1,3-Dichloro-2-Propanol: Looking Deeper at an Old Challenge
Historical Development
The tale of 1,3-Dichloro-2-Propanol, usually shortened to 1,3-DCP, links back to the long-standing industrial phase when synthetic organochlorines burst onto the chemical market. Chemists saw chlorinated propanols as useful bridges toward creating new materials, with 1,3-DCP snagging attention for how easily it entered reactions. Synthetic chemists in the mid-20th century identified chlorinated propanols almost by accident while trying to develop new intermediates for polymers and surfactants. The boom in industrial chemistry gave birth to substances like 1,3-DCP, showing just how quickly innovation could outpace a detailed understanding of safety.
Product Overview
Some call it by different names—from 1,3-dichloropropan-2-ol to glyceryl dichlorohydrin. The substance sits in that group of colorless liquids that sneak into a surprising number of applications. 1,3-DCP appears in the background of epichlorohydrin and surfactant manufacturing. Most people outside the chemistry sector rarely hear of it, but almost everyone has, at one point, come across goods whose processes involve this compound. The fact that this chemical slips so easily into daily products is what gives its story that extra layer of urgency and responsibility.
Physical and Chemical Properties
Anyone trained in a chemistry lab will recognize 1,3-DCP by its sharp, pungent odor and moderate volatility. Its solubility in water and organic solvents raises practical handling questions. With a moderate boiling point just above standard room temperature, the compound doesn’t take much to vaporize, reminding anyone nearby of its presence by its smell. Stability at ordinary conditions doesn’t always guarantee a lack of risk. The pair of chlorine atoms on its three-carbon backbone makes it reactive enough to find its way into further synthesis. Its physical data reads like a typical solvent, but the presence of chlorines changes how you treat it—never a good idea to rely on habits from working with alcohols alone.
Technical Specifications and Labeling
Industrial shipments of 1,3-DCP require rigorous attention to detail. Oddly enough, in my own time working around specialty chemicals, labeling errors rarely happen in the lab—mistakes often creep in during storage or because suppliers use outdated synonyms. Confusing one chloropropanol for another, especially when names overlap, sets the stage for accidents. Proper documentation of concentrations, batch numbers, and storage conditions prevents losses and keeps handlers safe. Regulated container standards reflect the lessons learned from decades of incidents where containers degraded or labeling wore off, giving way to costly mistakes down the line.
Preparation Method
Most technical-grade 1,3-DCP comes from controlled reactions between glycerol and chlorinating agents, often using phosphorus trichloride or thionyl chloride. This production method appears simple on the surface. Yet, the steps demand strong oversight to combat unwanted side products and reduce waste streams. Troubleshooting these reactions by small feed changes or temperature controls usually takes up more time than the actual run. From my own background in batch processing, I’ve seen just how easily the process can swing toward unwanted dichlorinated or trichlorinated compounds, which only ramps up purification headaches. High-yielding systems strike a delicate balance between conversion rate and keeping impurities at bay—a constant dance in chemical manufacturing.
Chemical Reactions and Modifications
The two chlorines on 1,3-DCP give this molecule a variety of reaction gateways. Nucleophiles approach either carbon, leading to functionalization, etherification, or opening the door to other syntheses—if steered with skill. At lab scale, chemists exploit this dual reactivity for custom syntheses, especially where a chlorinated allyl or propanediol unit forms a key structure. Larger operations need the same selectivity, only magnified by concerns over runaway reactions. Each chemical tweak inches the substance closer to value-added products like epichlorohydrin, which itself spins outward toward resins, elastomers, and more. The flexibility of 1,3-DCP speaks to the creativity, and sometimes the hazards, inherent in synthetic chemistry.
Synonyms and Product Names
A frustrating feature of 1,3-DCP’s reputation comes from its many aliases. Glyceryl dichlorohydrin, 2,3-dichloro-1-propanol, and more than a handful of trade names populate documentation from commercial sources. This naming confusion can slow research, spark legal debates over patent rights, or complicate analysis on lab safety audits. Training new chemists to check CAS numbers has become essential. As regulations tighten, transparent communication through labeling and coordinated databases gains new importance. I recall sorting through years-old chemical stocks and learning just how quickly confusion over synonyms can risk both productivity and safety.
