Understanding 1,3-Dioxolane: More Than a Simple Solvent
Historical Development
Curiosity has always pushed chemists to discover new ways of transforming simple compounds into tools for industry or research. 1,3-Dioxolane didn’t appear suddenly; its creation came from decades of growing knowledge in organic chemistry. Early organic chemists, hunting for reliable, stable solvents in the mid-20th century, noticed cyclic ethers offered unique abilities to separate and stabilize reactive materials. Amid a post-war boom in polymer science and electrical engineering, researchers homed in on compounds like 1,3-dioxolane for their stability and ease of manufacturing. Its emergence reflects a wider push to build better materials, connect synthetic steps, and unlock new areas of industrial chemistry rather than settling for what was already known.
Product Overview
A bottle of 1,3-dioxolane might look unremarkable, almost like water, with no color and a mild, pleasant smell. But anyone who has spent time at the bench knows its value. Used for stripping lithium batteries apart, cleaning up in labs, or making new polymers, 1,3-dioxolane stands out for dissolving both salts and organics. Unlike many mainstay solvents, it keeps going at lower temperatures and doesn’t break down easily, so it finds its way into tricky extractions or battery electrolytes. Its low boiling point also means it can be removed easily after finishing work—one less headache in purifying compounds.
Physical & Chemical Properties
Clear liquids like 1,3-dioxolane hide their strongest features at the molecular level. Its formula, C3H6O2, forms a five-membered ring with two oxygens. This ring stores some chemical tension, making it reactive in the right hands while staying safely stable under normal conditions. You can light it as it has a low flash point, so caution is vital in crowded labs. It dissolves well in both water and most organic solvents, bridging a tricky divide where many competitors fail. This makes it especially handy for formulations that need to pull both polar and non-polar ingredients together.
Technical Specifications & Labeling
Chemists and safety officers look carefully at technical details before a purchase. Purity ranks highest—typically, high-grade 1,3-dioxolane clocks in above 99 percent, often checked by gas chromatography. Water content, another big deal for battery and pharmaceutical work, stays below the half-percent mark in the best samples. Quality bottles include hazard warnings, clear expiry dating, and batch numbers to allow any safety alerts or tracking. Flammability warnings remain front and center. Labs relying on rigorous documentation often choose brands where those standards always stay the same.
Preparation Method
Making 1,3-dioxolane usually starts with ethylene glycol and formaldehyde in acidic conditions. Acid acts like a traffic cop, steering reactive intermediates together and out pops the ring-shaped molecule. Industrial production moved past open-flask reactions to closed, controlled vessels, increasing yields and cutting down on byproducts. Years of process refinement allowed large batches without raising risks, letting it become a common sight on factory floors and university storerooms. Good control at the preparation stage keeps impurities out, which makes a difference in high-stakes jobs—like electronics or drug research—where every contaminant counts.
Chemical Reactions & Modifications
Anyone who has spent time in synthetic chemistry knows cyclic ethers like 1,3-dioxolane can act as both partner and player. It behaves as a protective group for sensitive aldehydes and ketones, stepping in to shield delicate functional groups from harsher reagents. By building a temporary “shell” through acetal formation, it allows complex synthetic steps without losing a precious starting material. After the heavy work, a bit of acid or water cracks that shell, freeing the core chemical for analysis or next steps. In battery science, the molecule reacts cleanly with lithium salts, making it vital for safe, efficient non-aqueous electrolytes. While not often the main event, 1,3-dioxolane quietly connects many worlds—from material science to pharmaceuticals.
Synonyms & Product Names
Chemical catalogs come alive with alternative names for 1,3-dioxolane. Some may recognize it as 1,2-dimethylene oxide, methylene oxide, or by CAS number 646-06-0. Makers in Europe and North America list it under slightly tweaked spellings or catalog labels, but the basic five-membered ether ring always stands as the core. These varieties reflect global demand, where regulation or branding forces suppliers to tweak packaging or names, although the underlying compound doesn’t change. In the field, chemists grow familiar with all aliases to ensure smooth ordering and supply.
