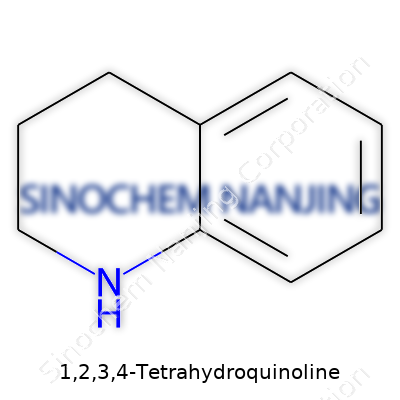
1,2,3,4-Tetrahydroquinoline: A Grounded Look at a Noteworthy Molecule
Origins and Historical Development
Chemists have eyed the quinoline family since the 19th century, scouring coal tar for clues on how life’s molecules work and how to make useful materials. 1,2,3,4-Tetrahydroquinoline popped up on the scene as researchers explored hydrogenation routes to tweak the aromatic system and bring new reactivity—opening doors to pharmaceuticals, dyes, and agrochemicals. I came across it in a university lab, puzzling over its synthesis for a project on antimalarial compounds. That project made it clear to me how foundational these building blocks are in synthetic chemistry, often acting as a springboard for designing modern therapies and specialty chemicals. The backbone has driven quite a bit of medicinal chemistry, as scientists chase after nitrogen-containing rings for their rich biological activity and potential to unlock new drugs.
Product Overview
Think of 1,2,3,4-Tetrahydroquinoline as the softened cousin of quinoline. The extra hydrogen atoms tucked into the ring structure make it more flexible chemically. Small tweaks to this core have paid dividends for researchers aiming to invent tuberculosis drugs and plant protection agents. In day-to-day lab work, it turns up as a pale yellow or colorless oily liquid, and over the years, suppliers have tightened up purity grades with ever-better distillation processes. The molecule is sold by its common name and pops up in journals and industry catalogs under aliases like “tetrahydroquinoline,” “THQ,” and systematic IUPAC conventions. Lab supply bottles speak the simple language of percent purity, boiling range, and safety precautions.
Physical and Chemical Properties
Pour 1,2,3,4-tetrahydroquinoline into a glass vial, and you’ll quickly notice the faint aroma, more pungent than offensive, letting you know that nitrogen is in there. At room temperature, it creeps along as a nearly colorless liquid, boiling near 243 degrees Celsius. It shows the solubility patterns you would expect for a heterocyclic ring: dissolve it in organic solvents, but don’t count on it mixing with water. The ring system resists oxidation better than its fully aromatic cousin, giving it a unique place in synthetic schemes. Stability remains a draw—in the right bottle, under cool and dry storage, this compound outlasts many other intermediates. It supports functionalization at several positions, tempting chemists to build more complex molecules on its framework.
Technical Specifications and Labeling
Lab work depends on clear specifications. Bottles of tetrahydroquinoline carry hazard labels for skin and eye irritation and advice for use in ventilated hoods. Material safety data sheets stress gloves and eye shields. Bench scientists keep a close watch on chemical purity—80 percent or higher purity for basic research, higher for pharmaceutical work. Handling instructions cut to the chase: store away from oxidizing agents and acids, keep lids tightly closed, and weigh small amounts using amber glassware to reduce light exposure. Reporting structure and labeling focus more on safety and quality than on marketing, so the same bottle could turn up in a particle physics lab or an agrochemical formulation workshop.
Preparation Method
The route into 1,2,3,4-tetrahydroquinoline usually starts with quinoline or its derivatives. Generations of chemists have tested different hydrogenation catalysts: nickel, palladium, platinum. I’ve sat at a hydrogenation reactor, monitoring pressure, temperature, and stir rate, waiting for the optimum conversion—enough to saturate the ring without touching the aromatic system. Catalytic transfer hydrogenation with safer reagents like formic acid and ammonium formate offers alternatives where compressed hydrogen isn’t available. Each approach trades off cost, scalability, and selectivity, though all roads require careful workup and purification since leftover catalyst or partial hydrogenation products can spoil downstream steps. Traditional batch reactors continue alongside automated flow systems in larger facilities, keeping this intermediate readily available for both research and industry.
