Taking a Closer Look at 1-Butylpyridinium Chloride: A Chemical that Caught Science’s Eye
Historical Development
Chemistry’s not short on fascinating moments, and 1-butylpyridinium chloride has had more than its share. Back in the mid-20th century, the focus sat squarely on simple salts and hydrocarbons, with little attention paid to ionic liquids outside an academic context. Then came breakthroughs with alkylpyridinium compounds, which changed the way chemists thought about liquid electrolytes. In industry, researchers started seeing real potential for 1-butylpyridinium chloride as early as the 1990s, especially as focus grew on green chemistry and low-volatility solvents. Over time, this chemical moved from a background curiosity to a centerpiece for people trying to push boundaries in both laboratory and industrial settings.
Product Overview
1-Butylpyridinium chloride doesn’t strike most people as memorable at first glance: it looks and handles much like other salts. Yet this compound turns heads among researchers, with its ability to dissolve a broad range of organic and inorganic molecules. Used in ionic liquids, it stands out because it’s not just stable, but flexible. The butyl side chain gives it enough organic character to change its physical behavior, without losing the essential ionic punch. Chemists who value materials that handle extremes of temperature, electrochemical stability, or functional modifications find themselves coming back to this compound again and again.
Physical & Chemical Properties
Despite its simplicity, 1-butylpyridinium chloride has a personality all its own. As a solid, it’s white to off-white, but it turns into a liquid at relatively moderate temperatures compared to many classic salts. Its melting point reflects a sweet spot for production of room-temperature ionic liquids. The chemical structure—a pyridinium ring with a butyl chain and a chloride counterion—creates an asymmetric charge distribution. This means its ions don’t crystallize as tightly, so they flow more freely and dissolve a remarkable range of partners. Solubility in water as well as certain organic solvents opens doors for both research and applications in electrochemistry, catalysis, and advanced material science.
Technical Specifications & Labeling
Reliable chemical sourcing has always mattered in my work, especially when a project’s outcome rests on consistent quality. 1-Butylpyridinium chloride typically gets labeled according to purity, water content, and residual solvent load. Purities above 98 percent are common in research-grade material, since stray ions and contaminants can ruin sensitive processes. Vendors follow strict labeling for hazard information, storage guidelines, and batch traceability. Chemists rely on these labels to prevent accidents and ensure results that can be repeated, something that patent offices and peer reviewers always check closely.
Preparation Method
Anyone who’s spent time at a synthesis bench knows the joy and frustration of organic prep. The most straightforward path to 1-butylpyridinium chloride starts with pyridine and 1-chlorobutane, reacting them under heat in a polar solvent. The resulting quaternization creates the pyridinium salt. Post-reaction, chemists filter and purify the solid, watching for side reactions or leftover reactants that rob yield or purity. This reaction, though simple in description, demands close attention to temperature, solvent ratios, and cleanliness. Skipping any detail can leave you with a mixture too dirty for serious electrochemistry.
Chemical Reactions & Modifications
What gets people excited about 1-butylpyridinium chloride isn’t just synthesis but flexibility. Chemists can swap the chloride for other anions using metathesis reactions, and the compound’s pyridinium ring can undergo further functionalization, giving rise to a library of ionic liquids. In my experience, this adaptability means a single batch can serve as a launching pad for dozens of custom solvent or electrolyte systems. Chloride’s nucleophilicity allows some niche organic transformations, though most researchers focus on the compound’s power as a liquid medium or an ion-pairing agent. Modifications to the butyl chain or the ring itself shape melting point, stability, and solvent properties.
Synonyms & Product Names
While shopping for or researching 1-butylpyridinium chloride, people will see it called by a handful of names. N-butylpyridinium chloride and 1-butylpyridinium chlorate pop up in literature, along with short-hand like [BPy]Cl. These variations tend not to confuse seasoned chemists, but in regulatory paperwork synonyms can create hair-pulling delays. A close eye on CAS numbers saves time and headache, especially since pyridinium-based ionic liquids sometimes go by trade names not always linked to their IUPAC designation.