Safety & Operational Standards
Handling solvents demands respect, and 1,3-dioxolane is no exception. Its low flash point and ability to produce explosive peroxides after long storage means that proper ventilation, fire safety, and regular monitoring become routine in any responsible lab. Experienced chemists know to date every bottle and ditch anything past its prime, avoiding dangerous byproducts from collecting. Many labs use peroxide test strips and keep stocks small, rotating through fresh supplies frequently. Safety data sheets urge gloves, eye protection, and usually a well-fitted fume hood when dealing with large volumes or heated reactions. Every mishap can cost more than just downtime; a laboratory fire or exposure can last a lifetime.
Application Area
Years in research and industry have shown that 1,3-dioxolane rarely stays in one spot. It acts as a solvent in lithium batteries, giving engineers options for building safer, longer-lasting energy storage. Polymer scientists reach for it during polyacetal resin production, while synthetic chemists use it to guard reactive carbonyls through tricky steps. Its dual polarity allows pharmaceutical processes to move smoothly from water-based mixes into organic syntheses without swapping out solvents between steps. In the restoration of old paintings, conservators use it for gentle cleaning where aggressive reagents threaten irreplaceable pigments. These examples don’t just showcase versatility—they reveal a substance increasingly hardwired into progress across disciplines.
Research & Development
Looking at recent research, much of the focus has shifted toward the role of 1,3-dioxolane in advancing energy solutions. Growing demand for lithium-ion and lithium-metal batteries pushes teams to investigate how electrolytes based on this molecule can help stabilize metal anodes and suppress dangerous dendrite growth. Academic studies dig into ways to blend it with cosolvents or additives, aiming to boost battery lives and withstand tough charging cycles. On the materials side, polymer research explores reactions that improve environmental profiles, searching for greener synthesis with less toxic waste. New methods in catalysis using cleaner conditions may soon open doors to safer, more scalable preparations, inviting even broader industrial acceptance.
Toxicity Research
Understanding chemical toxicity goes beyond trusting a label—lab safety depends on real knowledge passed through experience. Studies have shown that 1,3-dioxolane produces acute effects mostly at high exposure, with symptoms including dizziness, headaches, or respiratory irritation. Chronic studies remain limited; most data comes from animal models rather than human epidemiology. Regulatory agencies classify it as a flammable liquid with moderate health risks, not in the highest tier of dangers but always demanding attention. Keeping exposures low and air quality high can prevent health incidents. Air monitoring and personal protection, even for short jobs, reduce risk to those depending on a lifetime spent working with chemicals.
Future Prospects
The story of 1,3-dioxolane doesn’t freeze with current uses. As the world seeks better batteries, cleaner processes, and less toxic syntheses, materials with proven track records gain new attention. Startups and researchers look at its blend of solvency, volatility, and relatively safe profile, hunting for ways to incorporate it into advanced manufacturing. Environmental pressure will likely spur greener production or recycling programs, tightening standards to close the loop. New regulations may soon demand deeper toxicological studies or force a transition toward alternatives in consumer-facing goods. For now, anyone working at the boundary between chemistry, energy, or materials keeps a close eye on developments—with 1,3-dioxolane still playing a quiet but vital role in the search for the next technological leap.
What is 1,3-Dioxolane used for?
Chemistry’s Unsung Helper
Step into a lab or chat with a chemical engineer about versatile solvents, and you’ll likely hear stories about substances that punch above their weight. One of these is 1,3-dioxolane. This clear, colorless liquid often goes unnoticed outside specialist circles, yet it unlocks a surprising range of uses in research and industry. Its molecular structure gives it a knack for dissolving both polar and non-polar compounds, and this mix of flexibility and energy efficiency explains why it pops up where you least expect it.
Revving Up Lithium Batteries
Look at how modern devices power up, and you'll find lithium batteries. The push for lighter cars and longer-lasting phones drives the search for better battery solutions. 1,3-dioxolane enters the picture as a top pick for electrolyte solvents in lithium batteries. Its low viscosity helps ions move smoothly, and its chemical stability even under high voltage keeps tech safer. Battery makers reach for 1,3-dioxolane as they chase improved cycle life and capacity. Research out of Japan and Europe confirms that blending it with other solvents raises safety margins and lowers chances of shorts or thermal events.
Making Polymers Smoother
Factories crafting plastics use 1,3-dioxolane as a solvent whenever they need to spin up polyacrylonitrile fibers—core ingredients in carbon fiber and specialty textiles. It does more than dissolve stubborn polymers; it turns a messy job into a smooth flow in textile and plastics equipment. From my own time shadowing plant technicians, I noticed how it cut downtime and cleanup cycles. That means lower energy bills and less waste, two things any company likes to hear. The European Chemicals Agency backs its use for this reason, though they urge that handlers respect exposure limits and install good ventilation.
