The Ongoing Journey of 1,2-Propylenediamine: From Discovery to Future Frontiers
Historical Development
People first paid attention to 1,2-propylenediamine back when the chemical industry was staking out its territory in the late 19th and early 20th centuries. Early researchers saw the natural curiosity in molecules with two amine groups, and diaminopropanes soon caught the eye of scientists hoping to tweak chemicals for new medicines, materials, and industrial applications. At that time, a growing understanding of amines opened up new reactions, leading to more specialized synthesis routes. These scientists realized that modifying chain lengths or changing the position of the amine groups, even by a single carbon, could drastically alter reactivity and end uses. Over the decades, labs across the world confirmed that 1,2-propylenediamine wasn't just some sidekick to other, better-known amines but a versatile molecule in its own right. Industrial production increased during the 20th century as its value in the creation of resins, epoxy curing agents, and pharmaceutical intermediates became clear.
Understanding the Product: What Makes 1,2-Propylenediamine Distinct
Compared to its close relatives like ethylenediamine, 1,2-propylenediamine offers a slightly wonky carbon backbone that separates its two amine groups by one more carbon, changing both reactivity and solubility in practice. The molecule consists of a propane backbone with amine groups attached to adjacent carbons, giving it both versatility and, sometimes, stubbornness in certain chemical reactions. In daily use, it’s a clear liquid with a strong ammonia-like smell, reminding anyone handling it that even a minor spill needs a good ventilated workspace. The extra carbon gives it properties that chemists favor for some reactions where too much reactivity, or not enough, can ruin a process. It carves out a spot for itself where companies need both flexibility and reliability, most obviously in the complex world of polymer chemistry and specialty chemical synthesis.
Physical & Chemical Properties
Many who have worked at a lab bench know that getting the physical and chemical properties right makes or breaks a batch. 1,2-propylenediamine usually boils off at just above 120°C, evaporating quickly if left uncapped. With high solubility in water and a modest density, it mixes easily for reactions. The presence of two primary amine groups makes it an excellent starting point for all sorts of chemical transformations, from simple acid-base neutralizations to more complex condensation reactions. Its reactivity can outpace simpler diamines in select cases, owing to both its basicity and molecular arrangement. Chemical suppliers often list its refractive index, vapor pressure, and flash point to remind users of its flammability and tendency to vaporize faster than some would expect. Even minor contamination, such as water uptake on exposure to air, can affect how reliable it acts in those more sensitive processes.
Technical Specifications & Labeling
Those who order or handle chemical drums soon realize just how precise labeling needs to be for chemicals like 1,2-propylenediamine. Labels specify not just the chemical name but typical concentrations, purity percentage, and any stabilizers added for long-term storage. Specifications often reflect demands set by local authorities and global standards—companies want to avoid cross-contaminating batches with even traces of common solvents like ethanol or acetone. Suppliers tend to keep impurities like water, chlorides, and other residual amines below strict thresholds since even minor slip-ups can trigger out-of-specification product downstream. The right storage conditions help tame its volatility; rust-proof drums and tight gaskets stop vapor loss and oxidation, protecting workers and end-users alike from accidental exposure. Responsible companies print hazard statements, pictograms, and handling instructions on every container—good for safety, and essential for regulatory compliance in any lab or plant.
Preparation Method: A Trusted Roadmap
In practice, most large-scale production starts with the reaction of 1,2-dichloropropane and ammonia under pressure, a process controlled by temperature and reactant ratios to maximize yield. Ammonolysis routes remain mainstay methods, not least because they allow continuous operation at industrial scale. Side products can pose separation challenges, so distillation becomes part of the everyday routine for plant operators. Smaller batches or lab synthesis sometimes lean on alternative starting materials, such as nitropropanes reduced over catalytic beds, but commercial demand sticks mostly to the chloropropane approach. Recovery and purification bring up memories of long days fussing with fractional distillation columns, sometimes spending hours, if not days, perfecting a curve on the output to get purity high enough for the next stage.