Safety and Operational Standards
Toxicological studies made it clear that contact or inhalation brings significant risks. The eyes, respiratory tract, and skin react sharply even at low levels. Regulatory bodies like OSHA and the European Chemicals Agency list 1,3-DCP as hazardous, calling for gloves, fume hoods, and limited exposure periods. Reproductive toxicity and potential carcinogenic pathways have prompted greater study, with both lab-scale and industrial users urged to log exposure and seek substitutes where possible. In my early days, casual attitudes toward legacy solvents met harsh lessons from chemical spills. Regular training, quick engineering controls, and robust spill procedures have moved from “nice-to-have” to a baseline expectation in any responsible operation.
Application Area
Industry leans on 1,3-DCP mainly as an intermediate. Its role in epichlorohydrin manufacture, precursor to epoxy resins, puts it front and center in the plastics sector. Textile finishing, surfactant production, and some specialized pharmaceuticals all dip into this chemical pool, sometimes knowingly, sometimes through upstream intermediates. On paper, the compound opens many synthetic doors; in practice, the conversation shifts toward limiting direct use where possible, due to health concerns and shifting regulatory landscapes. Chemical companies now audit their supply chains more closely to track and, if viable, limit residual 1,3-DCP in final products.
Research and Development
Efforts in both academic and industrial research move toward refining synthesis, minimizing by-products, and inventing greener ways of working with or around 1,3-DCP. Enzymatic routes, for instance, now look more promising as groups investigate biocatalytic processes for halogenating glycerol with fewer emissions. Analytical chemists have become essential in developing better detection techniques—critical for tracking down low-level contaminants, especially in foods or environmental samples. Improvements in sampling and analytical chemistry move the needle when regulators tighten up acceptable limits in drinking water and food-grade supplies.
Toxicity Research
Toxicologists and public health officials have their work cut out with 1,3-DCP. Animal studies have shown liver and kidney trouble after chronic exposure. Human risk evaluation remains fraught with gaps, but data from accidents and workplace exposure keeps regulatory committees on alert. In food, minute traces have popped up as by-products of fat processing, prompting groups to petition for lower permissible levels. A multidisciplinary approach, including epidemiologists, molecular biologists, and industrial hygienists, continues to build a more complete picture. Most toxicological concern now centers on carcinogenic risks—driving companies to rethink not just end product safety but every step of the supply chain.
Future Prospects
Sustainability, safety, and regulatory friction all steer the future of 1,3-DCP. Green chemistry trends bring pressure to cut toxic intermediates, or at the very least, tighten up containment and waste controls. Companies with strong R&D arms invest in alternate synthesis routes or, better yet, drop-in replacements. Advances in detection science will likely keep risk management front and center, as the smallest misstep can trigger costly recalls or plant shutdowns. In my own experience, the more open a company’s culture is to listening to lab workers and frontline operators, the better it handles transition periods when phasing out risky intermediates. Strong collaboration across the regulatory, scientific, and business sectors usually turns potential crises into manageable projects. The case of 1,3-Dichloro-2-Propanol, after decades in the shadows, brings into sharp relief how modern industry must reckon with legacy chemistries using today’s knowledge, skills, and technology.
What is 1,3-Dichloro-2-Propanol used for?
What Is 1,3-Dichloro-2-Propanol?
1,3-Dichloro-2-propanol looks like a chemical you usually recognize only by a long label on a drum or a blurry line in a textbook. Most people will never see it at work, but it ends up influencing more than many realize. At its core, it’s a chlorinated alcohol, and the main setting for its story is industry, not your corner grocery shelf.
Industrial Chemistry and Manufacturing
Factories have turned to 1,3-dichloro-2-propanol for decades, particularly in making epichlorohydrin. This epichlorohydrin then helps build epoxy resins. You see those in construction, car parts, and the protective coatings on metal. These coatings stand up to heavy wear and tear, from harsh weather to chemical splashes. The demand for durable finishes keeps this chemical’s production moving along.
A lot of people touch or feel products treated with materials that trace back to 1,3-dichloro-2-propanol—items like circuit boards, flooring, and even marine paint. Epoxy resins almost always mean safer, longer-lasting surfaces. Epichlorohydrin’s role turns out crucial not just because of toughness, but due to its resistance to water, heat, and solvents.