Safety & Operational Standards
All chemicals deserve respect, and 1-butylpyridinium chloride definitely demands a cautious approach. Direct skin exposure may irritate, since the ionic nature messes with surface moisture and can sneak through gloves. Spills on counters or floors become sticky messes, so immediate cleanup makes sense. Inhalation of fine dust proves uncomfortable at best, hazardous at worst, so using a fume hood and proper respirators pays off. The chloride ion introduces the risk of forming potentially corrosive byproducts, especially in contact with acids or strong oxidizers. Standard chemical hygiene practices—labeling, secondary containment, proper waste handling—bring peace of mind and keep accidents rare.
Application Area
The list of uses for 1-butylpyridinium chloride is long and gets longer every year. Chemists working in green solvent technology appreciate its role in room-temperature ionic liquids, where it helps dissolve catalysts or electrolytes that water or traditional organics can’t handle. In electrochemistry, this ionic liquid finds use in supercapacitors and batteries, where its wide electrochemical window and thermal stability support higher energy densities. Synthetic organic chemists use it to run tricky transformations without the usual side reactions linked to traditional quats or simple salts. These applications hit the market slowly, moving first from university labs into pilot-scale setups for clean energy, fine chemical production, and specialty separations.
Research & Development
Many stories about materials breakthroughs start in research labs, and 1-butylpyridinium chloride is no exception. Academic labs worldwide keep exploring what this ionic liquid can do when it comes to dissolving biopolymers, supporting sustainable catalysis, or forming custom nanomaterials. My own experience isn’t unique: characterizing how the cation-anion structure affects melting point, toxicity, and solvation keeps graduate students and postdocs busy. Companies working at the intersection of pharmaceuticals, energy storage, and green chemistry value the “tuneable” nature of these salts. Patents for supercapacitor formulations, electrodeposition baths, and solvent-free reactions often mention 1-butylpyridinium chloride. The drive now is to tailor both the pyridinium backbone and the counterion for an expanding range of applications.
Toxicity Research
Any chemist thinking about scale-up pays attention to toxicity, and for 1-butylpyridinium chloride, data have started piling up. Studies show moderate toxicity to aquatic organisms and low acute toxicity to mammals, although chronic exposure info remains limited. High ionic strength can disrupt cell membranes, which matters if wastewater treatment lacks appropriate filters or collection. Nobody wants a wonder solvent that turns into a downstream pollutant. Chemical safety committees insist on best practices: avoid inhalation, minimize skin contact, and keep the compound out of drains. The need for detailed ecotoxicological and biodegradability studies grows as the scale of use creeps up from flask to factory.
Future Prospects
People in the chemical industry talk a lot about sustainability, and 1-butylpyridinium chloride could play a part in that story. As energy storage and clean manufacturing grow more popular, demand for highly stable, non-volatile liquids climbs too. Electrolytes based on ionic liquids promise higher safety and longer lifespans compared to old-school organic solvents. The next generation of batteries may not depend on lithium or classic salts but on materials built from pyridinium backbones. With that promise comes a challenge: large-scale synthesis that doesn’t leave behind dangerous byproducts, and full lifecycle analysis from cradle to grave. Researchers keep tuning the chemistry to squeeze out any weak points, driven by both scientific curiosity and real-world policy pressures. The story of 1-butylpyridinium chloride isn’t finished. Right now, it stands as a proof-of-concept that new ways of thinking can turn simple molecules into keys for a greener and more efficient future in both the lab and the market.
What is 1-Butylpyridinium Chloride used for?
A Look at Everyday and Industrial Uses
1-Butylpyridinium chloride. It sounds like something you’d encounter only in a chemistry textbook, but it actually turns up in some interesting places—more than most folks realize. People working in labs and factories have known about it for years. For them, it’s not just another obscure chemical on a shelf; it's part of the toolkit that keeps research and production running smoothly.
The Role of Ionic Liquids
Dig into its role and you’ll find it belongs to a group called ionic liquids. These are salts that melt below 100 degrees Celsius, and, in my own chemistry classes, we always looked at them as the “liquids that don’t evaporate like water.” This makes them reliable for processes where regular solvents would boil away or make a mess. 1-Butylpyridinium chloride carries a unique mix of qualities: it dissolves polar and nonpolar substances, and it doesn’t give off much odor or vapor. This means you don’t walk out of your lab smelling like a refinery.
