1-Butyl-3-Methylimidazolium Hexafluorophosphate: Insights Shaping a Modern Chemical Frontier
Historical Development
Chemists didn’t always have 1-butyl-3-methylimidazolium hexafluorophosphate at their fingertips. Ionic liquid research only picked up steam in the late 20th century. For decades, scientists steered clear of traditional salt derivatives that melted far above room temperature. The big shift came in the 1990s when chemists started exploring imidazolium-based salts. Unlike the clunky high-melting salts of earlier generations, these compounds—sometimes flowing like viscous oils—wrote a fresh chapter. They gave researchers the freedom to work with non-volatile, electrically conductive, and thermally stable media, changing up what was possible in labs and factories. The rise of 1-butyl-3-methylimidazolium hexafluorophosphate quickly followed. Its discovery sent shockwaves through green chemistry circles, sparking a flurry of papers and more than a few patent applications. This compound didn’t just arrive; it set off conversations about what “sustainable” solvents could look like, carving out a path for the massive library of room-temperature ionic liquids seen today.
Product Overview
From the start, 1-butyl-3-methylimidazolium hexafluorophosphate—often abbreviated as [BMIM][PF6]—stood out for being a liquid at room temperature, while many salts insist on solidifying. You end up with a clear, sometimes faintly yellow liquid, heavy and surprisingly dense. I’ve come across it in varied settings, from battery labs to organic synthesis benches, and the same traits tend to stand out: the stuff doesn’t evaporate like typical solvents, and it resists lighting up because its ignition point sits high above ambient temperatures. Core features that matter go beyond the label. [BMIM][PF6] stays non-volatile, reducing exposure and contamination risks. This ionic liquid’s ability to dissolve a mess of organic and inorganic compounds makes it more than a scientific curiosity; it unlocks processes that tradition-bound solvents still can’t pull off.
Physical & Chemical Properties
Any scientist working with [BMIM][PF6] gets a real lesson in contrast. On the one hand, its heavy, syrupy consistency couldn’t be further from the volatility of familiar solvents. Pour some out, and it clings to glassware in thick sheets. Practically, this density proves useful for layered separations and specialized extraction work. Thermally, it stays stable up to remarkably high temperatures, withstanding hundreds of degrees Celsius before breaking down. The compound’s ionic nature brings high conductivity, suiting it for electrochemistry experiments and futuristic battery prototypes. It doesn’t mix with water—a factor that allows it to form layers with aqueous solutions for extractions, though this hydrophobicity owes much to the hexafluorophosphate anion. The general lack of vapor means less concern for occupational inhalation in the workspace, though skin contact still deserves attention.
Technical Specifications & Labeling
Technical documentation for [BMIM][PF6] walks a fine line between clarity and caution. Labels emphasize not just purity but potential hazards. Purity often exceeds 98 percent, but traces of byproduct imidazolium salts or moisture can influence electrical and catalytic performance, making those numbers more than just bragging rights. Chemical suppliers note its CAS number and batch-specific moisture content, as hygroscopicity in some samples affects experimental outcomes. Labels also flag strong advice on glove use, goggles, and avoiding moisture—even in trace amounts—because water alters its properties and sometimes causes slow hydrolysis of the hexafluorophosphate ion. Every bottle comes with warnings about proper disposal and safe storage, part of mounting a responsible approach in labs and industry alike.
Preparation Method
Preparing [BMIM][PF6] means more than just mixing two chemicals and walking away. The most common route puts 1-butyl-3-methylimidazolium chloride together with potassium hexafluorophosphate in water or another compatible medium. After mixing, a telltale phase separation forms: [BMIM][PF6] drops out while potassium chloride heads for the aqueous layer. Extracting the ionic liquid can be tricky—water traces hide in the dense layer, so drying steps follow, often under high vacuum and gentle heat. I’ve watched chemists get burned by skipping purification; those impurities slow down reactions and complicate catalytic cycles, so a thorough washing and vacuum drying cycle makes the difference between a research-grade solvent and a disappointment. Synthesis at scale can mean more advanced apparatus and rigorous moisture exclusion, because the smallest slip can waste a whole batch.