Concerns in Food Processing
Stories surfaced over the years about 1,3-dichloro-2-propanol showing up in food or packaging. Traces appear during the processing of certain foods, like some soy sauces, thanks to contact with processed fats and acids. International health agencies called for tighter monitoring in response. The International Agency for Research on Cancer classifies it as possibly carcinogenic to humans based on animal studies. Regulatory agencies, including in Europe and the United States, set strict limits on allowable residue in food and packaging.
From my days working near food regulatory teams, the focus on detection and prevention stands out. Food manufacturers regularly run their equipment and finished goods through quality checks specifically for contaminants like 1,3-dichloro-2-propanol. No plant manager wants to risk consumer trust or stricter audits. Scientists keep fine-tuning how they spot even tiny amounts in complex mixtures. That’s pushed forward a new generation of sensitive test kits and lab protocols.
Worker Safety and Environmental Impact
My previous visits to chemical plants bring back memories of strong smells and the importance of safety routines. Workers in spaces that deal with 1,3-dichloro-2-propanol rely on specialized gear—gloves, masks, extensive ventilation. Safety training always covers accidental exposure since this chemical irritates the skin and mucous membranes quickly. Long-term contact can lead to more serious risks, so training and monitoring continue on the job.
Disposal holds its own challenge. While treatment systems can destroy most of the chemical before wastewater leaves a facility, surprises still show up downstream. Environmental watchdogs and local testing teams raise the alarm if levels run too high. This has pushed manufacturers and environmental engineers to focus on on-site capture and breakdown before anything escapes to water or soil.
Looking Ahead
Growing demand for safe products keeps pushing chemists to invent new methods that sidestep potential risks. Green chemistry now pushes companies to experiment with less hazardous intermediates for epoxy production. While it still plays a role in several sectors, tighter regulations and new technology could make older compounds less critical in the next few decades.
Is 1,3-Dichloro-2-Propanol hazardous to health?
Understanding the Basics
People don’t usually hear much about 1,3-dichloro-2-propanol (1,3-DCP) outside of industrial circles or food safety reports. This chemical starts as a colorless liquid, showing up during the production of other chemicals, like epichlorohydrin, or as a byproduct in some processed foods. It’s not covered nearly as often as well-known hazards like asbestos or pesticides, but concerns about its impact on human health keep surfacing—and for good reason.
Real Health Risks
The trouble with 1,3-DCP starts with its toxic profile. The International Agency for Research on Cancer flagged it as possibly carcinogenic to humans. Researchers saw clear signs of cancer and organ damage in animal tests. Even low-level exposure caused liver, kidney, and respiratory injuries. In my own work in food safety and environmental science, I've seen how even trace contamination in everyday products can trigger larger health anxieties, especially among families with small kids.
Stories often circulate about its detection in fatty foods, soy sauce, and some Asian condiments. A few years back, a food safety report caught my eye. Imported soy sauce sold in European markets contained 1,3-DCP far above recommended limits. Lab results showed this chemical popping up during high-temperature processing, particularly where hydrochloric acid hydrolyzed vegetable protein. While manufacturers streamline their processes, consumers rarely realize that roasting or hydrolyzing foods might leave behind more than just intense flavors.
Routes of Exposure
Direct contact doesn’t pop up for most people since 1,3-DCP isn’t sold on store shelves for home use. Still, workers in chemical manufacturing face higher risks from spills or mishandling. In my extended family, a cousin once had to deal with chemical burns after a workplace spill involving a similar compound—incidents like these force better training and faster safety response in plants. Most people, though, face risks through diet, with residues slipping into liquid seasonings and processed foods.
The Regulatory Gaps
Scientists agree on the dangers, yet policy sometimes drags behind. The European Union set strict limits on 1,3-DCP in foods, but global enforcement stays patchy. In the United States, lack of labeling or adequate testing carries consequences. Food recalls make headlines, but many people find out late, after they or their kids have already eaten the contaminated product.
This divide between regulation and real-world exposure calls for more than simple testing. I’ve watched small importers scramble to update their processes only to find little government support or consumer guidance. Education—both for workers on the factory floor and for people planning their meals—matters as much as tighter rules. Clearer ingredient labels would give everyone more agency.
Pushing for Safer Choices
We can’t pull 1,3-DCP out of the food supply by just waiting for industry self-regulation. Routine screening, stricter limits on food additives, and cross-border information sharing all push the needle forward. Community voices often drive real change; after a grassroots campaign, one local chain near me dropped all questionable imported seasonings, pushing suppliers to meet higher standards. These changes start with knowledge, pushed along by people who ask tough questions about what ends up in their food.
