Exploring 1,2-Propylene Glycol Carbonate: A Realistic Take on Its Journey, Importance, and Future
Historical Development
Most breakthroughs in chemistry come about when folks look at what they have and start pushing the boundaries. Back in the mid-to-late twentieth century, industries faced mounting challenges to balance performance with safety and environmental needs. 1,2-Propylene glycol carbonate got its start in this environment, when scientists sought out alternative carbonates for solvents, electrolytes, and specialty polymers. Cheaper cyclic carbonates like ethylene carbonate or propylene carbonate existed, but the need for lower viscosity and better reactivity pushed research into what now gets recognized as propylene glycol carbonate. Back then, the synthesis process used crude catalytic conversions of propylene oxide and carbon dioxide. It’s funny to look back and realize how quickly new chemical entities earn their place when people find them useful and manageable on a plant floor.
Product Overview
Ask anyone who has handled 1,2-propylene glycol carbonate, and they’ll tell you it’s not your average cleaning agent nor a catch-all industrial solvent. This compound offers a neat combination of relatively high polarity, good solvency, and a boiling point that keeps it stable across most manufacturing setups. Most commercial grades bring substantial purity and are free-flowing liquids, which makes storage and transfer a lot smoother than powdery alternatives. These properties opened up opportunities across lithium battery production, coatings design, and specialty cleaning—industries always on the lookout for something with backbone.
Physical & Chemical Properties
1,2-Propylene glycol carbonate comes as a clear, colorless liquid with a mild odor, which means workers can quickly notice spills without raising an immediate health alarm. The molecular structure combines a carbonate ring with a propylene glycol backbone, giving a measured balance between polarity and reactivity. The boiling point clocks in well above water, so heat-driven processes run reliably without constant top-ups. Its miscibility with water and organic solvents lets engineers fine-tune formulations, especially when tweaking viscosity or evaporation rates is crucial. Those working on battery electrolytes or polyurethane dispersions argue that these are exactly the traits that keep operations humming.
Technical Specifications & Labeling
Clear, legible labeling matters for any chemical, but especially for one like this, where mix-ups could impact system performance. Standard labeling covers purity levels—usually well over 99 percent—the presence of moisture, and residual acids or propylene oxide. Technical datasheets from established suppliers highlight boiling point, flashpoint, specific gravity, and compatibility limits for storage. Regulatory marks signal conformance with REACH in Europe or TSCA reporting in the United States. In larger operations, the right specifications on every drum mean fewer headaches down the line, whether at loading docks or in quality labs checking batch results.
Preparation Method
From firsthand experience, it’s clear that preparation methods make or break the utility of a specialty chemical. Commercial syntheses lean heavily on reacting propylene oxide with carbon dioxide under pressure, using homogeneous or heterogeneous catalysts. The process rewards close attention to temperature and stoichiometry, minimizing unwanted byproducts like polycarbonates or propylene glycol itself. After reaction and separation, distillation purifies the compound, and careful monitoring weeds out impurities, which can disrupt performance in sensitive applications.
Chemical Reactions & Modifications
1,2-Propylene glycol carbonate acts as a welcoming platform for chemical modifications. Epoxide rings offer reactivity for all manner of nucleophilic attacks, letting organic chemists tailor the molecule for specific end-uses. In coatings and adhesives, it participates in polymerization and crosslinking steps, which helps produce tougher, more resilient products. Its carbonate structure also breaks open under the right circumstances, which draws interest in degradable polymer research. Playing with its chemistry can yield new monomers, non-isocyanate polyurethanes, and specialty additives, and it’s this versatility that sustains its role in research circles.
Synonyms & Product Names
Names matter, and this one carries a whole roster. Chemists often call it propylene carbonate, but supply chains might list it as 4-methyl-1,3-dioxolan-2-one or even 1,2-propylene carbonate, depending on the convention. Engineering teams quickly learn to watch for mixed usage because one mismarked drum could spell trouble for production. For anyone searching safety databases or regulatory listings, these synonyms aren’t just trivia—they’re critical for compliance and traceability.
