1-Butyl-3-Methylimidazolium Bromide: Beyond the Ordinary Solvent
Historical Development
The story of 1-Butyl-3-Methylimidazolium Bromide, often mentioned as BMIMBr, begins in the search for solvents that don’t carry the same hazards as volatile organics. In my time working with new chemical technologies, I’ve seen how so much innovation starts with a simple need: safer, more reliable, and adaptable compounds. BMIMBr springs from the larger family of ionic liquids—a field that’s only been developed seriously since the late 1980s, despite early discoveries in the 20th century. The moment researchers realized that room-temperature liquid salts could offer conductivity, thermal stability, and tunable properties, the race to invent and commercialize was on. BMIMBr stands out as one of the poster children for this field, demonstrating the real-world value of a discipline that once looked mostly theoretical.
Product Overview
Mention BMIMBr in any research circle dealing with green chemistry or advanced materials, and you’ll encounter a sense of respect. This salt, liquid at room temperature, serves where common organic solvents fail—especially in places where evaporation, ignition, and toxicity stop other substances cold. Not every solvent lasts through multiple heating and cooling cycles, or dissolves both ionic and non-ionic species. My personal experience experimenting with cellulose processing showed firsthand how it beats out traditional systems for dissolving stubborn biopolymers with very little fuss.
Physical and Chemical Properties
At its core, BMIMBr is a chunky, colorless-to-pale yellow liquid that feels viscous between the fingertips, with a distinctly ionic feel—much like a thick, unusually oily syrup. Unlike water or alcohol, there’s nothing flammable here and almost zero odor. The melting point hovers below human body temperature, which means it doesn’t solidify on the lab bench in winter. Chemically, the cation carries a butyl chain and a methyl group on either side of the imidazolium core, which gives the compound stability in most lab environments. This structure leads to high ionic conductivity and an ability to dissolve polar and nonpolar compounds, making it unusually versatile. If you’ve handled it, you’ll remember how stubborn the stickiness remains on glassware. Hydrophilicity varies among imidazolium salts, but the bromide counterion in this case keeps solubility in check across several solvents without introducing unwanted reactivity.
Technical Specifications and Labeling
Walking into any chemical supply room, seeing a BMIMBr bottle instantly calls up thoughts about purity. You’ll see typical purity values above 98 percent, with labels warning about humidity, since the salt likes to suck up water from the air. In my own lab, I learned quickly to keep it capped once open. The labeling avoids dramatic warnings compared to old-school chlorinated solvents, but manufacturers rightly include notes on handling, storage, and transport. It’s not about satisfying regulations alone; researchers know that clear, honest labeling makes for consistent, reproducible results in both academic and industrial settings.
Preparation Method
BMIMBr comes from a straightforward reaction between 1-methylimidazole and 1-bromobutane, usually in a polar solvent under mild heat. For anyone who’s ever set up this synthesis, the process offers a kind of rewarding predictability. With a good setup—stirring, temperature control, and ample ventilation—the reaction moves smoothly, with purification requiring either washing or repeated crystallization. In my own experience, keeping moisture out prevents unwanted hydrolysis or cation degradation, so even small slipups can spoil a batch. Some research groups push for greener, solventless routes or promote microwave-assisted procedures to shave hours from synthesis, but the basics haven’t changed much since the earliest patents.
Chemical Reactions and Modifications
What fascinates me about BMIMBr is the sheer scope of what you can do after synthesis. You can swap the bromide anion for another by metathesis, creating a wide menu of other BMIM salts by mixing with different metal salts—an easy trick for tuning properties without changing the cation. Sometimes, researchers graft functional groups onto the imidazolium ring itself, chasing task-specific ionic liquids that handle unique dissolved species. In practical settings, BMIMBr reacts gently with most reagents, but strong bases or nucleophiles call for caution, as they can trigger side reactions leading to ring opening or substitution.
Synonyms and Product Names
Ask around, and you’ll hear BMIMBr go by a few different monikers: 1-butyl-3-methylimidazolium bromide for the chemists keen on formality, or BmimBr, and sometimes 3-methyl-1-butylimidazolium bromide in older literature. Major suppliers prefer the BMIMBr label, which appears on most commercial inventory lists and research papers. This consistency means less confusion, especially when working across borders or reviewing international data.