Chemical Reactions & Modifications in Industry
Chemists rely on 1,2-propylenediamine for its double-barreled chemical punch. Both amine sites react in slightly different ways, depending on the circumstances. You see this in polymer formation for epoxy curing, where cross-linking leads to tough and heat-resistant materials used in everything from wind turbine blades to coatings on floors and pipelines. Its structure nudges the curing speed and final characteristics of the polymer product; tweak the dosage or combine with other amines, and it’s possible to tune flexibility or hardness almost at will. In organic synthesis, it helps build heterocycles or acts as a ligand in catalysis, a detail that has made it a favorite in medicinal chemistry for drug candidate development. The two primary amines can be selectively protected and converted, opening up endless structural possibilities for downstream molecules. Reactions with acids, isocyanates, and aldehydes expand its reach into intermediate stages for pharmaceuticals and agrochemicals.
Synonyms & Product Names: A World of Aliases
In research papers and procurement databases, 1,2-propylenediamine wears a bunch of different hats—people call it 1,2-diaminopropane, propylene-1,2-diamine, or write out its IUPAC name. A few trade names appear in catalogs, though underlying chemistry stays the same no matter what’s printed on the invoice. Anyone who has worked a few years in a chemistry lab recognizes why keeping track of synonyms matters so much: mixing up two similar-but-distinct diamines can derail days of effort, ruin runs in an industrial process, or throw analytical results out of whack. Reliable sourcing depends on matching specifications, not just trusting a familiar product name.
Safety & Operational Standards: Hands-On Lessons
Handling 1,2-propylenediamine has always meant respecting its sharp vapors and reactive nature. The material earns hazard statements for both its irritancy and flammability. My own time in a chemical pilot plant taught the value of splash-proof goggles, gloves rated for amines, and keeping spill kits ready. Engineering controls such as fume hoods and explosion-proof storage cabins pay dividends, not just for peace of mind but to meet the strict expectations of regulators. Guidelines spell out thresholds for airborne concentrations and proper handling practices, as well as emergency protocols for accidental spills or exposures. Keeping air exchange rates high and labeling containers correctly reduces confusion and risk for shift workers or visiting researchers alike. Training and muscle memory help lower the odds of incidents—just as important as formal signage or periodic audits.
Application Area: Breadth Across Industries
Few chemicals weave into so many corners of the industrial and research community as 1,2-propylenediamine. It helps build the backbone of epoxy resin systems used in construction, electronics, and advanced manufacturing. Adhesive and sealant makers rely on its curing power, and researchers exploring new polymers appreciate its responsiveness in experimental systems. Outside the world of plastics, the molecule crops up as a chelating agent, a corrosion inhibitor, and as an intermediate in active pharmaceutical ingredient development. Its ability to anchor new functional groups attracts medicinal chemists who need to branch out from familiar skeletons. In water treatment and agrochemical research, its reactivity with metallic ions and organic acids extends its value far beyond the original application of making simple resins.
Research & Development: Innovation on Several Fronts
Academic and industrial labs keep pushing the boundaries for what 1,2-propylenediamine can achieve. Researchers design analogs to chase properties like higher selectivity or improved environmental compatibility, especially in specialty coatings or biodegradable polymers. In catalysis, chemists explore modified diamines as ligands to improve reaction yields, drawing on that same backbone while tweaking the molecule for steric or electronic effects. Pharmaceutical projects grow increasingly complex, with companies looking to 1,2-propylenediamine scaffolds for building novel bioactive compounds, making the most of its bifunctional nature. Even teams focused on green chemistry are testing new routes for synthesizing it from renewable feedstocks, addressing concerns about sustainability and long-term raw material security.
