1,1-Dichloroethane: Past, Present, and Paths Ahead
Historical Development
Few chemicals speak to the tangled relationship between industry, science, and health like 1,1-dichloroethane. Introduced in the early 20th century, this colorless, sweet-smelling liquid found its place during a time of explosive growth in chlorinated hydrocarbons. The conversation around solvents and intermediates kicked up as new materials like plastics and synthetic fibers gained momentum. 1,1-Dichloroethane, known among chemists as ethylidene chloride, fit right into this era. It landed in the toolbox of factories seeking reliable reactions and a dependable solvent with more stability than some earlier, more volatile choices. Chemists at the time saw an opportunity to simplify synthetic pathways, so they brought it in for polymer work, especially as demand for vinyl chloride and related products surged. Over the years, its use shifted, shadowed by the evolution of chemical safety regulations and rising awareness of volatile organic compounds’ impacts.
Product Overview
This compound carved a niche as both an industrial solvent and a chemical intermediate. Early industrial reports often highlighted its effectiveness in extracting oils and fats, stripping away grease, and deploying it as a component in various blended solvents. In manufacturing circles, it played its part on the pathway to vinylidene chloride, which helped form saran wrap and certain plastic films. Laboratories leaned on it for selective reactions, such as halogenation processes, where predictability and reaction selectivity mattered. Over time, 1,1-dichloroethane never dominated headlines the way some solvents did, but those who spent time with it understood its reliability and peculiarities.
Physical & Chemical Properties
Mix a sweet odor easily noticed in the air with a low boiling point, and the result is a solvent that evaporates quickly under everyday conditions. 1,1-Dichloroethane registers a boiling point around 57 degrees Celsius, making its volatility a practical consideration both for engineers designing systems and for safety teams crafting emergency protocols. The liquid’s density sits higher than water, so it forms a distinct lower layer in mixtures. Chemically, its two chlorine atoms hang on a single carbon, which sets it apart from its isomer, 1,2-dichloroethane. This structure affects how it behaves in the body and in industrial reactions; the chlorines make it less reactive toward bases, but surprisingly handy in certain nucleophilic substitution reactions. Its moderate solubility in water sets some operational limits and calls for tighter storage and handling rules.
Technical Specifications & Labeling
Regulatory labels highlight its flammability and potential health risks, a reflection of years of toxicology studies and research into environmental persistence. A history of mislabeling in the early days nudged authorities to require clearer hazard statements and handling guides. Product grades diverged based on downstream use: higher purities power labs and chemical synthesis, while technical grades occasionally leak into degreasing or cleaning operations. Compliance with updated regulations remains the only way to avoid costly recalls, legal challenges, and workplace injuries. The rise of the Globally Harmonized System (GHS) changed the language but underscored the same risks, reminding everyone from plant managers to shippers to handle it with care.
Preparation Method
A typical route brings together ethylene and chlorine in the presence of a catalyst, steering the reaction toward the 1,1-dichloro product. This chlorination process, refined over decades, relies less now on brute force and more on controlled reaction conditions. Trying to minimize byproduct formation became a point of pride in chemical engineering teams, who adjusted temperature and pressure parameters to steer away from 1,2-dichloroethane or unwanted tails. Recycling chlorine and managing vented gases mark the turning points in sustainable manufacturing: companies that invest in taller columns, better seals, and proper off-gas treatment now lead the way in responsible chemical production.
Chemical Reactions & Modifications
For those interested in chemical synthesis, 1,1-dichloroethane acts as a versatile building block. Its activation under certain conditions leads to dehydrochlorination, often a precursor to vinylidene chloride, crucial for plastics and barrier films. In reductions or with strong nucleophiles, it opens up routes to other dichloroethanes or simple hydrocarbon chains after dechlorination. The compound also lends itself to comparative reaction studies thanks to its structural simplicity. These qualities matter for chemists aiming to optimize yields, reduce waste, or pursue greener synthesis. Its reactions raise caution flags, as thermal or photolytic breakdown can release toxic fumes and persistent organochlorines, creating engineering and safety challenges that only rigorous protocols resolve.
Synonyms & Product Names
The chemical identity of 1,1-dichloroethane sometimes confuses those outside chemistry: it answers to names like ethylidene dichloride, 1,1-DCA, and even DCE, although the latter causes unwanted mix-ups with its 1,2-isomer. Older literature occasionally used brand or trade names, but the chemical structure always rooted product identity. For supply chain managers and researchers, double-checking these synonyms protects against misorders and accidental substitutions in sensitive processes.
