The Evolving Journey of 1-Butylpyridinium Hexafluorophosphate: From Early Discovery to Modern Application
Historical Development
Looking back on how 1-butylpyridinium hexafluorophosphate came about, it’s hard to miss the impact that curiosity-driven research had on advancing ionic liquids. Chemists in the late twentieth century hunted for salts that could stay in liquid form at room temperature, mainly to sidestep the volatility and flammability of common organic solvents. The butylpyridinium family emerged from this experimental playground. Once research groups noticed its stability and low vapor pressure, adoption in academia and industry picked up pace. Efforts to expand the list of viable ionic liquids pushed 1-butylpyridinium hexafluorophosphate into the spotlight, especially as environmental concerns grew over traditional solvent use.
Product Overview and Properties
1-butylpyridinium hexafluorophosphate usually shows up as a colorless to pale yellow liquid, almost odorless, often catching newcomers off guard since many solvents have strong smells. Its structure, built from a butyl chain attached to a pyridinium ring and paired with a hexafluorophosphate anion, leads to some unique features. Its melting point sits well below many salts, offering flexibility in different labs and industrial settings. It rarely evaporates under ambient conditions, one of the reasons researchers lean toward it when seeking alternatives to volatile organic compounds.
Physical and Chemical Nuances
On the bench, the viscosity stands out. Stirring or pouring takes more effort than with typical organic solvents, slowing down mixing in large setups. The low volatility means those who work with it don’t fill the lab with fumes, and that contributes to safer workspaces. Its solubility profile often piques chemists’ interest: it mixes nicely with many polar solvents but struggles with very non-polar ones. Electrochemical stability also puts it ahead of a lot of other ionic liquids, and that resilience helped it carve a niche in battery research.
Technical Specs and Labeling in Practice
Seeing the detailed technical labels in laboratory supply rooms, I’m reminded of the long string of numbers and symbols that define this compound. 1-butylpyridinium hexafluorophosphate usually arrives with purity above 98%, with moisture content tightly controlled to prevent unwanted reactions. In daily tasks, small changes in moisture sometimes compromise results, driving chemists to treat their bottles with extra care. Barcodes, hazard pictograms, and systematic names fill the sides of containers, targeting both safety and traceability.
Preparation: Realities of Synthesis
Crafting 1-butylpyridinium hexafluorophosphate, especially in scale-up settings, often starts from pyridine or butyl halides, and manufacturers rely on straightforward alkylation reactions followed by anion-exchange steps. Using hexafluorophosphate salts such as potassium or sodium forms, the desired ionic liquid forms as an oily layer above an aqueous waste phase. Laboratory synthesis brings challenges, especially regarding handling, since hexafluorophosphate salts decompose in the presence of water or acids, sending skilled workers into strict glovebox routines. Purification often runs through repeated washes and drying under reduced pressure.
Chemical Reactions & Avenues for Modification
The chemical backbone of butylpyridinium allows room for functional tweaks. Changing the alkyl chain length, swapping in functional substituents, or altering the anion transforms its solubility and thermal properties, letting researchers tune the liquid for specific challenges. Electrochemists take advantage of its strong cathodic and anodic windows in supercapacitor and battery projects. As catalyst supports, one simple modification can open up opportunities for immobilizing complexes or enzymes, increasing efficiency or recyclability. The dense fluoride shell on the hexafluorophosphate unit tends to keep it unreactive, but with clever design, chemists steer the reactivity of the overall molecule to benefit from both stability and selectivity.
Names, Synonyms, and Industry Usage
In day-to-day lab discussions, shorthand names like “BuPy PF6” or “1-BuPy PF6” flow easily, reflecting how chemists trim down multi-word names. Sometimes the parent compound gets name-dropped as N-butylpyridinium hexafluorophosphate, but confusion almost never arises given the small pool of similar ionic liquids in regular use. Synonyms appear in patent filings, journal articles, and safety documentation as a way to link older literature to current research.