Chemical Reactions & Modifications
[BMIM][PF6] sometimes becomes more than a solvent; it can step up as a participant. Chemists use it in catalysis, sometimes tweaking the imidazolium ring or trading out the butyl side chain for other groups. Its reactivity can surprise those who treat it purely as inert. The imidazolium cation occasionally acts as a weak acid, affecting the course of base-sensitive reactions. The hexafluorophosphate anion offers strong resistance to hydrolysis, though prolonged exposure to water, acids, or bases eventually releases toxic byproducts. Reaction products can sometimes dissolve into the ionic liquid, so extra care during extractions is key. Attempts to tailor its features include swapping anions or cations, creating analogous liquids with better solubility, stability, or environmental profiles depending on the task.
Synonyms & Product Names
Conversations about [BMIM][PF6] get tangled if you don’t know the alternate names. Chemical suppliers and journal articles use formats ranging from the full tongue-twister, “1-butyl-3-methylimidazolium hexafluorophosphate,” to shorter forms like BMIM-PF6 or [C4mim][PF6]. Shorthand helps in the lab and around the conference table but can also generate confusion if readers mistake similar compounds. Variant spellings with methyl and butyl reversed turn up in older or foreign-language texts. Regulatory lists and shipping manifests often lock in a single version—usually just BMIM PF6—even though purists will insist on clarity. Desktop databases use unique identifiers like CAS numbers to cut through ambiguity, especially as similar ionic liquids reach market.
Safety & Operational Standards
Workplace safety for [BMIM][PF6] often includes training above normal solvent handling. Though nonflammable and practically nonvolatile, accidental spills leave persistent residues. Experience has taught many chemists never to skimp on protective gloves, eye protection, and well-ventilated workspaces—their efforts go beyond box-ticking. Absorption through skin and accidental contact can cause irritation or longer-term effects still under study. Proper storage means keeping containers sealed and away from moisture sources, since water contamination degrades stability. Disposal of [BMIM][PF6] must avoid drains or regular trash bins, with organic waste streams marked for proper incineration. Regulatory guidance sometimes lags behind new research, so those working with it must balance official recommendations against up-to-date academic findings. Each use adds another layer of practical experience to the rulebook, nudging best practices forward over time.
Application Area
Walk into research labs and you’ll find [BMIM][PF6] scattered across surprising fields. Electrochemistry takes it up for its wide electrochemical window and conductivity, trialing it in supercapacitor prototypes and novel batteries. In organic synthesis, researchers lean on its solvent strength and recyclability, skipping traditional volatile organic compounds for greener goals. It’s played a role in difficult separations, helping isolate valuable molecules from complex mixtures or acting as a medium for enzyme-catalyzed reactions. Chemical engineers look at its function as an extraction medium for metal ions or aromatic compounds, capitalizing on attributes that water and standard organic solvents lack. The absence of smell and low evaporation cuts down on workplace hazards, which means fewer headaches for those sticking with traditional solvents. Some groups are pushing it into solar cell prototypes or as a part of advanced reaction media in pharmaceutics, betting on untapped potential still hiding in the wings.
Research & Development
I’ve watched [BMIM][PF6] become a recurring theme in chemical innovation workshops and academic conferences. Research labs dive into improving synthesis routes, often seeking greener byproducts or less energy-hungry drying steps. Collaborative projects have expanded into life cycle assessments, where teams evaluate the long-term impact of wide adoption. On the science front, breakthroughs hinge on understanding solvation properties and optimizing performance for specific applications like catalysis or battery electrolytes. Universities and private R&D groups test new anion and cation substitutions, striving to maintain benefits while cutting toxicity and cost. Studies track recyclability to keep up with circular economy principles and reduce operational waste. Data-driven approaches feed into computational studies that predict properties for designer ionic liquids, narrowing the search for optimal formulations and shaving years off trial-and-error efforts.
