Understanding 1-Butyl-3-Methylimidazolium Tetrafluoroborate in a Changing Scientific Landscape
Historical Development
From my days in the university lab, I remember the buzz around ionic liquids in the early 2000s. Chemists loved these salts that stay liquid at room temperature, for good reason. The old way relied on volatile organic solvents, which meant headaches—sometimes literally—from fumes, and always worries about environmental cleanup. The introduction of compounds like 1-Butyl-3-Methylimidazolium Tetrafluoroborate, often called BMIM BF4, marked a real shift. French and Russian scientists worked on these imidazolium-based salts decades ago, but mainstream research only caught up once green chemistry became more than a fashionable idea. As publications on BMIM BF4 piled up through the 2000s and 2010s, the chemical community realized this wasn’t just a one-off curiosity. Ionic liquids, BMIM BF4 included, allowed safer, cleaner alternatives for synthesis, separation, and even energy storage. The growth of the field mirrored the shift in industry’s focus: from raw performance at all costs, to performance that also respected people and the planet.
Product Overview
So, what makes BMIM BF4 stand out in the lineup of ionic liquids? I’ve seen it turn up in labs dedicated to catalysis, electrochemistry, organic synthesis, and even in folks’ batteries or capacitors. At the core, it’s a salt made from pairing a butyl-methyl-imidazolium cation with a tetrafluoroborate anion. Molecularly, it’s chunky enough to stay liquid under mild conditions. Its non-flammable, stable nature appeals not just to researchers, but also to industries chasing practical, safe materials. I’ve poured BMIM BF4 myself, and the stuff feels slicker than water, a touch alien even. That odd combination of slipperiness and stability is what hooks people.
Physical & Chemical Properties
BMIM BF4 skips a lot of the issues seen with typical solvents. Its melting point lands well under ambient, letting folks work at room temperature with ease. You can count on it not to boil or evaporate under regular lab conditions, thanks to its extremely low vapor pressure. It resists catching fire, and it won’t produce whiffs of noxious odor. Density runs higher than water, which you’ll notice immediately by weight. As an ionic liquid, its ability to dissolve a variety of organic, inorganic, and even some polymeric materials puts it somewhere between a Swiss Army knife and a faithful old workhorse. BMIM BF4 also features wide electrochemical windows, opening the door for all sorts of modern applications. I’ve dropped metal salts and some stubborn organic compounds into this stuff, watching them dissolve efficiently. The virtue is clear to anyone frustrated by sluggish or incomplete dissolutions in traditional solvents.
Technical Specifications & Labeling
Those who work with chemicals know that label accuracy can mean the difference between a successful experiment and wasted time. For BMIM BF4, suppliers report purity levels commonly above 99 percent, with low water content emphasized. Trace metal analysis and halide content regularly get checked, as both impact electrochemical and catalytic performance. Even a chemist more interested in synthesis than in analytical nitpicking becomes fastidious about water content with this ionic liquid; moisture creates hiccups, so many bottles come vacuum-sealed. Exact specs depend on the intended use—battery researchers are fussier than organic chemists, for instance. All this attention stems from the expectation that BMIM BF4 works reliably, without hidden contaminants that could skew results or prompt safety incidents.
Preparation Method
BMIM BF4 comes from a fairly straightforward route. Butyl and methyl groups connect to the imidazole ring via alkylation, usually under mild to moderate heating. Reacting the resulting 1-butyl-3-methylimidazolium chloride with sodium tetrafluoroborate completes the process, with sodium chloride dumped out as a side product. Purification includes water washes and drying, since any leftover water or impurities spell trouble in technical work. Researchers interested in scaling up the process watch for byproduct management, solvent recovery, and quality consistency. At bench scale, I always found the reaction manageable; scaling it into safe, cost-effective production remains more complex, and pushes producers to refine their protocols to avoid exposure to potentially hazardous reagents and side streams.
Chemical Reactions & Modifications
BMIM BF4 works like a chemical chameleon. In my own lab experience, using it as a green solvent in multiple step synthesis shaved time and cut waste. Its ionic environment helps catalyze organic reactions that usually take longer or give lower yields in conventional media. Swapping out the tetrafluoroborate anion for others—hexafluorophosphate, bis(trifluoromethane)sulfonimide—lets chemists tune properties like viscosity and solubility. Sometimes, the imidazolium core gets more exotic modifications for specialty applications, such as enhanced thermal stability or unique electronic properties. Electrochemists use BMIM BF4 for electrodeposition and supercapacitors, since its stable electrochemical window reduces risk of side reactions. After years working with traditional solvents, the ability to customize BMIM-derived ionic liquids feels liberating for chemists who want more control over their systems.