Solvents for Green Chemistry
In recent years, researchers have shared real excitement about moving toward greener chemistry. I’ve seen more people choosing 1-butylpyridinium chloride for chemical reactions because it reduces the need for old-fashioned, polluting solvents—stuff like benzene or toluene, which health experts have flagged for their risks. Instead of the chemical smell I remember from college, labs now feel a lot less harsh on the lungs. Published studies point out how this ionic liquid helps chemists design cleaner reactions that put less strain on both people and the planet.
Battery Innovation and Energy Storage
Over the past decade, I’ve seen colleagues hunting for new materials to make batteries safer and last longer. 1-Butylpyridinium chloride shows up in this search. Its stable, low-volatility nature makes it attractive for use as an electrolyte or additive in lithium batteries and fuel cells. Picture battery fires in the news—companies want to move past those headlines. Scientists have tested this chloride for improving the lifespan and safety profile of battery cells, aiming to match our growing reliance on clean energy.
Catalysis and Organic Synthesis
The world of catalysis and organic synthesis has its own demands. Researchers using 1-butylpyridinium chloride as a catalyst—not only as a solvent—have found that it often helps push stubborn reactions past their usual stall points. This versatility gives scientists a flexible hand in building everything from pharmaceuticals to agricultural chemicals. I worked on a project years ago where the team spent weeks trying to carry out a tricky reaction, only to hit our goal within days after switching to an ionic liquid system. The shift was that effective.
Challenges and the Path Forward
Every new tool comes with trade-offs. Working with ionic liquids, people watch costs, environmental impact, and issues with recycling or disposal. It’s true that some ionic liquids can linger in soil or water, so companies keep looking for ways to break them down more quickly. Government watchdogs and chemical companies must keep an eye on long-term risks. Teams around the globe are searching for drop-in substitutes and safer variants.
Balancing safety, sustainability, and affordability isn’t easy. Clear, evidence-based oversight and honest reporting help everyone spot trouble early. When I check the news or scientific journals, it’s obvious the story of 1-butylpyridinium chloride keeps evolving, driven by people hungry for progress but cautious about the cost to health and nature.
What are the safety precautions when handling 1-Butylpyridinium Chloride?
The Everyday Truth about Chemical Risks
Careless mishandling of chemicals leads to real harm, often not just in obvious lab accidents, but by repeated little exposures that sneak up on you over the years. I’ve seen people underestimate risk, thinking only corrosive acids or obvious toxins matter. In reality, almost every chemical carries potential hazards, and 1-butylpyridinium chloride serves as a good reminder: its hazards don’t always show up right away. This ionic liquid—a favorite for specialty synthesis and electrochemistry—asks for clear respect for safety, and a willingness to take real steps instead of shortcuts.
Understand What You’re Dealing With
Forget any idea of “harmless” just because it’s not a classic strong acid or big brand-name solvent. 1-Butylpyridinium chloride causes irritation to the skin, eyes, and respiratory tract. It’s possible to become complacent, but everyone from seasoned scientists to students should treat this compound like any other potentially hazardous chemical. I’ve seen younger researchers skip gloves “just for a second,” only to regret it. Every exposure counts.
Gloves serve as your main line of defense. Not all gloves work—use chemical-resistant nitrile or neoprene; latex won’t cut it. Before handling even small samples, goggles need to go on and lab coats come out. Some folks still go for open shoes—this is dangerous, even if a splash seems unlikely. Spills find their way to skin far more often than you think.
Ventilation Guards Your Health
Another thing people underestimate: invisible risk. Even without much of a smell, chemicals release vapors or fine particles. Fume hoods matter for every transfer or weighing step. I remind anyone I mentor—just because you can’t see fumes doesn’t mean they can’t harm your lungs. Coughing or mild eye irritation sometimes get brushed off, but those are your body’s early warning signs. Share the importance of clean air with your team, and fix the hood if it’s broken rather than working around it.
Watch for Reaction Risks
It’s easy to focus on handling chemicals “as-is,” but 1-butylpyridinium chloride reacts with strong oxidizers. Never store it near bleach, peroxides, or nitric acid. An accidental mix-up can cause hazardous decomposition, sometimes with heat or toxic byproducts. Never transfer chemicals to an unlabeled bottle—it only takes one distracted colleague to cause trouble. Clear communication saves everyone from dangerous surprises.
Quick Action Beats Regret
I’ve known researchers embarrassed to pull the safety shower or eye wash after a spill, thinking it’s an overreaction. Don’t hesitate. Even small splashes need immediate attention. Wash the affected area with lots of water for at least 15 minutes. Remove contaminated clothing without delay. Accidents lose their sting when you treat the situation seriously right away.