Safety Standards People Actually Follow
Despite the appealing physical properties, 1-butylpyridinium hexafluorophosphate demands respect when handling. Manufacturers point to the risks associated with fluorinated anions—when overheated or mixed with strong acids, these salts can release hazardous gases. In practice, most laboratory teams stick to using gloves and protective eyewear, with careful waste collection to block accidental hydrolysis or decomposition. Over the years, training lab newcomers to look out for leaks and drips has become a routine part of onboarding, just as important as teaching them how to measure or store the material.
Application Stories from the Field
Electrochemistry circles often celebrate the rise of ionic liquids thanks to this compound’s ability to support robust current flow at low temperatures. Some battery projects make use of its stability and conductivity in electrolytes. In organic synthesis, it frequently acts as a “green” solvent, offering an alternative to hazardous reagents and opening new pathways for selectivity. This adaptability breathes life into a range of industries, from plastics and chemical engineering to pharmaceutical R&D, where its mildness benefits delicate reactions.
Research & Development: Where Curiosity Drives Breakthroughs
Researchers push deeper into the use of 1-butylpyridinium hexafluorophosphate as both a medium and a component of new materials. Work in nanoengineering leverages its ionic properties to assemble advanced electrode materials. Biotechnologists explore enzyme activity in ionic liquids, hoping to improve reaction rates and selectivity. In collaboration with environmental scientists, some groups examine recycling strategies and lifecycle impact, treating it as a model for sustainable practices across the broader class of ionic liquids.
What We Know about Toxicity
The presence of the hexafluorophosphate anion has brought this ionic liquid under the toxicological lens. Rat studies and aquatic organism trials reveal measurable effects at high concentrations, with chronic exposure raising concerns over organ toxicity. Lab protocols emphasize minimizing skin contact and inhalation, and researchers working with these substances learn quickly to avoid long-term exposure. Waste regulations increasingly call on facilities to develop strategies for collection and disposal, reflecting the caution needed when managing compounds with persistent anions. Regulatory bodies in Europe and North America track new data, and changes in guidelines mirror the evolving scientific understanding.
Looking Forward: Future Prospects and Addressing Gaps
Future growth relies on balancing technical performance with safety and environmental impact. As new regulations restrict PF6-based compounds, synthetic chemists search for alternative anions that maintain performance without the toxic legacy or potential for environmental persistence. Some promising directions point to organic or bio-derived anions. As battery technology and pharmaceutical processes continue to advance, the lessons learned from working with 1-butylpyridinium hexafluorophosphate shape the next wave of safer and more effective ionic liquids. Industry-academic partnerships hold promise in scaling up production while shrinking waste footprints and toxicity hazards.
What is 1-Butylpyridinium Hexafluorophosphate used for?
Getting To Know 1-Butylpyridinium Hexafluorophosphate
Most people outside a chemistry lab rarely give a second thought to the likes of 1-butylpyridinium hexafluorophosphate. This ionic liquid pops up in research papers that get shared among scientists. If you’ve ever followed new trends in sustainable chemistry or battery technology, you’ve likely seen this name buried somewhere. It’s no household cleaner, but it touches some important areas that shape new technology.
The Role in Green Chemistry
Think about the push to use less toxic, more sustainable solvents in chemistry labs. For a long time, volatile organic compounds filled the bill, since they do their job well but tend to evaporate or pollute the air. 1-butylpyridinium hexafluorophosphate dropped into the scene as part of the movement toward greener options because it doesn’t evaporate at room temperature and rarely catches fire like old-school solvents. The chemical world appreciates features like that. In my time learning about lab safety, knowing that a flask filled with this ionic liquid wasn’t going to explode or fill the air with fumes mattered a lot.
Helping Out in Electrochemistry
In the race for a better battery, lots of compounds get their shot at the spotlight. This ionic liquid can serve as an electrolyte, sitting between the battery’s positive and negative ends and allowing ions to move. I remember reading about test cells, where researchers swapped out the typical lithium salt solution for one based on this chemical. What happens? More stability in the battery, more resistance to heat, and a chance to recharge more often before throwing the cell away. Industry still wants cheaper answers, but the potential for safer, longer-lasting batteries helps push research forward.