Synonyms & Product Names
Anyone looking for BMIM BF4 in literature or catalogs will see an alphabet soup: 1-butyl-3-methylimidazolium tetrafluoroborate, BMIM BF4, [BMIM][BF4], or shortened as RTIL (room temperature ionic liquid) with its full chemical name. Synonyms sometimes create confusion for younger lab members, so double-checking registry numbers and supplier details avoids mix-ups. Different suppliers market under brand names or abbreviations, so knowing your acronyms makes ordering and cross-referencing much less of a headache.
Safety & Operational Standards
While BMIM BF4 gets attention for its safety compared to volatile organics, it’s no free pass. Gloves and goggles remain a must; the compound isn’t known to catch fire easily, but contact with skin or eyes still means trouble. Long-term biodegradability and aquatic toxicity continue to raise concern among environmental scientists. Safety data sheets flag the need for waste management protocols that don’t just send solutions down the drain. In crowded academic labs, I found researchers sometimes grew too casual, treating ionic liquids as harmless alternatives. Training needs to keep up with new materials. Regulatory frameworks should adapt to cover ongoing toxicity research and environmental impact, rather than sitting comfortably in categories designed for much older chemicals.
Application Area
BMIM BF4 has shown up in all sorts of unexpected places. Catalysis stands out: reactions like alkylations, oxidations, and cross-couplings often benefit from its ability to promote phase transfer and stabilize reactive intermediates. Supercapacitor and battery research leans heavily on BMIM BF4 for electrolytes that function beyond water’s limitations. Separation scientists use it for extraction steps involving metal ions or organics thanks to its selective solubility. I’ve seen material engineers test films and membranes produced from blends containing BMIM BF4, searching for new properties. Clean energy and environmental cleanup researchers explore its use for CO2 capture and solvent recycling. The breadth speaks to its flexibility; it’s rare to see one chemical pop up in so many distinct scientific communities.
Research & Development
Academic and corporate R&D pour significant effort into improving BMIM BF4 and related ionic liquids. Labs work on reducing residual halide content and pushing for greener, less energy-intensive synthesis methods. The hunt for more biodegradable or less toxic ionic liquids amps up each year. I’ve worked with research teams who combined computational chemistry with benchwork to design next-gen ionic liquids tuned for specific reactivity or environmental safety. Direct applications in carbon capture, industrial catalysis, and electronics drive commercial investment. The research output still trends upward, as young scientists grab at the chance to shape technologies not weighed down by legacy pollution and waste.
Toxicity Research
Toxicity questions always shadowed the celebration of ionic liquids like BMIM BF4. Initial studies overlooked long-term ecological impact. More recent papers measure aquatic toxicity and effects on microbial life, showing BMIM BF4 breaks down slowly and can bioaccumulate in certain organisms. The risks don’t compare to classic solvents like benzene or chloroform, but they’re real enough to warrant care. Environmental chemists argue for designing new salts with the same technical punch but shorter environmental lifespans. Responsible disposal and tracking of emissions should go hand-in-hand with expanding use, to avoid repeating errors of the twentieth century chemical industry. I see opportunity for better stewardship, and hope regulatory agencies match pace with the science so the field doesn’t trade one set of problems for another.
Future Prospects
The pathway for BMIM BF4 stretches far beyond its original uses. Battery, supercapacitor, and solar cell sectors look to ionic liquids for safer, high-performing electrolytes. Sustainable chemistry continues to seek alternatives to classic solvents, and BMIM BF4 fits the bill—with room for improvement in terms of lifecycle and toxicity. Collaborative projects now focus not just on raw technical performance but on recyclability, sourcing, and ultimate biodegradation. The drive for decarbonized energy and green chemical production keeps BMIM BF4 and its derivatives in the spotlight. No single solution will tick every box, but experience shows that listening to feedback from every corner—bench chemists, industry safety officers, environmental watchdogs—pushes the development of ionic liquids towards safe and sustainable futures. For those still learning the ropes, BMIM BF4 offers a real-world lesson in how chemistry can adapt to challenges without falling back on past mistakes.
What is the chemical formula of 1-Butyl-3-Methylimidazolium Tetrafluoroborate?