Pushing the Envelope in Medicine
Some pharmaceutical labs trust 1,3-dioxolane to speed up key reactions, especially for forming sensitive ring structures and extracting plant-based ingredients. Organic synthesis can tie up hours if the right solvent is out of reach. By using 1,3-dioxolane, chemists often trim lab time and cut back on harsher solvents like dichloromethane or chloroform. Regulatory agencies flag 1,3-dioxolane for careful control since traces sometimes sneak into finished drug products, but methods like vacuum drying and purging drop residual levels well below risk thresholds.
Tackling the Safety Puzzle
Praise for 1,3-dioxolane’s versatility comes with reminders about responsible use. Its rapid evaporation means it won’t linger in workspaces, but it can irritate eyes or lungs if workers skip proper masks or fume hoods. Storage with good labeling and spill control helps keep accidents rare. Health agencies in the US and EU list it as a potential health hazard at high exposures, linking it to headaches or dizziness. Industry pushes for better monitoring and more robust personal protective equipment so teams stay safe. Getting alternatives ready that match its performance without added risks keeps researchers busy—some are testing dimethyl carbonate and related solvents to see if they measure up.
Looking Forward
The push for electric vehicles, safer batteries, and greener factories means solvents like 1,3-dioxolane will keep filling important gaps. Its future depends on transparent risk management, honest reporting on health data, and investment in next-generation solvents. In the meantime, anyone working with it knows firsthand how one chemical can smooth out big challenges in labs, factories, and product design.
Is 1,3-Dioxolane hazardous or toxic?
Looking at the Chemical in the Real World
Anyone who’s spent any time in a lab or on a factory floor has come across chemicals with names like 1,3-dioxolane. Most folks see a clear liquid, give it a respectful nod, and hope someone up the chain knows exactly how risky it is. 1,3-Dioxolane shows up in a lot of places—solvent in the plastics industry, cleaning agents, even in some pharmaceutical synthesis. Its low boiling point and ability to dissolve a range of stuff draw attention far beyond academic labs.
Real Hazards for Regular People
Getting specific, 1,3-dioxolane will irritate skin and eyes if it spills or splashes. Based on my own years of dealing with solvents, most folks try to minimize direct contact simply because the burning or stinging is immediate and memorable. Inhalation raises extra worries. The liquid tends to evaporate at room temperature, turning into invisible vapors you can breathe in without knowing it. That’s the point where headaches, dizziness, or nausea can sneak up—classic signs the body’s telling you to step outside and get some air. Long stretches in poorly ventilated spaces increase the risks, as the body builds up exposure over time.
Digging into Toxicity and Environmental Impact
Toxicity isn’t just about whether something kills you on the spot. For 1,3-dioxolane, data from animal studies shows problems with bigger doses: central nervous system trouble, and some organ toxicity in severe cases. In most workplaces, exposure doesn’t hit those levels, but the U.S. Environmental Protection Agency and European regulators recommend limits on how much can be floating around. That tells us enough—to ignore routine safety is to roll the dice with long-term health. Water contamination also warrants focus. This solvent dissolves well in water, so an accidental spill can travel far, impacting soil and aquatic life. Breakdowns in wastewater treatment don’t always fully remove these solvents, and fish or invertebrates down the stream can pay the price.
The Role of Regulation and Workplace Safety
Most manufacturing outfits treat 1,3-dioxolane like a flammable, moderately toxic substance. That means strict control on how much stays in the air, along with gloves, goggles, and sometimes even respirators. Emergency response guidelines spell out clear steps for cleaning up spills, and chemists, engineers, and line workers get trained not because they expect something to go wrong, but because hoping nothing happens doesn’t stop accidents. Agencies keep updating guidelines as new research comes in—setting exposure limits and updating safety data sheets. It’s not paranoia or overreaction; it’s the result of enough evidence showing that repeated low-level exposure adds up.
What Makes a Real Solution?
No single fix applies everywhere, but some choices always help: Better ventilation makes the biggest difference. Fume hoods, exhaust fans, and open doors aren’t fancy, but nothing beats fresh air for reducing vapor. Safe handling shows up in habits—folks double-checking labels before pouring, storing containers tightly sealed, and keeping chemical spill kits within reach. If greener alternatives turn out to work just as well, many industries switch. Regulatory watchdogs support that shift by flagging chemicals with safer profiles and encouraging companies to reconsider what they use.
