The Heavy Legacy and Uncertain Future of 1,2,3-Trichloropropane
Historical Development
Anyone with an eye on the evolution of synthetic chemistry runs into 1,2,3-Trichloropropane sooner or later. This small molecule, with three chlorine atoms decorating its three-carbon backbone, first caught the attention of industrial chemists after the Second World War. Mass production crept up as the chemical industry ballooned, with TCP showing up as a byproduct during the manufacture of epichlorohydrin, a precursor for epoxy resins. For decades, no one paid much mind to this stowaway. Factory runoff and groundwater quietly absorbed whatever leaked out. It wasn’t until worrying patterns in environmental tests and health surveillance gave hints of its toxic burden that researchers and regulators started taking a much closer look. The chemical’s history underscores a familiar lesson: industrial convenience rarely squares with environmental and health security, especially when waste takes priority over care.
Product Overview
1,2,3-Trichloropropane stands out among chlorinated hydrocarbons for reasons both practical and troubling. Though not sought for itself on a grand scale, TCP rides along as a byproduct and contaminant where chlorinated solvents or chemical intermediates take center stage. For decades, manufacturers disposed of TCP-rich waste without a second thought, and the chemical drifted through soils and aquifers. Some industrial processes kept TCP fractions high enough that waste streams carried significant concentrations into unlined pits and waterways. While TCP once peaked in certain niche pesticide formulations, mounting toxicity data and tighter regulation forced producers out of most intentional applications. Today, unwanted TCP still shows up wherever chlorinated chemistry handles old platforms or cross-contaminated feedstocks, calling for serious reevaluation of legacy practices.
Physical & Chemical Properties
TCP’s physical form doesn’t turn heads. It's a clear, colorless, oily liquid at room temperature with a mildly sweet odor—boiling high enough that you don’t see it steam away, but volatile enough to mean trouble for indoor air. Chemically, it's less reactive than many cousins but far from inert. The three chlorines make it dense and poorly soluble in water, but not so hydrophobic as to stick just with oils. Once in the soil, it's tough to get rid of, drifting into groundwater in ways that still frustrate remediation experts. The vapors pose more than a slight nuisance, and the chemical lingers for years if left undisturbed.
Technical Specifications & Labeling
In my own work handling hazardous materials, accurate labels never looked so essential as they do with TCP. Regulations classify it unmistakably as a hazardous substance, especially for inhalation and dermal exposure. Industrial containers might show the synonyms allyl trichloride or glycerol trichlorohydrin, but the health pictograms leap out. Its safety data sheets demand gloves, splash-resistant goggles, and tight engineering controls, and seasoned handlers watch for even small leaks with TPS. Labs focus on the chain-of-custody and traceability parts, since one slip can taint a whole batch of soil or product testing. The technical paperwork never promises convenience—just precision in hazard recognition and safe containment.
Preparation Method
Most of the world’s TCP didn’t start as someone’s target product. Instead, it lurks in the shadows of epichlorohydrin synthesis, usually from reacting allyl chloride with chlorine in an aqueous medium. Producers once treated TCP as a routine byproduct, separating it from main lines only because it gummed up catalysts or poisoned reactors. The chemical’s preparation is simple but rarely clean, explaining why “traces” keeps popping up in environmental monitoring. Efforts to reduce TCP formation involve adjusting process conditions, but even today, legacy plants in some markets keep TCP’s ghost haunting groundwater surveys one decade after another.
Chemical Reactions & Modifications
TCP’s chemical backbone opens up some routes for transformation, though none offer easy salvation for contaminated sites. The molecule can hydrolyze under extreme conditions, swapping out chlorines for hydroxyls, but this process remains energy-intensive and slow in nature. In synthetic labs, conversion pathways include dehydrohalogenation—a method to wring out a double bond, but again, hardly a silver bullet for waste clean-up. TCP resists most forms of natural decay in groundwater and soil, which means it’s stubborn, sticking around long past most other volatile organics. Cleanup chemists tried advanced oxidation and bioremediation. Results mixed at best in practical field settings. TCP’s reluctance to break down mirrors the bigger question about what sort of molecules we design—and how much future cleaning we’re willing to shoulder for convenience today.