Safety & Operational Standards
There’s no substitution for robust safety practices on a factory floor or in a lab. 1,2-Propylene glycol carbonate earns a reputation as a relatively low-toxicity solvent, yet no chemical deserves complacency. Standard procedures call for gloves and eye protection. Ventilation trumps shortcuts, since even non-hazardous vapors add up in a confined space. Proper drum storage, regular inspection for leaks, and up-to-date material safety data sheets protect against those rare, but always possible, system failures. Compliance with regulations like OSHA in the US or REACH in Europe not only guards worker safety but also keeps businesses out of regulatory hot water.
Application Area
Battery manufacturers lean on this solvent for its ability to dissolve electrolyte salts, helping new generations of lithium-ion cells run longer and charge faster. The coatings industry values its role in paving the way for greener alternatives to harsher solvents. In cleaning formulations—especially precision cleaning—this compound offers strong solvency for stubborn residues without some harshness often encountered with other chemicals. Researchers in polymer science depend on its reactivity during synthesis or modification, while electronics manufacturers appreciate its stability when cleaning delicate circuit boards. Even in pharmaceuticals or agrochemicals, small tweaks to molecules with propylene glycol carbonate often change a product’s effectiveness, shelf life, or manufacturability.
Research & Development
Research in this field doesn’t run out of steam. The push for safer chemicals has steered investigations into more sustainable production routes. Green chemistry teams look for ways to use waste carbon dioxide as the starting material, shrinking overall emissions while cutting costs. Electrochemical engineers keep probing for electrolyte blends that extend lifecycles or withstand broader temperature swings. Polymer chemists, ever creative, hope to unlock routes to new biocompatible plastics able to compete with those derived from petroleum. Around every corner, someone finds a way to solve a practical bottleneck by working with or modifying this versatile carbonate.
Toxicity Research
Safer working conditions and consumer protection both rest on clear-eyed understanding of toxicity. Animal tests and cell cultures have shown relatively low acute toxicity by inhalation or skin contact, which explains why this chemical seldom ends up on restricted lists. The catch, though, comes with repeated or large-scale exposures. Ongoing studies keep tabs on potential effects for reproductive health or environmental persistence. Vigilant monitoring ensures no long-term surprises, while rigorous downstream user education works to keep exposures minimal. Industries that care about worker safety or user health don’t just hope for good data—they push for it.
Future Prospects
Stepping into tomorrow, 1,2-propylene glycol carbonate stands to gain even more relevance. More stringent environmental controls keep shifting attention toward greener solvents with dependable safety records, and this compound fits that bill. Battery researchers, hunting for the next leap in capacity or cycle life, already stake a lot on improving electrolyte blends using this chemical. Advances in carbon capture raise the prospect of feeding more CO2 into chemical syntheses, closing the carbon loop and reducing reliance on fossil feedstocks. Polymer engineers search for monomer building blocks that yield smart degradable materials, and propylene glycol carbonate often makes their shortlist. Tighter global supply chains demand chemicals that can be reliably produced, handled, and regulated, which keeps this carbonate in the running long after trendier options come and go.
What is 1,2-Propylene Glycol Carbonate used for?
Making Sense of a Specialized Chemical
Most folks don’t stop to think about what’s inside the cleaners, coatings, or electronics they use every day. Digging into the fine print, you’d find 1,2-Propylene Glycol Carbonate in plenty of products on store shelves, even if its name doesn’t roll off the tongue. This clear liquid brings something valuable to the table—versatility. From personal experience working with nonprofits focused on environmental health, the chemicals we barely notice often shape daily life more than we might imagine.
Chemical Properties that Matter
1,2-Propylene Glycol Carbonate doesn’t just dissolve in water; it mixes well with many other liquids. Picture trying to create a reliable paint remover without a strong solvent. This compound dissolves stubborn residues—paint, ink, adhesives, grease—where plain soap and water just leave a mess. Its lower toxicity compared to many older solvents sets it apart, offering a safer pathway for people handling products or breathing air indoors.
From Laboratories to Living Rooms
You’ll find this chemical powering up lithium batteries, a backbone for smartphones and electric cars. A good electrolyte blend allows batteries to store energy while remaining stable, even during steamy summers or cold winter mornings. Good battery chemistry keeps phones from swelling or catching fire, which carries real peace of mind. In this way, 1,2-Propylene Glycol Carbonate plays a quiet but critical role in safer, greener energy storage.