Safety and Operational Standards
Despite the buzz around BMIMBr’s green credentials, it doesn’t get a total free pass on safety. Anytime I’ve worked with this salt, gloves and eye protection became habit, since prolonged skin contact can trigger irritation. Few ionic liquids are truly harmless—research has shown some toxicity toward aquatic species, so dumping waste down a drain is off the table. Labs with good chemical hygiene treat it like any specialty compound, using ventilated hoods and dedicated waste disposal. What sets BMIMBr apart is the lower risk of explosive vapor formation, but no process allows total complacency.
Application Area
BMIMBr began turning heads in cellulose processing, dye chemistry, and electrochemistry. In my time consulting for a renewable energy startup, this compound cropped up as a component in battery electrolyte formulations—highly conductive, nonvolatile, and stable to a range of temperatures. It serves in catalysis, extracting rare earth metals, and making tough biopolymers dissolve more gracefully than with traditional solvents. Chemical engineers in pharmaceuticals lean on BMIMBr when they want selective extraction without fear of product degradation. This isn’t just a niche chemical for academics tinkering in a back room; its reach spreads across everything from separations to specialized coatings in microelectronics.
Research and Development
You’ll find hundreds of papers published each year on new uses, property tweaks, and cleanup methods for BMIMBr and its relatives. My experience collaborating with a university team opened doors to new approaches—combining BMIMBr with deep eutectic solvents or biodegradable additives, exploring reactions that were unthinkable with volatile organic solvents. Funding agencies sink money into ionic liquids because the path forward isn’t just about replacing old solvents; it’s about offering platforms for entirely new reactions that benefit from tunable polarity, viscosity, and ion-pairing tendencies. Every time researchers think they've found the limits of BMIMBr, a new synthesis or unexpected use proves otherwise.
Toxicity Research
There’s a lot of optimism about BMIMBr and its family of ionic liquids, but looking through the toxicity data paints a more sobering picture. A handful of studies flag concerns about chronic exposure in aquatic environments—persisting harm to basic organisms at low concentrations shows up in both European and Asian research. I’ve watched environmental chemists wrestle with these questions, attempting to thread the needle between utility and ecological risk. While no major disasters linked to BMIMBr have made headlines, ignoring these warning signs would be poor science and even worse policy. Safe-use protocols, closed-loop systems, and proper waste management keep risks in check, but long-term monitoring remains non-negotiable for anyone serious about sustainability.
Future Prospects
What excites me most about BMIMBr is the sense that we’re only scratching the surface. More companies look to swap dangerous solvents for options like this, banking on lower emissions and improved process safety. There’s room for versions made from renewable building blocks or tailored for complete biodegradability. In energy storage, as battery chemistries continue to evolve, BMIMBr and close relatives seem likely to step out from the shadows of lab-scale experiments to support commercial-scale technology. Future researchers need to nail down the toxicity puzzle and devise scalable, low-impact synthesis. If that happens, this salt, once a boutique curiosity, could transform large-scale production in industries keen to balance performance with planetary health.
What is 1-Butyl-3-Methylimidazolium Bromide used for?
The Role of 1-Butyl-3-Methylimidazolium Bromide in Today’s Labs
Over the past decade, the research world started swapping out old-school solvents and salts for something far less familiar: ionic liquids. 1-Butyl-3-methylimidazolium bromide is an example, and it’s changed the way chemists, engineers, and anyone tinkering with molecular recipes approach their work. Not every fancy-sounding chemical gets much attention, but this one belongs in a separate category. It's more than a tool; it’s a formula for keeping pace with environmental standards and scientific demands.
Green Chemistry’s Friend
If you’ve ever spent time in a chemistry lab, you get why solvent choice matters. Many solvents are volatile, toxic, and tough on the people who use them. 1-Butyl-3-methylimidazolium bromide doesn’t vaporize easily. It doesn’t fill a lab with eye-watering fumes or go up in flames if left on the counter. Scientists lean on it to run reactions at room temperature or just a little warmer, which slashes energy bills and lowers risks. The push for greener chemistry relies on substitutes like this—compounds that allow for cleaner results without painful trade-offs.
Bringing Efficiency to Organic Synthesis
Colleagues who use this ionic liquid in organic synthesis talk about speed and precision. In cross-coupling reactions, for example, it helps dissolve tricky substances and creates an environment where catalysts shine. The same applies in the pharmaceutical world, where chemists often chase selectivity while trying to avoid waste. Instead of throwing away liters of dirty solvent for each step, you can sometimes recycle and reuse these ionic liquids with simple treatments. That helps streamline drug development and pilot plant testing, where every saved day matters.