Toxicity Research: Unpacking the Risks
Long-term studies aim to map the real risks—people know that 1,2-propylenediamine can irritate the skin, eyes, and respiratory tract, yet the chronic exposure picture has some unanswered questions. Toxicologists study its effects on laboratory animals, looking for clues about potential organ toxicity, carcinogenicity, and ecological impact, especially as environmental regulations continue to tighten. Peer-reviewed studies have flagged concerns over acute contact, while repeated low-level exposures push industry to strengthen safeguards and protocols. This chemical joins a long list of common-use reagents that demand both respect and ongoing research to clarify safe exposure levels. Information-sharing among manufacturers, researchers, and regulators helps weed out outdated assumptions and guides the next generation of safety standards.
Future Prospects: Where the Molecule Goes Next
Looking forward, innovations in both manufacturing methods and application areas will likely shape the next chapter. Demand in composites and sustainable polymer systems keeps rising, especially as industries worldwide shift to greener materials that must still deliver toughness and consistent performance. Synthesis pathways drawing from renewable carbohydrates or bio-based feedstocks could help lower the carbon footprint, aligning with new global environmental goals. Ongoing breakthroughs in pharmaceutical chemistry and catalysis point to 1,2-propylenediamine scaffolds as sources for more complex drug candidates. Regulatory landscapes and public expectations drive continuous improvement in worker safety and environmental controls, adding extra push toward safer, smarter chemical production. Pure technical curiosity—driven by both industry veterans and new researchers—will keep this once old-school amine in the spotlight, promising new surprises as decades pass.
What is 1,2-propylenediamine used for?
Understanding the Role of 1,2-Propylenediamine
1,2-Propylenediamine, also called propylene-1,2-diamine, pops up in more than one corner of the chemical industry. I first came across it in a college organic chemistry lab, trying to wrap my head around its fishy smell and its use in synthesis reactions. Later on, after working with manufacturing facilities, I noticed it played a much bigger role than textbooks let on.
Behind the Scenes in Chemical Manufacturing
Manufacturers rely on 1,2-propylenediamine to create bigger, more complex molecules. As a building block, it links up with other chemicals, often serving as a starting point for pharmaceuticals, textile additives, and water treatment agents. The most common job this compound handles involves making chelating agents, those molecules that grab on to metals and pull them out of solutions. These play a big part in cleaning industrial waste streams and softening water.
Chemists lean into 1,2-propylenediamine’s two amine groups because they react quickly with acids and other electrophiles. Industries making pesticides and dyes use it to build molecules that stand up to harsh conditions. Pharmaceutical chemists turn to it to piece together medications and research new treatments, counting on its reactivity to open the door to more complex drugs. I’ve spoken to process engineers who say that switching to 1,2-propylenediamine sometimes brings new efficiencies and cuts costs compared to older, less reactive amines.
Tackling Wastewater Problems
Across water treatment plants, engineers add 1,2-propylenediamine-based chelating agents to remove metal ions like lead or copper. Left unchecked, those metals slip into drinking water and cause health issues for entire communities. I remember when the EPA flagged a local water utility for lead contamination; solutions like chelating agents were on the list for urgent upgrades. Using compounds made from this diamine, facilities can meet safety standards more reliably and at a lower dose—protecting folks downstream.
Safety and Sustainability Concerns
It’s not all good news. Like many amine chemicals, 1,2-propylenediamine is toxic to aquatic life and can irritate skin or lungs. Industrial facilities have to use tight controls—ventilation, gloves, and containment—to manage worker exposure. A 2022 report from the European Chemicals Agency raised concerns about the environmental persistence of amines, pushing manufacturers toward safer handling and better waste treatment. Chemical engineers told me that with proper scrubbers and containment, risks can drop sharply, but “proper” always depends on training and investment from management.
Moving Toward Better Solutions
Many companies explore greener chemistry these days. Researchers work on alternatives that break down faster in the environment or present less risk for workers. Some experiments swap out 1,2-propylenediamine for bio-based amines, aiming to cut the long-term impact. Change moves slowly—supply chains and processes don’t shift overnight—but regulatory pressure and better technology both drive improvement.
