emerging nanomaterial platforms. The future of this molecule rides as much on practical safety and smart development as it does on chemistry itself—a mix of traditional know-how and the drive for sustainable industry.
What is 1-Chloro-2-Propanol used for?
More Than Just Chemistry: Why This Compound Matters
Science classrooms often make chemistry sound distant from daily life, like the formulas only belong on a chalkboard. We tend to forget that chemicals form the foundation for countless things we touch, build, or heal with. 1-Chloro-2-propanol is a good example. It might have a name straight out of a research journal, but it ends up flowing through plenty of essential industries.
Production of Pharmaceuticals
I once talked with a pharmacist who explained how active ingredients for common cold medicines and allergy pills come from lengthy steps in chemical synthesis. 1-Chloro-2-propanol gets added to pharmaceutical batches because of the way it helps build intermediate molecules. In certain cases, it steps in as a building block for beta-blockers or antiviral medicines. The safety profile for any chemical used around human health gets long scrutiny, which tells us how important this compound's stability and predictable reactivity are for companies that make these vital products.
Specialty Chemicals and Solvent Uses
Factories that blend specialty chemicals rely on solvents that dissolve ingredients cleanly without bringing unwanted side effects. 1-Chloro-2-propanol checks this box for producers of dyes, coatings, and adhesives. Some formulations use this chemical to fine-tune viscosity and help spread pigments evenly. Even in small amounts, its presence shapes the outcome for paints or industrial lacquers destined for construction and automotive plants.
Manufacturing of Plasticizers and Epoxides
If you've worked in building materials or plastics, you know how chemicals like this one help tweak flexibility. When raw material prices rise, or rules change about plasticizer safety, manufacturers look for ways to reduce risk without sacrificing toughness. 1-Chloro-2-propanol feeds into reactions that create certain glycidyl ethers and other additives that end up in flexible PVC or polyurethane foams. Without these additives, your phone cable would crack easily, and your yoga mat would turn brittle after a few uses.
Laboratory Research and Synthesis
Back in graduate school, lab benches filled up with little bottles labeled with long names like this one. 1-Chloro-2-propanol showed up during organic synthesis experiments where precision matters. Because it reacts in predictable ways, chemists rely on it to test theories, grow small batches of new molecules, or develop cleaner synthesis strategies. Even though the general public rarely sees what happens at this stage, smooth-running research brings new inventions to market faster. Safer, more efficient production can spark ripple effects across medicine, electronics, and sustainability.
Health and Environmental Concerns
Exposure to any chlorinated compound raises eyebrows for a good reason. Mishandling can lead to toxic fumes or contaminated water, something I've seen news crews report after accidental plant leaks. The Centers for Disease Control and Prevention (CDC) and other agencies monitor safe handling guidelines for workers and recommend spill procedures. Producers and end users need to commit to proper containment, recycling, and waste treatment to prevent trouble down the road.
Looking Ahead: Making Safer Choices
As industries shift toward greener processes, 1-Chloro-2-propanol faces pressure from stricter regulations and customer demand for safer alternatives. Chemical engineers keep testing bio-based routes and recycling strategies. Some firms set up closed-loop systems to keep emissions in check, while researchers in Europe and Japan test non-chlorinated substitutes that offer similar flexibility and reactivity without the same risks. Real breakthroughs will come when companies and labs pool knowledge and invest in safer supply chains. It’s a slow process, but I’ve learned that stubborn problems need patience and creative thinking on every level.
What are the safety precautions when handling 1-Chloro-2-Propanol?
Recognizing the Risks
1-Chloro-2-Propanol shows up in labs and workplaces, often as a solvent or intermediate. Its clear liquid form looks harmless at first glance, but just a small misstep can set off trouble for skin, eyes, lungs, or even deeper inside. Years in the chemical industry taught me the hazards that can sneak in from a tiny splash or overlooked whiff of vapor.
Protecting Yourself: Simple Steps That Matter
Lab coats, splash goggles, and chemical-resistant gloves form the basics. Forgetting gloves, even for a minute to check a bottle or open a door, exposes skin to burns or rashes. I’ve seen coworkers miss a patch on their wrist and end up with red, blistered skin. Eye exposure brings stinging pain, redness, and lasting damage that doesn’t always heal. Goggles should fit snug against your face, and I double-check mine by shifting my head around before every job.
Fumes creep into lungs quickly, even when you don’t smell anything in the air. Having solid ventilation makes a difference. Fume hoods and exhaust fans keep vapors away from your nose and mouth. If a lab doesn’t have them, I reach for a respirator with cartridges rated for organic chemicals—no shortcuts there. Chemical information sheets warn that 1-Chloro-2-Propanol can irritate the nose, throat, and lungs, and studies point toward nerve damage with repeated exposure.