Toxicity Research
Careful scrutiny surrounds [BMIM][PF6], especially as early “it’s green” claims met new data head-on. Toxicity research has become a regular concern, with animal model studies and environmental assessments now sharing top billing next to application studies. Experts have found that persistence ranks as a larger worry than acute toxicity. While it resists immediate breakdown, leaked quantities in waterways or soil linger, concentrating through repeated use or accidental spills. Chronic effects from long-term low-level exposure remain under investigation, keeping risk assessments on cautious footing. Human health data stem mostly from lab incidents—skin irritation, possible longer-term organ effects—so safety protocols call for careful handling until broader epidemiological evidence grows. Calls for further research mirror patterns seen in other “green” chemistry booms, where initial optimism runs up against more complex realities.
Future Prospects
The outlook for [BMIM][PF6] bridges excitement and responsibility. I see the compound set to drive significant advances, especially where existing solvents fall short. Its use in batteries and clean energy technology holds real promise. Yet, future popularity ties directly to environmental track records and regulatory response. Ongoing work seeks to curb persistence and develop more biodegradable or less toxic analogs. As industries up their investment in sustainable chemistry, there’s demand for sharper guidelines, expanded toxicology data, and better waste management systems. Chemists will keep searching for formulations that preserve the performance edge without ecological drawbacks that cloud long-term adoption. Unlocking this balance means more years of patient, methodical research—something the current generation of scientists, and those just starting out, are ready to take on.
What is 1-Butyl-3-Methylimidazolium Hexafluorophosphate used for?
Getting Familiar With a Complex Name
Talking about chemicals often feels like entering a world with its own language. Take 1-butyl-3-methylimidazolium hexafluorophosphate. The name alone packs a punch, but its story goes much deeper than a long string of syllables. In research labs and industry, this compound steps up as a versatile ionic liquid—a class of salts that remain liquid at room temperature. People don't reach for it because of its fancy name; they use it because it offers real, tangible improvements in science and technology.
The Role in Chemical Labs
A lot of the work in chemistry comes down to solvents. Many classic solvents, like acetone or dichloromethane, give off strong fumes and raise environmental red flags. 1-butyl-3-methylimidazolium hexafluorophosphate sidesteps a lot of those problems. It hardly evaporates, making it less hazardous to breathe in, and it's not flammable. Labs need better ways to dissolve, mix, or help reactants meet, all without putting technicians at risk. This ionic liquid answers that call, especially during tough organic reactions and electrochemistry processes.
Better Batteries and New Tech
Electrolytes make or break battery performance. 1-butyl-3-methylimidazolium hexafluorophosphate found a role in this space. While I spent some time in materials research, I saw how old-school lithium batteries rely on flammable, sometimes toxic, solvents. This chemical goes in another direction. By swapping out standard ingredients for this ionic liquid, developers tighten up battery safety and sometimes unlock higher efficiencies, especially in lithium-ion and supercapacitor tech. That opens the path for safer electric cars and longer-lasting gadgets.
Cleaning Up the Environment
Another area where this chemical shines: green chemistry. Since it’s stable and reusable, it prompts scientists to rethink how they separate metals, recycle rare materials, or clean water supplies. I've watched teams struggle with extracting precious metals from e-waste. Traditional solvents create headaches with disposal and health risks. By shifting to 1-butyl-3-methylimidazolium hexafluorophosphate, recovery rates often climb, and the whole process leaves a smaller footprint. Still, no solution comes without concern—the environmental impact of disposal still asks for attention from chemists and regulators alike.
Pushing Sustainable Solutions Forward
Even though 1-butyl-3-methylimidazolium hexafluorophosphate brings lots of benefits, its manufacture depends on fluorinated substances, which sometimes come with hefty environmental costs. Adopting ionic liquids broadly means industry leaders and labs must probe deeper into life-cycle impacts. Researchers look for improved variants—less fluorine, biodegradable options—while keeping the performance that makes these liquids stand out. Balancing progress in green tech with genuine sustainability stands as the next big step.