Chemical Reactions and Modifications
What makes tetrahydroquinoline captivating is its ability to serve as both a chemical sponge and a building scaffold. The molecule goes through N-alkylation, oxidation, and aromatic substitution, letting chemists expand its role into antimalarial, antiviral, and anti-inflammatory agents. The amine nitrogen at position one lets you plug on different functional groups—methyl, ethyl, benzyl—and each tweak changes how it interacts with biological targets. Beyond pharmaceutical leads, derivatives serve as ligands in transition metal catalysis, or as starting materials for material science projects. The large-scale synthesis of advanced materials sometimes begins with a bench-top reaction of this very compound. Chemistry papers keep exploring new reactions on this skeleton, showing an energy that points to its continued utility.
Synonyms and Product Names
While its tongue-twisting official name, 1,2,3,4-tetrahydroquinoline, tends to dominate, the molecule travels under different banners. I’ve seen it called “THQ,” “tetrahydroquinoline,” and just “hydroquinoline” in reaction notebooks. Regulatory registries and supplier inventory sheets also use CAS numbers for clarity in ordering and compliance checks. No matter the label, the compound draws attention for its chemical reactivity. Those naming variations sometimes trip up newcomers, but a few minutes with a chemical database sorts out the confusion.
Safety and Operational Standards
Working with tetrahydroquinoline means respecting its risks. Lab protocols set ventilation, splash guards, and face shields as non-negotiables. If a drop gets on your skin, it can cause irritation, and inhaling vapors isn’t wise. Disposal must meet strict standards because of its nitrogen content and potential to make problematic byproducts when burned or oxidized. No wonder chemical hygiene courses drum this point in with stories of lab accidents—a reminder that responsible use isn’t just about following the rules, but about protecting everyone in the lab. In industry, automated handling reduces direct exposure, with robotic pipettors and remote monitoring. The trend in research circles pushes for greener solvents and closed systems, cutting back on waste and environmental impact.
Application Area
See the fingerprints of tetrahydroquinoline in the pharmaceutical industry, crop protection, dye synthesis, and catalysis. Medicinal chemists lean on it to build both licensed and experimental drugs, hunting for molecules that slip past bacterial defenses or disrupt cancer pathways. Agrochemical researchers check how ring modifications tackle fungal blights and insect pests. Material scientists adapt its chemistry for organic electronics, including light-emitting diodes and photovoltaic cells. In every application, the nitrogen ring does more than offer structure—it tweaks how compounds behave in the environment or living systems, changing activity, stability, and absorption. Having worked with this molecule across several projects, I can say that any researcher bumping into a challenge with nitrogen rings ends up considering tetrahydroquinoline as a scaffold or intermediate before long.
Research & Development
Today’s research explores greener ways to make tetrahydroquinoline, pushing for catalysts that use less energy or leave minimal waste. High-throughput screening and machine-learning models speed up the design of new derivatives, shaving months off the slow grind of bench experiments. Pharmaceutical companies invest in analogs with tweaks for treating mental health and infectious diseases. Chemoinformatics studies map out possible modifications, exploring which tweaks boost biological activity before running expensive animal trials. Even in academic circles, grant calls look for eco-friendly and cost-effective syntheses, since cost and scale can decide whether a molecule moves from idea to industrial drum. With more automation in labs, the workflow shifts to data analysis and less repetitive hands-on grunt work—all helpful conditions for scientists fixated on using tetrahydroquinoline's versatile ring to solve pressing health and material challenges.