Pushing Forward in Catalysis and Synthesis
A chemist spends plenty of time figuring how to coax molecules to come together just right. 1-butylpyridinium hexafluorophosphate helps as a solvent that won’t annoy metal catalysts or sensitive ingredients. In organic synthesis labs, I saw its use when the recipe required both high temperature and zero water content. Reactions that would fizzle or create toxic fumes in water or traditional solvent, ran cleaner and safer. That’s an important improvement. A lot of pharma companies look at these outcomes and start to wonder how industrial synthesis might change for the better in the next decade.
Concerns and Considerations
Every new chemical comes with trade-offs. 1-butylpyridinium hexafluorophosphate takes a long time to break down in the environment, and it doesn’t play well with water systems. Lab disposal methods can keep it contained, but larger-scale industry use would need careful waste handling. Researchers look for ionic liquids that will biodegrade if spilled. Sometimes, the best use for this compound isn’t the most obvious. With each promising lab report, there’s a call for better understanding and less toxic alternatives.
Paths Forward
Shifting away from fossil-derived solvents matters as industries push for smaller ecological footprints. By testing out compounds like 1-butylpyridinium hexafluorophosphate, people in chemistry open the door for safer factories and better energy storage. That means economic gain and less risk for workers. Like every step in science, it takes careful study about risks, benefits, and smarter disposal methods.
What are the safety precautions when handling 1-Butylpyridinium Hexafluorophosphate?
The Realities of Working with Chemicals
My days in the lab showed me that chemical safety is not just a manual-hugging exercise. It’s about respect for the substances we work with, especially ionic liquids like 1-butylpyridinium hexafluorophosphate. This salt finds a place in advanced batteries, catalysis, and electrochemistry, but its pleasant-sounding name hides real dangers. Mishandling can lead to burns, poisoned air, or even costly downtime from spills.
Why Personal Protective Equipment Matters
Experience taught me a lot about skin contact and chemical burns. 1-Butylpyridinium hexafluorophosphate in particular can irritate the skin and eyes. Once, a simple splash on my glove sent me straight to the sink. Nitrile gloves created a decent barrier, but goggles—sealed against the face—kept my eyes safe during every transfer. Long sleeves and a sturdy lab coat helped, but I never skipped the basics: frequent hand washing and no eating or drinking in the working zone.
Breathing in this material’s dust or fumes can cause respiratory irritation. A fume hood became my best friend whenever handling this compound, even in low quantities. Ventilation, I realized, means more than just opening a window. A high-efficiency hood, regularly checked for airflow, stopped invisible exposure that could otherwise threaten my health.
Storage and Safe Handling
Improper storage can turn even small amounts of this chemical into a hazard. I learned fast to label every single bottle and always keep containers tightly sealed. Cabinets lined with acid-resistant materials kept leaks from spreading, while segregating reactive compounds eliminated the chance of nasty surprises. Keeping the workspace uncluttered and containers off the edge of the bench cut down on spills. Any spill, big or small, got treated with respect—absorbent pads, immediate clean-up, and waste bags designed for hazardous materials.
Health and Environmental Considerations
Accidental exposure poses more risks than just an itchy rash. Chronic exposure to hexafluorophosphate ions can damage vital organs, so simple milk runs for medical attention were replaced by informed and proactive behavior. Quick access to showers and eyewashes in the lab always took priority during setup checks. Proper waste management matters too. Pouring remnants down the drain could poison local waterways. By using approved disposal containers and pairing up with regulated chemical waste companies, I felt confident that my safety extended to the world outside the lab.
Teamwork and Training
No worker is an island. I looked to experienced colleagues, training sessions, and trusted resources like the CDC and European Chemicals Agency. Regular drills on spill response and first aid made everyone sharper. Building a culture of open communication—where asking, “do we know how this behaves when heated or mixed?”—led to fewer accidents and smoother workdays. Fact-based decisions drove trust in our safety routines, pushing us to revisit protocols when something unfamiliar showed up in a shipment.
Improving Safety Culture
Using chemicals with long names and complex risks urges everyone to stay vigilant. Reviewing safety data sheets before each new process, asking hard questions, and setting up feedback circles built an environment where caution and curiosity worked together. It’s not just about avoiding burns and stains—respect for chemicals protects health and builds stronger labs.