Trust, Transparency, and Staying Informed
My experience says never to rely on gut instinct with chemicals. Data and transparency matter more. The story of 1,3-dioxolane shows how practical measures keep harm in check, and how workplace culture—education, shared responsibility, reporting near-misses—often sets the tone for actual safety. If you’re unsure about exposure or the best ways to avoid harm, asking questions and demanding clear guidance means fewer regrets later. Trust comes from open information, and real-world health grows out of small, daily choices by people who pay attention.
What are the storage and handling requirements for 1,3-Dioxolane?
The Real Risks in Everyday Work
Ask anyone who’s worked in a chem lab or a facility that uses solvents, and you’ll hear the same thing: 1,3-Dioxolane can cause real trouble if ignored. Its sweet ether-like smell tricks new people into thinking it’s harmless. It isn’t. This liquid flashes at low temperatures, making fire a constant risk. Leaving a container open, even for a short time, quickly fills the air with vapors. Ignoring established rules not only places workers at risk, but can cause damage to expensive equipment—and more importantly, lives.
Storing For Safety
During my years spent around small-scale chemical production, “dry, cool, and well-ventilated” was repeated so often I started seeing it in my dreams. Why? Humid or warm storage helps dioxolane break down or form explosive peroxides. Nobody wants that surprise. Locking up this compound in a tightly closed steel drum and placing it in a chemical storage shed away from sunlight and ignition sources became standard in every workplace I knew. Forget once, and you could be replacing a whole inventory after contamination. Acids and bases should sit in separate rooms. Dioxolane eats through soft plastics, so companies stick with stainless steel or compatible glass. Each storage area displays clear hazard labels—some for insurance, some by law, but always to keep new hires sharp.
Handling: Routine, Not Casual
Careless moments don’t just damage health—they cost jobs and reputations. The liquid soaks right through hold-tight gloves if you pick the wrong material. In my teams, we always used nitrile or butyl rubber gloves, and changed them often. During transfers and pouring, face shields or goggles and flame-retardant coats aren’t just for show. Even with years behind you, dioxolane splashes happen quickly. Fume hoods run every time a bottle opens, never skipped. Even trace vapor clouds irritated my lungs more than any instructor’s warning said they would.
Spills belong in the “deal with it now” category. Granular absorbents, not shop towels, stop spread and keep reactions in check. Disposal takes a special touch. We learned early not to just toss old dioxolane down the drain—the local water authority once fined our shop after a worker thought the small amount “wouldn’t matter.” It mattered plenty. Disposal contracted to licensed handlers became policy, and it stuck for good reason.
Learning From Regulations—And Real Life
Regulatory bodies, from OSHA to the EU’s REACH system, carry heavy fines for missed hazard communication and unsafe storage. They built those rules after years of real-world accidents. This punchy solvent counts as both flammable and a health hazard. Training refreshers every six months kept old hands and newcomers aware of the basics. From reviewing the Safety Data Sheet (SDS) before working to knowing the nearest fire extinguisher, basics never get old. Fire drills felt excessive at first, but the day a storage drum leaked and set off an alarm, we emptied the hall in seconds with no injuries.
Improvements on the Horizon
Chemical manufacturers have started offering safer, pre-mixed blends to limit risk. Engineer friends mention push-to-connect transfer systems and smart sensors tracking vapor levels as up-and-coming fixes. Simple steps like double-checking permanganate test strips for peroxide formation every month can save thousands down the line. People benefit from peer pressure—in our workplace, nobody mocked double-checkers, because we saw what a slip-up does. Experience, solid planning, and enforced rules guard both workers and property.
What is the chemical formula and structure of 1,3-Dioxolane?
What 1,3-Dioxolane Looks Like on Paper and in Practice
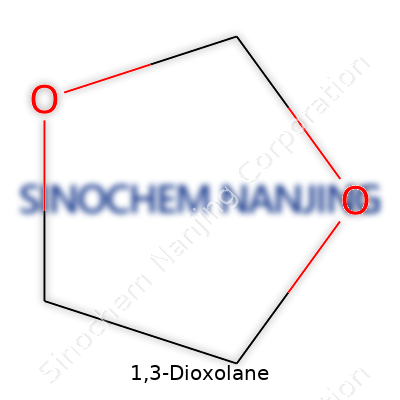
People working in any chemical lab or industrial environment eventually run into 1,3-dioxolane. This five-membered ring molecule, often shown as C3H6O2, carries a punch both in synthetic chemistry and various industrial processes. Its cyclic structure, with two oxygen atoms set apart by a carbon atom, creates a shape that opens up doors for all kinds of reactions and uses.