Final Thoughts From Experience
After seeing this compound from lab bench to industrial shop floor, I can say that 1,2-propylenediamine stands out for its reliability and flexibility. Its risks matter, though, and ignoring proper safety or environmental steps doesn’t just hurt workers—it can cost communities down the line. This story isn’t just about making chemicals; it’s about balancing progress with responsibility. Smart handling and a look toward safer substitutes should guide any business that relies on this important ingredient.
Is 1,2-propylenediamine hazardous or toxic?
Understanding 1,2-Propylenediamine
1,2-Propylenediamine shows up in chemical plants, research labs, and even in some industrial products. This chemical brings a strong, ammonia-like smell and comes as a clear liquid. Most folks outside of chemistry circles won’t run into it, but those who handle it in a lab or a factory might wonder if it’s safe—or something best kept behind thick gloves and goggles.
What Happens If You Get Exposed?
If 1,2-propylenediamine splashes on your skin, you feel a sharp sting pretty quick. It irritates skin, and some people can break out in a rash or get red, sore spots. Breathing in the fumes, especially without ventilation, can leave you coughing and gasping. The chemical can bother your lungs. As for eyes, even a little contact burns. Long-term effects have not been mapped out as well as with some bigger-name chemicals, but the risk of asthma-like symptoms or long-term skin problems sits on the list, based on what’s known about similar amines.
Is It Poisonous?
Toxicity depends on dose and exposure. 1,2-Propylenediamine is not among the worst offenders. For example, household bleach and ammonia are far more common in poisonings, and a lot of pesticides carry a higher acute risk. Still, chemicals classified as amines—including this one—do carry toxic effects. Studies in animals show that high doses cause problems with organs like the liver and kidneys. It won’t vaporize easily at room temperature, but in tight spaces, the fumes can build up enough to cause headaches, dizziness, and nausea.
Regulations and Precautions
Workers usually depend on safety sheets, training, and protective gear. The US Occupational Safety and Health Administration (OSHA) and the European Union keep tabs on the risks. Right now, there’s no nationwide exposure limit in the US for this chemical, though the National Institute for Occupational Safety and Health advises minimizing inhalation and contact. Germany sets a workplace exposure limit at 1 ppm. These standards come from years of seeing what happens in real workplaces, not just in a controlled lab.
What Can Be Done to Stay Safe?
I’ve worked around some pretty nasty stuff in university and industrial settings. Best practice means strong gloves, full goggles, and a solid lab coat at the very least. A well-ventilated room, a fume hood running, and proper storage go a long way. Never taste, touch, or sniff a clear liquid with a warning label. I once watched a colleague brush off face protection while cleaning glassware. He spent the day at a clinic with burning, red skin because of an accidental splash from a similar chemical.
Knowledge is the first layer of protection. Read up on what’s in your workplace. Call your safety manager with questions, and don't skip training. If a spill happens, know the chemical’s properties, locate the showers and eyewash stations ahead of time, and never work alone with something dangerous.
Paying Attention to the Bigger Picture
Too many chemicals slide under the radar because bigger, more dramatic hazards draw the headlines. 1,2-Propylenediamine won’t poison you with light exposure, but it demands respect. Workers, safety managers, and chemical manufacturers all play a role in keeping risk low. More openness about risks, clear safety standards, and smarter engineering controls can shrink the dangers and save people from painful emergency room visits, or worse.
What are the storage and handling guidelines for 1,2-propylenediamine?
Understanding the Risks
1,2-Propylenediamine carries a reputation in the chemical industry for being useful yet edgy. It brings a strong, ammonia-like smell and a tendency to irritate eyes, skin, and respiratory tracts. It can catch fire easier than many folks realize. I remember my first job in a university research lab—early on, I thought the glass jars on the back shelf posed little danger, but a sharp whiff and a burning tingle in my sinuses set me straight.
Safe Storage Counts
Forget about leaving this chemical around in open or mixed-use storage areas. Always pick a cool, dry spot, away from heat sources, direct sunlight, and anything that sparks. Flammable liquids cabinet or flameproof locker—that’s where 1,2-propylenediamine belongs. You don’t want to mess with damp basements or shelves near radiators.