Safety & Operational Standards
Safety thought has come a long way since the days when solvent use drew little scrutiny. 1,1-Dichloroethane brings its own risks: inhalation exposure, skin absorption, and metabolic breakdown into potentially toxic products. Modern plants adopt closed-system engineering, robust ventilation, and sensor-driven leak monitoring. Safety training shifted from compliance exercises to skills drills, worth every second when handling substances that cross paths with pressurized lines or heated surfaces. Regulatory agencies periodically update permissible exposure limits, forcing refineries and synthesis shops to audit procedures and invest in PPE upgrades. Waste handling remains a sticking point, with on-site chemical neutralization or transport to approved incineration facilities often required, driven both by environmental rules and genuine concern for groundwater and air quality.
Application Area
This chemical finds its main uses as a solvent and as an intermediate in synthesizing vinylidene chloride. Industrial teams value its solubility profile and volatility where rapid removal of solvent matters, like coatings or adhesives. Its limited but persistent role in some extraction processes keeps it on certain equipment lists, while outside chemical manufacturing, new uses don’t frequently arise. Laboratories sometimes include it in specialty halogenation or elimination reactions, especially in syntheses where the specific reactivity of chlorinated intermediates helps drive selectivity. The steady decrease in allowable emissions and discharge has led many manufacturers to phase it out or substitute less hazardous chemicals, especially in products touching consumers.
Research & Development
Ongoing research explores alternatives and process improvements, fueled by environmental and health priorities. Green chemistry teams direct attention toward minimizing formation, capture, and ultimate destruction or recycling. Some technical projects focus on photocatalytic breakdown, leveraging new materials that break down chlorinated ethers or ethanes under sunlight or UV, hoping to find solutions that work at industrial scales, not just in small pilot setups. Continuous improvement in reactor design and automation, too, offer hope for tighter process control, higher selectivity, and energy efficiency. Academic labs keep tracking transformation pathways in water, soil, and air, helping regulators and engineers close knowledge gaps about byproducts and fate.
Toxicity Research
Early on, workers and neighborhoods near production plants reported symptoms that caught regulators’ attention: headaches, nausea, even organ damage after long, unprotected exposure. Contaminated surface water and air samples demanded studies on both acute and chronic effects. Researchers pinpointed how the molecule absorbs into the body, metabolizes, and forms reactive intermediates that trigger cellular stress or DNA changes. Animal studies raised cancer alarms, prompting classification as a possible carcinogen by several authorities. Ongoing work searches for better exposure biomarkers, mitigation strategies, and deeper understanding of low-level, long-term impacts. Environmental monitoring, influenced by citizen science and stricter reporting, reveals residues in groundwater near some sites years after production ended, spotlighting the need for responsible legacy management.
Future Prospects
Over the next decade, regulatory forces and public health data will likely continue to limit the practical footprint of 1,1-dichloroethane. Sectors relying on it will need to invest in safer substitutes, process upgrades, or in some cases, full redesigns that eliminate its use altogether. Legacy contamination, still visible at certain cleanup sites, remains a thorny challenge requiring both technical fixes and public engagement. Green chemistry and sustainable engineering hold promise. As new discoveries in catalysis and solvent design spread from lab to factory floor, the chemical’s role will diminish. Yet, the arc of 1,1-dichloroethane tells an important story: how innovation, unintended consequences, and persistent curiosity drive both industry and science toward safer, smarter solutions.
What is 1,1-Dichloroethane used for?
A Closer Look at This Industrial Chemical
1,1-Dichloroethane doesn’t turn up in daily conversations, but it’s tucked into a surprising number of industrial practices. If you’ve ever been around a manufacturing plant or read a chemical inventory sheet, you’ll spot this compound close to solvents and intermediates. Factories often lean on 1,1-Dichloroethane for its properties as a solvent, especially in the production of other chemicals. It doesn’t take center stage like some bigger-name solvents, but its role behind the scenes carries weight.
Why Industry Reaches for 1,1-Dichloroethane
This chemical works as a solvent, meaning it can dissolve or extract certain substances. Formulators involved in the production of plastics, adhesives, and cleaning agents turn to 1,1-Dichloroethane for its ability to thin out or mix certain resins and waxes. The paint and coatings business also dips into this resource. I’ve seen manufacturers use it during degreasing steps, cleaning metal surfaces before they move down the line. Its volatility, while not quite like lighter solvents, lets it evaporate relatively fast after doing its job.
