Reflections on 1,3-Propylenediamine: Past, Present, and Future Directions
A Historical Perspective on 1,3-Propylenediamine
Chemistry has a rich tradition of unlocking essential building blocks for modern industries, and 1,3-propylenediamine sits right among those breakthroughs. Back in the mid-20th century, as chemists expanded the family of polyamines, this particular molecule drew attention for its simple structure and di-functional nature. Those early days saw researchers experimenting with catalytic hydrogenation and amination reactions, looking for efficient routes to synthesize versatile diamines that could kick off polymer chains or crosslink proteins. It did not take long before people recognized the value of 1,3-propylenediamine beyond the laboratory, and over time it planted roots in industrial processes—showing up in resins and hardeners. Watching the evolution of its role has given a sense of how organic synthesis drives real-world progress.
Product Overview: Beyond the Name
In everyday language, 1,3-propylenediamine gets called by several names, including trimethylenediamine and propane-1,3-diamine. This compound packs a punch with two amino groups flanking a three-carbon alkyl chain, giving rise to a primary diamine with clear reactivity. People who work in chemical manufacturing appreciate how its straight chain and symmetrical placement of amines avoid clutter, opening doors for reliable transformations and easy integration into polymer backbones or specialty synthesis applications. I have seen first-hand how its structure simplifies design in curing agents and chelating treatments, reducing the unpredictability that sometimes comes with more complex amines.
Physical and Chemical Realities
1,3-Propylenediamine appears as a colorless to pale yellow liquid, with a faint fishy odor—a telltale signature of alkyl amines. What stands out in the lab is its intense affinity for water, a trait that underscores its usefulness in aqueous reactions and cleaning formulations. The boiling point hovers just above most room temperatures, so unless tightly sealed, it tends to evaporate in open vessels. Chemically, the presence of two primary amines at both ends of a short carbon skeleton produces high basicity and sharp nucleophilicity—meaning it jumps into reactions with acids or electrophiles at the first opportunity. Whether working on coordination compounds in a research setting or balancing properties in an epoxy formulation, these behaviors drive decisions.
Understanding Technical Specifications and Labeling in Practice
Even if you walk into the warehouse or open a chemical inventory, strict protocols surround the labeling of 1,3-propylenediamine. Containers spell out concentration, storage instructions, and hazard statements in plain terms. The specifics—such as purity expressed as percentage values, moisture limitations, or residual solvent levels—matter on the production floor where a small deviation can disrupt downstream reactions. Over the years, improvements in standardization have tightened definitions, making it easier for professionals to anticipate product consistency, safeguard quality control, and avoid unpleasant surprises in formulations. This transparency helps forge trust with customers and regulatory agencies alike.
Preparation Methods Shaping Industrial Practice
People often start their journey with 1,3-propylenediamine through catalytic hydrogenation of acrylonitrile or reaction of 1,3-dichloropropane with ammonia. Both processes reflect decades of process optimization—minimizing byproducts, ensuring operator safety, and upgrading yields. Early on, batch reactors held sway; now continuous flow setups and more selective catalysts deliver higher outputs with smaller environmental footprints. Staying up to speed with greener process chemistry demands constant vigilance, especially as emission standards and waste management rules keep evolving. I’ve followed process upgrades as fine-tuned reactors and modern automation reduced both energy usage and hazardous emission profiles.
Reacting and Modifying: The Chemistry That Drives Progress
With two reactive amino groups, 1,3-propylenediamine punches well above its weight class in organic transformations. As a core building block, it joins up with acids to form stable salts or hooks into epoxies and isocyanates to produce robust thermosets. In the hands of a skilled chemist, it finds life as a chelating ligand—wrapping around metal ions with ease. Surface modifications of nanoparticles or enzymes often rely on bifunctional amines just like this, taking advantage of their bridging ability. Every time a team in pharmaceuticals tweaks the backbone of a new molecule or an engineer crafts additives for coatings, you see not just the versatility of 1,3-propylenediamine, but also how these connections can push innovation into new areas.
Understanding Synonyms and Naming Conventions
Names matter in the chemical world. Anyone who’s sorted through catalogues knows how 1,3-propylenediamine’s array of synonyms—like trimethylenediamine or propane-1,3-diamine—reflects the habits and conventions of different regions and industries. These overlapping terms can trip up even the most experienced hands during procurement or regulatory reporting, especially when product labeling and technical sheets don’t align. Setting clear communication standards and cross-referencing identifiers can cut down on errors and save time—a lesson learned many times on busy projects or audits.