Toxicity Research
Toxicity remains a top concern, since nitrogen heterocycles can affect the liver, kidneys, or nervous system if mishandled. Regulatory data shows that small-scale exposure tends to cause irritation but not lasting harm; animal testing at high doses has uncovered organ effects, especially if the structure gets metabolized unevenly. Toxicologists keep a close watch on chronic exposure and breakdown products, since nitrogen rings sometimes form persistent pollutants or bioactive compounds. In my time working with risk teams, routine air monitoring in workspaces proved essential—not just for compliance, but for uncovering hidden dangers. Scientists now map toxicity early in research, pushing for safer alternatives or tweaking molecular structures to avoid unwanted side effects in potential drugs. The pressure to meet global safety standards grows year by year as international regulatory bodies tighten the rules for chemical handling, documentation, and downstream application.
Future Prospects
Looking ahead, the future for 1,2,3,4-tetrahydroquinoline features plenty of promise. Demand for nitrogen-containing intermediates won’t slow, especially as the hunt for targeted pharmaceuticals and high-performance materials keeps picking up pace. Sustainable chemistry drives efforts to reinvent its synthesis using plant-based raw materials or renewable catalysts. Cross-disciplinary applications beckon—outside of medicine and agriculture, teams investigate it for sensors, battery electrodes, and even antiviral coatings. The challenge: keeping one eye on safety, environmental footprint, and regulatory shifts as new uses appear. Community conversations at conferences and in journal editorials keep surfacing the same point: progress comes from better processes, mindful use, and relentless curiosity about what this little ring can do. From the first flask to the latest high-throughput screen, this molecule looks set to keep its seat at the table.
What is 1,2,3,4-Tetrahydroquinoline used for?
Chemistry’s Unsung Workhorse
1,2,3,4-Tetrahydroquinoline doesn’t star on the evening news or spark dinner-table debates, but in many labs, it’s a solid staple. It’s a nitrogen-containing heterocycle, closely related to quinoline, only this version carries four extra hydrogen atoms. That subtle tweak changes how researchers and manufacturers approach a variety of challenges. I remember in my early days during university lab sessions, hands-on with aromatic compounds, realizing the potential packed into such structures. Scientists worldwide treat this molecule as a versatile building block.
Medicinal Chemistry: More Than Just a Building Block
Pharmaceutical labs rank 1,2,3,4-tetrahydroquinoline high up on their list of starting materials. This compound often ends up as the scaffold for drug candidates. Medicinal chemists tweak its structure to create new products. Some drugs under development, such as antihypertensive or antimalarial agents, use derivatives formed from this very molecule. Researchers highlight its unique arrangement because it helps mimic biological molecules, offering a shortcut for better drug-receptor fit or bioavailability. Drug patents regularly reference analogues, thanks to the scaffold’s flexibility and compatibility in synthesis.
The Colorful Side: Dyes and Pigments
Factories focused on dyes have plenty of reasons to order this compound. Its chemical structure paves the way for developing new dyes and pigments, especially ones meant for textiles or specialty inks. The quinoline backbone shines in color chemistry—add hydrogens, adjust side chains, and suddenly the array of colors widens. Textile industries demand high-quality, stable pigments, and tetrahydroquinoline-derived dyes can stand up to sunlight or repeated washing much better than simpler colorants.
The World of Materials Science
Beyond medicine and colors, there’s another less-publicized corner: materials chemistry. Researchers working on organic electronics or specialized polymers explore compounds like 1,2,3,4-tetrahydroquinoline as monomer units. Certain liquid crystals, which power everything from calculators to flat-screen TVs, use quinoline derivatives. Scientists keep searching for better combinations to improve flexibility, conductivity, and stability. I once joined a seminar where a chemist showed prototypes of organic solar cells—one of the molecular tweaks involved a tetrahydroquinoline core, pushed for efficiency and durability.
Chemical Synthesis and Catalysis
This compound helps chemical engineers and synthetic chemists build targets that aren’t easy to reach otherwise. Its ability to donate or accept electrons makes it useful in catalysts and in making complex molecules. Many reactions depend on efficient scaffolds to carry out steps cleanly. This one stands as one of those reliable, mid-range tools that professionals count on to get chemical transformations right. Scientific journals fill their experimental sections with procedures that use this molecule—some geared for large-scale synthesis, some for precise, small-batch experiments.