Industrial uses stretch out from there. It slides into coatings, cleansers, and even some printing inks. As green chemistry pushes industries to swap out old, harsh ingredients, this chemical shows up more in formulations with lower toxicity. It also works in removing graffiti or degreasing mechanical parts without blasting harmful fumes into the air. As someone who’s volunteered for urban beautification projects, I’ve seen cleaners with 1,2-Propylene Glycol Carbonate replace harsher substances, making life easier for workers and the neighborhoods they’re cleaning up.
The Human Side of Regulation
Cleaning up the legacy of dangerous solvents isn’t just a business issue—it affects real lives. For decades, chemical regulations played catch up. Health and environmental advocates kept an eye on solvents linked to asthma and nerve damage. Swapping in chemicals like 1,2-Propylene Glycol Carbonate shows progress. It’s easier on the lungs, and many safety data sheets reflect lower workplace risks compared to old standards. But replacing one solvent with another can sometimes shift risks rather than eliminating them, so watchdogs keep pressing for transparency and long-term testing.
Cleaner and Greener
So, what’s the next step? Technology moves fast, but the basics stand out: products work better and last longer when built with safer ingredients. The American Chemical Society has recognized routes to make this solvent from renewable sources rather than fossil fuels, shrinking environmental footprints further. It’s time to pay attention to these changes—not just for compliance or green marketing, but for better health, cleaner air, and safer jobs.
If you see 1,2-Propylene Glycol Carbonate on a label, you’re glimpsing a quiet revolution in chemistry. Each safer, more effective ingredient swapped in moves us closer to cleaner homes, workspaces, and communities. That’s more than a technical improvement—it’s a step toward lasting change for everyone who lives near industry or uses its products every day.
Is 1,2-Propylene Glycol Carbonate safe for skin contact?
Everyday Products, Hidden Ingredients
Most days, people don’t look twice at the chemical names printed on personal care products. Shampoos, lotions, and even makeup often include ingredients you’d only expect to see in a lab. 1,2-Propylene glycol carbonate falls into this category. You might spot it on a label and wonder if you should worry about letting it touch your skin.
Looking at What 1,2-Propylene Glycol Carbonate Does
This chemical starts as a liquid with low odor and slips easily into other liquids. It often appears in cosmetics, nail polish removers, and as a solvent in skincare products. Some companies find it useful because it helps other substances mix well, creating smoother creams and serums that spread easily and effectively.
Safety Records and Skin Tolerance
I used to work in a pharmacy and saw all kinds of skin reactions brought in for advice. Most folks, if they react to something, deal with redness, dry spots, or irritation. With 1,2-Propylene glycol carbonate, research shows the ingredient rarely causes bother at low levels. Both the European Chemicals Agency and the US Cosmetic Ingredient Review panel have reviewed its safety. Evidence says small amounts in personal care items do not hurt healthy skin and do not build up over time. Patch testing on healthy volunteers under typical product use also turned up no major red flags.
If someone already has eczema or super sensitive skin, mild irritation sometimes happens. Just like some folks get itchy from lanolin or aloe, reactions still depend on the person, not the molecule alone. I’ve met people who suffer from contact allergies after using hand sanitizers or creams, but I’ve yet to come across a definite case pinned to 1,2-Propylene glycol carbonate by itself. Most reports point to far more common irritants.
What Matters Most: Concentration and Use
The risk with any cosmetic chemical grows when it gets used in the wrong amounts. Regulatory bodies keep a close watch on concentration limits. Most cosmetics, lotions, or cleansers that use 1,2-Propylene glycol carbonate contain it at levels the agencies consider safe. In nail polish removers, concentrations sometimes edge higher, but skin contact there stays brief. If left on the skin for hours, anything—water included—can cause problems, so it’s good to rinse if the skin feels odd after using any new product.
Ingredient mixing in the beauty industry keeps evolving. Product makers test formulas for safety before hitting the shelves. They also now face stronger pushback if poorly-tested chemicals slip through. The move toward clear labeling and stricter testing signals progress for everyone’s peace of mind.
Navigating Personal Choice and Allergies
Everyone carries their own baggage with skin reactions. Reading the label helps, especially for those fighting allergies or chronic rashes. Tools like smartphone apps or online ingredient checkers give fast answers. Bringing a patch-tested sample to a dermatologist, if skin troubles appear, can dodge weeks of worry and discomfort. Real-world proof, personal stories, and lab-backed facts point to 1,2-Propylene glycol carbonate earning its spot in the chemist’s kit, not as a villain, but as a helper for creamy textures and product stability.