Looking for Smarter Uses
This chemical already reshaped how scientists think about solvents, batteries, and recycling. To keep momentum, more collaboration across chemical, environmental, and materials sciences needs to happen. Publishing real-world results (not just lab data) and sharing open safety information help sharpen best practices. By staying critical and curious, we steer innovations like 1-butyl-3-methylimidazolium hexafluorophosphate toward a future that values both progress and responsibility.
Is 1-Butyl-3-Methylimidazolium Hexafluorophosphate safe to handle?
Real Risks, Not Just Numbers
People love to talk about “safer solvents.” This compound, often called BMIM-PF6, falls under the ionic liquids category—fancy chemicals used in labs for their unique solubility and low vapor pressure. Low vapor pressure sounds reassuring, but these labels sometimes hide the practical dangers. I’ve watched seasoned chemists drop their guard thinking newer reagents mean fewer headaches, only to regret it later.
BMIM-PF6 isn’t volatile, so it doesn’t jump out of the bottle and into your lungs like acetone or ether. On the bench, it feels oily, sticky, and curious for anyone used to handling traditional solvents. Yet lurking beneath that slippery texture, there’s a dangerous flaw: the hexafluorophosphate anion. If you spill this chemical on a wet lab bench and try wiping it up with a damp towel, you quickly learn about what those safety data sheets mention. Water contacts the salt, and hydrolysis kicks in, generating tiny amounts of hydrogen fluoride (HF). HF has a fearsome reputation among chemists because a small splash on skin, undetected, can cause severe burns or bone damage.
Not Your Everyday Spill
In my early days, someone in the lab dropped a BMIM-PF6 vial. After cleanup, only hours later did the faint stinging on their wrist begin. Bare skin, sweat, and a tiny splash had combined with trace moisture to produce a problem. All the gloves and goggles kept the rest of us safe, but it taught everyone a quick lesson: treat this stuff with as much respect as something obviously hazardous, like strong acids.
Glass, nitrile gloves, and proper airflow make up the front line of defense. Chemists who work regularly with BMIM-based liquids avoid latex gloves, since ions can sneak through cheaper glove materials. Bench work goes smoother using disposable liners, so cross-contamination doesn’t cause headaches for others. I recommend splash goggles, not just safety glasses, because BMIM-PF6 can flick up if pipetting gets squirrely. For those pouring or weighing in larger quantities, face shields become a smart addition.
Waste, Fumes, Future Trouble
Stability tricks new users. BMIM-PF6 doesn’t reek, doesn’t boil away, doesn’t leave obvious marks—but residues stick to beakers and gloves. If a beaker sits damp for hours before washing, little bits of PF6 hydrolyze, and hazardous gases could build up. Good labs wash glassware promptly and bag solid waste, preventing lingering exposure. People sometimes believe that “green solvents” mean “harmless,” yet PF6 anions complicate waste disposal, since incineration and simple down-the-drain methods can’t be used.
Researchers face the temptation to skip hazard training when working with trending materials. Reliable supervision and realistic safety drills do more than written policies. Remembering simple facts, like the ability of PF6 to generate HF, makes a difference. Responsible labs also plan for change. Better substitutes exist, such as ionic liquids with less problematic anions. Switches to these alternatives take effort, but the long-term payoff involves fewer stories of unexpected burns or tricky cleanup sessions.
Building Safer Habits Generates Trust
Chemists survive because we learn from mistakes—ours and those of old-timers who spent decades with dangerous compounds. Real trust in any workplace grows from showing respect for every new substance, asking questions, reading the latest journals, and challenging the myth that “non-volatile” means “no problem.” BMIM-PF6 brings innovative chemistry to the table, but nobody should treat it like water or oil paints. Gloves, goggles, clean benches, and clear waste protocols turn dangerous jobs into everyday routines that still get everyone home safe.
What is the chemical structure of 1-Butyl-3-Methylimidazolium Hexafluorophosphate?
What Exactly Makes Up This Chemical?