Confronting chemical hazards like 1,3-DCP means more than accepting risks. It means pushing for transparent science, better safety practices, and a shared focus on public health over cheap shortcuts. That's where real progress happens.
What are the storage requirements for 1,3-Dichloro-2-Propanol?
Why Anyone Should Care About Chemical Storage
My time working with industrial chemicals taught me one thing: the details around storing every compound really do matter. 1,3-Dichloro-2-propanol, often used for manufacturing and research, packs a punch in terms of hazards. People sometimes think chemicals stay tame when they’re just sitting on a shelf. That outlook leaves room for mistakes that can affect people, property, and the environment.
Direct Exposure and Physical Risk
1,3-Dichloro-2-propanol comes with risks tied to its health effects. A splash or inhaling vapors can trigger skin burns, irritation of the eyes and lungs, or even more serious long-term concerns. I’ve watched new lab staff brush off safety sheets, only to regret it after accidental contact. No one enjoys an emergency eyewash visit—never mind the panic if someone can’t breathe properly. Keeping the chemical in tightly sealed, unmistakably labeled containers, stored well below eye level, cuts those accident chances to nearly zero.
Controlled Environments and Fire Hazards
Temperature and ventilation matter. 1,3-Dichloro-2-propanol isn’t something to keep next to fan heaters or in sunlight. Warm spots let vapors build up and raise the risk of fires. Chemical-specific storage cabinets—fire-rated and vented—keep both temperature and vapors in check. Keeping this substance away from ignition sources is just as important as storing gasoline far from open flames.
Material Incompatibility and Corrosion
Any chemical that reacts unpredictably with regular metals or bases belongs far away from those types of containers. Glass or compatible plastics make safer choices for 1,3-Dichloro-2-propanol. Over time, incompatible storage can mean leaks, damaged shelving, or fumes escaping. I’ve seen corroded drum lids and sticky shelving from those who did not think twice about compatibility—or who hoped a few months in the wrong canister might slide by. It rarely does.
Spill Control and Secondary Containment
Spills don’t respect intentions. Even a sealed container sometimes cracks. Simple trays or secondary containment bins, made from materials that handle the chemical, give a second line of defense. The cost difference between a decent spill tray and managing a building-wide contamination event makes the safer choice clear. I always insist that no one skip this step, no matter how tight storage space gets.
Handling Documentation and Access
Every storage area needs updated safety data sheets, clear signage, and easy access to emergency equipment like eyewash stations and fire extinguishers. Frequent checks make sure labels stay readable and containers aren’t decaying over time. Restricting who can reach hazardous chemicals to trained people sets a culture of care. That prevents both theft and accidents from curious but ill-prepared visitors.
Improving Storage Systems
No setup is perfect from day one. Regular audits, updated training, and a willingness to budget for better cabinets or more spill kits always pay off. Where possible, rotating older stock helps avoid surprise leaks from aging containers. Digital tracking systems—simple as a shared sheet or as advanced as a full chemical inventory platform—make it hard to lose track of what’s stored and where.
Making Storage Work—Every Day
Safe storage isn’t about paranoia—it’s about daily habits and respect for the risks that come with the work. Mistakes around chemicals rarely give second chances. By setting up the right physical safeguards, choosing compatible materials, and limiting access, anyone can avoid avoidable accidents and protect both people and the environment. That’s not only a smart move—it’s a duty for anyone working with chemicals like 1,3-Dichloro-2-propanol.
How should 1,3-Dichloro-2-Propanol be handled safely?
The Stakes of Chemical Safety
Nobody wants to end a shift with a trip to the emergency room. You open a drum of 1,3-Dichloro-2-Propanol, a chemical used for syntheses in many labs, and the sharp scent reminds you why you looked up those handling guidelines in the first place. This isn’t the kind of compound to take lightly: 1,3-DCP carries enough risk to remind anybody with sense that accident prevention starts before the bottle opens.