Store 1-butylpyridinium chloride at room temperature in a dry, well-ventilated spot, with the lid tightly sealed. Regular safety checks catch leaks or deteriorating bottles before they turn into emergencies. It’s easy to ignore the chemical shelf until something leaks, but frequent inspections pay off. Good labeling and clear records avoid confusion down the line.
Shared Responsibility in the Lab
Training makes a real difference, especially for those new to the lab. Go beyond the basics—read the safety data sheet together, talk through “what would you do if” situations, and double-check that everyone has personal protective equipment that fits. Real trust builds when team members look out for one another and speak up at the first sign of trouble. The safest labs are communities, not isolated individuals.
What is the chemical structure and formula of 1-Butylpyridinium Chloride?
Getting to Know 1-Butylpyridinium Chloride
1-Butylpyridinium chloride has grabbed the interest of chemists and researchers in recent years, and not just as a chemical curiosity. This compound belongs to the family known as ionic liquids, a group that keeps making waves in green chemistry for its unique properties. To truly understand why folks care about its formula and arrangement, it's helpful to look closer at the structure.
Chemical Structure and Formula
At its core, 1-butylpyridinium chloride is a salt. The cation—1-butylpyridinium—has a five-member ring (pyridine) with one nitrogen atom tucked in, and a butyl group hanging off that nitrogen. The anion? Just good old chloride. The chemical formula for the complete salt reads as C9H14ClN. If you draw it out, you see a pyridine ring (imagine a hexagon with one corner swapped out for nitrogen instead of carbon), and a straight chain of four carbon atoms (the butyl group) attached to the nitrogen. The chloride ion floats nearby, ready to form ionic bonds.
Back in college, I spent a lot of time staring at molecular models to get a feel for this layout. Visualizing the butyl chain coming off the ring helps highlight how these organic groups can change the chemical’s personality, including solubility and melting point, compared to regular salts. Chemists often rely on these tweaks in real-world applications.
Why Structure Matters
At first glance, this formula might just look like a bunch of letters and numbers. In the lab, though, those atoms and their arrangement shape everything from the compound’s feel to its utility. The long butyl tail attached to the pyridine ring gives the cation extra flexibility, and that changes the way molecules interact. This is a big reason why 1-butylpyridinium chloride can stay liquid at room temperature while sodium chloride forms crystals.
There’s growing interest in these structures because ionic liquids like this avoid the volatility and flammability of many organic solvents. This opens new doors for safer chemical processes—fewer harmful fumes, less risk of fire, and often more control over reactivity. The butyl chain isn’t just window-dressing; it tunes the compound for specific uses, from dissolving cellulose in processing plants to stabilizing catalysts in battery design.
Applications and Responsible Use
This compound has found uses across several sectors. I’ve seen it pop up in electrochemistry, organic synthesis, and even biomass processing. Researchers look for ways to swap out more dangerous solvents and embrace alternatives that don’t pack the same environmental punch. Of course, safety still matters—chloride ions can lead to corrosion, and any new solvent needs to be examined for environmental persistence.
Moving forward, industries should keep digging into the physical and toxicological profile of compounds like 1-butylpyridinium chloride. Companies need to weigh performance against health and environmental impacts. Academic partnerships and transparent reporting help build trust, and long-term studies offer insights into how these substances interact with air, water, and living systems.
Looking for Solutions
It’s not enough to celebrate innovation for its own sake. Cost, recyclability, and disposal practices must be front and center. Chemistry classrooms and boardrooms alike have a stake in balancing progress with responsibility. By pushing for greener synthesis methods and safer handling, researchers and manufacturers can unlock the potential of 1-butylpyridinium chloride while protecting people and ecosystems.
How should 1-Butylpyridinium Chloride be stored?
Understanding 1-Butylpyridinium Chloride
Everyone working in a lab knows chemicals come with their own set of rules. 1-Butylpyridinium chloride ranks among those liquid compounds you see in research, batteries, and greener chemistry projects. Its usefulness really shines when folks want alternatives to more hazardous solvents. Still, its unique character brings up questions: where do you keep it, and what keeps it from breaking down or causing problems?
Why Care About Storage?