Safety and Operational Standards: Lessons in Vigilance
Handling 1,3-propylenediamine calls for respect and diligence. Its volatility and irritant nature pose risks for skin, eyes, and lungs—so splash goggles and gloves always become part of the uniform, along with well-managed ventilation. Over the years, tightening workplace safety regulations pushed improvements in air monitoring, containment, and spill response protocols. I have seen how a slip-up with labeling or a lax approach to indoor air quality lead to unnecessary incidents, reinforcing the habit among colleagues to never cut corners. As safety guidance evolves, ongoing training and risk assessment play a bigger role in keeping teams safe, healthy, and confident at work.
Application Areas: Real Impact, Not Just Potential
The reach of 1,3-propylenediamine goes well beyond textbooks or chemical plants. In polyamide and polyurethane resins, it drives polymer chains that turn up in automotive, electronics, and even sporting goods. Its readiness to form chelates supports water treatment and metal recovery operations—directly reducing waste and bolstering sustainability efforts. I have observed teams use this diamine in specialty surfactants for personal care and cleaning products, balancing mildness with effectiveness by leveraging its molecular structure. Down at the bench, biochemists deploy it as a crosslinking agent in protein analysis or enzyme stabilization, pushing the frontiers of diagnostics and therapeutics.
Research and Development: Adapting to Modern Demands
Progress never stands still, and R&D around 1,3-propylenediamine mirrors this reality. Teams explore new derivatives that blend higher selectivity with reduced toxicity. Innovations in catalysis and greener synthesis routes reflect growing environmental concerns and stricter regulation. Some projects focus on bridging the gap between lab-scale synthesis and full-scale manufacturing, aiming to maintain purity without ramping up cost or complexity. Collaborations between university groups and industry players push creative boundaries with functional polymers, novel chelators, or medical-grade linking agents. Watching these developments, it’s clear that flexibility and openness to change are as critical as technical prowess in making a difference.
Toxicity Research: Balancing Performance and Human Health
Toxicology never stops evolving, and attention to 1,3-propylenediamine’s impact stays strong. Repeated exposure risks—like respiratory irritation or contact dermatitis—drive research into dose-response relationships, safe handling thresholds, and long-term effects. Regulatory agencies regularly revise permissible exposure limits as new studies surface. The drive to replace traditional amines with safer alternatives, or at least implement engineering controls to minimize exposure, draws from both ethical responsibility and business continuity. These conversations, often grounded in firsthand experience, emphasize the importance of worker education, regular medical monitoring, and vigilance in manufacturing audits.
Future Prospects: Toward Sustainability and Innovation
Looking ahead, the future for 1,3-propylenediamine lies in weaving sustainability into every step—from raw materials sourcing to end-of-life strategies for polyamide products. Researchers eye bio-based feedstocks as an alternative to fossil-derived inputs, aiming for a lower carbon footprint and better circularity. Next-generation polymers and smart materials reshape consumer demands, and 1,3-propylenediamine faces competition from newer, safer designs. Its inherent versatility and robust performance keep it in the spotlight for now, though the pressure mounts to prove its place through constant innovation and environmental stewardship. In every meeting, professionals navigate a tightrope: meeting the needs of today without compromising tomorrow—a challenge that keeps the field vital, compelling, and worthy of attention.
What is 1,3-Propylenediamine used for?
Looking Into the Practical Uses
Nobody talks about 1,3-propylenediamine at the dinner table, but you can trace its impact back to real products we use every day. If you walk into a hardware store or pick up household goods, chances are chemists somewhere had their hands in the mix, using this substance as a key building block.
Key Role in Chemical Manufacturing
1,3-Propylenediamine shows up in factories where big tanks and whirring reactors turn raw ingredients into things that last and do the job right. The chemical helps companies make resins and polymers, those tough materials in adhesives and coatings. They coat floors in warehouses, keep furniture looking sharp for years, and seal materials protected against water and wear. The strength that comes from cross-linking these chemicals makes everyday goods, from wood glue that holds a cabinet together to paint that doesn’t peel after a few summers.