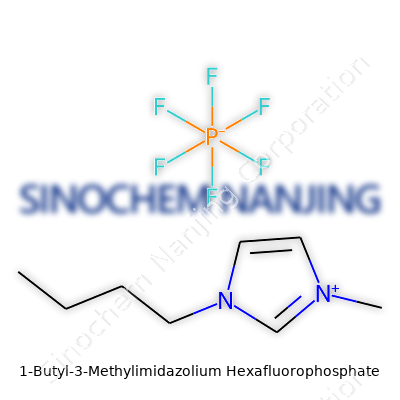
1-Butyl-3-methylimidazolium hexafluorophosphate carries a name that's more at home in a graduate chemistry lab than in everyday conversation. But look past the scientific jargon and you discover a compound with a structure that's actually pretty approachable. Start with the basics: it’s made up of two parts—a positively charged organic ion and a negatively charged inorganic counterion.
The first part, called 1-butyl-3-methylimidazolium, comes from the imidazole ring—a five-membered ring with two nitrogen atoms. Add a methyl group to one nitrogen and a butyl chain on the other, and you’ve got the organic shell of this compound. The second half, hexafluorophosphate, consists of one phosphorus atom surrounded by six fluorine atoms, forming a stable, bulky anion (PF6-).
Why Does This Structure Matter?
Real value doesn’t just stem from knowing the atoms, but from understanding how they act together. 1-Butyl-3-methylimidazolium hexafluorophosphate (often called BMIM-PF6) forms what chemists call an "ionic liquid." These are salts that stay liquid at room temperature. Imagine table salt, but’s in a glass like water, not sprinkled on your fries. This unique state shapes what the compound can do for everyday life and industries alike.
I’ve watched energy researchers pick up BMIM-PF6 because it barely evaporates and refuses to catch fire, even under high heat—unlike traditional organic solvents, which often come with risks and headaches. In the world of green chemistry, where finding alternatives to hazardous solvents turns into a full-time occupation, ionic liquids like BMIM-PF6 are a breath of fresh air.
Putting It To Work: Applications and Safety
Chemists have put this compound to the test in battery research, fuel cells, and chemical separations. Take batteries: solvents often break down or endanger workers with their toxicity. BMIM-PF6 steps in, offering a safer, more robust solution. In labs, it helps separate hard-to-handle substances or supports catalysts in complex industrial reactions. Factories could someday transition from volatile hydrocarbons to this new class of liquids, if costs line up.
Still, handling chemicals always brings challenges. Some studies, including ones from reputable scientific sources, raised environmental questions about hexafluorophosphate ions. They don’t break down easily in nature, and the fluorine content means some persistence in water supplies. That’s a real trade-off: less risk during use, but long-term worries for disposal.
What Steps Can Move It Forward?
Innovators shouldn’t just pat themselves on the back for swapping one risk for another. Researchers now push for greener ionic liquids by tweaking molecular structures—maybe swapping out PF6 for less persistent alternatives. Companies working with these chemicals can explore safer waste handling, tighter process controls, and tougher environmental standards.
Expertise, care, and ongoing experimentation will steer the future of these advanced materials. A chemical’s molecular makeup doesn’t just live on a textbook page—it shapes real outcomes for safety, sustainability, and daily technology. Knowing the structure means knowing how to use it responsibly, making room for better science and a cleaner world.
How should 1-Butyl-3-Methylimidazolium Hexafluorophosphate be stored?
Why Storage Choices Shape Lab Safety
Standing in front of a crowded shelf in the chemicals cabinet, it’s easy to underestimate the impact of a single bottle’s placement. Some chemicals, like 1-Butyl-3-Methylimidazolium Hexafluorophosphate, remind us that storage decisions in the lab aren’t just about convenience. They’re about safety and long-term integrity.
Understanding This Ionic Liquid’s Personality
This liquid, often called BMIM PF6, looks harmless—clear, with hardly any scent—but it packs a chemical punch in the wrong conditions. Its appeal comes from acting as a solvent and electrolyte, stable under certain rules. A humid or unprotected shelf flips stability into a risk. Hexafluorophosphate complexes break down with water. I learned fast to never shrug off humidity controls after one bottle ruined a set of costly reference electrodes by turning cloudy and corrosive after a steamy summer week.