On top of that, 1,1-Dichloroethane steps in as an intermediate, or building block, when creating bigger, more complex chemicals. Producers chasing vinyl chloride—the stuff at the core of PVC pipes and flooring—often use this compound as part of the journey. Its presence cuts down several steps in the process, offering a shortcut in a world where time and cost both matter. Chemical plants have kept it in their playbooks for decades, as it’s reliable and available through a fairly inexpensive synthesis route.
Concerns and Community Conversations
Chemicals like 1,1-Dichloroethane shape more than just products, they influence the air in our neighborhoods and the water beneath our feet. During my visits to older industrial sites, I’ve learned that leaks or dumping from before tighter regulations left this compound in soil and groundwater. Research from the EPA and other public health groups points to health risks from breathing or drinking contaminated water—ranging from dizziness with short exposure to more severe impacts after years of contact.
Communities near manufacturers sometimes face lingering pollution tied to historic practices. Remediation is tricky because volatile organic compounds like this can wiggle through soil and spread quickly. The EPA’s Superfund program has flagged 1,1-Dichloroethane at certain cleanup sites, showing how mistakes with storage or disposal can echo for decades.
Rethinking Chemical Management
Using this chemical safely depends on containment, solid engineering, and effective disposal. Closed-loop systems that catch vapors or leaks before they reach people or groundwater work better than older, open-air methods. I’ve seen newer plants invest heavily in automated detection—that way, a minor leak triggers alarms before turning into environmental trouble.
Successful transitions away from riskier chemicals start with transparency. Companies now share more data with neighbors, listing what’s on site and explaining their plans for waste. Pushing for safer alternatives, investing in new technology, and building habits around regular checking go a long way. Ultimately, the story of 1,1-Dichloroethane highlights both the usefulness and the responsibility that comes with handling industrial chemicals. Learning from past mistakes, industries can keep workers safe and nearby families confident about the air and water they rely on.
What are the safety precautions when handling 1,1-Dichloroethane?
Understanding The Risks Up Front
1,1-Dichloroethane comes up a lot in labs, factories, and places that shape plastics or produce solvents. This liquid doesn’t look threatening—clear, with a slightly sweet smell. That smell tricks people into thinking things are fine, but the truth is, you don’t want to mess around with exposure. Inhaling vapors or letting the stuff touch your skin can set off headaches, dizziness, and breathing trouble. Some research ties chronic exposure to liver and kidney damage, as well as links to cancer. The National Institute for Occupational Safety and Health (NIOSH) and the American Conference of Governmental Industrial Hygienists pay close attention, spelling out strict limits for workplace air.
Personal Protective Equipment (PPE): The Real Line of Defense
Rubber gloves and goggles seem obvious, but folks often cut corners, especially if they’re in a rush or feel the job is routine. It takes one splash to remind you why you’re supposed to use PPE. Not all gloves block chemicals the same way; nitrile or butyl rubber gloves create a solid barrier. Cheap disposable ones actually soak up solvents over time, so quality counts. Splash-proof goggles or, better yet, a face shield, block vapors and stray drops. If you’re pouring or transferring the liquid, a full lab coat, long pants, and closed footwear offer key backup.
Ventilation: Fresh Air Saves Lives
A lot of people underestimate vapor buildup, especially if they work in older buildings. Small amounts of 1,1-Dichloroethane in the air will not just irritate your nose; over time, that stuff builds up in your body. Good ventilation matters more than most people realize. Fume hoods pull vapors away from your face before they settle in your lungs. At the very least, open windows and fans help, but nothing beats a proper exhaust system rated for volatile organic compounds (VOCs).
Handling and Storage: Keep Surprises to a Minimum
You never want to leave open bottles or spill-prone containers sitting out. I once saw a shelf collapse and send a couple of unlabeled bottles crashing down—no one knew which solvent they had to mop up, and the panic set in quick. So, label everything and store 1,1-Dichloroethane in tightly sealed containers made from thick glass or chemical-resistant plastic, away from heat or flame. The chemical has a pretty low flash point, and it will ignite near open flames, sparks, or static. Keep all sources of ignition out of the way, and never smoke near storage areas.
Spill Cleanup: Plan Ahead, Don’t Scramble
No one expects a spill, but panicking and grabbing a random towel won’t help. My old supervisor drilled us on always using spill containment pads. They soak up solvents fast and don’t react. If you get any on your skin, wash right away with lots of water. Ventilate the area, and if things get out of control, call for help and leave cleanup to professionals. It’s not just about preventing rashes; inhaling solvent fumes during cleanup can knock you out.
Waste Disposal: Follow Local Rules
Dumping 1,1-Dichloroethane down the drain or into any regular trash isn’t just illegal—you risk groundwater contamination, fires, and toxic exposure down the line. Designated disposal containers, marked “Halogenated Organic Waste,” make a real difference. Companies should hold regular training refreshers and have clear procedures for waste transfer and removal.