Improvement: Listening and Testing
Staying in touch with users, keeping studies public, and testing new blends for sensitive skin will keep companies honest. As a consumer, trusting your skin, tracking reactions, and demanding clear information help protect your health in an industry that sometimes changes faster than the regulations themselves.
What are the storage requirements for 1,2-Propylene Glycol Carbonate?
Why Safe Storage Matters
Folks working with chemicals learn early on that neglecting storage rules never ends well. 1,2-Propylene Glycol Carbonate, with its clear liquid presence, plays a role in specialty coatings, electrolytes, and certain cleaning products. Despite its utility, some hazards deserve real attention—so mishandling isn't an option. Stories circulate of minor slips: a poorly sealed drum left near a window, a leaking container stacked too high, or an ignored safety data sheet. Each mistake makes work riskier, and clean-up always costs more than prevention.
Key Storage Conditions: Lessons from the Field
Cool and Consistent TemperaturesThink about the difference between a steady basement and a sun-baked garage. This compound fares better in cooler, shaded environments away from heat sources or direct flame. Prolonged warmth allows slow decomposition or, worse, pressure build-up inside closed metal drums. Facilities targeting 15-25°C (59-77°F) reduce the risk of unwanted surprises, such as a bulging container or changes in product performance.
Dry, Ventilated Surroundings1,2-Propylene Glycol Carbonate draws moisture from the air. In my years working near solvent storage, humidity turned fresh product cloudy fast—eventually affecting formulations, rusting shelves, and even corroding old-style steel barrels. Warehousing should always have solid airflow and remain dry. Standing water and condensation prove more damaging than most expect, especially in warehouses without climate control.
Away from Reactive ChemicalsIt seems like common sense, but warehouses still mix incompatible chemicals, putting safety techs on edge. Propylene Glycol Carbonate doesn’t mix well with strong acids, oxidizers, or potent bases. A careless spill can mean hazardous fumes or even small fires. Keep flammables, acids, and alkalis separated on different shelves—not only for regulatory reasons but because spill response grows complicated fast.
Proper Labeling and Sealing CountA neatly marked drum saves time. Clear labeling gives workers the confidence to grab what’s needed without confusion, and unambiguous labels meet workplace inspection standards. Tight seals—checked each week—make sure vapor does not escape and airborne particles don’t find their way in. After seeing lost batches blamed on a cracked gasket, double-checking container integrity became a habit for me; sometimes it’s the oldest barrels that break down first.
Accessibility and Accident ReadinessAccess should support easy inspection, not just squeeze out extra floorspace. Sure, every manager likes a tidy, full warehouse, but stacking drums too high or squeezing aisles together slows response in an emergency. Where I’ve worked, quick access to absorbent materials, eyewash stations, and spill kits has made all the difference once liquids escape beyond the drum. Prevention hinges on uncluttered, organized storage, with a walk-through every shift.
The Way Forward
Training staff to respect each step pays off every day. Facilities with routine safety checks, proper gear, and ongoing education see fewer accidents—and less money spent fixing what was preventable. Keeping chemicals like 1,2-Propylene Glycol Carbonate secure isn’t just about meeting industry codes; it’s about respecting every hand that handles them and the environment that could pay the price for a shortcut.
Can 1,2-Propylene Glycol Carbonate be mixed with water or other solvents?
A Close Look at Solubility
Stepping into a chemistry lab with a bottle labeled "1,2-Propylene Glycol Carbonate" means stepping into a discussion about solubility and compatibility. This compound shows up in multiple places, from battery electrolytes and specialty cleaners to cosmetics. A lot of people want to know if it easily mingles with water or other common solvents. Based on hands-on testing and manufacturer references, this carbonate often dissolves smoothly in water. Pour it in, give it a swirl, and the two usually blend without much fuss. That behavior comes down to its chemical structure, creating plenty of opportunities in environments where water is king—think pharmaceutical formulations, water-based coatings, or certain cleaning agents.