Challenges and Safety
Every tool has its downsides. Waste management stands as a real concern for chemical manufacturers handling this compound, considering environmental protection and worker safety. Companies planning new production lines follow strict regulations to keep exposure in the factory low. Researchers design greener methods, cut down on solvents, and recover as much starting material as possible. It isn’t just about getting results—it’s about responsibility.
Grow More, Waste Less
Looking ahead, the future for 1,2,3,4-tetrahydroquinoline revolves around creativity and efficiency. Labs focus on greener synthesis, cleaner disposal, and expanding its use in fields such as renewable energy, where demand for efficient materials keeps growing. Each discovery in one area can open doors in others, driving both innovation and sustainability.
What is the chemical structure of 1,2,3,4-Tetrahydroquinoline?
Looking at 1,2,3,4-Tetrahydroquinoline
People working with organic molecules often come across rings and fused rings in chemical drawings. 1,2,3,4-Tetrahydroquinoline lands right in that territory. Chemists know this molecule features a fused bicyclic system, where a benzene ring shares a pair of carbon atoms with a partially saturated six-membered nitrogen-containing ring. Its structure reads like a short chapter of organic chemistry’s endless novel: six aromatic carbons on one side, tied to a heterocycle where four carbons and a nitrogen round out the second ring. The “1,2,3,4-tetrahydro” bit signals four extra hydrogens saturating the quinoline backbone, reducing half of the second ring to a structure closer to piperidine.
Why Chemists Care About This Structure
The arrangement isn’t just a quirky feature; it makes this compound unique and useful. Fused aromatic and heterocyclic rings show up in a lot of pharmaceutical research. These skeletons get used as building blocks for drugs because the nitrogen atom at the ring junction offers a convenient site for chemical tweaks—things like adding functional groups or building larger molecules.
I’ve seen this backbone used as a scaffold for medicinal chemistry. Researchers like it because it’s sturdy and can anchor various chemical groups. Some antihistamines and potential anti-cancer drugs draw on quinoline and its partially hydrogenated cousins, including 1,2,3,4-tetrahydroquinoline. The fused system interacts well with enzymes, binding snugly in pockets where fully aromatic or fully saturated rings won’t fit as neatly. I once worked in a lab where a whole year went into making tiny changes on this backbone, seeking a version that tackled malaria parasites in vitro.
Connecting Structure, Health, and Application
A strong foundation of chemistry concepts helps here. The double-ring spine with the nitrogen atom often supports both hydrogen bonding and π-stacking in biological systems. What happens in a flask doesn’t always happen in the bloodstream, but drug developers depend on molecules like this as a starting point. Mélange of aromaticity and saturation boosts solubility and lets the quinoline core enter cells more easily.
On the industrial side, the compound shows up in dye manufacturing, agrochemical development, and as an intermediary for other specialty chemicals. Chasing color fastness in textiles or maximizing pest control sometimes goes smoother with the help of structures like this, especially because it’s possible to add reactive sites or tailor side chains according to end needs.
Room for Improvement and Solutions
Some problems persist, though. Access to pure, well-characterized 1,2,3,4-tetrahydroquinoline isn’t universal. Lab synthesis can run up against scalability issues, and impurities sometimes derail research or process development. Academic reports often talk about purity, but pharmaceutical-grade batches demand more rigorous controls. Collaborating with suppliers and investing in better purification tech helps. As green chemistry advances, finding cleaner and more efficient ways of building these bicyclic backbones also matters. Fewer byproducts, less waste, and cheaper feedstocks will take this molecule from an occasional specialty to a mainstay in sustainable chemical manufacturing.
Unraveling the full potential of 1,2,3,4-tetrahydroquinoline starts with understanding its structure, but it moves ahead through careful research, good partnerships, and a willingness to experiment at the bench and in the plant.
Is 1,2,3,4-Tetrahydroquinoline hazardous or toxic?