Dioxolane doesn’t feel esoteric. Chemists find its structure straightforward: two oxygens at positions 1 and 3 in a ring joined by three carbons. This arrangement might look simple, but it shifts the properties enough to carve out its place as a workhorse in research, manufacturing, even battery technology. Anyone handling solvents or electrolytes will recognise its distinct, slightly ether-like smell and quick-to-evaporate character.
Why the Structure Actually Matters in Real Life
Chemical structure isn’t just textbook trivia. Those two oxygens in the ring don’t sit idle—they inject reactivity and a measure of polarity. When I worked in a small formulation lab, dioxolane turned out to be the only solvent that dissolved certain stubborn electrolytes fast enough to run same-day tests. Its unique geometric arrangement helped stabilise intermediates, especially when we needed anhydrous conditions. You notice performance differences between a generic cyclic ether and dioxolane the first time you try purifying delicate compounds—losses drop, and columns run faster without clogging.
Electrochemists talk about dioxolane as a star in lithium battery research. Its ring structure supports high permittivity and rapid ion movement, essential for keeping a steady stream of current. Battery engineers used to fight breakdowns at high voltages—dioxolane made it possible to push the chemical limits, drawing longer run times out of test cells. Specific structure here shapes the molecule’s ability to resist chemical attack while still dissolving lithium salts, something not every ether brings to the table.
Risks, Knowledge, and Safer Paths with Dioxolane
Benefits don’t erase the hazards. Dioxolane’s volatility—directly tied to its structural features—means fast evaporation and easy ignition near open flames or hot surfaces. It evaporates so quickly that spills dry up before you grab a towel, but that means it’s in the air, and inhaling those vapours never sits well with health and safety officers. In our lab, we learned to push for better ventilation, even basic fume hoods, before cracking open a fresh bottle. Publications point to its irritant properties, and the flammability class lands around the same spot as other low-weight ethers.
Waste management teams run into trouble if protocols ignore the ring closure feature—burning dioxolane takes more oxygen than some linear ethers. Here experience changes habits: fire marshals recommend storing it in explosion-proof cabinets, away from oxidising agents, based on real-world incident reports, not just rulebooks.
Smart Use and Future Prospects
Demand for dioxolane reflects its structure. People working in green chemistry have started hunting for alternatives with similar geometry but improved biodegradability, partly because regulations in Europe and California stare down volatile organics. Recent literature shows research teams tweaking the dioxolane ring, inserting side groups, hoping for lower toxicity and better retention in solvent recycling systems. This approach doesn’t just sound green on paper; it saves money and cuts down hazardous waste streams for companies forced to keep a close eye on compliance and disposal costs.
Can 1,3-Dioxolane be used as a solvent in chemical reactions?
Getting Practical with Lab Work
In my experience, chemists tend to combine pragmatism with creativity. The tools and solvents we use often shape our reactions, not just in yield but in feasibility. 1,3-dioxolane isn’t a household name, even among scientists, yet it’s got a certain versatility that makes it stand out for people willing to work beyond the usual ethyl acetate or THF.
Solubility and Reaction Compatibility
This solvent dissolves both polar and nonpolar compounds. Polymers, organometallics, and some stubborn inorganics part ways in 1,3-dioxolane, even when other choices stall. A colleague once told me how she saved an entire batch of lithium batteries in development by swapping out another ether for dioxolane, because it handled salts better. Journal articles back up that flexibility, especially in Grignard and borohydride reductions.
Safety Considerations Don't Disappear
Schoolbooks and safety sheets always mention flammability and carcinogenic risks with organic solvents. 1,3-dioxolane joins the crowd here, so nobody cuts corners. Fume hoods, gloves, and constant checks for peroxide formation become routine. This isn’t just box-ticking, either—I’ve seen peroxide crystals form at the neck of a bottle left open too long, and it spooked everyone in the lab. Using stabilizers or testing for peroxides before distillation isn’t negotiable. Researchers need to approach with respect and a plan for safe disposal, not just hope nobody notices if something smells odd.