Keeping the Workspace Safe
I never work alone with strong chemicals, no matter how basic the task. Sharing a room with a colleague means someone can help in case of a spill or splash. Spills bring the biggest headaches. Absorbent pads or vermiculite work fast, but using bare hands to scoop up mess leaves you open to burns or skin uptake. I remind coworkers to use chemical-resistant tools and never rags meant for water or general cleaning.
Storage often gets overlooked. Bottles should seal tightly and stay out of sunlight or heat. Locking cabinets help prevent accidental access, especially in workplaces where visitors might not recognize hazard warnings. It’s easy to trust that containers will hold up, but old, cracked caps and faded labels bring risk. In my experience, a sharpie mark and a date on every bottle go a long way toward safe tracking.
Learning and Training That Sticks
Experience doesn’t excuse anyone from ongoing training. Chemical suppliers include updated safety data sheets with every order for a reason, and reviewing these sheets saves trouble. Regular safety drills keep everyone sharp on eyewash stations, showers, and emergency response. A three-minute walk-through on where to find showers and how to flush eyes beats panicking if the real thing happens.
Companies and schools set their own procedures for emergencies, but every routine should cover the same ground—immediate washing after exposure, calling for medical backup, and reporting any symptoms of dizziness or breathing trouble. Those steps almost always mean the difference between a quick fix and a lasting injury.
Building a Safety-First Mindset
The safest labs and workshops I’ve worked in never see protective gear as a chore or afterthought. Nobody feels shy about reminding a coworker to put on goggles or swap out old gloves. That kind of openness creates a culture where accidents turn rare, and people go home the way they showed up. Mistakes still happen, but prevention and preparation always help catch them early.
Every bottle of 1-Chloro-2-Propanol in a cabinet or on a lab bench comes with lessons memos or trainings might overlook. Day-to-day routines, a habit of double-checking, and looking out for each other bring the most effective protection.
What is the chemical formula and structure of 1-Chloro-2-Propanol?
Understanding the Formula and Structure
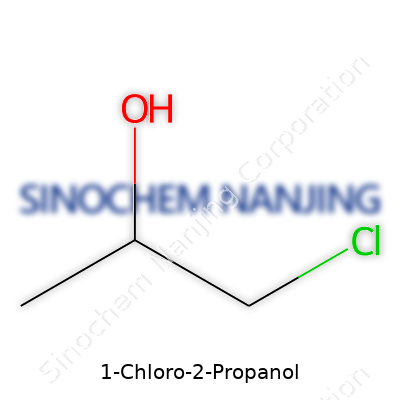
1-Chloro-2-propanol shows up in labs and industry, sometimes called simply as an intermediate for making other chemicals. At its core, it’s a three-carbon molecule. One carbon connects to a chlorine atom in place of a regular hydrogen—hence “chloro.” Another carbon holds onto a hydroxyl (OH) group. The chemical formula is C3H7ClO. For anyone familiar with organic chemistry, the structure lines up like this:
Start with propane’s backbone, which stacks three carbons in a row. Swap one hydrogen on the first carbon for a chlorine atom, and on the adjacent carbon, replace a hydrogen with a hydroxyl group. In a line, the layout looks like this: Cl–CH2–CH(OH)–CH3. The way these atoms bond makes the compound a “secondary alcohol” because the OH is attached to the second carbon, not the first or last.
Industry and Research Value
Small changes in a molecule can shift its behavior dramatically. 1-Chloro-2-propanol’s structure turns it into a valuable building block for synthesizing more complex chemicals. I’ve read about uses in pharmaceuticals, inks, and even some surfactants, thanks to the way that chlorine and hydroxyl groups guide further reactions.
Chemists rely on its ability to react with nucleophiles (compounds that want to share electrons) because the chlorine can be swapped out. This property makes it easier to stick new groups onto the carbon backbone. Picture a busy workshop, where a team can pop off the chlorine and plug in whatever tool the job requires—anything from amines to organic acids.
Handling Hazards and Safety
It’s one thing to know what a formula means in the abstract. It’s another to see safety warnings in bold red on the side of the container. From my hands-on experience, working with compounds like 1-chloro-2-propanol calls for real care. The chlorine not only makes the molecule reactive but also brings some risk. The compound’s volatility means it can enter the air—so proper ventilation, gloves, and eye protection make a real difference.
Direct contact or inhalation can irritate the eyes, skin, and lungs. Some reports and studies in chemistry journals have flagged potential toxicity, especially after long-term exposure. The U.S. EPA and similar agencies pay attention to chlorinated organics because they can persist in the environment or break down into more harmful substances if not handled right.
Responsible Chemistry and Solutions
Several chemistry professors I’ve worked with push for better methods: using less hazardous starting materials or adopting greener routes, for example. Some innovations swap out chlorine altogether or use catalysts that limit waste. The worldwide push for green chemistry isn’t just buzzwords—it comes from cases where accidental spills or improper disposal led to groundwater problems.