Synonyms & Product Names
TCP wears many hats in industrial and regulatory language. Chemical registries list it as 1,2,3-trichloropropane, but you’ll spot names like allyl trichloride, omega-trichloropropane, or trichlorohydrin drifting through the literature. Each comes with its own history and usage quirks. Arguments about synonyms make it clear that clarity saves lives once monitoring and compliance get serious. Overlapping names trip up field sampling teams and regulatory filings. In my own experience, more than one laboratory wasted days puzzling over historic waste manifests, only to learn that “glycerin trichloride” meant TCP all along. Regulatory harmonization of synonyms—insisting everyone calls it by the same name—could have saved headaches, if not lives.
Safety & Operational Standards
Handling TCP safely draws on hard-earned lessons. Routine exposure leads to chronic health fallout among workers, and acute exposure triggers nausea, headaches, and more severe effects at high doses. The EPA and agencies worldwide flag it as a likely human carcinogen, and the Occupational Safety and Health Administration lists stringent guidelines for facility engineering controls and required personal protective equipment. In real-world settings, relying on fume hoods, closed paints, and regular leak checks makes the difference between a safe shop and a toxic mess. Site audits, often driven by legal action or regulatory consent, show that companies that ignored hazard signs often ended up footing enormous clean-up and compensation bills decades later. Better practice means respecting stated exposure limits, treating all waste with suspicion, and keeping emergency controls updated—otherwise, communities wind up bearing a toxic cost stamped with a company’s old logo.
Application Area
TCP once played a minor role in specialty applications, but its main presence today shows up in places nobody wants: contaminated sites. Historical pesticide formulations and old solvents carried it into fields and groundwater, particularly in the big agricultural valleys. Environmental executives would rather their company never showed up in the same sentence as TCP. Where it is present, the cleanup costs driven by Superfund designations and litigation regularly break budgets. The drive for “green chemistry” draws a hard line against anything structurally similar. Some fringe research still uses TCP as a model contaminant to test new remediation technologies, but outside of legacy contamination, no modern industry wants to market new uses for this molecule.
Research & Development
A big part of today’s research on TCP focuses on remediation and detection. Scientists in groundwater chemistry and toxicology devote years trying to develop better sensors and automated systems that sniff out TCP at parts-per-trillion in complex soils. Bioremediation studies test whether specially bred bacteria or clever gene editing can coax environmental microbes to chew up TCP faster and safer than chemicals can. Public health researchers keep drilling down on long-term, low-level exposure to understand the true population risks from drinking water and inhalation. Studies sometimes reach into the courtroom; for example, community-driven research spurred class-action lawsuits that forced polluters to install filtration or foot medical bills. Bringing together chemists, engineers, and health specialists broke old academic silos. It also nudged companies to rethink waste and process byproducts before regulators show up years later with fines.
Toxicity Research
Evidence piles up showing how even trace TCP can mark a community with unexplained clusters of liver and kidney problems. EPA hazard listings didn’t materialize from one or two outlier studies—this came after reviewing hundreds of pages’ worth of rat studies, epidemiology, and chemical analysis. Drinking water alerts kicked in after detection at just a few parts per trillion in some California towns; lawsuits and remediation stretched into years of bitter legal wrangling. Handling TCP risk means accepting that past convenience turned up in someone’s tap, with kids facing higher cancer risks because clean-up was too slow or underfunded. Public health communication improved, but trust does not recover overnight. Full knowledge of the mechanism by which TCP triggers toxicity remains a research target, but animal models point clearly at DNA damage and chronic organ stress. In my opinion, recognizing TCP’s harm requires constant humility about what industry once didn't know—or chose not to monitor.