Getting Real About Lab Chemicals
Walk into any chemistry lab, and it’s easy to get used to rows of bottles full of names that sound like tongue twisters. 1,2,3,4-Tetrahydroquinoline falls into that category. For most people, the name never pops up outside research circles, but those who have worked with it know the smell, the oily feel, and the need for gloves and good ventilation. Getting information straight means cutting through jargon, looking at toxicology data, taking seriously the practical stories from lab veterans, and not just sticking to technical language lifted from a data sheet.
The Facts on Toxicity
[^1]To get the basics straight: 1,2,3,4-Tetrahydroquinoline counts as an organic compound in the nitrogen heterocycle family. Reports from animal studies and some workplace safety guidelines call attention to potential hazards. Swallowing or breathing in this stuff brings a risk of harm. Animal studies have noticed signs like reduced activity, respiratory issues, and even death at high doses. Chemicals with similar structures show signs of damaging the liver, kidneys, or nervous system after repeated exposure. The science here suggests caution, even if someone can’t remember all the mechanistic pathways or every symptom in the toxicology reports.
Skin Contact and Chronic Risks
People who have worked with this compound often remember the way it can sneak past thin gloves or hang in the air after a spill. Absorption through the skin leads to irritation. Even small splashes sting, and rashes stay for days. Eyes take the brunt even after a quick mist. From experience, direct handling without goggles nearly guarantees trouble. Short-term, the risk means painful irritation. Stay around those vapors or get it on your hands repeatedly, and there’s talk of more lasting issues—think headaches, dizziness, or skin running dry and starting to crack. No one wants to gamble with nerve symptoms or damaged organs, and stories from industrial plants repeat that lesson.
Environmental Side
This compound doesn’t just stop with people. Dispose of it carelessly, and soil or water pick up the slack. Nitrogen heterocycles, in general, aren’t the sort you want leaking into riverbeds or groundwater. Some studies point out toxic effects on aquatic organisms. Nature doesn’t always bounce back quickly, and pollution hangs around in some ecosystems for years. The costs of cleanup—fish kills, fines, damage to clean water—hit small communities the hardest. Good stewardship asks more than just tossing leftovers down the drain or venting fumes into the open air.
Safer Practices and Real Solutions
Pure caution beats apologies after the fact. Thick nitrile gloves, splash goggles, and a working fume hood form the basics of defense. In the lab where I worked, the routine was simple: bottle stays sealed, spills get cleaned with real urgency, and everyone double-checks their protective kit before they even uncap the bottle. Training for everyone on the team keeps mistakes down, especially with new hires who’ve yet to learn the hard way. Waste heads into the right hazardous chemical containers, not into the regular trash. No corners cut, no half-measures.
Buy only as much as needed. Don’t stockpile. Challenge the recipe in research or manufacturing—ask if solvents or processes could swap this compound for something safer. Supply companies stock alternatives for a reason, and skipping a more hazardous step can speed up cleanup work and prevent injuries. Staying updated on hazard assessments and reviewing safety data keeps people honest about risks. I’ve seen labs change direction entirely after a single mistake.
Final Thoughts
No chemical ever stays entirely risk-free, but information, training, the right equipment, and plain common sense cut the dangers down where it counts. Treat 1,2,3,4-tetrahydroquinoline with respect, and its hazards lose the power they hold over inexperienced hands.
What are the storage conditions for 1,2,3,4-Tetrahydroquinoline?
Understanding Chemical Sensitivity
1,2,3,4-Tetrahydroquinoline shows up in many organic chemistry labs. It’s a solid example of a compound that sits between basic organics and more reactive specialty chemicals. Controlled storage can make the difference between trouble-free handling and unexpected risk. Over the years, I’ve seen labs lose valuable samples to simple mistakes—humidity in the wrong place, or a forgotten open vial during a busy workday.
The Real-World Risks
The compound has a mild odor, but don't let that fool you. Prolonged or careless exposure can irritate skin and eyes. Inhalation isn’t a wise idea, either. A spill on a hot day spills more than trouble. That's more than an inconvenience; once, an open bottle in a shared drawer led to complaints about the odor, which led to a scramble to find proper storage information.