Bromide Salt for Extraction and Separation
I’ve watched researchers turn to 1-butyl-3-methylimidazolium bromide for extracting metal ions from wastewater or separating out tough-to-purify compounds. The way these ionic liquids grab and hold onto metal ions puts them on the list for environmental cleanup. Heavy metals like cadmium or lead, which usually require expensive processes to capture, can be pulled from solution using carefully designed ionic liquid treatments. There’s no perfect solution for pollution, but practical progress looks like replacing hazardous chemical steps with something less disruptive and easier to handle.
A Catalyst’s Sidekick in Electrochemistry
Electrochemistry has always struggled with stability. Many solvents tear themselves apart under voltage; batteries and sensors pay the price. 1-Butyl-3-methylimidazolium bromide gives chemists a way to boost stability and shelf life for experimental batteries, dye-sensitized solar cells, and more. It supports smooth ion flows, reduces the odds of unwanted side reactions, and gives engineers leeway for testing new ideas. This means bolder R&D, safer device assembly, and less downtime from failed runs.
Stepping Up to New Industry Standards
Too many industries still rely on legacy chemicals, whether due to habit or lack of safer alternatives. The slow adoption of ionic liquids often boils down to cost or inertia, but those barriers are fading. Regulatory trends keep nudging manufacturers to reconsider their risk profiles and resource management strategies. By offering a compound that works across laboratories and pilot lines—without adding more headaches—this ionic liquid takes a step towards a safer, cleaner, and more efficient future.
Looking Toward Smarter Practices
The solution isn’t in one compound, or a magic formula, but in shifting habits. People trust tools they know, yet science keeps changing the game—offering up new, safer, and sometimes even cheaper methods. 1-Butyl-3-methylimidazolium bromide has earned its reputation in the fields of synthesis, separation, and electrochemistry through results rather than hype. The more scientists and decision-makers look for practical, green innovations, the more these forward-looking alternatives catch on—and start setting the standard everyone else wants to reach.
What is the chemical formula and structure of 1-Butyl-3-Methylimidazolium Bromide?
From Lab Bench to Real Life: Why This Salt Matters
Chemistry often sparks curiosity with its long strings of letters and numbers. Strip away the complexity and you get molecules with real impact. One that deserves a closer look is 1-butyl-3-methylimidazolium bromide. You find it in research labs and sometimes even in classrooms, serving as an ionic liquid with a unique profile.
Chemical Formula and Molecular Details
The formula: C8H15N2Br. Breaking it down, you see that this compound carries two main parts. The cation—1-butyl-3-methylimidazolium—holds five atoms in the imidazole ring (three are carbon, two are nitrogen), one methyl group (a single carbon with three hydrogens), and a butyl side chain (four carbons strung together). The anion is a bromide ion, just a simple Br−.
The structure gets more interesting up close. The imidazolium ring is a flat, five-membered ring: there’s a nitrogen at the 1 and 3 positions. Attach a butyl group (–C4H9) to one nitrogen, stick a methyl group (–CH3) to the other—now you’ve got a cation with flexibility and enough bulk to stop the ions from packing too tightly. This ‘slipperiness’ at the molecular level keeps it liquid at room temperature, which paves the way for some unusual properties.
Real-World Relevance and Insights
Ionic liquids like this one catch attention because they don’t behave like your everyday table salt. Pour it from a bottle and you see a transparent, sometimes oily liquid—no crystals. Back in grad school, we would measure its viscosity and marvel at how it barely evaporated, even under a vacuum. This stability comes from the way those butyl and methyl groups shield the positive part from the negative bromide, making crystal formation pretty unattractive.
Chemists value this compound as more than just a curiosity. It dissolves metal salts, organic molecules, and even some polymers, which makes it a big help in green chemistry. Swapping out volatile organic solvents for ionic liquids can reduce air pollution in industry. And in the lab, it smooths out chemical reactions that just won’t run in water.
Potential Hurdles and Responsible Use
The story of a chemical doesn’t end at its benefits. Years ago, our lab group worried about cost and environmental safety. Ionic liquids can be pricey, sometimes ten times the cost of traditional solvents. And while they promise greener chemistry, questions have popped up about biodegradability and toxicity. Some ionic liquids linger in the environment longer than you’d expect, and bromide ions at high concentrations can mess with certain ecosystems.