Future Prospects
TCP’s days as a manufacturing input or intentional additive finished long ago. Looking forward, the real prospect comes from cleanup: finding ways to pull this persistent molecule out of water, soil, and air more efficiently and at lower cost. Smart money goes into advanced filtration and promising new biological approaches, from genetically engineered microbes to photolytic breakdown that harnesses sunlight. Government funding should follow, since the alternative means leaving rural towns and city wells vulnerable for decades. Spreading awareness across industry stakeholders remains critical, nudging investment into waste reduction and smarter chemical design at the start. Progress means less TCP production in future years, with long-term sustainability measured not just by balance sheets, but by cleaner groundwater tables and healthier families downstream from the old mistakes. Perhaps the most powerful lesson TCP teaches is that chemistry’s legacy never stays buried; clean production starts with an honest reckoning with the past.
What is 1,2,3-Trichloropropane used for?
The Chemical’s Real-World Purpose
1,2,3-Trichloropropane (TCP) pops up in a few surprising places, though most people won’t ever spot it on a label. People working in chemical manufacturing or who grew up near farmlands might have heard about it. TCP hasn’t found a starring role as a product by itself; it generally ends up in the world as an impurity when making other chemicals, especially chlorinated solvents and soil fumigants that farmers used for decades.
Factories that make products like dichloropropenes for pest control or solvents for cleaning industrial equipment sometimes produce TCP as a by-product. In former decades, little attention got paid to these leftovers. Farmers in California’s Central Valley, for instance, used soil fumigants containing TCP to protect crops from nematodes, with neither producers nor regulators fully aware of the consequences. Research and lawsuits later revealed that TCP lingers in groundwater long after use, raising health alarms.
Unintended Trouble in the Water
Anyone who has ever worried about what’s in their glass of tap water might want to listen up. Studies by the Environmental Protection Agency show that even tiny amounts of TCP in drinking water can raise cancer risks. It’s not just a theoretical concern — communities across California and several other states have detected levels above what scientists consider safe. TCP doesn’t break down easily, so it sticks around long after the original source is gone.
My own family got a notice about TCP in our water a few years ago. That letter came with a lot of worry, plus the sudden hassle of using filtered water for everything from drinking to making coffee. After talking with neighbors, it turned out several towns in the area faced similar contamination, all linked to decades-old farm practices and lax rules around industrial waste.
Health Risks and Industry Pushback
Health agencies are pretty clear: long-term exposure to TCP could raise the risk of cancer, and there’s emerging evidence linking it with kidney and liver problems too. Commercial interests pushed back on tighter regulations, arguing that stricter cleanup requirements meant big costs and litigation from affected towns. Some companies dumped the old soil fumigants long ago, profited, and moved on. Now, ordinary families have to foot the bill for treatment or bottled water. That’s a hard truth for a lot of folks around the country.
Industry sometimes points out that TCP production today is safer and that better safety rules are in place. Yet, the legacy of past contamination sticks around. And for communities stuck with polluted water — often farm towns and low-income neighborhoods — trust evaporates fast when they ask who will cover the cleanup.
Facing the Future
The TCP story throws a spotlight on how even small by-products in industrial processes can create outsized problems. Strict monitoring, transparent reporting, and investing in water treatment technology all matter. At the same time, holding polluters accountable and supporting affected communities should sit at the front of every discussion about chemical use and contamination.
I’ve come to see this not as a problem of the past, but a clear reason for tougher rules, honest labeling, and investment in public health. Only then will people stop hearing about cancer threats buried in something as simple as a glass of water.
Is 1,2,3-Trichloropropane hazardous to health?
What is 1,2,3-Trichloropropane?