For labs and factories, investing in full training and up-to-date safety data pays off long-term. Clear labeling, reliable ventilation, and emergency response plans help keep both workers and the environment safer. For students and researchers, having easy access to molecular models and risk data helps bridge the gap between textbook knowledge and real-world application.
Why It Matters
1-Chloro-2-propanol’s straightforward formula unlocks a lot of complexity down the line. The right information—both about how it works and how to use it safely—lets people innovate without needless risk. Chemicals like this remind us that even small details in structure and handling echo out into broader impacts, both for industries and communities nearby.
How should 1-Chloro-2-Propanol be stored?
What 1-Chloro-2-Propanol Brings to the Table
I remember handling chemicals in cramped college labs, always anxious about what each liquid could do—some good, some downright risky. 1-Chloro-2-Propanol falls into that risky side. Anyone who’s spent time with this colorless, sometimes pungent material knows: you don’t want carelessness within arm’s reach of your workspace. This stuff burns the skin and eyes, and its vapors wreak havoc in poorly ventilated rooms. At home or in the workplace, it never acts like a friendly desk plant.
Solid Practices Keep People Safe
Chemistry culture often skips practical advice. My old mentor would holler from across the storage room, “Keep it cool and keep it shut!” He knew that vapor pressure in the wrong bottle wrecks everyone’s morning. The place for 1-Chloro-2-Propanol is always away from flames, sparks, and heat. Below 25°C (77°F) is a smart upper limit—think cool, shaded rooms before thoughts drift to fridge space. Sunlight pushes up the temperature and boosts breakdown, so no window ledges or car trunks.
Humidity and fresh air matter. No one wants leaky containers, so glass or sturdy plastic bottles with tight lids prove dependable every time. Avoid using metal. This chemical chews through softer metals and lines, turning what looks like a secure stash into a slow leak. I’ve seen folks store solvents alongside acids or bases like they’re interchangeable—1-Chloro-2-Propanol absolutely doesn’t play nice with strong oxidizers or acids. Mixing means more than a terrible mess; it runs the risk of toxic fumes or fire.
Why Small Errors Add Up
Small labs, garages, and supply closets aren’t immune to disaster. Take spills. It’s not just about a slick floor; this chemical evaporates and fills the air with harshness, catching nose and throat in just a couple breaths. Ventilated rooms, fitted with exhaust hoods or at least an open window, block most problems before they start. Storing bottles on lower shelves, away from edges, prevents accidental falls. Even a well-placed absorbent pad or chemical tray beneath the bottle stops small leaks from turning big.
Training shapes safer habits. That story from a friend who opened a bottle without gloves still sticks with me. She learned fast—cool water and a trip to the urgent care. Posting labels and instructions mounts reminders for new folks and old hands alike. Regular audits, reading checklists, and quick reminders at the start of the day build a kind of muscle memory around safe handling.
Building a Healthier Chemical Culture
People deserve to come home from work in the same shape they left. Chronic exposure, even at low levels, sets up chances for headaches and liver strain. Regulations—from OSHA’s directives to local statutes—guide the best storage setups and emergency actions. By sticking to these standards, I’ve seen even small operations keep serious incidents off the books entirely. First step: buy only what you actually need, so half-used bottles never linger past their best days.
Proper disposal closes the circle. Holding onto expired product tempts fate. Partner with certified chemical waste handlers and skip the temptation of dumping drain or trash can. The planet needs that kind of respect, and so do all the folks down the line.
What are the physical and chemical properties of 1-Chloro-2-Propanol?
Physical Characteristics Worth Knowing
1-Chloro-2-propanol sits among the simpler organochlorine compounds. In the flask, it looks like a clear, oily liquid. Pour it out and you’ll catch a noticeable odor—sharp but not overpowering. It feels slippery between your fingers, and if you leave it open, it creeps through the room, hinting at its somewhat volatile nature.
Boiling picks up at around 127°C, so it’s more robust than some solvents but still manageable under standard lab ventilation. It chills to a solid only below -70°C. Seeing it bead on a benchtop makes you notice its density—around 1.17 grams per cubic centimeter, heavier than water and, because of that, it’ll settle at the bottom if you mix the two. Toss a few drops in water, and it’ll dissolve without a fight; its partial polarity bridges the gap between fully water-loving and oil-leaning properties.
Spotting Reactivity Patterns
Its reactivity stands out because of the chloro group attached to a molecule with a hydroxyl (alcohol) group. In day-to-day terms, this means both patience and caution come into play. That chlorine—bonded to the first carbon—acts suspiciously in the presence of nucleophiles; it likes to swap out when the right chemical comes along. Water, strong bases, or certain metals can trigger substitution or elimination reactions. The hydroxyl side introduces the chance for dehydration, willingness to form esters, and it can play in the world of hydrogen bonding.