Careful design offers a way forward. Teams are working on alternatives that break down more easily after use, which keeps unwanted buildup under control. Recycling ionic liquids after reactions can lower costs and waste. In my own work, simple filtration and distillation loop ionic liquids back into new experiments, saving money for the department and minimizing what we toss out.
Why Understanding Structure Still Matters
Digging into the structure guides every big advance. The right mix of atoms affects how well this liquid dissolves compounds or speeds up chemical reactions. For those using it for battery technology or industrial extraction, tiny tweaks at the molecular level can mean higher yields and safer processes. Chemistry doesn’t live on paper—it shapes the world, one formula at a time.
Is 1-Butyl-3-Methylimidazolium Bromide hazardous or toxic?
What is 1-Butyl-3-Methylimidazolium Bromide?
1-Butyl-3-methylimidazolium bromide, often called BMIM Br, shows up in chemical labs where ionic liquids play a big part. Researchers use it to dissolve other substances, work on extractions, and even try out greener chemical reactions. This doesn’t always mean it’s completely harmless just because it’s popular in green chemistry circles.
What Do We Know About Its Hazards?
It’s easy to think something used in research so widely must be safe, but safety data and toxicology studies tell a more complicated story. BMIM Br can cause irritation if breathed in or if it gets on skin or in eyes. People often overlook risks with exotic-sounding chemicals until they see real-world effects. I’ve seen seasoned chemists caught off guard by mild but irritating symptoms after handling chemicals that seemed routine.
Animal studies point out more: aquatic organisms tend to suffer from exposure to some imidazolium salts, and BMIM Br is no exception. Water fleas and fish exposed to this compound show reduced movement and survival rates at enough concentration. This compound sticks around in the environment longer than many common solvents. Green chemistry doesn’t always guarantee a low footprint on ecosystems if mishandled.
Human Toxicity and Everyday Exposure
There isn’t a ton of human toxicity data on BMIM Br compared to familiar hazards like acetone or toluene. Handling a new chemical without much of a track record means exercising extra caution. Standard safety sheets give it hazard warnings for respiratory and skin irritation, and suggest gloves, goggles, and good ventilation. For me, following lab protocol always comes before skipping steps for convenience’s sake. There’s never a shortcut to safety.
No one expects hobbyists or the public to run into this substance but anyone working in research, industry, or waste management can encounter it. Proper disposal and spill management stand out because these ionic liquids resist breaking down in the environment.
What Can Be Done For Safer Use?
Being careful with BMIM Br comes down to understanding what you’re working with and taking simple steps that protect health. Gloves, eye protection, and a fume hood aren’t up for debate. Anyone involved in training newcomers in a lab owes it to them to focus on hazard awareness, not just lab technique.
Companies and labs using these compounds should develop solid waste protocols and emergency strategies. From what I’ve seen, strong communication and updated safety training keep slip-ups to a minimum. Regulatory bodies can help by pushing for more toxicology studies, since making substitutions without thorough vetting can backfire environmentally.
Scientists love to chase new ideas, but history proves that short-term convenience sometimes brings long-term trouble. BMIM Br isn’t the most hazardous chemical on the block, but its staying power in the environment and effects on smaller creatures make its use a decision to weigh carefully. Clean chemistry means more than marketing buzzwords; it means checking claims against real evidence, watching for overlooked risks, and keeping safety at the front of every experiment.
How should 1-Butyl-3-Methylimidazolium Bromide be stored and handled?
What Makes This Chemical Worth Respect
1-Butyl-3-Methylimidazolium Bromide doesn’t show up on everyone’s shelf, but I’ve worked with it in labs that value ionic liquids for their unique uses. It's a chemical favored for its low melting point and strong solvating power, especially in green chemistry. This drives interest in safe storage and handling, since it’s not as harmless as table salt.
Getting Storage Right
A shelf isn’t just a shelf for this compound. I store 1-Butyl-3-Methylimidazolium Bromide in sealed containers, away from moisture and sunlight. Any hint of humidity can mess with purity. I've opened a poorly kept bottle to find clumps, which signals trouble for sensitive experiments later. Airtight containers, glass or quality plastics, keep air and water vapor out. Direct sun also causes problems — too much heat can degrade this compound or create safety risks nobody wants to clean up.