What is the chemical formula and molecular weight of 1-Butylpyridinium Hexafluorophosphate?
Breaking Down the Formula: C9H16F6N·P
Some chemicals make a splash not just because of what they do, but because of how they’re built. 1-Butylpyridinium hexafluorophosphate is one of those substances scientists and tech developers have found especially useful. Getting to know its formula sheds light on much more than just its basic structure. The formula reads: C9H16F6N·P. That spells out nine carbon atoms, sixteen hydrogens, six fluorines, a single nitrogen, and a phosphorus. This isn’t your run-of-the-mill salt—there’s a positively charged butylpyridinium ion matched with a big, stable anion: hexafluorophosphate (PF6−).
Molecular Weight Revealed
Plugging these atomic numbers into a calculator, the molecular weight comes out to about 285.20 g/mol for 1-butylpyridinium, and 144.97 g/mol for hexafluorophosphate. Added together, the full unit tips the scale at around 430.17 g/mol. Not exactly lightweight, which helps explain some of its unique physical properties.
Why Knowledge of Formula and Weight Matters
A formula is more than just a string of letters and numbers. This data goes straight into real-life decisions in the lab. If someone’s mixing ionic liquids for batteries or catalysts, precise weighing matters. Errors can burn resources or lead to sketchy results. With its relatively high molecular weight, 1-butylpyridinium hexafluorophosphate brings thick textures and steady ionic behavior, which makes it attractive for uses where stability and low volatility matter, like in advanced batteries and green solvents.
The Real Science: Where Facts Meet Application
Industrial labs care about details—like knowing this chemical’s structure leads to strong ionic interactions. Factoring in the high fluorine content, it becomes clear the substance resists reacting with water, reducing the risk of hydrolysis. Engineers in the field often report that the predictability you get from this chemical plays well with sensitive electronics, which explains why it keeps popping up in papers about next-generation capacitors.
Challenges and Solutions in Working With This Chemical
The impressive properties of this ionic liquid do not come without bumps in the road. Purity remains a constant concern. Even small traces of water or contaminants reshape the way ionic liquids perform. Researchers aiming for reliable outcomes count on suppliers who rigorously test batches and provide transparent purity data. It’s not enough to trust a label; verifying via NMR or similar analytical tools becomes the smart move. That mindset catches on in most university teaching labs, where careful technique is hammered home from day one.
There’s also the trouble of cost and environmental burden. Fluorinated chemicals, for all their advantages, create tough questions about long-term impacts. It’s hard to ignore that fluorine compounds stick around in the environment. Chemists and environmental scientists explore recovery and recycling processes to soften this impact, often by building infrastructure into labs to recapture and reuse residuals. Universities push for green chemistry grants precisely to support work in this area.
Why Get the Basics Right?
Having tried to synthesize and purify ionic liquids in academic labs, I’ve seen firsthand how knowing the correct formula and molar mass prevents headaches in real projects. Without these, recipes flop and data loses meaning. Reliable sourcing from peer-reviewed literature, safety data sheets, and hands-on confirmation keeps progress on track. This kind of careful attention pays real-world dividends, both in products that work and in reduced environmental harm over time.
How should 1-Butylpyridinium Hexafluorophosphate be stored?
Why Proper Storage Matters
Anyone who’s spent time hauling boxes of laboratory chemicals knows securing them isn’t just about obeying rules, it’s about keeping accidents out of the headlines. 1-Butylpyridinium hexafluorophosphate has carved out its own space in electrochemistry circles and research settings. But it doesn't play gently if mixed with moisture or exposed to the wrong materials. The hexafluorophosphate part can react with water and start releasing toxic gases like hydrogen fluoride. Once, early in my own research days, I saw poorly stored ionic liquids create a headache straight out of chemistry’s own version of Murphy’s Law.
Keep It Dry, Keep It Tight
Moisture spells trouble. Even in a place where air feels dry, you can’t bet against humidity. 1-Butylpyridinium hexafluorophosphate demands an airtight seal—anything less can let in just enough water to turn a shelf into a hazards list. If you’ve got a desiccator or a good dry box, use it. Throw in fresh desiccant packs. Once, a colleague skipped this step, and condensation inside the bottle left crystals clumped at the bottom. Cleanup burned up most of a morning and morale.