Making Safety a Habit
People in the field know that quick shortcuts come back to bite. Taking time to train, using proper gear, and setting up safety routines build habits that protect everyone. Safety with chemicals like 1,1-Dichloroethane isn’t about paranoia—it’s about respect. Respect for your health, coworkers, and community. Even if you aren’t handling this compound every day, all it takes is one mistake.
Is 1,1-Dichloroethane hazardous to health or the environment?
Behind the Chemistry: What’s at Stake?
People don’t commonly recognize 1,1-Dichloroethane outside of science labs and industry supply rooms, but this clear liquid pops up in factories, adhesives, and cleaning products. Smell it, and you’ll notice a sweet chemical edge. Work around it, and you quickly realize it’s not something to take lightly. I once spent a summer in a plant that used chlorinated solvents, and the rules around handling 1,1-Dichloroethane stuck with me. Respirators, gloves, and strict ventilation weren’t overkill—they kept us safer.
Risks and Health Concerns
Breathing in the vapors can irritate eyes and skin fast. You might feel dizzy or queasy after exposure. Extended contact brings serious risks. Studies on lab animals have pointed to liver and kidney problems after long-term exposure; the United States Environmental Protection Agency (EPA) lists it as a possible human carcinogen. Occupational Safety and Health Administration (OSHA) places strict limits on airborne concentrations, acknowledging that chronic exposure stacks up and causes harm.
Drinking water with 1,1-Dichloroethane above safety standards adds another worry. It seeps into groundwater at old manufacturing sites. The EPA tracks contamination closely—some communities have had to shut down wells and spend big on filtration. People living near these locations get worried, sometimes angry, when they see “volatile organic compound” showing up in water reports. Trust can be fragile.
Environmental Impact
Spilled or dumped, 1,1-Dichloroethane does not stick around in soil like some chemicals, but it travels far in groundwater. Once there, it spreads, polluting aquifers beyond the original spill site. Wildlife that drinks contaminated water faces health risks too. Large spills turn small mistakes into long-term problems. Local governments often need to act quickly to protect drinking supplies, and cleanup costs climb fast.
Air pollution comes into play, too. Factories releasing vapors can affect neighborhoods downwind. Inhalation isn’t the only problem—chemical breakdown products, like vinyl chloride, add to the trouble. Vinyl chloride holds a place on the list of known human carcinogens, making every release a little more dangerous over time.
Regulation and Solutions
Government standards limit how much 1,1-Dichloroethane workers and communities face each day. Manufacturers can switch to alternative solvents, but some industries still use this chemical for specialized tasks, especially where old processes linger. Modern factories set up closed systems and scrubbers to keep vapor levels down. Many regions require groundwater monitoring where past use created contamination. It’s possible to trap and destroy vapors using carbon filters or incinerators before release.
Public health depends on regular testing and quick communication. If water tests detect anything over EPA’s maximum contaminant level, communities need answers right away. After seeing the unease in people’s faces during town hall meetings, I know how important it is for companies and regulators to admit problems openly and address the risks with real solutions, not empty promises.
Education, stricter controls, safer alternatives, and persistent cleanup—these are the tools that keep both workers and the environment safer from the dangers 1,1-Dichloroethane brings to the table.
What are the storage guidelines for 1,1-Dichloroethane?
Why 1,1-Dichloroethane Demands Respect in Storage
Storing chemicals has always called for a cautious approach, and 1,1-dichloroethane isn’t any different. This colorless, sweet-smelling liquid shows up in various labs and manufacturing spots, but as anyone who’s spent time handling solvents knows, overlooking safety leaves you open to headaches — sometimes literally. The big concern with 1,1-dichloroethane boils down to its flammability and health risks. It evaporates quickly, giving off vapors that not only catch fire but can also cause dizziness and long-term harm with repeated exposure.
Shelving Choices and Storage Locations Matter
On the job, a few things always seem to separate a well-run chemical storage area from a disaster waiting to happen. You want a spot that stays cool and dry, away from direct sunlight. Temperatures staying below 30°C help dodge pressure build-up inside containers, which cuts down on leaks and accidental releases. One workplace I know kept their chemical cabinets outdoors. Even with ventilation, hot days pushed indoor temperatures up, making it riskier than folks realized. Moving the storage indoors, out of sunlight and away from exterior walls, lowered the chance of these problems.