Active Ingredient in Agriculture
Out on the farm, this chemical makes another appearance. It’s not a fertilizer, but it plays a part in making herbicides. The diamine acts like a piece of a puzzle in agrochemical synthesis. Farmers rely on weed killers made using intermediates like this one to help protect their crops. Even in the age of organic buzzwords, fields around the world still depend on crop protection, and ingredients like 1,3-propylenediamine help maintain steady yields. According to the Food and Agriculture Organization, effective weed management prevents losses that would slash global crop output by as much as 20 to 40 percent.
Medicine, Research, and Other Surprising Places
The story doesn’t end at paint and plants. Chemists use this diamine to create molecules for medicine. Think of medicines for the heart or nervous system. While you won’t find it in a pharmacy, labs blend it into precursor compounds that undergo further steps, tweaking its chemistry to target diseases or assist in diagnosis. Researchers pursue molecules using 1,3-propylenediamine, aiming for better drugs or materials with antibacterial properties.
Academic journals confirm its role, with studies mentioning its usefulness in designing polyamides and novel materials. The aim isn’t always profit; sometimes, it’s about cleaner water or safer packaging. In one project, teams used propylene-based diamines to craft membranes to filter contaminants. Safe water isn’t guaranteed worldwide, and creative use of basic chemicals changes that equation.
Industrial Safety and Environmental Concerns
Responsible handling matters. Anyone using it wears the right safety gear—goggles, gloves, ventilated rooms. Long-term contact with amines can irritate skin or airways, so proper engineering controls and training make the difference. Regulators flag improper storage or accidents as real risks that need constant attention. According to the U.S. Environmental Protection Agency, facilities handling volatile amines—including this one—must monitor air quality and prevent leaks.
Pathways Moving Forward
There’s always room to improve. For companies using 1,3-propylenediamine, investing in greener methods to reduce waste and emissions matters. Industry researchers now look at bio-based alternatives or tweak reactions to lower side-products. Training for workers and regular safety drills remain essential. Partnerships with academics open further doors, driving discovery at the border where chemistry meets real life.
I’ve seen the difference firsthand—the way a small change in a formulation means fewer barrels shipped, less waste by the end of the day, or even new partnerships between industry and community. Every upgrade counts, because the story of a molecule isn’t just about beakers and patents—it’s about people and places, too.
What are the safety precautions when handling 1,3-Propylenediamine?
Understanding the Risks
Anyone working with chemicals like 1,3-Propylenediamine knows that respecting the hazards makes the difference between an ordinary day and a nightmare. This stuff gives off strong fumes and can bite into skin or eyes before you realize what’s happened. I’ve seen coworkers land in the clinic because they thought a splash on their gloves was no big deal, only to find out how fast irritation sets in.
Ventilation Isn’t Optional
Packing people and volatile chemicals into a cramped room leads to trouble. From what I’ve seen, good ventilation matters more than any checklist. Opening a door does a bit, but proper fume hoods and local exhaust systems pull those sneaky vapors out of your breathing space. One shop I knew tried to cut corners with desk fans and paid for it with headaches and sick days. Airflow saves you from fumes building up and masking the danger until it’s too late.
PPE: Gear Up Every Time
Certain risks just don’t get any easier with experience. Gloves rated for chemicals, snug-fitting goggles, sturdy lab coats—these aren’t just for decoration. 1,3-Propylenediamine eats through thin rubber pretty fast. Some shops keep nitrile gloves by the door and swap them out every hour or so, even if they look fine. Long sleeves and splash-proof aprons keep any drips off your skin, especially since a careless spill near your wrists or neck can get ugly fast.
Handling and Storage Make or Break Safety
Even well-trained folks get caught off guard by unlabeled containers or half-closed lids. Storing 1,3-Propylenediamine in tightly sealed, clearly marked bottles shields everyone in the area. Locking up the supply keeps wandering hands out of danger. We kept all the reactive stuff in ventilated cabinets, and grouped acids, bases, and amines apart to stop reactions before they started. Even one mix-up can send fumes through the whole building. Always double-check labels and never reuse containers, no matter how “clean” they look.
Spill Cleanup Isn’t Just Mopping Up
The right cleanup gear must be close by. Neutralizing agents, absorbent pillows, and heavy-duty disposal bags should be standard on every workbench. I remember a guy who thought paper towels would do the trick—a week later, yellow stains still marked the spot on the bench. Emergency showers and eyewash stations need regular tests, not just a “working fine” sticker from last year. Quick reaction stops a minor splash from turning into chemical burns or scarring.