1,2,3-Trichloropropane, often shortened to TCP, does not turn up in many household chats. Even so, this colorless liquid finds its way into the news, mostly on the back of industrial spills or concerns about drinking water. Used in manufacturing and as a cleaning solvent, TCP also appears as a byproduct from pesticide production. The chemical has no known use in food, but it ends up leaching into soil and groundwater.
Health Risks of Chemical Exposure
Any time I’ve researched chemicals in drinking water, certain names seem to reappear, and TCP is among them. Public health groups and scientists have warned about it for years. According to the U.S. Environmental Protection Agency (EPA) and the International Agency for Research on Cancer, TCP qualifies as a probable human carcinogen. This means studies point to an increased risk of cancer in lab animals, and in some communities, tests have tied higher rates of certain cancers to contaminated drinking water. No one wants to discover that their tap is delivering more than just H2O.
Health effects from TCP exposure do not just end at cancer. Ingesting or inhaling high amounts, even for a short time, may cause irritation of the respiratory tract, headaches, dizziness, and nausea. Liver and kidney damage become real concerns with long-term exposure. There’s bitter truth that even small concentrations over a long period can build up, especially in areas where the chemical sits tight in the soil and groundwater for decades.
Why Safe Water Matters
Access to clean water never feels negotiable. I remember the fear some families in California faced after groundwater samples revealed TCP well above safe limits. Folks had no clue that decades-old pesticide runoff could still cast a shadow over their kitchens and showers. Companies responsible for this chemical often left local government and taxpayers to deal with the aftermath. Costly filtration projects and legal battles drag on, always highlighting the real-world costs beyond the lab reports.
Regulatory Oversight and Solutions
The slow-moving wheels of regulation do not always match the urgency of a health threat. In 2017, California set one of the nation’s strictest limits for TCP in drinking water at five parts per trillion. More states are waking up to the risks. It takes strong oversight, regular testing, and quick action to keep water supplies safe. Reverse osmosis and granular activated carbon filters show promise for removing TCP, but installation and maintenance carry steep price tags.
People need regular updates about what’s in their water, not just a letter in the mail. Local agencies could build online dashboards or even community alerts so families have real-time information about their taps. If contamination happens, clear support—like bottled water and prompt filter installation—should follow. Stepping up enforcement against polluters and demanding that companies clean up their messes send a powerful message. These actions protect those who have no say in what chemicals pass through their pipes.
Building Trust Through Transparency
Trust in water depends on more than invisible regulations. Open reporting of chemical detections and breakdowns of clean-up progress let residents see that their health takes priority. As awareness grows, more towns and rural areas call for independent research and honest answers. Trusted scientists and local leaders can bridge the gap, turning complex terms into practical steps neighbors can take to stay safe. Ensuring water never poses a silent risk should always count as a community’s most basic right.
How should 1,2,3-Trichloropropane be stored and handled?
Understanding the Risks
1,2,3-Trichloropropane (TCP) rarely lands on the news, yet it deserves a frank discussion. I’ve spent years around industrial chemicals, and it’s always the quiet ones like TCP that turn up in the toughest headlines when something goes sideways. This clear, colorless liquid comes with a weighty reputation for toxicity and persistence in the environment. Direct contact can damage the skin and irritate the eyes. Breathing its vapors brings out headaches, dizziness, and long-term harm nobody wants. Hard facts show TCP doesn’t just evaporate and disappear; it hangs around in groundwater, and the EPA classifies it as a likely human carcinogen.
Storage: No Cutting Corners
Don’t play games with TCP storage. I’ve seen operations try to stretch aging containers to save a buck, but with this chemical, that shortcut leads to spills and major liability. Stores use high-integrity, corrosion-resistant steel drums or tightly sealed containers designed for halogenated solvents. These need to stay away from direct sunlight and heat. Temperatures swing enough in an uninsulated shed to warp drums and spark leaks. Secure, well-ventilated indoor storage—not a lean-to or a rusty metal box—means coworkers sleep better at night. Life gets a whole lot easier when secondary containment, like spill pallets, stands ready to catch drips or flood from a punctured drum.