Shelving Choices and Container Savvy
I keep this chemical in glass bottles with Teflon-lined caps. Some plastics break down or leach when used for storage. Glass sidesteps these issues—plus, you can spot clouding or sediment. Write a sharp label with the contents and the date opened, since nobody wants a guessing game a year down the line. Keep it off low shelves, since a dropped bottle can end the workday fast. Medium-height shelves, free of vibration, lower the risk of accidents but still keep things within reach.
Temperature and Light: Controlled Darkness
Room temperature storage usually suffices if you can trust your building’s climate control. Avoid direct sunlight or heat sources, since extra warmth accelerates chemical changes. A dark, locked cabinet away from windows works best. Once, a summer blackout shut off the air conditioning in our lab for hours; some reagents, including this one, didn’t come out quite the same. Loss like that hits budgets and productivity harder than most folks expect.
Avoid Unwanted Reactions
Labs love to crowd reagents together, but that’s a mess for things like this. Store away from acids, bases, oxidizers like permanganates and peroxides, and anything prone to emitting moisture. Store only compatible chemicals on the same shelf. Pairing up with something reactive only adds risk. Years ago, a friend discovered a leaky bottle next to strong acid—the smell alone sent us running to the safety shower. Segregation saves more than just supplies—it protects people.
Handling Spills and Waste
If you drop or spill even a small amount, don’t swipe at it with a paper towel. Protective gloves, goggles, lab coat, fume hood—it’s not overkill. Clean up spills with an inert absorbent, like vermiculite, and place residues in clearly labeled hazardous waste containers. Ventilate the area and alert staff right away.
Tools and Training: The Best Safeguards
Regular training tops any equipment. New students or staff need hands-on demonstrations rather than just SOP handouts. Outdated habits or laziness creep in fast. I’ve seen the difference a quick refresher makes—increased vigilance and fewer close calls.
Personal Wisdom
Good storage habits for 1-butylpyridinium hexafluorophosphate may look like overkill, but a little care keeps science safe and productive. Mistakes stick with you, and chemicals don’t forgive shortcuts. Follow clear labeling, controlled access, and solid housekeeping. Protect people first; results follow naturally.
What are the main physical and chemical properties of 1-Butylpyridinium Hexafluorophosphate?
Getting to Know This Ionic Liquid
People sometimes call 1-butylpyridinium hexafluorophosphate by a mouthful—BMPyPF6. On the shelf, this chemical shows up as a colorless or pale yellow liquid. It doesn’t smell much, probably because it hardly evaporates. Liquids like this handle high temperatures without boiling away, which lets researchers pull off lab work that would give regular solvents a meltdown. I’ve spilled a drop on my glove before and noticed right away it doesn’t feel greasy or oily, just a bit heavier than water.
How It Behaves In the Lab
Test a bit between your fingers (with gloves!) and you’ll notice its high viscosity. This is not water; it moves slower. Its melting point sits well below freezing, usually around -40 °C, so you can stick it in a cooler without freezing it solid. The boiling point is hard to catch because it breaks down into other chemicals if you heat it past 320 °C. That sort of thermal stability makes it useful for jobs that need heat resistance.
The density lands just over 1.2 grams per cubic centimeter, which feels heavier than a sugar solution but not quite by much. If you’ve tried mixing it with water, you’ll see they don’t blend. This ionic liquid keeps to itself and won’t dissolve easily in most non-polar solvents like hexane either. Hit it with some acetonitrile or acetone and you’ll get a little mixing, but not as much as some other ionic liquids.
Chemical Stability and Safety
The hexafluorophosphate anion makes this chemical sturdy. It refuses to react with lots of common compounds, which is a huge deal in electrochemistry. You can push and pull electrons through it without breaking it down. Batteries and capacitors rely on that kind of toughness. In my experience, people always pay attention to storage: moisture ruins it. Even a whiff of water in your flask and you start getting HF, and nobody wants that. For safety and storage, I always reach for tight-sealing bottles and stash them in a dry box.