Temperature control plays a big part. I stick to room temperature, usually under 25°C. Too much heat or even freezing doesn’t preserve the chemical and might change its properties. I've seen freezers used for some chemicals, but 1-Butyl-3-Methylimidazolium Bromide prefers dry, cool spots instead.
Handling Techniques That Make a Difference
Gloves and goggles top my list every single time. The skin reaction from a spill might not be as obvious as an acid splash, yet evidence and MSDS sheets point toward irritation and possible respiratory discomfort. On days with lots of weighing or mixing, a fume hood keeps me from breathing in any stray dust or vapors, even if they seem mild.
Spills don’t always look dramatic, but I treat even a small amount with caution. I grab paper towels or spill pads and seal up waste in dedicated bins. Pouring or transferring? A little care with spatulas or scoops works better than rushing. Cross-contamination can ruin months of work and threaten safety.
Why Good Practices Matter
Ignoring best practices catches up to even experienced chemists. I once saw a project stall for weeks after someone forgot to cap a bottle tightly. Traces of water got inside, and the chemical clumped up — we had to order more, losing valuable time and money. On another occasion, not wearing gloves led to minor but lasting hand irritation, even after quick washing.
Well-documented reports highlight the dangers of neglect. Some ionic liquids like this one may trigger allergic responses, especially for those exposed often. Poor ventilation adds risk, especially with scale-up activities. The science community recognizes these risks and has built a habit of safety through years of incidents and study.
Better Solutions, Practical Steps
Training always comes up first. Newcomers should see storage and handling demos and receive easy-to-read instructions. I write out steps and make sure everyone checks storage lapses at week’s end. Good signage around storage rooms and hoods helps remind folks — even experienced ones — of risk points.
Label containers with the date opened and any unusual changes in appearance. I’ve caught contamination early this way. If something looks off — discoloration, odd smell, or clumping — I set it aside for disposal and don’t take chances. Companies making this compound update safety recommendations as new data comes in, so I check for guidance each year.
Practice and awareness keep the lab productive and safe. Mistakes cost more than just chemicals — sometimes health and careers get hurt too. Putting effort into proper care of chemicals like 1-Butyl-3-Methylimidazolium Bromide just makes sense, every day.
What are the common applications of 1-Butyl-3-Methylimidazolium Bromide in research or industry?
What Makes 1-Butyl-3-Methylimidazolium Bromide Stand Out
Walking through research labs or specialty chemical plants, you'll often spot bottles labeled with long names like 1-Butyl-3-Methylimidazolium Bromide (BMIM-Br). On the surface, it’s just another ionic liquid—basically, a salt that stays liquid at room temperature. What stands out about this compound is how it tackles problems traditional solvents struggle with. It doesn’t evaporate quickly, handles moisture without breaking down, and lets researchers work with a much wider temperature range. These traits matter both at the lab bench and in industrial tanks.
Tool for Green Chemistry
Researchers want safer, cleaner ways to separate chemicals or run reactions. BMIM-Br answers a lot of those concerns. Unlike volatile solvents, it gives off almost zero fumes, which means researchers breathe easier, literally. Chemists in my circle appreciate not having to worry about the fire department showing up over a simple spill. Cleanup is easier, too, since the compound doesn’t evaporate and can be reused several times. This reuse factor helps both the environment and the budget, especially in academic labs with limited funding.
Sharp Edge in Synthesis and Extraction
Synthetic chemists often lean on BMIM-Br for tough reactions. The ionic liquid can boost the rate of certain organic reactions, including tough-to-handle alkylations and oxidations. Some labs even use it to anchor sensitive catalysts, since the liquid can help stabilize precious metals or reactive transition states. Biomass researchers find it useful for breaking down cellulose into biofuels, something traditional solvents rarely manage without wrecking the final product.
Pharmaceutical production gets a boost, too. Fine chemical companies have embraced BMIM-Br in selective extraction steps and even in the crystallization of active drug ingredients. If you’ve seen how tightly regulated drug plants are, you understand why having a non-volatile, robust solvent means less headache meeting safety standards.