BMIM PF6 reacts to water, releasing toxic and corrosive products like hydrofluoric acid. A single drop can turn a benchtop into a safety incident. The only good place for this chemical is a dry, tightly sealed bottle. Placing it next to water-loving solvents or acids just tempts fate. Any leak or accidental contact sets off a chain reaction that can leave lasting damage, not just ruined results.
Fresh Air—But Not Too Fresh
A lot of colleagues think room air is fine for most organics. Not in this case. BMIM PF6 belongs in an air-tight glass container with a sturdy screw cap. Some teams favor nitrogen-filled glove boxes, especially if they're working in a humid climate or handling large volumes. The point is to limit interaction with moisture and keep oxygen out. Every lab tech should know that even a few minutes of open exposure can change the chemistry, often producing results nobody wants or expects.
Temperature—Low, But Not the Freezer
It’s tempting to put every sensitive chemical in the freezer “just to be safe.” BMIM PF6 doesn’t need below-zero storage. Room temperature works, if you avoid direct sunlight and hot spots, like those near heating vents. Cooler, dark cabinets extend shelf life by slowing down any sneaky side reactions. I’ve seen colleagues regret storing chemicals near windows—sunshine sometimes shifts the entire contents from a consistent liquid to a gloopy mess that’s no longer usable.
Labels, Logs, and the Human Factor
Not every chemist has the luxury of a custom storage facility. Labeling becomes a lifeline. Date received, last opened, and any odd odors or changes in texture let the next person know what to expect. In crowded shared labs, it helps to keep a written log for checking and rotating stock. Using old, contaminated BMIM PF6 transforms a painstaking synthesis into a wasted afternoon or something far worse.
Proper handling of BMIM PF6 makes the lab a safer and more reliable place. Keeping it dry, sealed, and away from heat or sunlight isn't just a rule—it’s a daily choice that shows respect for everyone in the space. Solid chemical storage habits start as chores but become habits that stop accidents before they start.
What are the physical properties of 1-Butyl-3-Methylimidazolium Hexafluorophosphate?
A Liquid with a Long Name and a Long Story
Let’s break down this tongue-twisting chemical, 1-Butyl-3-Methylimidazolium Hexafluorophosphate, often called BMIM PF6. Anyone who has spent time in a lab knows these so-called “ionic liquids” can change the game compared to old-school solvents. Unlike most familiar liquids, BMIM PF6 stays liquid over a surprisingly broad temperature range. It doesn’t freeze until around minus 8 degrees Celsius and won’t boil off under normal use, since it does not have a proper boiling point — it decomposes before boiling. That means no clouds of vapor drifting around the hood, which makes storage and handling a little less of a headache.
Slippery, Heavy, and Not Your Average Solvent
Try pouring BMIM PF6, and it feels heavy for a liquid, with a density around 1.37 grams per cubic centimeter. It pours more like olive oil than water. This density makes a difference if you work with mixtures that settle out or react in layers. With viscosity that runs thicker than water, BMIM PF6 doesn’t slosh around quickly. The thicker texture affects mixing and transfer, so the right equipment comes in handy—something I learned the hard way during a long afternoon with a stubborn pump.
Makes Water Avoid It
BMIM PF6 won’t dissolve much water. In fact, it’s hydrophobic enough that water just forms blobs in it instead of blending. This trait isn’t just a curiosity; it means BMIM PF6 can pull off some neat tricks with chemical separation. Labs can use it to yank organic molecules out of water-based mixtures, no need for a fume-hood full of flammable stuff. That’s not just safer, it also saves on costs tied up in ventilation and fire safety.
Stability and Breakdown: The Fine Print
People often treat ionic liquids like they’ll last through anything, but BMIM PF6 does have limits. Persistent exposure to water or moist air can break it down, forming hydrofluoric acid—never something you want to handle casually. If you’re working in humid environments or storing BMIM PF6 in open bottles, corrosion becomes real, and not just for the bottle. That’s the kind of oversight that ruins experiments and damages gear. Dry boxes or desiccators keep it intact, a lesson most researchers pick up fast after one ill-fated mishap.