Those sorts of mistakes chip away at trust. Proper storage isn’t just about checking off boxes—it protects everyone in the room, and it keeps expensive materials from going to waste. No matter how seasoned you are, basic respect for the chemical shelf saves headaches down the road.
Core Storage Rules
A dry, well-ventilated spot knocks out most hazards with 1,2,3,4-Tetrahydroquinoline. Temperatures around typical room conditions—roughly 20°C to 25°C—work well, so long as things don’t swing wildly. I’ve never lost a bottle to a cool, shaded cabinet, but I’ve seen solvents spoil when someone left them near a window.
Humidity deserves attention. This compound holds up in dry air, but long-term exposure to moisture leaves a mark. Always cap bottles tightly, and store containers far from sinks or steam sources. I learned this the hard way. A leaky pipe once ruined half our reference stock—simple oversight, but costly.
Direct sunlight also causes issues. Bright light can, over time, start slow degradation, especially if someone leaves a clear glass bottle by mistake. Better to use amber glass, though not absolutely necessary for short storage.
Labeling and Isolation
A legible, up-to-date label beats even the best memory. Include the date received, concentration, and who’s responsible for the chemical. It’s tempting to cut corners on busy days, but those handwritten notes save stress during safety audits or emergencies.
Keep away from strong oxidizers and acids. This isn’t just a theory—mixing quinoline derivatives with the wrong neighbors can start dangerous reactions. Store it on a separate shelf, not next to the bleach, and avoid shared bins with unknowns.
Safe Handling Tips
Putting on gloves and safety glasses feels like second nature after a while, but these habits defend against those “just a few drops” moments that lead to hospital visits. If you spill any, clean up right away with proper absorbents and notify coworkers. An eyewash station nearby isn’t an option; it’s a priority.
Check stored containers every few months. Look for changes in color, texture, or smell. Early intervention catches problems before they get worse. Disposal should always follow local hazardous waste protocols—never pour leftovers down the sink.
Building a Culture of Respect
People sometimes ask why storage rules get repeated so often. The truth: nobody wants to be the person responsible for an accident that was preventable. I’ve worked in places where a strong safety culture didn’t just prevent problems; it built trust across the team. For 1,2,3,4-Tetrahydroquinoline, a few minutes spent storing it properly saves hours of cleanup and risk down the line. That’s responsibility in action, not just compliance.
What is the molecular weight of 1,2,3,4-Tetrahydroquinoline?
Taking Apart the Numbers
Chemistry often feels like learning a new language. Behind every name, there’s a story about structure and utility. With 1,2,3,4-Tetrahydroquinoline, the molecular weight lines up at 133.19 g/mol. That figure isn’t just for trivia nights. It feeds right into any work done with this compound, from precise measurements in the lab to scaling up reactions in the industry.
Work in the Lab Takes Precision
In my experience preparing mixtures, nothing throws off a project faster than vague weights. For researchers or chemical manufacturers, this molecular weight matters. It lets chemists calculate how much of the compound to add to achieve the right chemical balance. Get the math wrong, waste materials, and run up expenses or even get poor results.
Handling 1,2,3,4-Tetrahydroquinoline, the difference between success and a failed experiment can hang on a precise number. If, say, you plan a reaction where a set number of moles determines whether you get a working drug candidate or just a gummed-up flask, this molecular weight becomes the anchor.
Big Players Know Their Numbers
Pharmaceutical research leans on this kind of detail. 1,2,3,4-Tetrahydroquinoline pops up as a building block in drug development and synthesis of dyes. Knowing its weight means chemists can swap it in and out, combine it with other molecules, and forecast exactly how much starting material to buy or store.
Some years ago, I watched a colleague troubleshoot a batch of compounds. The chain reaction would only work with the right ratio of starting materials. The first runs failed, and it turned out the original calculations leaned on a typo in the molecular weight. After fixing the math, yields spiked, costs dropped, and the mood in the lab improved.
Staying Accurate: Why Trust Matters
Nobody wants a lab notebook full of unreliable data. One thing I've learned: chemical suppliers and scientific databases keep a close eye on published values. Publishing correct information, backed by reliable sources like PubChem and Merck Index, builds trust. For 1,2,3,4-Tetrahydroquinoline, the consensus lands on 133.19 g/mol, backed by peer-reviewed literature and reputable databases.