Training Sets the Tone
Every time we started with a new crew, hands-on training beat out any manual or wall poster. Walking through a simulated spill or accidental splash makes everyone much more alert to risk. People remember what they feel in the heat of the moment, not what they skimmed over on a safety sheet. Regular refreshers and spot checks keep those habits ingrained. Nobody learns to respect 1,3-Propylenediamine overnight, but real stories and live practice make it stick better than lectures.
Watching Out for Each Other
Chemical safety often falls apart when people rush or try shortcuts. I’ve watched teams pull together, checking up on forgotten gloves or reminding each other before pouring or mixing. That culture of looking out for coworkers pays off over time. Fewer accidents, more healthy days on the job. Building that trust through honesty and sharing lessons—good and bad—turns a potentially risky shop into a genuinely safe team. That’s how you keep everyone protected when there’s no room for guessing.
What is the chemical formula and structure of 1,3-Propylenediamine?
The Basics: Formula and Structure
1,3-Propylenediamine, known in some labs as trimethylenediamine, brings to mind chemistry lessons that stay glued in your memory because of their simplicity and impact. Its formula, C3H10N2, tells a story that reaches right into the core of molecular building. Imagine a three-carbon backbone, almost like a sturdy spine, with an amino group (–NH2) clinging to each end. Chemists like to draw it as H2N–CH2–CH2–CH2–NH2. These two terminal amine groups open the door to a host of applications.
Why Structure Matters in Chemistry and Industry
With two amine groups anchored at each end, 1,3-Propylenediamine steps up in more ways than one. Each of those amines acts like a handshake—willing to bond, eager to help forge new materials. This flexibility changes the game for a chemist looking to craft polymers or work up new surfactants. From personal experience handling diamines, I’ve seen how vital that structure is when mixing with acids or aldehydes. Get the backbone wrong, results stray far from expected.
The symmetrical nature of 1,3-Propylenediamine shapes its reactivity profile. Since both ends look and behave the same, reactions using it tend to stay straightforward—a relief for anyone working in a bench-top setting looking for consistent, reproducible results. Years back, someone in my lab tried to swap a different diamine into a synthesis, just to shave a step. The process twisted out of shape with side products clogging the glassware. Only 1,3-Propylenediamine fit the bill.
Usage in Everyday Products
This simple molecule threads its way into plenty of products. Manufacturers use it as a building block for polyamides—materials that appear in fibers, coatings, and resins. Take nylon: a staple in clothing and industrial fabrics, relies on amines just like this one. Because it boasts two primary amine groups, it acts as a reliable connector in these polymer chains, giving finished plastics strength and flexibility. If you’ve ever handled a kitchen spatula or a piece of outdoor gear, odds are it owes durability partly to molecules like 1,3-Propylenediamine.
Beyond plastics, cleaning formulations often benefit from its twin functional groups. It links up to give cleaning agents real staying power on both grease and water-based grime. Those amines won’t shy away from binding with grime or other reactive partners. The same property shows up in water treatment processes, helping capture or neutralize impurities through strong covalent bonds.
Safety and Environmental Considerations
Any chemist worth their salt respects the dual nature of small amines: useful, but also reactive enough to demand care. Direct exposure to 1,3-Propylenediamine causes irritation. Gases evolved from misuse or accidental heating leave an industrial floor far from pleasant. Standards suggest gloves and face shields whenever handling it—a lesson often underscored the hard way during long hours in the lab. Over years, I’ve seen the push for tighter PPE and better labeling. Accurate information saves skin, literally and figuratively.
On the environmental side, 1,3-Propylenediamine doesn’t hang around for long. Bacteria present in soil and water grab onto it and break it down, limiting long-term risk. Still, responsible handling—tight drum seals, spill plans, secondary containment—remains important, especially near waterways or in large-scale set-ups. Risk management means fewer headaches for both chemists and communities living downstream.
Meeting Future Needs
The structure and utility of 1,3-Propylenediamine open up clear pathways for new polymer design and chemical research. While it’s tempting to look for substitutes, its reactivity profile and track record keep it in rotation for now. Ongoing research into safer derivatives hints at future advances, especially as industries seek greener options and tighter control over end-of-life impact.
How should 1,3-Propylenediamine be stored?