In hands-on use, these features sneak up during storage or transport. Moisture or traces of acid or base can push the compound toward side products—sometimes to epoxides, sometimes to more chlorinated byproducts. This unpredictability matters in larger operations, especially where waste and byproduct control kick into regulatory territory.
Safety Matters
Experience around halogenated organics tells me not to take shortcuts—this goes double for anything with both a chloro and alcohol group. Splash it on skin and you’ll notice irritation or even blistering with enough exposure. Breathe the vapors and your eyes and lungs won’t thank you. There’s evidence from industry and academic reports that repeated handling without care can lead to liver and kidney problems. Looking at European Chemicals Agency data, the compound shows moderate toxicity to aquatic organisms. This brings environmental stewardship into play, not just personal protection.
Smart Handling and Practical Solutions
Improvement chances lie in engineering controls. Well-designed fume hoods and locked chemical storage keep vapors at bay. Gloves that resist chlorinated solvents—nitrile over the thin latex fare—hold up well. Better labeling and education in lab spaces help young chemists and workers avoid routine mistakes. On a large scale, chemical manufacturers use closed systems to limit fugitive emissions. Waste streams run through activated carbon to snatch up organics before effluent leaves the building.
There’s room to cut down on hazardous residues with process improvements. Some groups switch over to safer alcohols for similar synthesis steps, only using chlorinated intermediates when truly needed. Using modern analytical equipment—GC, LC-MS—lets labs track where the chemical ends up, so clean-up and recycling don’t become afterthoughts.
Looking Ahead Responsibly
Working with 1-chloro-2-propanol gives reminders about balancing chemistry’s benefits against real risks. Its physical and chemical quirks make it valuable for certain syntheses and transformations. Safe practices and smart regulation keep the benefits while limiting harm to health and the environment. Built from personal experience, careful handling and continuous learning remain the best protection.
| Names | |
| Preferred IUPAC name | 2-Chloropropan-1-ol |
| Other names |
1-Chloropropan-2-ol 2-Hydroxy-1-chloropropane Monochlorohydrin β-Chloropropanol 1-Chloro-2-hydroxypropane |
| Pronunciation | /ˈklɔːroʊ-tuː-proʊˌænɒl/ |
| Identifiers | |
| CAS Number | 78-89-7 |
| Beilstein Reference | 1209225 |
| ChEBI | CHEBI:51700 |
| ChEMBL | CHEMBL16852 |
| ChemSpider | 16318 |
| DrugBank | DB14096 |
| ECHA InfoCard | 100.004.016 |
| EC Number | 200-949-6 |
| Gmelin Reference | 8214 |
| KEGG | C02642 |
| MeSH | D015242 |
| PubChem CID | 8055 |
| RTECS number | TC9625000 |
| UNII | J730B2F1UL |
| UN number | UN1992 |
| CompTox Dashboard (EPA) | DTXSID0022273 |
| Properties | |
| Chemical formula | C3H7ClO |
| Molar mass | Molar mass: 78.54 g/mol |
| Appearance | Colorless liquid |
| Odor | mild, chloroform-like |
| Density | 1.07 g/mL at 25 °C(lit.) |
| Solubility in water | miscible |
| log P | 0.16 |
| Vapor pressure | 3.7 mmHg (at 25 °C) |
| Acidity (pKa) | 15.93 |
| Basicity (pKb) | 1.86 |
| Magnetic susceptibility (χ) | -9.73×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.421 |
| Viscosity | 5.1 mPa·s (20°C) |
| Dipole moment | 2.05 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 330.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –120.5 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -586.6 kJ/mol |
| Pharmacology | |
| ATC code | D08AX10 |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin irritation, causes serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07, GHS08 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H312, H315, H319, H332 |
| Precautionary statements | P210, P261, P264, P271, P280, P301+P312, P330, P304+P340, P305+P351+P338, P311, P303+P361+P353, P370+P378, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0 |
| Flash point | Flash point: 93 °C |
| Autoignition temperature | 225 °C |
| Explosive limits | 4.8–18.0% |
| Lethal dose or concentration | Lethal dose or concentration (LD50/LC50): Oral rat LD50: 2600 mg/kg |
| LD50 (median dose) | LD50 (median dose): 2070 mg/kg (rat, oral) |
| NIOSH | SN2100000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Chloro-2-Propanol: "No specific OSHA PEL established |
| REL (Recommended) | REL: 9 mg/m³ |
| IDLH (Immediate danger) | IDLH: 100 ppm |
| Related compounds | |
| Related compounds |
1,2-Propanediol 2-Chloropropanol Isopropanol 2-Propanol Glycidol |