It’s not just about containers. Every storage zone for TCP requires clear labeling, locked access, and regular checks for leaks or corrosion. Some folks assume fire won’t approach a liquid with a high flash point, but fire finds ways. Keep the fire suppressant gear close by, with chemical foam or dry powder suitable for organic solvents. Fire departments should know exactly where this chemical lives on-site, whether that’s a sheet at the entrance or a digital map they can pull up during an emergency.
Handling: Gloves On, Eyes Protected
Tossing on thin latex gloves never cut it with TCP. Nitrile or butyl rubber gloves—checked for pinholes—keep hands protected. Chemical splash goggles or face shields save eyesight from vapor clouds or accidental sprays. Workers suit up in coveralls, and vented hoods stop vapors from drifting into lungs. Every time someone cracks open a container, mechanical ventilation pulls fumes away. Respirators find their place if readings climb close to exposure limits, not after someone notices symptoms.
Spills demand attention right away. Absorbent pads and neutralizers sit within arm’s reach, not buried under shelves. Workers trained to respond act fast instead of running for the phone. Waste gets scooped into labeled hazardous containers; nobody tries to flush TCP down a drain or toss rags in the trash. On-site safety officers remind teams often: one slip turns into contaminated soil and lawsuits that last for years.
Taking the Lead on Responsibility
Ignoring TCP’s dangers brings consequences that stretch beyond the job site. I’ve seen small mistakes lead to months of environmental remediation, legal battles with neighbors, and even criminal charges. The chemical business trusts experience, and institutions like OSHA and the EPA set storage and handling rules for good reason. Following their guidelines means potential accidents stay rare. Double-checking training and equipment every month beats explaining the aftermath of a toxic spill in court.
Practical Steps for a Safer Workplace
Invest in sturdy storage, run routine hazard training, keep safety gear updated, and review procedures with every new batch. Document everything. Auditors want proof that workers stay safe and communities stay protected. Stakeholders—whether they’re employees, neighbors, or investors—expect nothing less than careful, responsible management of chemicals as persistent and dangerous as 1,2,3-Trichloropropane.
What are the environmental impacts of 1,2,3-Trichloropropane?
Looking at the Water We Drink
City tap water looks clean, but stories from farming towns often tell a different tale. In California’s Central Valley, farmers used soil fumigants and nematicides loaded with 1,2,3-trichloropropane, or TCP, throughout the 20th century. Most folks didn’t think too much about it back then. Wells now show TCP levels so high that a simple glass of water becomes a risk. People can’t always see or smell this chemical, but the consequences track back to the decisions made before anyone connected the dots.
Persistence and Spread
Once TCP lands in soil or groundwater, it rarely leaves. The compound resists breakdown by sunlight or bacteria, sticking around for decades. Rain doesn’t wash it away; neither do droughts. Heavy rainfall sends it deeper into aquifers. Almost every time someone studies a contaminated site, TCP levels stubbornly stick. United States Geological Survey maps show plumes spreading under city blocks, sometimes into neighboring regions. Clean-up plans often stretch generations, making TCP one of those problems handed down like unwanted heirlooms.
Health Risks Tied to Contamination
Cancer experts call TCP a “likely human carcinogen.” Drinking water with high TCP brings a higher risk of cancer after years of exposure. The number isn’t just a statistic — towns like Arvin, Lamont, and Bakersfield have faced lawsuits and real fear among residents who trust their kitchen taps less each year. A mother, filling a bottle for her child, wants answers. She should not have to parse chemical reports to know if water is safe.
Impact on Cropland and Wildlife
Crops don’t absorb much TCP, but the bigger story comes from its journey through soil and water. Frogs, insects, and fish crossing contaminated ground touch it, too. Irrigation ditches that carry runoff to rivers pass along traces far downstream. No one intends for a pesticide byproduct to linger in a trout’s habitat, but that’s what happens. Lessons learned with DDT echo here: long-lasting chemicals often show up where no one expects.