People might overlook storage directions if they haven’t seen what a neglected bottle can do. In chemical storerooms I’ve seen, poor placement means fumes, leaks, or labels peeling off. Lab teams have a responsibility not just to their projects but also to safety and the shelf life of what they’ve bought. If somebody leaves a polar ionic liquid like 1-butylpyridinium chloride out in the wrong spot, humidity creeps in, or it gets too hot, unwanted reactions happen. Moisture can start a chemical change, and that means waste, contamination, or even risks like corrosion.
Common Problems Without Proper Storage
Moisture isn’t just an annoyance. Ionic liquids love absorbing water from the air, so a loose cap or broken seal becomes an open invitation. Over time, even a small amount of slip-up means the product’s properties can shift. I’ve watched eager researchers lose a week of experiments because invisible contamination changed their results. That never hits the budget or timeline favorably. In bigger facilities, careless mistakes can even lead to mixing with incompatible substances, risking toxic fumes or equipment damage.
What Works: Practical Storage Tips
A dry, ventilated, and cool space deserves top priority. Most chemical storerooms already have these traits, but the trick lies in following through.
Sealed Containers MatterUsing airtight bottles keeps humidity out. Always double-check the lid after use. Switch out container types if the original looks damaged or starts showing discoloration. Labels need to stay readable — fading ink or chemical smudges will only lead to confusion later, and mistakes multiply fast in a rush.
Keep It Cool, Not ColdTemperature doesn’t need to drop to freezing, but heat shortens shelf life. Short exposure to direct sunlight or sitting near heat sources changes the chemical itself, rendering it less stable. Trust laboratory refrigerators or well-ventilated cabinets, wherever sudden temperature swings stay rare.
Separate From IncompatiblesEven though 1-butylpyridinium chloride performs well against many reactive substances, acids and bases at close quarters spell trouble. Segregating it from common acids, alkali metals, and oxidizers brings risk way down. I’ve found color-coded bins helpful for visual reminders, especially in busy environments.
Handling ProtocolsGloves and eye protection always belong in the protocol, not only because you avoid skin contact but also because great handling often stops spills before they start. Clean spills up immediately since even a few drops left behind can attract unwanted reactions or foul up shared workspaces.
Building Good Habits
Anyone using or managing chemical inventories holds responsibility for tracking shelf life. Out-of-date inventory audits keep storerooms uncluttered. Digital logs work if everyone checks them, but nothing replaces the value of a quick in-person inspection for dusty caps or leaks.
The effort to store 1-butylpyridinium chloride well grows more rewarding the longer you keep it. Every lab can cut costs, prevent accidents, and protect results with attention to these details — storage isn’t just a chore, it’s infusing daily work with care and expertise built from experience and attention.
Is 1-Butylpyridinium Chloride soluble in water?
What Stands Behind 1-Butylpyridinium Chloride’s Solubility?
The chemistry world can get tangled in long words and dense formulas, but clear answers help everyone—especially in the lab, where mistakes get expensive. 1-Butylpyridinium chloride, an ionic liquid, draws attention for its ability to mix with water. This solubility trait shapes how it’s used in real research.
A molecule like this, where a pyridine ring hooks up to a butyl group and snags a chloride ion, doesn’t surprise anyone who’s worked with ionic liquids. It’s got a polar head and a relatively short nonpolar tail. Pause and think about how salt—the kitchen kind—slips easily into water. That’s about the same principle. Researchers find that 1-butylpyridinium chloride dissolves in water, often without drama or need for extra steps. The ionic bond between the cation and the chloride ion interacts strongly with water molecules, opening the door to clear solutions in a matter of minutes with stirring.
Why Solubility Matters for Chemistry Projects
Solubility isn’t an idle curiosity for chemists. Pure, dry, and undissolved chemicals stick out as troublesome. Full solubility means putting an accurate amount in and knowing it’ll react or act as needed. Folks working in catalysis, phase transfer reactions, or extraction appreciate a substance that teams up with water handily. It’s often about speed and avoiding side problems like incomplete mixing, gunk buildup, or lost material. It’s annoying to run a reaction, then realize bits of the ionic liquid just sat at the bottom; full solubility cuts down on troubleshooting and wasted supplies.