Getting to the Basics
Chemistry classrooms sometimes seem worlds away from life outside a lab. But right now, ionic liquids like 1-butyl-3-methylimidazolium tetrafluoroborate are changing not just industries, but small bits of daily experience that most folks would never think link to chemical formulas. The formula for this substance, known as BMIM BF4, is C8H15N2BF4. Even without a chemistry degree, it’s clear that knowing what’s inside a material—and how its atoms mesh together—gives real insight into why certain technologies are moving in surprising directions.
Why This Formula Isn’t Just Alphabet Soup
Ionic liquids aren’t just chemical curiosities. They stay liquid at room temperature, don’t evaporate easily, and handle electricity differently than most standard solvents. I remember working in a lab where we tried to switch to greener solvents and ended up using derivatives of this very compound. Its formula—C8H15N2BF4—reminds us that tweaking molecules slightly, like adding a butyl or methyl group, totally changes a liquid’s use or safety profile.
The imidazolium ring, for example, is more than a pretty structure. It lets the compound dissolve a range of substances. Pair that with a stable tetrafluoroborate anion, and the whole mixture resists breaking down and opens up possibilities across electrochemistry and catalysis. You see these liquids in research on better batteries, safer solar cells, and as possible replacements where waterless and non-volatile solutions can cut accidents and waste.
Practical Importance: More Than Just a Formula
Most people won’t think twice about what’s inside their phone batteries, but these ionic liquids deserve attention for safety and resource reasons. The components in BMIM BF4 avoid flammability issues shared by ordinary organic solvents. More industries experiment with it because the chemical formula points to a reduced risk of fires and explosions. The less you have to worry about a dangerous vapor cloud, the more practical and sustainable innovation becomes.
From a safety engineer’s perspective, this compound has made some industrial sites easier to manage. Less hazardous material in the air means fewer long-term health worries. Data from the European Chemicals Agency shows ionic liquids tend to stick around, giving them new potential as durable solutions for challenging reactions and processes.
Where the Challenges Still Sit
Not everything about this formula spells progress without hurdles. Many ionic liquids, even BMIM BF4, cost more to make than common solvents. Disposal rules haven’t caught up to their newness, and scientists are still tracking their long-term effects beyond the lab. In one project, we dealt with cleanup protocols that didn’t fit these liquids. It took trial and error—plus a bit of stubbornness—to keep waste out of places it didn’t belong.
Looking Forward
More people in research, waste management, and policy are teaming up to build strategies that catch up to this chemistry. Training chemists and engineers to handle these compounds safely, and designing recycling processes before the materials pile up, feels urgent. Seeing the impact of one clever formula like C8H15N2BF4 ripple out through energy, environment, and safety gets real when you recognize just how much the right combination of atoms can change the world around us.
What are the main applications of 1-Butyl-3-Methylimidazolium Tetrafluoroborate?
Diving Into Real Uses
Anyone who’s handled solvents in chemical labs knows just how restrictive and flammable traditional options can be. 1-Butyl-3-Methylimidazolium Tetrafluoroborate, a type of ionic liquid, changes that by offering a safer alternative with its low volatility and non-flammability. I remember sweating over a fume hood in graduate school, trying to coax a reaction to finish while worrying about breathing in fumes. This ionic liquid, often called BMIM BF4, lets chemists sidestep many of those headaches. Workflows get smoother and safety improves in both teaching labs and R&D centers.
Pushing Clean Chemistry Forward
The push toward greener processes in industry didn’t happen overnight. BMIM BF4 has staked a claim thanks to its ability to dissolve an array of compounds that regular solvents can’t touch. Groups working on pharmaceuticals use it to synthesize tricky molecules without turning to harsh acids or bases. It creates a friendlier path for making drugs, reducing corrosive waste and worker exposure. Green chemistry isn’t just for the idealists — it cuts costs related to waste disposal and regulatory compliance.
Sharpening Electrochemical Tech
Rechargeable batteries keep phones and cars moving, but their efficiency and safety rely on the fluids shuttling ions inside. BMIM BF4 steps up as a stand-out electrolyte in supercapacitors and experimental batteries. Its electrochemical stability window widens the performance envelope, so devices charge faster and run longer. Researchers at MIT and other institutions have published studies showing that ionic liquids like this one enable flexible, safer energy storage, crucial for anything off the grid or carried in your pocket. Anyone using consumer electronics, from headphones to tablets, will eventually benefit.