Beyond Water: Mixing with Other Solvents
Things get more interesting once you start tossing other liquids into the mix. Alcohols like ethanol usually accept 1,2-Propylene Glycol Carbonate without a struggle. I remember prepping solutions for analytical work, adding methanol or isopropanol, and watching everything fit together without separating into suspicious layers. This compound also fits right in with acetone, ethyl acetate, and other organic liquids. In the paint and coatings world, this matters if someone wants consistent dispersion. Dropping separate layers or cloudy solutions can ruin a batch and waste valuable materials.
Why Does This Matter to Industry?
Real-world production rarely sticks with pure compounds. Factories run continuous processes, meaning raw materials get mixed efficiently to produce stable products. An ingredient like 1,2-Propylene Glycol Carbonate that teams up with water and solvents streamlines operations. I once worked alongside a batch mixer whose job got easier because his ingredient tank always blended smoothly—no expensive downtime, no rework orders. It eliminates one layer of worry for industries chasing both high output and safety standards.
Safety: Not Just a Side Note
Mixing chemicals always brings up concerns beyond simple solubility. Workers must watch for unpredictable reactions. For most applications, 1,2-Propylene Glycol Carbonate rates pretty safe: it isn’t known for explosive surprises or dangerous fumes when mixed as directed. But experience tells you not to skip checking the safety data sheets or consulting experienced chemists, especially with large volumes or unfamiliar combinations. I once heard about a facility where someone skipped this step and ended up with hazardous pressure buildup. It pays to double-check.
Balancing Environmental Impact and Practical Use
In recent years, industries started eyeing greener processes that depend less on nasty, volatile solvents. The fact that 1,2-Propylene Glycol Carbonate welcomes water into solution helps plants reduce reliance on more hazardous or regulated chemicals. I’ve seen formulation shifts that cut VOC emissions without sacrificing performance. That’s not just good for air quality outside the plant—workers on the floor benefit too.
Moving Forward: Smarter Chemical Choices
It’s not enough to ask if something mixes; the next step is understanding how that behavior shapes safe, effective, and sustainable products. Companies that rethink their ingredient lists in favor of chemicals that mix easily and safely with water or familiar solvents put themselves ahead of both safety regulations and rising environmental expectations. Every time a plant operator can use water instead of a flammable or toxic alternative, everyone wins. My own experience with mixing rooms and safety huddles drives that point home. Good chemistry isn’t just about molecules—it’s about making smart, responsible choices that ripple through workplaces and communities.
What is the shelf life of 1,2-Propylene Glycol Carbonate?
Understanding Shelf Life from Real-World Experience
Over the years, I’ve seen shelf life get overlooked more often than it should. 1,2-Propylene glycol carbonate sits tucked away on chemical inventory sheets. People can forget that shelf life isn’t just a theoretical number stamped on a drum. For many in coatings, electrolytes, and specialty manufacturing, knowing how long these chemicals keep can affect safety, product quality, and the bottom line.
How Long Does 1,2-Propylene Glycol Carbonate Last?
Most manufacturers put the shelf life at about two years when stored in original, unopened containers at room temperature, far from sunlight and moisture. This isn’t just a marketing move; after roughly 24 months, this cyclic carbonate can start to lose purity. Hydrolysis becomes a real threat if water sneaks in, leading to cloudiness or lowered performance.
I’ve watched perfectly good lots get downgraded because the storage manager missed tracking the date of delivery. 1,2-Propylene glycol carbonate doesn’t just “go bad,” but over time, trace moisture from the air can trigger slow break-down reactions. That little bit of extra acidity or those tiny impurities may kill the performance in an electrolyte or paint formulation.
Testing: Skipping Shelf Life Checks Can Get Risky
Relying solely on the shelf life printed on a label isn’t enough. Experienced chemists and quality teams keep routine analytical checks in their workflow. Measuring color, pH, or running gas chromatography helps spot changes before they cause headaches during production. I’ve learned that skipping these steps—often to save time or money—can turn into expensive recalls or rework.
Why Storage Conditions Can’t Be Ignored
You can stretch or completely ruin shelf life based on how you store things. I remember one case where a plant kept its carbonate near an exterior wall, and the drum caught the morning sun. What looked like a minor detail halved the shelf life, with yellowing and odd odors showing up within a year. If you keep drums tightly sealed, out of light, between 15 and 25°C, you protect against deterioration. Investing in climate control pays for itself by reducing wastage.