Lab techs and experienced researchers like working with 1-butylpyridinium chloride partly because consistent results matter. It’s tough to hit reliable numbers if a reagent plays hide-and-seek with water. Papers published in well-known journals support these observations, noting that this chloride salt goes quickly into water at standard temperatures, without needing heating or solvents. The resulting solution usually comes out clear, and concentration tweaks rely only on how much salt goes in, not on extra tricks.
Environmental Safety and Storage Thoughts
Some see ionic liquids as the next step in green chemistry. Not all are perfect, though. Soluble chemicals can sneak into waterways if spilled or binned carelessly. Parts-per-million levels show up fast in environmental labs. For a hydrophilic liquid like this, treating waste with respect reduces later headaches. Simple steps, such as collecting solutions for specialized disposal or using smaller batch sizes, make a difference in routine work. At my old university lab, we’d run reaction waste through basic water treatment and hand over extra solutions to hazardous waste experts. Even when a substance seems safe, minimizing exposure and runoff makes practical sense—both for colleagues and the world outside the window.
Practical Steps in Daily Use
Once a team knows that 1-butylpyridinium chloride loves water, preparing buffer solutions, extracting products, or carrying out electrochemical runs gets smoother. There’s less checking and second-guessing. Projects stay on schedule. Mixing this ionic liquid into water involves basic tools—nothing more complex than a stir bar. For researchers hitting stubborn solubility issues elsewhere, switching to this compound can open new doors. Chemists tend to share tips, and word spreads when a chemical both works well and avoids typical dissolving headaches. I’ve watched colleagues run side-by-side comparisons: substances that behave this way save time, especially on tight research deadlines.
The takeaway for scientists and students is clear. Bet on compounds that give full solubility and reliable behavior in water. 1-Butylpyridinium chloride fits that bill—speeding up experiments, shrinking supply waste, and helping clean science stay on course.
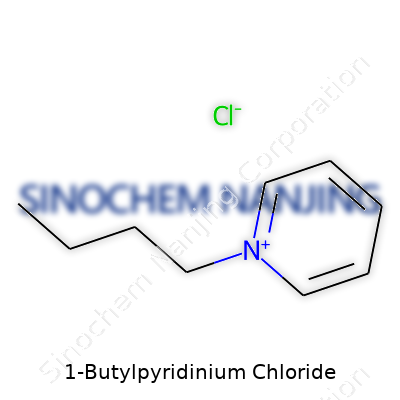
| Names | |
| Preferred IUPAC name | 1-butylpyridin-1-ium chloride |
| Other names |
1-Butylpyridinium chloride N-Butylpyridinium chloride 1-Butylpyridinium chloride (1:1) Butylpyridinium chloride BPC |
| Pronunciation | /ˌwʌnˌbjuːtɪl.pɪˈrɪdiːniəm ˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 1124-64-7 |
| 3D model (JSmol) | `6MRUWVXMGGXXHR-UHFFFAOYSA-M` |
| Beilstein Reference | 3936892 |
| ChEBI | CHEBI:73093 |
| ChEMBL | CHEMBL3183317 |
| ChemSpider | 20457376 |
| DrugBank | DB11106 |
| ECHA InfoCard | 100.149.113 |
| EC Number | 202-956-0 |
| Gmelin Reference | 110110 |
| KEGG | C19668 |
| MeSH | D000071277 |
| PubChem CID | 4695936 |
| RTECS number | UJ0700000 |
| UNII | 67U1I87S2J |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | 'DTXSID8045063' |
| Properties | |
| Chemical formula | C9H14ClN |
| Molar mass | 179.68 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 0.968 g/mL |
| Solubility in water | very soluble |
| log P | -1.2 |
| Acidity (pKa) | 12.5 |
| Basicity (pKb) | -4.02 |
| Magnetic susceptibility (χ) | -68×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.520 |
| Viscosity | 89.6 cP (25 °C) |
| Dipole moment | 8.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 224.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -336.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5768.7 kJ/mol |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| Flash point | Greater than 230 °F (110 °C) |
| Lethal dose or concentration | LD50 (oral, rat): 400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 > 2000 mg/kg |
| NIOSH | B1425 |
| PEL (Permissible) | Not established |
| REL (Recommended) | N/E |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
1-Butylpyridinium bromide 1-Butyl-3-methylpyridinium chloride 1-Ethylpyridinium chloride 1-Butyl-4-methylpyridinium chloride 1-Hexylpyridinium chloride |