1,3-Propylenediamine and Why Storage Matters
Anyone working with chemicals has probably learned quite early that safety doesn’t begin with wearing goggles; it starts in the storage room. 1,3-Propylenediamine—a clear, colorless liquid with a sharp odor—brings its own challenges. It’s not something to keep near your lunch or in a spot that sees a lot of traffic. This compound can irritate the skin, eyes, and respiratory system, which means small mistakes in storage can lead to plenty of regret.
Lessons From the Chemical Shack: Keep Containment Top Priority
I’ve seen chemical stores where things look more like a puzzle than a plan. You don’t want 1,3-Propylenediamine on a shelf next to oxidizers or acids. Mixing those by accident can end up worse than just a mess on the floor. Segregated storage, with well-labeled shelves, prevents disasters that happen not from bad intentions, but simple mix-ups after a tired day.
How Temperature and Ventilation Keep Problems Small
Leaving this amine in a hot, unventilated space can mean pressure builds in the container, releasing vapors that no one wants to breathe. Fumes irritate the senses and, with longer exposure, can hit the central nervous system. So, the room needs air circulating and steady, moderate temperatures—below 30°C keeps the risks lower. Fire suppression nearby helps. A flammable spill’s the last thing anybody wants with a substance like this.
Seals and Containers: More Than a Lid
One summer, a colleague kept chemicals in reused soft drink bottles, thinking “plastic is plastic.” He learned quickly, as the seals warped and chemicals began to evaporate. 1,3-Propylenediamine reacts with some plastics and absorbs moisture, so factory-sealed drums or thick-walled containers designed for amines keep it stable. Metal drums with proper lining handle storage best. Each should have clear hazard labeling: “Corrosive,” “Irritant,” and the correct chemical name—because nobody needs extra puzzle-solving during a leak.
Mitigating Accidental Spills and Leaks
Spills can happen even with the best routines. Setting up secondary containment like spill trays keeps leaks away from the rest of the room. Chemical absorbents made for amines end the cleanup without spreading more vapors around. Eye wash stations, showers, and sorbent material nearby aren’t luxury—they’re what stand between a mishap and a hospital visit.
Rules, Responsibility, and Routine Checks
Written storage protocols matter more than checklists to please an inspector. They remind staff about the dangers and what steps to follow. Regular storage inspections catch leaks, deformed containers, or containers stored in the wrong spot. Staff training should include hands-on sessions with spill kits. I’ve seen emergency drills save the day more than once.
Good Habits Outlast Fancy Equipment
It’s tempting to rely on fancy monitoring devices, but the real defense comes from careful handling and respect for the rules built by people who’ve seen things go wrong. Record-keeping helps track how long chemicals sit on shelves. Rotating stock means older material doesn’t get forgotten and start degrading. These habits build a safer workplace and protect everyone who has to handle the tough stuff.
Expertise and Sources Build Trust
The best storage guidelines come from a blend of experience and reference to trusted sources. OSHA, NIOSH, and material safety data sheets give technical depth, but hands-on know-how fills in the gaps between good advice and the practical realities of a busy workplace.
What are the physical properties of 1,3-Propylenediamine?
Understanding a Simple But Significant Chemical
1,3-Propylenediamine draws attention in labs and industries because of its straightforward structure and practical uses. This colorless liquid brings a sharp, ammonia-like scent, which has a way of lingering in the air long after the cap closes. Handling this chemical always calls for careful ventilation. A boiling point sitting around 140°C means it won’t turn to vapor as quickly as you think, but wandering molecules still carry a punch for your nose and eyes. Not everyone who deals with it will recognize the name, but the smell hangs around in memory.
Density, Solubility, and What Water Means
This liquid packs a density near 0.87 g/cm³ at room temperature. It will float if poured onto water, moving around instead of sinking, making clean-up clearly more of a headache during spills. Watching how fast it dissolves in water helps you see its versatility. Given its two amine groups, propylenediamine mixes easily with water, whipping up clear solutions no matter the ratio. That ease of dissolution paves the way for it to travel through pipes, hoses, or even accidentally on your own skin—which is why gloves matter every single time.
Why the Melting Point Matters
At temperatures as low as -13°C, expect it to freeze up. Easy to store above this, but facilities in cold places find themselves dealing with solid blocks, far from convenient. That melting point also sets it apart from similar chemicals, making it a go-to for processes running at moderate temperatures. No one wants to wait for a frozen drum to thaw mid-production shift.