Containers should stay tightly sealed. I’ve seen folks slap worn-out lids on bottles, then get surprised by strong odors and wasted material. Go for robust, corrosion-proof containers. Forged steel or quality plastic both work, as long as the material stands up to strong bases. Most storage accidents happen not from big leaks but small seeps through lousy caps and old seals.
Workplace Ventilation Is a Must
This chemical evaporates quick, and its vapors hurt the lungs. Always use it under a fume hood, not in the open lab or a poorly-ventilated storeroom. In small engineering shops, even a portable extraction fan helps, but fume hoods knock down the risks best. Never forget that good airflow saves people from both headaches and serious injuries. The National Institute for Occupational Safety and Health sets guidelines for minimum air changes—using their advice always serves well.
Handle With Respect—PPE Is Non-Negotiable
Personal protective gear matters here—nitrile gloves, splash goggles, and lab coats keep skin and eyes safe. One of my old coworkers ignored gloves just once, thinking a quick transfer wasn't a big deal. His hands burned for hours and he learned the hard way. No one should ever treat handling 1,2-propylenediamine like they’re pouring lemonade.
Spills and Clean-Up—Don’t Wing It
Even small spills demand careful cleanup. Sprinkle absorbent materials like vermiculite or sand over the puddle. Never use cotton rags, since strong bases eat right through them. Gather the waste in a chemical-safe bag and send it to hazardous waste disposal, not the regular trash. A simple mistake—like tossing contaminated paper towels into a regular bin—can spark big headaches for janitorial staff and anyone down the disposal chain.
Label Everything, Communicate Risks
Every bottle and canister should have a clear, printed label listing the contents, hazards, and date of receipt. Confusion causes trouble; I once saw a technician mistake amine for distilled water due to lazy labeling. Training and culture build safety, not just rules. Teach new staff the signs of leaks and safe cleanup routines, not just chemical names. Share firsthand experiences about problems that happened in the past.
Reducing Danger Isn’t Rocket Science
Simple changes make a big difference: right storage, good airflow, the right gloves and goggles. A little attention to these habits brings down the risk level. It shows respect—for the people working, the neighbors, and the wider world. Legislation and best-practices guides give the backbone, but it’s the choices made day-to-day that keep everything safe.
What is the chemical structure or formula of 1,2-propylenediamine?
Chemical Structure and Formula
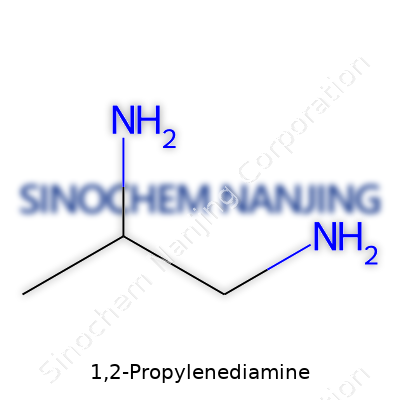
In the world of industrial and research chemistry, 1,2-propylenediamine gets noticed for its versatility. The chemical formula looks simple at first glance: C3H10N2. Laying out the structural details, you get a chain of three carbon atoms, with amino groups attached to the first and second carbon. The traditional name, 1,2-diaminopropane, gives away the position of the two NH2 groups on the straight chain. Draw it out, and you’ll see something like H2N–CH(CH3)–CH2–NH2. Small differences in how those amino groups attach can make a world of difference, especially in reactivity and usefulness in more complex reactions.
A Closer Look at Its Importance
Lots of people outside chemistry labs rarely hear about 1,2-propylenediamine, but it finds work in places ranging from curing agents for resins to building blocks for pharmaceutical ingredients. That matters because many industries lean on intermediates like this to produce goods and materials we rely on every day. Building polymers, creating chelating agents, or blending specialty chemicals—all these processes can tap into the structure of this diamine. You might not see it listed on a consumer label, but your epoxy adhesives hold together a little better because of smart molecules like this one.