Direct Health Effects
Years in the lab have taught me to read the scary stuff before grabbing a pair of gloves. International Agency for Research on Cancer classifies 1,3-Dichloro-2-Propanol as possibly carcinogenic to humans. Skin, eyes, lungs—exposure attacks fast, causing irritation and, in higher concentrations, even more serious trouble. Swallowing it by mistake or breathing in the vapor can mean serious medical complications, including kidney and liver issues. No experiment or routine procedure is worth gambling with those odds.
Gearing Up for Safety
One fact stands out—protection is personal. Short sleeves and cotton gloves offer no defense; you want chemical-resistant gloves, goggles, and a well-fitted lab coat. Respiratory protection also matters if the room isn’t well-ventilated. Over the years, I’ve found protective gear uncomfortable in the summer, but it is worth every minute when splashes and spills do happen.
Ventilation and Storage Count
A fume hood does more than keep odors down. Working in the open means risking lung irritation or more, especially with chemicals that evaporate easily. Vent hoods pull harmful vapors away, giving those inside a better shot at avoiding accidental exposure. Storage also sets the tone for safety. Anything flammable, reactive, or even just unfamiliar shouldn’t sit on a crowded shelf. 1,3-Dichloro-2-Propanol should always live in a tightly sealed, clearly labeled container, somewhere cool, shaded, and locked away from incompatible substances like strong bases or oxidizers.
Planning for Spills
Even when you do everything right, a bottle tips over. Panic doesn’t clean chemicals off the bench or floor. Absorbent materials, proper neutralizers, and a clear protocol make a world of difference. Some labs I’ve worked in post step-by-step spill response guides right by chemical storage zones, so nobody fumbles for next steps if things go sideways. Waste disposal deserves the same attention to detail—no pouring down the drain, ever. Designate a waste container and log what goes in.
Training and Accountability
Reading a material safety data sheet isn’t just paperwork—it’s the start of every safe interaction. Regular safety drills and refreshers matter more than new equipment. I’ve seen colleagues grow complacent until an incident snaps everyone back to caution. Empowering every member of a lab or plant to speak up keeps lapses from snowballing into disasters. Involving the whole team in safety decisions and training breaks down the “someone else will handle it” attitude.
Building a Culture of Care
Respect for hazardous chemicals begins with education and builds as colleagues watch out for each other. Safety equipment, storage rules, and spill protocols matter, but what counts most is an honest understanding of the risks and open teamwork. This approach, backed by real-world facts and reinforced by steady routines, turns good intentions into habits that save limbs, organs, and sometimes lives.
What is the chemical structure and formula of 1,3-Dichloro-2-Propanol?
Looking Closely at the Structure
Every time I handle organic molecules in the lab, a clear structure makes the difference between a routine experiment and discovering a problem before it gets serious. Take 1,3-Dichloro-2-propanol. It shows up in industrial chemistry, often connected with synthetic processes—sometimes with baggage, since improper use can mean environmental and health risks. The structure deserves a walkthrough for anyone wanting to get their hands dirty in chemistry work or environmental science.
1,3-Dichloro-2-propanol carries the formula C3H6Cl2O. Visualizing that, it means a backbone with three carbons. Here’s the actual map: at carbon one, there’s a chlorine; at carbon two, you spot a hydroxyl group (–OH); carbon three ends with another chlorine. Thinking hands-on, imagine a three-step ladder with the first and third steps sticky from chlorine, and the middle slick from hydroxyl. This arrangement goes by the IUPAC name 1,3-dichloropropan-2-ol. A person using early chemistry kits might picture the propanol backbone and then tag chlorines at the outermost carbon atoms.
Applications and Concerns Right Now
Plenty of manufacturers use this compound as a steppingstone, especially in epichlorohydrin production—a mainstay for making epoxy resins. Resins end up in everything from smartphone cases to wind turbine blades. That’s where safety comes in: not every batch reaches completion, and leftovers sneak out from careless waste management. A structure with two chlorines and an alcohol tends to hang in water and resist natural breakdown. My experience tells me the stakes rise fast, because dichloro compounds show up in groundwater, and they aren’t easy to remove once they’ve made their way in.
Reports from places with rapid industrialization, like certain districts in China and eastern Europe, back this up. Regular detection of dichloro-propanol residues puts drinking water at risk, since compounds with chlorine atoms often carry high toxicity. The World Health Organization and Environmental Protection Agency both label this molecule as a probable carcinogen—meaning even trace amounts need a hard look, not just a casual test.