Separating and Cleaning Up
Refining and recycling both need smart separation techniques. In my time researching rare earth recycling, we chased selective separation methods to pull valuable elements from e-waste. BMIM BF4 offers an effective solvent for liquid–liquid extractions, picking out metals like copper, cobalt, or lanthanides from industrial streams. Cleaner separation routes satisfy government rules on emissions and open the door to more efficient urban mining operations. The price of rare metals isn’t dropping, so anything that stretches supply or cuts waste makes a difference.
Potential Pitfalls and Moving Forward
One challenge stands out: ionic liquids still come at a price. They’re usually more expensive than old-school solvents, so they don’t work for every budget or every reaction. Some ionic liquids can also pose toxicity issues if handled carelessly, and early research flagged their persistence in the environment. Industry and academia respond with new synthetic tweaks to cut costs and bump up biodegradability. It takes honest collaboration between chemists, environmental scientists, and industrial engineers to keep progress going without trading new concerns for old ones.
Practical Solutions
Tighter industry standards and ongoing research can drive safer, more affordable ionic liquids. Open publication of safety and toxicity data helps labs pick the right options for their needs. Funding joint projects between universities and private companies can accelerate the discovery of greener, specialized alternatives to BMIM BF4, pushing the benefits out of niche R&D and into everyday products. If downstream users — from recyclers to battery makers — demand environmental transparency and low-impact processes, suppliers will follow suit.
Is 1-Butyl-3-Methylimidazolium Tetrafluoroborate safe to handle and what precautions should be taken?
What It Means to Work with Ionic Liquids
1-Butyl-3-methylimidazolium tetrafluoroborate pops up in labs and industrial operations for a reason. This ionic liquid dissolves a range of things, doesn't catch fire easily, and lets chemists push reactions in new directions. Handling it safely doesn't call for rare gear or secret know-how, but some risks deserve respect.
Why Precautions Matter
I spent time in university labs where these types of chemicals show up on the shelves. On paper, they look less scary than old-school solvents like benzene. Fewer fumes, not as flammable, and you don't run from the room after a drop spills. Still, common sense works better than trust. Toxicology research tells us 1-butyl-3-methylimidazolium tetrafluoroborate causes irritation if it touches skin or eyes. A few studies point to damage after long exposure, either through skin contact or breathing in droplets. Labs in Europe set exposure limits for a reason—chronic toxic effects can add up.
That all lines up with what my classmates worried about, even during routine work. Gloves and goggles go on before the bottle opens. Good ventilation means you breathe easier. The safety sheet for this stuff puts it clearly: direct skin contact can cause redness, swelling, or worse, especially with open cuts. Reports from workers who forgot gloves and touched large amounts back this up—rashes and irritation can hit hard and fast. No one needs a chemical burn from a substance that looks almost harmless.
Real Hazards in the Lab
Handling spills becomes a big moment to show responsibility. Unlike water or alcohol, this liquid hangs around on surfaces and slowly evaporates. That leaves more time for others to touch it. Its high boiling point adds to the risk in labs that run hot equipment—slow leaks or drops won't vanish. I saw cleaning crews step in within minutes, scooping up small puddles with absorbent pads followed by lots of soap and water. Waste goes into the hazardous bin, not down the drain, since local water treatment plants can’t handle this combination of fluorine and organic components safely.
Looking After Yourself and Colleagues
Chemical handling always goes better with basics: nitrile gloves, closed lab coats, long pants. Protective goggles matter more than people think because splashes reach eyes in an instant. Testing emergency eyewash stations isn't just formality—after a spill, every second helps. Keeping hands away from your face avoids surprises, too.
If spills land on your skin, wash off with plain soap and water much longer than you’d guess—at least several minutes. That quick rinse my classmates sometimes tried doesn’t cut risk. Reports from US OSHA and European ECHA suggest using chemical-resistant gloves, preferably long ones, cuts accidental exposure well below harmful levels.
Improving Safety Culture
It surprises me how easy it is for groups to take shortcuts as familiarity grows. Supervisors should keep safety discussions regular and honest, not just check off training once a year. Updated safety data sheets should stay on hand, and anyone confident with this chemical should show their co-workers best practices. Even small oversights cause big problems—the cost of new gloves beats a time-consuming accident report every time.
Taking It Seriously Pays Off
No chemical makes headlines by itself, but nobody wants to risk long-term health for a shortcut. Sharing knowledge about risks, showing how to handle a spill calmly, and insisting on protective gear builds habits that stick. I’ve watched junior lab members gain confidence working next to people who take these rules seriously. Health matters to everyone, and a few simple steps create a safer workplace for all.
What is the purity specification of this product?