The Role of Stability and Environmental Demand
Some solvents fight moisture. 1,3-dioxolane holds its ground better than tetrahydrofuran when it comes to resisting hydrolysis. That extends shelf life in storage rooms with changing seasons and questionable humidity, and it eases the headaches with reactions that can’t tolerate water. Still, ethereal solvents carry environmental baggage. I’m careful when I open a new drum, knowing there’s increasing pressure to green up lab and industrial processes. Groups like the ACS Green Chemistry Institute highlight alternatives, but dioxolane tends to pop up when chemists weigh risks against benefits in electrochemistry or polymer synthesis.
Cost and Accessibility Shapes Choices
Academic labs usually keep budgets tight, so the price tag sometimes trumps ideal performance. Dioxolane costs a bit more than THF, but less than other “exotic” choices. Most chemical suppliers stock it in multiple grades. That kind of access speeds up research and troubleshooting—less time waiting, more time experimenting.
Real-World Applications and Some Cautions
In a pilot plant setting, 1,3-dioxolane allows for scale-up of reactions that would otherwise stall in larger reactors. It doesn’t just play a support role—it can make the chemistry work where other options give inconsistent results. That said, experienced chemists talk shop about the dangers of substitution without deeper analysis. Swapping solvents changes pressure, temperature, and kinetics. Pulling a method from a paper and copying it with dioxolane without test runs has led to ruined projects or, worse, dangerous situations.
Looking Ahead
Regulators and research leaders both watch solvent selection carefully. The next generation of lab pros keeps an eye on safety, sustainability, and performance all at once. 1,3-dioxolane brings possibility, but also risk. My best advice: don’t use it just to “try something different.” Weigh risks, check the literature, run pilot reactions, and get genuinely curious about what works. That’s what keeps real progress moving—and people safe.
| Names | |
| Preferred IUPAC name | 1,3-Dioxolane |
| Other names |
Dioxolane Methylenedioxyethane 1,3-Dioxacyclopentane Formal ethylene acetal 1,3-Ethylene formal |
| Pronunciation | /ˌwʌn.θriː.daɪˈɒk.sə.leɪn/ |
| Identifiers | |
| CAS Number | 646-06-0 |
| Beilstein Reference | 1207193 |
| ChEBI | CHEBI:28347 |
| ChEMBL | CHEMBL54559 |
| ChemSpider | 4978 |
| DrugBank | DB11125 |
| ECHA InfoCard | ECHA InfoCard: 100.005.864 |
| EC Number | 206-016-6 |
| Gmelin Reference | 8217 |
| KEGG | C07125 |
| MeSH | D003994 |
| PubChem CID | 7921 |
| RTECS number | JL5950000 |
| UNII | I8X9NYA2JF |
| UN number | UN1165 |
| CompTox Dashboard (EPA) | DTXSID2020227 |
| Properties | |
| Chemical formula | C3H6O2 |
| Molar mass | 74.08 g/mol |
| Appearance | Colorless liquid |
| Odor | Ether-like |
| Density | 1.06 g/mL at 25 °C (lit.) |
| Solubility in water | Miscible |
| log P | -0.37 |
| Vapor pressure | 56.3 mmHg (20°C) |
| Acidity (pKa) | pKa = 18.4 |
| Basicity (pKb) | -1.29 |
| Magnetic susceptibility (χ) | -61.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.407 |
| Viscosity | 0.589 mPa·s (25 °C) |
| Dipole moment | 1.66 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -368.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2116 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H319, H336 |
| Precautionary statements | P210, P261, P280, P304+P340, P312, P403+P233 |
| NFPA 704 (fire diamond) | 1,2,0 |
| Flash point | -2 °C (28 °F; 271 K) |
| Autoignition temperature | 183 °C |
| Explosive limits | 2.5–25% (in air) |
| Lethal dose or concentration | LD50 oral rat 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 3200 mg/kg |
| NIOSH | YN8225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,3-Dioxolane: 100 ppm (360 mg/m³) |
| REL (Recommended) | 50 ppm |
| IDLH (Immediate danger) | 3000 ppm |
| Related compounds | |
| Related compounds |
1,2-Dioxolane 1,3-Dioxane Tetrahydrofuran Ethylene glycol Acetals |