What to Watch For and Possible Challenges
Handling BMPyPF6 in the lab, you sometimes spend more time worrying about contamination than doing experiments. Water absorption lowers its performance. Hexafluorophosphate salts always come with the worry of releasing toxic fluorine compounds if mishandled or heated too much. Wearing proper gloves, goggles, and using a fume hood isn’t just a box to check—it really matters with this stuff.
Waste management gives folks headaches, too. Ionic liquids brought excitement as “green” alternatives, but the disposal of fluorinated ones like this creates trouble. Regular lab waste channels won’t cut it. Facilities with strict hazardous waste handling step in, which isn’t cheap or convenient. Some teams look into recycling processes, but getting a clean product back remains tricky.
Solutions and Best Practices
Trouble with water contamination can be managed with molecular sieves and glove boxes—the expense is real, but so is the benefit. For waste issues, ongoing research looks promising. Some groups explore swapping PF6- with safer anions. Others try recycling spent liquid with activated alumina and other filter setups. These approaches could eventually shift BMPyPF6 from a specialty lab solvent to a real workhorse for greener chemistry.
BMPyPF6 shines in a few specific fields, especially where thermal stability and non-volatile behavior matter more than raw convenience. With good handling, safe storage, and responsible disposal, it offers a real upgrade for certain challenging jobs.
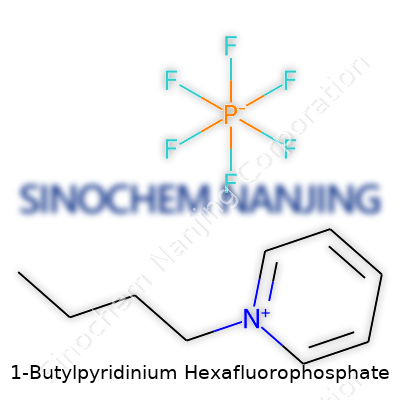
| Names | |
| Preferred IUPAC name | butyl(pyridin-1-ium) hexafluorophosphate |
| Other names |
N-Butylpyridinium hexafluorophosphate 1-Butylpyridinium PF6 1-Butylpyridinium hexafluorophosphoric acid salt BuPyPF6 1-Butyl-pyridinium hexafluorophosphate |
| Pronunciation | /ˈwʌn ˌbjuːtɪl.pɪˈrɪdiːniəm ˌhɛksəˌflʊəroʊfəˈsfeɪt/ |
| Identifiers | |
| CAS Number | 143314-17-4 |
| Beilstein Reference | 1858739 |
| ChEBI | CHEBI:38580 |
| ChEMBL | CHEMBL565430 |
| ChemSpider | 22460291 |
| DrugBank | DB11297 |
| ECHA InfoCard | 100.128.157 |
| EC Number | EC 695-314-1 |
| Gmelin Reference | 135970 |
| KEGG | C18835 |
| MeSH | D079430 |
| PubChem CID | 2994218 |
| RTECS number | UX8571000 |
| UNII | S8YHH0805T |
| UN number | UN3488 |
| Properties | |
| Chemical formula | C9H16F6NP |
| Molar mass | 284.22 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | 1.18 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -0.87 |
| Vapor pressure | 0.01 mmHg (20 °C) |
| Acidity (pKa) | -2.0 |
| Basicity (pKb) | 8.7 |
| Magnetic susceptibility (χ) | −9.72×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.447 |
| Viscosity | 86 cP (25 °C) |
| Dipole moment | 7.5642 Debye |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 211.3 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -576.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -6898.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P210, P261, P264, P271, P272, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1-1-0-⚗ |
| Flash point | > 212 °F |
| Lethal dose or concentration | Rat oral LD50 > 2000 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): >2000 mg/kg |
| NIOSH | Not listed |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Butylpyridinium Hexafluorophosphate: Not established |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
1-Butylpyridinium chloride 1-Butylpyridinium bromide 1-Butyl-3-methylimidazolium hexafluorophosphate Pyridinium hexafluorophosphate 1-Butylpyridinium tetrafluoroborate |