Pushing Past Numbers on a Specification Sheet
A purity specification seems simple—just a number, right? But those digits mean a lot more than most realize. In the chemical industry, purity defines if a supplier deserves your trust and if a material fits a role where precision rules the day. Whether it’s building pharmaceuticals, electronics, or food ingredients, calling a product "pure" doesn't just boil down to some lab result. There’s a lot riding on that guarantee.
Why Purity Matters for Real-World Decisions
People working in labs or workshops don’t treat purity like fine print. If you’re a chemist pipetting reagents, a small impurity can wreck years of research. I’ve seen graduate students comb through data, only to learn a batch gone wrong traces back to a contaminated starting material. The worst part isn’t just wasted money—it’s time you don’t get back. This pain hits in manufacturing where margins get tighter every day. With advanced materials, trace impurities flip success to failure on a massive scale. Fail a pharmaceutical specification, and regulators will block every bottle. Miss a metal standard, and that fancy chip stops working.
Behind the Label: Testing and Verification
Plans on paper tell part of the story, but purity testing adds the real weight. Reliable suppliers don’t just stick a purity guarantee on a label; they back it up with data. You want clear Certificates of Analysis—made for the actual batch, not a copy-paste from last year. Methods like chromatography, spectrometry, and titration pull the curtain back on what’s really there. It takes human judgment to spot patterns, compare against known standards, and call out what doesn’t belong. Every veteran chemist has opened a data file and felt a wave of relief, or dread, seeing real-world results.
Pitfalls Hiding Below the Surface
Some producers cut corners hoping nobody notices. This isn’t always about malice—even honest mistakes add risk. Lax cleaning routines, careless storage, and switching manufacturers without notice all inject uncertainty into the equation. In one case I saw, what should have been pure sodium chloride for a biology project showed signs of calcium lurking from reused containers. Suddenly, the lab lost days of work tracing failures back to a routine order.
Building Trust: Reliable Sourcing and Open Communication
Relationships drive reliable sourcing. It’s not just a transaction—it’s about knowing the people standing behind those barrels or jars. Many seasoned buyers take the time to tour supplier factories, pressing for details about quality checks. Some insist on random sampling rather than taking the cheapest offer. Others ask tougher questions, pushing for third-party verification or extended testing. This pays off in the long run, especially for products that shape health, safety, or hefty investments.
The Push for Transparency
With global trade blending far-flung inputs, traceability grows ever more complicated. “Purity” means nothing if you can’t trace raw material back to the source. Audits, digital tracking, and blockchain tools all promise to add layers of verification. More companies now share detailed data about their purity controls, letting customers dig deeper when stakes run high.
The Path to Fewer Surprises
In my years moving between research labs and production floors, one thing stood out: the people who asked tough questions up front lost less sleep later. Purity specification isn’t just a technical point—it shapes outcomes, builds reputations, and keeps progress moving forward.
How should 1-Butyl-3-Methylimidazolium Tetrafluoroborate be stored and handled?
Understanding the Substance
1-Butyl-3-Methylimidazolium Tetrafluoroborate falls under the class of ionic liquids, making it popular in labs for its unique properties, such as low volatility and great ability to dissolve a lot of different compounds. This doesn’t mean it gets a free pass on safety. Its chemical makeup, especially the tetrafluoroborate part, can cause trouble if not managed right.
Storage Tips That Protect Both People and Quality
I’ve worked with a variety of chemicals over the years, and some of the worst incidents I’ve seen happened because someone got too casual with storage. For this compound, exposure to moisture triggers the release of toxic gases like hydrogen fluoride. I always recommend keeping it in tightly sealed containers. Glass works well, but make sure the cap is resistant to corrosion. Don’t store this chemical near water sources, acids, or bases. Even in a well-organized lab, stray vapors or spills can happen, and humidity sneaks in faster than you expect.
Temperature also makes a big difference. I’ve noticed that keeping chemicals at room temperature in a well-ventilated storage area prevents surprises. Some folks overdo it and stick everything in the fridge, but this can backfire by making containers sweat or get brittle. Stick to a dry, cool shelf kept out of direct sunlight. Don’t let it become just another bottle at the back, forgotten between ventilation checks.
Handling Practices That Matter
In my experience, shortcuts in chemical handling tempt even seasoned researchers. For 1-Butyl-3-Methylimidazolium Tetrafluoroborate, gloves rated for chemical resistance make a big difference. Nitrile gloves manage well, but don’t forget eye protection and a lab coat. Accidents can come down to a single careless splash.