It’s easy to see why Google and regulators talk about E-E-A-T standards. Accurate information keeps experiments reproducible and investments safe. Small errors can cascade through whole research and development chains, making the right molecular weight essential.
Linking Small Errors to Big Solutions
Chemists, especially beginners, run into trouble if they skip over details. Some classrooms push the point: double-check values, cross-reference reputable sources, and make sure every calculation holds up. Digital lab management tools have made it easier to check molecular weights in real time, cutting down on human error.
Teaching students how to find, double-check, and use these numbers in context would go a long way in raising quality across the field. It’s not about memorizing every weight, but learning where and how to confirm the right data. Giving researchers access to up-to-date digital libraries and direct supplier verification helps guard against old or inaccurate listings.
So, 1,2,3,4-Tetrahydroquinoline may just seem like another name out of a textbook, but its molecular weight represents more than an entry on a label. It’s a number that keeps experiments honest, products safe, and progress on track.
| Names | |
| Preferred IUPAC name | 2,3,4,4a-Tetrahydroquinoline |
| Other names |
Benzene, 1,2,3,4-tetrahydro- Tetrahydroquinoline 1,2,3,4-Tetrahydro-1-quinoline Quinoline, tetrahydro- THQ |
| Pronunciation | /ˌwʌn,ˌtuː,ˌθriː ˌpɔɪnt ˌfɔːr ˌtɛtrəˌhaɪdrəʊkwɪˈnəʊliːn/ |
| Identifiers | |
| CAS Number | **635-46-1** |
| 3D model (JSmol) | `3D model (JSmol) string` for **1,2,3,4-Tetrahydroquinoline**: ``` c1ccc2c(c1)CCCN2 ``` |
| Beilstein Reference | 136873 |
| ChEBI | CHEBI:35561 |
| ChEMBL | CHEMBL15408 |
| ChemSpider | 69278 |
| DrugBank | DB01899 |
| ECHA InfoCard | 100.011.074 |
| EC Number | 208-743-2 |
| Gmelin Reference | 82722 |
| KEGG | C09649 |
| MeSH | D015847 |
| PubChem CID | 7059 |
| RTECS number | XU4025000 |
| UNII | 5984558Q5V |
| UN number | UN2305 |
| CompTox Dashboard (EPA) | DTXSID6041797 |
| Properties | |
| Chemical formula | C9H11N |
| Molar mass | 145.22 g/mol |
| Appearance | Colorless to yellow liquid |
| Odor | amine-like |
| Density | 1.091 g/mL at 25 °C(lit.) |
| Solubility in water | slightly soluble |
| log P | 1.86 |
| Vapor pressure | 0.19 mmHg (25°C) |
| Acidity (pKa) | 5.70 |
| Basicity (pKb) | 5.73 |
| Magnetic susceptibility (χ) | -66.5·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.597 |
| Viscosity | 13.2 cP (20°C) |
| Dipole moment | 2.33 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 170.9 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | 10.3 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3255 kJ mol⁻¹ |
| Hazards | |
| Main hazards | Harmful if swallowed. Causes skin irritation. Causes serious eye irritation. Suspected of causing cancer. Toxic to aquatic life with long lasting effects. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P264, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 1,2,3,4-Tetrahydroquinoline: 2-2-0 |
| Flash point | Flash point: 115°C |
| Autoignition temperature | 515°C |
| Explosive limits | 6.6–14% |
| Lethal dose or concentration | LD50 oral rat 400 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 800 mg/kg |
| NIOSH | RN6569 |
| PEL (Permissible) | PEL: 2 ppm |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | 100 ppm |
| Related compounds | |
| Related compounds |
Quinoline 2-Methyl-1,2,3,4-tetrahydroquinoline N-Methyl-1,2,3,4-tetrahydroquinoline 1,4-Dihydroquinoline Indoline |