What Matters Outside the Lab
Folks talk big about “green chemistry,” and BMIM PF6 often lands in those conversations. Traditional organic solvents can catch fire or poison workers. BMIM PF6 dodges those issues—barely flammable and less volatile, so less gets into the air. That reduces the risk for people nearby. Still, it’s not a free pass. The breakdown products can be toxic. Responsible disposal and minimizing leaks into the environment are just as important as with any other chemical. Praising its safety only makes sense if labs respect proper handling and disposal best practices. The substance doesn’t do the safety for you.
Building a Smarter Lab
This stuff opens up creative ways to design chemical reactions. I’ve seen teams use BMIM PF6 to recover catalysts that would’ve gone to waste. Reusability lies at the center of cost-effective and less wasteful work. That kind of step can save thousands over a year and sidestep a lot of the mess tangled up in hazardous waste. So, BMIM PF6 isn’t just a neat curiosity—it pulls its weight by changing how researchers think about separations, safety, and sustainability, provided the risks and quirks don’t get ignored.
| Names | |
| Preferred IUPAC name | 1-butyl-3-methyl-1H-imidazol-3-ium hexafluorophosphate |
| Other names |
BMIM PF6 1-Butyl-3-methylimidazolium hexafluorophosphate(V) 1-Butyl-3-methylimidazolium hexafluorophosphate 1-Butyl-3-methylimidazolium hexafluorophosphoric acid 1-Butyl-3-methyl-imidazolium hexafluorophosphate |
| Pronunciation | /wʌn-ˈbjuːtɪl θriː ˌmɛθɪl ɪˌmɪd.əˈzoʊ.li.əm ˌhɛksəˌflʊəroʊˈfɒs.feɪt/ |
| Identifiers | |
| CAS Number | 174501-65-6 |
| Beilstein Reference | 3955046 |
| ChEBI | CHEBI:39026 |
| ChEMBL | CHEMBL142064 |
| ChemSpider | 21543976 |
| DrugBank | DB11238 |
| ECHA InfoCard | ECHA InfoCard: 100.127.042 |
| EC Number | 205-280-2 |
| Gmelin Reference | 84262 |
| KEGG | C19599 |
| MeSH | D000077248 |
| PubChem CID | 6918490 |
| RTECS number | TX1100000 |
| UNII | 3S568W9A8E |
| UN number | UN3488 |
| Properties | |
| Chemical formula | C8H15F6N2P |
| Molar mass | 284.18 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Odorless |
| Density | 1.367 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.6 |
| Vapor pressure | 0.0000163 mmHg at 25 °C |
| Acidity (pKa) | 24.0 |
| Basicity (pKb) | pKb = 15.21 |
| Magnetic susceptibility (χ) | -77.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | nD 1.418 |
| Viscosity | 72-86 cP (25 °C) |
| Dipole moment | 5.93 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 471.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -788.9 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -5646.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS05, GHS07 |
| Pictograms | GHS07,GHS05 |
| Signal word | Danger |
| Hazard statements | H226, H302, H314, H319, H332, H412 |
| Precautionary statements | P210, P262, P273, P280, P301+P312, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-1-0-W |
| Flash point | 85 °C (closed cup) |
| Autoignition temperature | 360 °C |
| Lethal dose or concentration | LD50 oral rat 300 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 300 mg/kg |
| NIOSH | SAF086 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1-Butyl-3-Methylimidazolium Hexafluorophosphate: Not established |
| REL (Recommended) | Not established |
| IDLH (Immediate danger) | 300 mg/m3 |
| Related compounds | |
| Related compounds |
1-Butyl-3-methylimidazolium tetrafluoroborate 1-Butyl-3-methylimidazolium chloride 1-Butyl-3-methylimidazolium bromide 1-Butyl-3-methylimidazolium iodide 1-Butyl-3-methylimidazolium trifluoromethanesulfonate |