Community Burdens and Solutions on the Table
Water suppliers must choose between expensive filtration and warning their customers. Granular activated carbon filters and advanced oxidation processes help remove TCP from water, but these fixes cost millions. Small towns can’t always keep up. State regulators now call for stricter rules and enforceable limits. In California, legislation recently required water systems to notify customers when contamination exceeds the legal threshold.
Communities keep fighting for corporate responsibility. Companies that sold these fumigants now face lawsuits. Residents argue that the folks who profited should bear the cleanup costs. Such legal fights bring attention, but also remind us cleanup never feels quick or easy.
Choosing a Different Path
Options exist for heading off the next problem like TCP. Smarter pesticide regulation, thorough chemical testing before market release, and investments in modern farming methods can cut down future pollution. Consumers play a role by asking questions about food and water sources. As cities expand and new chemicals find their way into use, we need more transparency and accountability from both industry and regulators.
TCP’s lingering presence becomes a reminder to make decisions with tomorrow’s children in mind. Tech fixes alone can’t erase mistakes that last a lifetime, but listening, learning, and acting with more foresight can steer us toward a cleaner future.
What safety precautions are required when working with 1,2,3-Trichloropropane?
Why 1,2,3-Trichloropropane Matters in Safety Discussions
Handling chemicals like 1,2,3-Trichloropropane brings with it a heavy responsibility. Working with this compound goes beyond lab coats and goggles; misplaced steps and shortcuts put lives on the line. This chlorinated hydrocarbon carries some pretty nasty risks—skin burns, lung damage, and evidence points to possible cancer links in humans. My years in industry remind me that shortcuts with chemicals often lead to mistakes nobody plans to make.
Recognizing the Real Hazards
Liquid or vapor exposure to 1,2,3-Trichloropropane hurts more than people realize. A single splash can burn skin or irritate eyes severely. Breathing in the fumes—think carelessness with lids or poor ventilation—affects breathing, sometimes causing a persistent cough or worse. Chronic exposure in workplaces saw workers suffer liver and kidney trouble. The U.S. EPA and OSHA recognize this chemical as a hazard for potential cancer risk. Real people have ended up in hospitals from missed warnings, so these dangers aren't just lines in a manual.
Working Smart: Personal Protection First
Chemical-resistant gloves make a huge difference, with nitrile over latex. Too many times, I’ve seen folks grab the nearest gloves without checking compatibility—many gloves melt right through. Splash goggles with side protection stop a single drop from costing an eye. Even on hot days, wearing a proper chemical-resistant apron and sleeves has kept burns from becoming stories. Inhalation risk doesn’t get the attention it deserves; half-face respirators with organic vapor cartridges come out whenever fumes might build up. No one stands over an open container without one unless they feel like rolling the dice with their lungs.
Keeping the Workspace Safe
Tight, locked-down storage matters more than appearance. I store 1,2,3-Trichloropropane in fire-resistant cabinets—never in crowded fume hoods or on open benches. Every time someone skips labeling or forgets to check vent fans, spills and leaks get easier. More than a few accidents trace back to poor housekeeping. Facilities need working showers and eye wash stations within arm’s reach, not just somewhere in the next building. Regular, honest training doesn’t just tick off boxes; it shares stories where mistakes led to real harm, so new workers feel the reality behind the rules.
Reducing the Bigger Risks
Training and constant reminders have kept people safer than any stack of safety manuals. Peer checks on PPE before handling hazardous chemicals can catch missed steps. Some plants have adopted double-lock controls and sign-ins for access to certain solvents, cutting down on unauthorized or distracted handling. If a substitute with lower toxicity exists, use it—even if it slows down a process. Engineering fixes like sealed transfer systems and isolated mixing stations take people out of harm’s way more than any personal gear can. Risk drops when businesses choose safety over squeezing out a few more dollars.