Building Safer Systems
Relying on facts and hands-in-the-dirt experience, I see robust chemical management as a key fix. Companies able to swap in greener synthesis routes—ones that skip harmful intermediates—usually cut long-term costs and avoid legal headaches. Where substitution won’t fly, tight air and water monitoring steps in. Waste treatment that taps advanced oxidization or structured filtration works; that includes pumping effluent through activated carbon or using UV light to crack chlorine bonds.
Education plays a big role. Lab workers trained to recognize structural risks from compounds like 1,3-dichloro-2-propanol not only shield themselves, but also protect the environment downstream. I’ve seen toolkits that combine structural models and hazard rankings turn new hires into safety champions faster than any lengthy manual could.
The Bottom Line for Everyday Use
This is more than a chemical formula scribbled on a board. Understanding the arrangement of a molecule like 1,3-Dichloro-2-propanol links directly to safer workspaces, cleaner water, and smaller medical bills for everyone nearby. For regulators, builders, and chemists, knowing the layout isn’t just academic—it's a shield, a warning sign, and sometimes an open door to a better solution.
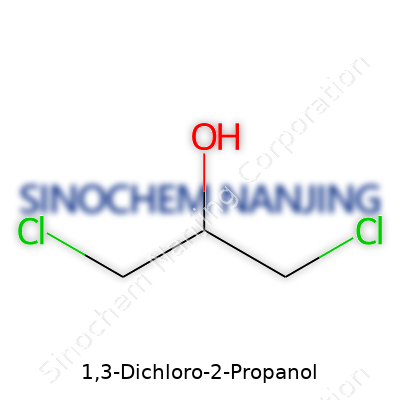
| Names | |
| Preferred IUPAC name | 2-chloro-1-(chloromethyl)propan-1-ol |
| Other names |
Glycerol dichlorohydrin 1,3-Dichloro-2-hydroxypropane beta, gamma-Dichloropropanol beta,gamma-Dichlorohydrin 1,3-DCP |
| Pronunciation | /ˌwaɪˌænθriːˌdaɪˈklɔːroʊˌtuːˈproʊpənɒl/ |
| Identifiers | |
| CAS Number | 96-23-1 |
| Beilstein Reference | 1209223 |
| ChEBI | CHEBI:34750 |
| ChEMBL | CHEMBL1507460 |
| ChemSpider | 56478 |
| DrugBank | DB08336 |
| ECHA InfoCard | 03d3e503-bd73-404a-bb5e-dabe3ee1dfa5 |
| EC Number | 202-474-5 |
| Gmelin Reference | 82222 |
| KEGG | C08360 |
| MeSH | D003982 |
| PubChem CID | 8017 |
| RTECS number | TX9625000 |
| UNII | X41C1C72AO |
| UN number | UN1992 |
| Properties | |
| Chemical formula | C3H6Cl2O |
| Molar mass | 127.00 g/mol |
| Appearance | Colorless to pale yellow liquid |
| Odor | chloroform-like |
| Density | 1.32 g/mL at 25 °C |
| Solubility in water | miscible |
| log P | 0.48 |
| Vapor pressure | 0.46 mmHg (25 °C) |
| Acidity (pKa) | 14.46 |
| Magnetic susceptibility (χ) | -53.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.464 |
| Viscosity | 22.1 mPa·s (20 °C) |
| Dipole moment | 2.64 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 171.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -277.0 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -728.7 kJ·mol⁻¹ |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H311 + H331, H315, H319, H351, H373, H411 |
| Precautionary statements | P261, P264, P270, P271, P301+P312, P304+P340, P305+P351+P338, P312, P330, P337+P313, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 1,2,2,3 |
| Flash point | 85°C |
| Autoignition temperature | 605 °C (1,3-Dichloro-2-Propanol) |
| Lethal dose or concentration | LD₅₀ Oral Rat: 110 mg/kg |
| LD50 (median dose) | LD50 (median dose): 160 mg/kg (oral, rat) |
| NIOSH | TX7525000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,3-Dichloro-2-Propanol: "No OSHA PEL established |
| REL (Recommended) | REL (Recommended): 75 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
1,2-Dichloro-3-propanol 2,3-Dichloro-1-propanol 3-Chloro-1,2-propanediol Epichlorohydrin Glycerol 1,3-Dibromo-2-propanol 1-Chloro-2-propanol |