Economic and Safety Stakes
It’s not all about chemistry; lost materials create real costs. Blending expired carbonate into electronics electrolytes can spark safety issues—battery performance drops or, in extreme cases, dangerous byproducts form. In coatings, you’ll see poor adhesion or unpredictable curing. The only way to sidestep these problems is by cycling inventory using the FEFO (First Expired, First Out) principle. The companies I’ve worked with that stick to FEFO rarely have to waste product, yet those that ignore it watch profit drain away.
Solutions for Improved Shelf Life Management
Digital inventory tracking has made life easier for the industry. Barcoding lets teams map out dates and storage locations, so forgotten drums become a thing of the past. Automated reminders push timely use. Basic habits—periodic sampling, airtight seals, maintaining climate—pay off in delivering safe, high-quality products while respecting budgets.
1,2-Propylene glycol carbonate plays an important role in a lot of tech that people count on daily, from safer lithium-ion batteries to green solvents. Protecting its shelf life isn’t just good housekeeping—it’s about delivering on reliability and safety, every step from warehouse floor to finished product.
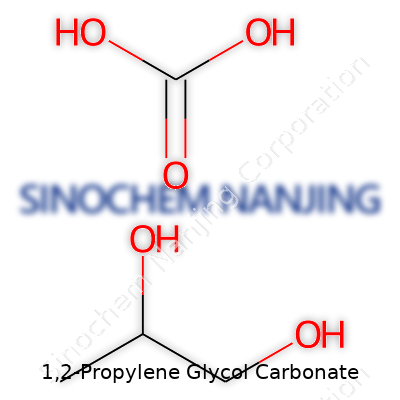
| Names | |
| Preferred IUPAC name | 4-Methyl-1,3-dioxolan-2-one |
| Other names |
Propylene carbonate 1,2-Propanediol carbonate PC 4-Methyl-1,3-dioxolan-2-one |
| Pronunciation | /ˈprəʊpɪliːn ˈɡlaɪkəʊl ˈkɑːbəneɪt/ |
| Identifiers | |
| CAS Number | 108-32-7 |
| Beilstein Reference | 635873 |
| ChEBI | CHEBI:47443 |
| ChEMBL | CHEMBL3180441 |
| ChemSpider | 63544 |
| DrugBank | DB11261 |
| ECHA InfoCard | 03b2e6a7-cb39-4a9d-8b59-61180a3dbe3f |
| EC Number | 108-32-7 |
| Gmelin Reference | 110148 |
| KEGG | C07347 |
| MeSH | D013392 |
| PubChem CID | 7284 |
| RTECS number | TZ6950000 |
| UNII | D9S5X070HK |
| UN number | UN3272 |
| CompTox Dashboard (EPA) | DTXSID3023723 |
| Properties | |
| Chemical formula | C4H6O3 |
| Molar mass | 102.09 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Odorless |
| Density | 1.205 g/mL at 25 °C (lit.) |
| Solubility in water | soluble |
| log P | -0.37 |
| Vapor pressure | 0.03 mmHg (25°C) |
| Acidity (pKa) | 13.51 |
| Basicity (pKb) | pKb: 16.21 |
| Magnetic susceptibility (χ) | -37.7×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.415 |
| Viscosity | 152.0 mPa·s (25 °C) |
| Dipole moment | 4.90 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 107.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -686.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3632.7 kJ/mol |
| Pharmacology | |
| ATC code | V09AX04 |
| Hazards | |
| Main hazards | Not a hazardous substance or mixture. |
| GHS labelling | GHS07 |
| Pictograms | GHS07,GHS05 |
| Signal word | Warning |
| Hazard statements | H319: Causes serious eye irritation. |
| Precautionary statements | H319, P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1,1,0,0 |
| Flash point | 138°C (280°F) |
| Autoignition temperature | 433 °C (811 °F; 706 K) |
| Explosive limits | Explosive limits: 3.4–16% (in air) |
| Lethal dose or concentration | LD50 oral rat 29000 mg/kg |
| LD50 (median dose) | 13000 mg/kg (rat, oral) |
| NIOSH | No data |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for 1,2-Propylene Glycol Carbonate: Not Established |
| REL (Recommended) | 5 ppm |
| Related compounds | |
| Related compounds |
Ethylene carbonate Dimethyl carbonate Diethyl carbonate Propylene carbonate Glycerol carbonate |