Chemical Reactivity and Exposure
With both ends acting as amine groups, it reacts quickly with acids, producing salts that get used in many downstream products. I’ve seen this versatility used so often in making resins and polymers, folks almost take it for granted. As a strong base, it can corrode skin and eyes, so quick rinses are not just a good idea—they are critical. This property has shifted workplaces to double-check protective gear and improve emergency showers. OSHA data backs up these risks, showing moderate to severe irritation in unprotected workers. Chemicals with such physical bite need safety processes in place—no exceptions.
Fire Risk and Storage Tips
Flammability creeps up in conversations around amines like this one. With a flash point near 80°C, it won’t ignite as readily as gasoline, but there’s a reason we keep sources of ignition away. Vapors caught in a confined area reach explosive mixes fast, which fire departments and safety officers watch closely, especially during hot summer months or in poorly ventilated storage.
Tools for Safe Handling and Moving Ahead
Industry leaders use containment trays and chemical hoods nearly every day, only trusting bulk storage in containers rated for amine resistance. Regular inspections, MSDS training, and clear warning signs all keep people safe. Local water treatment centers also keep their eyes open for spills, since the high solubility makes accidental releases tough to contain.
Looking Toward Fewer Risks and Smart Applications
With more labs developing alternatives and smarter engineering controls, risks can drop over time, especially if everyone involved values training and follows guidelines strictly. Newer generations of PPE and chemical sensors provide real-time protection when used well. Regulators use updates in physical property data to nudge standards higher. Making sure everyone understands the realities of what this liquid can do—both its hazards and its strengths—remains the best way to keep innovation moving while health and safety stay front and center.
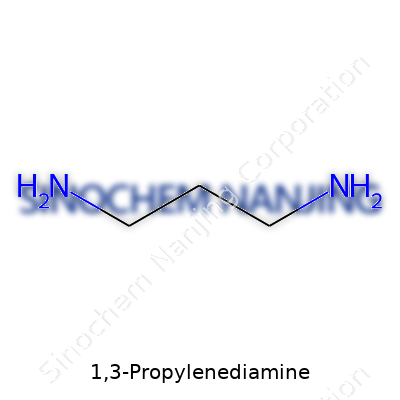
| Names | |
| Preferred IUPAC name | propane-1,3-diamine |
| Other names |
1,3-Diaminopropane Trimethylenediamine Propane-1,3-diamine |
| Pronunciation | /ˈproʊ.piːliːn.daɪˌæmin/ |
| Identifiers | |
| CAS Number | 107-00-6 |
| 3D model (JSmol) | ``` NH2CCCNH2 ``` |
| Beilstein Reference | 1100125 |
| ChEBI | CHEBI:13136 |
| ChEMBL | CHEMBL71538 |
| ChemSpider | 16052 |
| DrugBank | DB02709 |
| ECHA InfoCard | EC Number 203-822-7 |
| EC Number | 203-792-1 |
| Gmelin Reference | 7909 |
| KEGG | C02570 |
| MeSH | D017255 |
| PubChem CID | 10436 |
| RTECS number | UF9100000 |
| UNII | C0H741ZK53 |
| UN number | UN2672 |
| Properties | |
| Chemical formula | C3H10N2 |
| Molar mass | 74.12 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | Ammonia-like |
| Density | 0.872 g/cm³ |
| Solubility in water | Miscible |
| log P | -1.5 |
| Vapor pressure | 0.23 mmHg (25°C) |
| Acidity (pKa) | 10.68 |
| Basicity (pKb) | 5.70 |
| Magnetic susceptibility (χ) | -12 × 10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.435 |
| Viscosity | 7.1 mPa·s at 25 °C |
| Dipole moment | 3.24 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 95.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −54.9 kJ mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2164.7 kJ/mol |
| Pharmacology | |
| ATC code | C07CA06 |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes severe skin burns and eye damage. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Harmful if inhaled. |
| Precautionary statements | P261, P280, P304+P340, P305+P351+P338, P310, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-0 |
| Flash point | 85 °C |
| Autoignition temperature | 630 °C |
| Explosive limits | 4.1–22% |
| Lethal dose or concentration | LD50 oral rat 552 mg/kg |
| LD50 (median dose) | 780 mg/kg (Rat, oral) |
| NIOSH | UW0175000 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Ethylenediamine 1,2-Propylenediamine Putrescine Diethylenetriamine Hexamethylenediamine |