Exposure to a substance like 1,2-propylenediamine needs careful attention. Regulations such as REACH in Europe require manufacturers to assess risks and register chemicals. Eye and skin irritation stand as common problems if you handle the neat material, so gloves and goggles remain must-haves in any basic lab. Remember, industrial safety guidelines only count if people follow them. In my own graduate lab experience, a teaching assistant once picked up a bottle of this stuff without gloves and learned quickly about proper precaution. No serious harm, just a memorable lesson—precaution beats regret.
Balancing Benefits and Risks
Companies and researchers want materials that balance efficiency, availability, and environmental impact. 1,2-propylenediamine gets produced from propylene oxide, which ties its supply firmly to the oil and gas industry. Sustainable sourcing becomes a hot topic whenever a raw material depends on non-renewable feedstocks. Green chemistry aims to shift production toward renewable or less hazardous starting points. Factoring in carbon footprint—even in hidden intermediates—pushes everyone in the supply chain to take stock of raw material choices. Some pilot projects already look at bio-based propylene oxide, but large-scale adoption takes time and significant investment.
Paths Forward: Better Practice and Smarter Design
Chemicals like 1,2-propylenediamine prove useful beyond the bench, yet their flow through production lines poses questions about waste and efficiency. Streamlined synthesis that cuts down by-products or recycles solvents stands as a practical way forward. Wastewater treatment facilities look for ways to catch nitrogen-rich compounds before they cause downstream problems. As more regulations kick in around the world, small and mid-sized companies get nudged into compliance by public expectation and legal mandates. Open discussion between chemists, manufacturers, regulators, and consumers tends to lead to better outcomes, not just safer products. In my own consulting work, I’ve seen real progress come from workshops that include everyone in the room—engineers, end-users, even local policymakers. Transparency pushes industry standards higher, making sure lab discoveries become safe, usable materials in our everyday lives.
How should 1,2-propylenediamine spills or exposure be managed?
The Reality of Chemical Spills on the Ground
In busy factories and research labs, chemicals like 1,2-propylenediamine play a behind-the-scenes role. Sometimes, spills happen—sometimes a splash, maybe a leaky drum. This clear liquid can irritate the skin and eyes or even cause breathing trouble if fumes build up. If you’ve ever worked in a warehouse where someone fumbled with drums or noticed a puddle forming around a storage pallet, you know half the risk comes from surprise, the other half from not knowing what to do next.
Why 1,2-Propylenediamine Deserves Respect
1,2-Propylenediamine is used for making pharmaceuticals, resins, coatings, and water treatment chemicals. But just because something plays an important part in industry doesn’t mean you let your guard down around it. Repeated skin contact can set off irritation or allergic reactions. Inhaling the vapor can sting the airways and even trigger dizziness or headaches. If someone’s not careful, a spill can bring real harm—not just to people, but to soil and water, because this compound moves fast and mixes well with water.
Common-Sense Steps Everyone Can Follow
Heading off problems starts with knowing what’s on hand. Safety training loses its punch when it’s just a poster on a wall. Workers who roll up their sleeves and handle chemicals every day benefit from regular, real-life drills. It pays off to label containers clearly and make sure safety data sheets never collect dust. Keeping eye-wash stations and showers easy to find (and not blocked by boxes or gear) often makes the difference between a close call and a serious injury.
Responding Swiftly to a Spill
If a leak happens, get people clear of the area—nobody should play the hero with chemicals. Putting on gloves and goggles can save skin and eyes. Ventilating the area using exhaust fans helps keep fumes from building up. Sealing off floor drains with pads or absorbent socks stops the chemical from slipping into pipes or wastewater. Spilled liquid gets soaked up with special absorbent granules or pads, never sawdust or clay which can heat up and cause extra risk. Once the mess is cleaned up, it should go in a sealed drum for hazardous waste disposal, not tossed with regular trash.