Always work under a chemical fume hood, especially during transfers. Tetrafluoroborate-containing substances sometimes release fumes that aren’t obvious right away. If the lab doesn’t have a hood, portable local exhaust systems can bridge the gap, but that’s not an excuse to skip PPE. I once saw someone get complacent and pop open a similar compound at their desk—within minutes, the smell hit, and it took hours to clear.
Spill Cleanup and Disposal
Cleaning up spills should never become routine. Small puddles on the bench or floor deserve a full response with absorbent pads. Use agents designed for ionic liquids if available. Bag up everything, label the waste, and get it to hazardous disposal right away. Pouring leftovers down the drain puts everyone downstream at risk—tetrafluoroborate breakdown can create persistent toxins.
Training and Culture Matter, Too
Relying only on safety sheets or instructions taped to a cabinet doesn’t cut it. Regular drills and honest conversations about what each person’s seen work or fail prevent incidents. If something seems off with the way the bottle looks or smells, trust that instinct. The most valuable lab safety resource I know is shared memory—learning from near-misses builds a safety net that policy alone won’t.
Moving Forward with Confidence and Care
Lab safety never gets old-fashioned, especially with chemicals like this. Staying prepared with basic tools like dry storage, proper PPE, and constant situational awareness protects both your team and the integrity of the work. Experience shows that routines, not luck, make the difference.
| Names | |
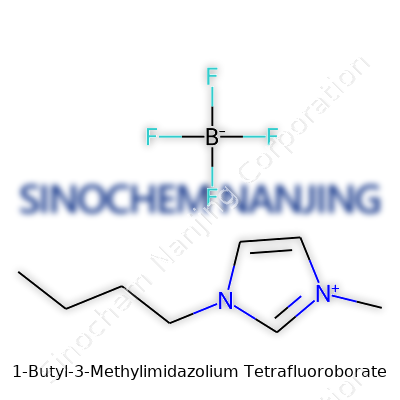
| Preferred IUPAC name | 1-butyl-3-methyl-1H-imidazol-3-ium tetrafluoroborate |
| Other names |
BMIM BF4 1-Butyl-3-methylimidazolium tetrafluoroborate 1-Butyl-3-methylimidazolium fluoroborate 1-Butyl-3-methylimidazolium tetrafluorate BMIM Tetrafluoroborate |
| Pronunciation | /ˈwʌn ˈbjuːtɪl θriː ˈmɛθɪl ɪˌmɪdəˈzoʊliəm ˌtɛtrəˌflʊəroʊˈbɔːr.eɪt/ |
| Identifiers | |
| CAS Number | [174501-65-6] |
| Beilstein Reference | 3917254 |
| ChEBI | CHEBI:39297 |
| ChEMBL | CHEMBL1231864 |
| ChemSpider | 795723 |
| DrugBank | DB11100 |
| ECHA InfoCard | 100.216.820 |
| EC Number | 325299-90-1 |
| Gmelin Reference | 77853 |
| KEGG | C20870 |
| MeSH | D000077559 |
| PubChem CID | 69111 |
| RTECS number | DY6550000 |
| UNII | GS3X6L539N |
| UN number | UN3474 |
| Properties | |
| Chemical formula | C8H15BF4N2 |
| Molar mass | 227.03 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | Odorless |
| Density | 1.12 g/cm³ |
| Solubility in water | miscible |
| log P | 0.78 |
| Vapor pressure | 0.0213 mmHg at 25 °C |
| Acidity (pKa) | pKa > 14 |
| Basicity (pKb) | pKb ≈ 13.8 |
| Magnetic susceptibility (χ) | χ = -87.0×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.428 |
| Viscosity | 241 cP (25°C) |
| Dipole moment | 5.96 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 326.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -469.2 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -5867 kJ/mol |
| Hazards | |
| GHS labelling | GHS07, GHS05 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302 + H315 + H319 + H335 |
| Precautionary statements | P210, P280, P305+P351+P338, P370+P378 |
| Flash point | > 113 °C |
| Autoignition temperature | > 362 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 300 mg/kg |
| NIOSH | MW4020000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | N.D. |
| IDLH (Immediate danger) | No IDLH established. |
| Related compounds | |
| Related compounds |
1-Butyl-3-methylimidazolium chloride 1-Butyl-3-methylimidazolium hexafluorophosphate 1-Ethyl-3-methylimidazolium tetrafluoroborate 1-Butyl-3-methylimidazolium bromide 1-Butyl-3-methylimidazolium iodide |