Moving Forward Together
Respect for chemicals like 1,2,3-Trichloropropane grows out of experience and learning from mistakes. When companies put safety into daily habits—from the layout of workspaces to open conversations about close calls—workers go home whole and healthy. Anyone who spends time with dangerous chemicals owes it to themselves, and everyone around them, to demand more than the bare minimum: better practices, ongoing education, and a culture where looking out for each other isn’t just encouraged, it’s expected.
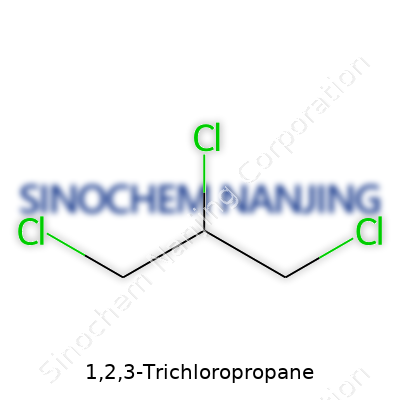
| Names | |
| Preferred IUPAC name | 1,2,3-Trichloropropane |
| Other names |
Allyl trichloride Glyceryl trichlorohydrin Trimethylene trichloride TCP Trichlorohydrin |
| Pronunciation | /ˌwaɪ.tuː.θriː.traɪˌklɔːr.oʊ.proʊˈpeɪn/ |
| Identifiers | |
| CAS Number | 96-18-4 |
| Beilstein Reference | 1730767 |
| ChEBI | CHEBI:17140 |
| ChEMBL | CHEMBL44438 |
| ChemSpider | 8257 |
| DrugBank | DB06800 |
| ECHA InfoCard | ECHA InfoCard: 03b254e4-580c-45b1-92c8-5c8739b02b4e |
| EC Number | 202-486-1 |
| Gmelin Reference | 824 |
| KEGG | C06581 |
| MeSH | D014268 |
| PubChem CID | 6576 |
| RTECS number | TR5250000 |
| UNII | NFY0XIC13C |
| UN number | UN2810 |
| CompTox Dashboard (EPA) | DTXSID8020345 |
| Properties | |
| Chemical formula | C3H5Cl3 |
| Molar mass | 147.44 g/mol |
| Appearance | Colorless liquid |
| Odor | Chloroform-like |
| Density | 1.388 g/mL at 25 °C(lit.) |
| Solubility in water | Moderate (7.5 g/L at 20 °C) |
| log P | 2.41 |
| Vapor pressure | 3.1 mmHg (20°C) |
| Acidity (pKa) | 13.6 |
| Magnetic susceptibility (χ) | -6.13·10⁻⁶ |
| Refractive index (nD) | 1.488 |
| Viscosity | 2.02 mPa·s (20 °C) |
| Dipole moment | 2.68 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 309.6 J⋅mol⁻¹⋅K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –165.0 kJ·mol⁻¹ |
| Std enthalpy of combustion (ΔcH⦵298) | -2064.3 kJ/mol |
| Hazards | |
| GHS labelling | GHS07, GHS08, GHS05 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H331, H350 |
| Precautionary statements | P210, P261, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P308+P313, P501 |
| NFPA 704 (fire diamond) | 2-4-1 |
| Flash point | 71 °C |
| Autoignition temperature | 370 °C |
| Explosive limits | 4.0–10.5% |
| Lethal dose or concentration | LD50 (oral, rat): 187 mg/kg |
| LD50 (median dose) | > Rat oral LD50: 187 mg/kg |
| NIOSH | TTQ |
| REL (Recommended) | 1 ppm (7 mg/m3) |
| IDLH (Immediate danger) | 75 ppm |
| Related compounds | |
| Related compounds |
Chloroform 1,3-Dichloropropane Epichlorohydrin Glycerol Allyl chloride |