Engine for Electrochemistry and Renewable Energy
BMIM-Br doesn’t just live in glass beakers—it finds a home in batteries and energy storage. Electrochemists turn to it for its wide electrochemical window and good ionic conductivity. Additives like this help shape new generations of batteries, supercapacitors, and solar cells. I’ve seen research teams push their lithium-ion prototypes harder using ionic liquids because they can avoid nasty side reactions caused by water or traditional organic solvents.
Challenges and the Path Forward
Not every story is perfect. BMIM-Br costs a fair bit more than basic organics, especially if purity matters. That hits home in bigger industrial settings. There’s also an ongoing conversation around how biodegradable these liquids really are. While they’re more sustainable up front, researchers keep working on ways to recycle or break them down even further.
With all the world’s focus turning toward greener, smarter manufacturing, the appeal of BMIM-Br keeps growing. Cost will likely come down as more production ramps up and recycling strategies become mainstream. If the chemical industry commits to proper handling and end-of-life practices, this compound’s future looks solid—not just in the lab, but across a spectrum of industries.
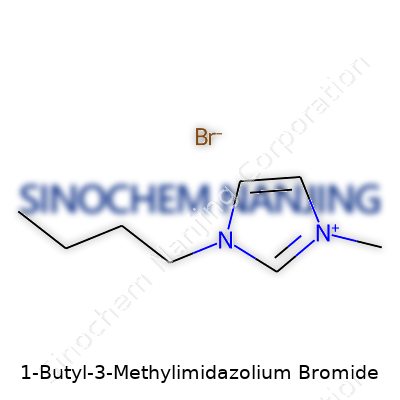
| Names | |
| Preferred IUPAC name | 1-butyl-3-methyl-1H-imidazol-3-ium bromide |
| Other names |
BMIM Br 1-Butyl-3-methylimidazolium bromide 1-Butyl-3-methylimidazolium bromide salt 1-Butyl-3-methylimidazolium bromide ionic liquid |
| Pronunciation | /ˈwʌn ˈbjuːtɪl ˈθriː ˈmɛθɪl ɪˌmɪd.əˈzoʊli.əm ˈbroʊmaɪd/ |
| Identifiers | |
| CAS Number | 85100-77-2 |
| Beilstein Reference | Beilstein Reference 3902338 |
| ChEBI | CHEBI:39297 |
| ChEMBL | CHEMBL69760 |
| ChemSpider | 26534313 |
| DrugBank | DB11125 |
| ECHA InfoCard | 07c3c2b4-8015-40c5-b4bb-29abc30b93f5 |
| EC Number | 85100-98-9 |
| Gmelin Reference | 82294 |
| KEGG | C210872 |
| MeSH | D000071300 |
| PubChem CID | 85783 |
| RTECS number | PA9846000 |
| UNII | 5Y58U29ZLO |
| UN number | Not regulated |
| CompTox Dashboard (EPA) | DB11106 |
| Properties | |
| Chemical formula | C8H15BrN2 |
| Molar mass | 219.12 g/mol |
| Appearance | White to off-white solid |
| Odor | Odorless |
| Density | 1.32 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -0.23 |
| Vapor pressure | <0.01 mmHg (20°C) |
| Acidity (pKa) | 14.0 |
| Basicity (pKb) | 9.94 |
| Magnetic susceptibility (χ) | -77.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.489 |
| Viscosity | 87.1 mPa·s (at 25 °C) |
| Dipole moment | 4.44 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 341.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -143.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -684.8 kJ/mol |
| Hazards | |
| Main hazards | May cause respiratory irritation. Causes serious eye irritation. Causes skin irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | P264, P270, P273, P280, P301+P312, P305+P351+P338, P337+P313, P330, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 176 °C |
| Autoignition temperature | > 373 °C |
| Lethal dose or concentration | LD₅₀ (oral, rat): >2000 mg/kg |
| LD50 (median dose) | LD50 (median dose) = 400 mg/kg (Oral, Rat) |
| PEL (Permissible) | Not established |
| REL (Recommended) | REL (Recommended Exposure Limit) for 1-Butyl-3-Methylimidazolium Bromide: "Not established |
| IDLH (Immediate danger) | Not listed |
| Related compounds | |
| Related compounds |
1-Butyl-3-methylimidazolium chloride 1-Butyl-3-methylimidazolium iodide 1-Butyl-3-methylimidazolium tetrafluoroborate 1-Butyl-3-methylimidazolium hexafluorophosphate 1-Butyl-3-methylimidazolium bis(trifluoromethylsulfonyl)imide |