Supporting Health and the Environment
Every person exposed, even for a few minutes, needs a check from a health professional. If chemicals get on the skin or in the eyes, thorough rinsing with clean running water should start straight away, then seek medical advice. For anyone feeling short of breath or dizzy, get help—breathing issues don’t wait for appointments. Reporting even small exposures pays off for workplace safety records and prevents trouble from being swept under the rug.
Reducing Risks is Everyone’s Job
Every workplace handling chemicals should run regular risk assessments. Double-check storage containers and holding tanks for signs of weakness or leaks. Personal stories often show that accidents drop in places where people know each other’s names and look out for each other—not just from written rules, but from a shared sense of responsibility. Investing in ongoing training and spending the extra cash for safer valves or spill kits protects both people and the bottom line.
From Personal Experience: Staying Ready Saves Trouble
Having worked around industrial chemicals, I’ve seen how a little spill can rattle nerves, even among seasoned teams. The best-run teams take these moments in stride because they’ve prepared ahead of time. Open conversations about near-misses help everyone catch problems early. Clear instructions, hands-on training, and teamwork don’t just avoid fines or inspections—they keep workers healthy and bring peace of mind to families waiting at home.
| Names | |
| Preferred IUPAC name | propane-1,2-diamine |
| Other names |
1,2-Diaminopropane Propane-1,2-diamine 1,2-PDA PDA 1,2-Propylenediamine |
| Pronunciation | /ˌwʌn,tuːˌproʊˈpiːlɪndiəˌmiːn/ |
| Identifiers | |
| CAS Number | 78-90-0 |
| 3D model (JSmol) | `Jmol._Canvas2D (JSmol)` model for **1,2-Propylenediamine** (also known as 1,2-diaminopropane) as a string is: ``` NC(C)CN ``` This is the **SMILES** string used by JSmol (and many viewers) to render the 3D model. |
| Beilstein Reference | 1209246 |
| ChEBI | CHEBI:41285 |
| ChEMBL | CHEMBL15740 |
| ChemSpider | 17535 |
| DrugBank | DB02288 |
| ECHA InfoCard | 03b0e6e5-6a87-406a-9a7d-5a3ed1b3e122 |
| EC Number | 202-457-9 |
| Gmelin Reference | 8286 |
| KEGG | C00334 |
| MeSH | D011374 |
| PubChem CID | 15938 |
| RTECS number | UE9100000 |
| UNII | 81A278W4FK |
| UN number | UN prot. no. 2734 |
| CompTox Dashboard (EPA) | Q409587 |
| Properties | |
| Chemical formula | C3H10N2 |
| Molar mass | 74.12 g/mol |
| Appearance | Colorless to light yellow liquid |
| Odor | ammonia-like |
| Density | 0.867 g/mL at 25 °C |
| Solubility in water | Miscible |
| log P | -1.5 |
| Vapor pressure | 2.7 mmHg (20°C) |
| Acidity (pKa) | 10.47 |
| Basicity (pKb) | 4.28 |
| Magnetic susceptibility (χ) | -13.2·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.431 |
| Viscosity | 1.005 mPa·s at 25 °C |
| Dipole moment | 1.58 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 81.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -39.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -4360 kJ/mol |
| Pharmacology | |
| ATC code | D06BB06 |
| Hazards | |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H301, H311, H314 |
| Precautionary statements | P280, P261, P305+P351+P338, P310, P303+P361+P353, P304+P340, P405, P501 |
| NFPA 704 (fire diamond) | 1*3*3*W |
| Flash point | 80 °C |
| Autoignition temperature | 410 °C |
| Explosive limits | 2.7–15.5% |
| Lethal dose or concentration | LD50 oral rat 1470 mg/kg |
| LD50 (median dose) | LD50 (median dose): 530 mg/kg (oral, rat) |
| NIOSH | SD4725000 |
| PEL (Permissible) | PEL: 2 ppm (parts per million) |
| REL (Recommended) | 10 ppm (25 mg/m3) |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Ethylenediamine Trimethylenediamine 1,3-Propylenediamine Isopropanolamine Diethylenetriamine |