Matching Container Types with Chemical Properties
Not every storage container holds up to strong solvents like 1,1-dichloroethane. Glass or high-density polyethylene containers, with tight-fitting caps, keep leaks at bay and stop the liquid from eating through the container walls. Metal cans aren’t smart here — this solvent can corrode certain metals over time. Labeling isn’t just for paperwork, either; in a rush, clear, bold labels save time and prevent confusion, especially for those who don’t handle these chemicals every day.
Separating Incompatibles and Avoiding Accidents
One thing that has stuck with me: storing 1,1-dichloroethane with acids or oxidizers is asking for trouble. I once saw a shelf collapse from overcrowding, sending incompatible chemicals spilling onto each other. The cleanup was tedious, and those vapors really are as harsh as they sound. Keep groups of chemicals apart using different cabinets, and never stack containers higher than is safe. An accidental bump or quake could lead to a far messier — and more hazardous — situation.
Ventilation Prevents Buildup and Keeps Workers Safe
Stuffy rooms and strong solvents don’t mix. Fume hoods or rooms dedicated to chemical storage with proper ventilation protect both workers and the building itself. I have worked with setups where inadequate airflow left the whole room smelling of chemicals, causing anyone who stayed too long to complain of headaches. Good fans and exhaust systems clear the air and make sure small leaks don’t build up to dangerous levels.
Support for Safe Storage
Many people look up regulations, but personal experience counts, too. Consult OSHA, NIOSH, or your country’s chemical safety boards; they lay out guidelines for storing and handling 1,1-dichloroethane based on real case studies and incident reports. Staff training pays off, not just for routine handling, but when an emergency hits, no one wastes time figuring out what to do.
Final Tips
Never ignore the small things: keep spill kits nearby, fix worn-out seals, and update storage logs regularly. Over the years, attention to these details has prevented more than one close call. Good storage isn’t about paranoia, but about respect — for the chemicals, your coworkers, and your own health.
How should 1,1-Dichloroethane be disposed of properly?
A Chemical with Consequences
Many people know 1,1-Dichloroethane gets used in industry as a solvent or in chemical manufacturing. Fewer realize how dangerous it can be in the wrong place or handled with little care. I remember working in a lab as a student, where even the smallest spill of something like this drew sharp words from supervisors. Nobody wanted to mess up. It quickly stuck with me how little separates a useful tool from a persistent environmental problem and a health risk.
This chemical evaporates easily and releases fumes that can damage the liver and nervous system. The EPA has flagged it as likely to cause cancer. If poured into the trash, drainage, or soil, 1,1-Dichloroethane doesn’t just disappear. It seeps into groundwater, it lingers in air, and it builds up where it shouldn’t, impacting communities long after the original user walks away. Not everyone lives near a chemical plant, but water travels, and what seems far from home eventually circles back.
Practical, Safe Disposal Steps
People working with 1,1-Dichloroethane need more than a handbook — they benefit from tough routines that treat every drop as a potential hazard. Closed containers made from compatible material keep fumes in and accidental leaks out. Storing those containers in ventilated, labeled areas sets a clear line: this stuff gets special treatment, every time.
Disposal shouldn’t end with a label or a locked cabinet. Specialized hazardous waste companies pick up containers and follow strict incineration processes that destroy the chemical at high temperatures. These professionals train for unexpected leaks, fires, or adverse reactions that no untrained worker should try to solve in a pinch. From my days in research, I saw what happened when people tried shortcuts — fumes triggered alarms, and everyone learned to trust the systematic approach.
Transport also matters. Laws in the U.S. and many other countries limit who can carry dangerous chemicals like this. Unmarked vans or casual trips in a trunk open up more than one line of risk — fines for companies, risk to the driver, threat to anyone in the area if there’s a collision or spill.
Why Ordinary People Should Care
Most people think only labs or plants deal with such chemicals. But 1,1-Dichloroethane can show up in pressure-sensitive tape, some old solvents, even certain types of paint removers. Household hazardous waste programs in many cities now accept small amounts, often for free. Dropping off old solvents during a community hazardous waste collection event helps keep them far from landfill leaks and accidental burns in backyard trash barrels. A friend once ignored this step, tossed a half-empty can in the garbage, and weeks later felt real regret when the neighborhood learned a landfill fire was traced to volatile chemicals.
Safe disposal depends on knowing what’s inside containers, reading labels, and calling waste collection services if uncertain. One phone call to the right agency or professional turns a risk into a routine safety step. The small effort beats cleanup disasters every time.
Setting Stronger Systems in Motion
Companies with ongoing use of 1,1-Dichloroethane face the biggest duty. Regular audits, mandatory staff training, and real penalties for shortcuts push compliance beyond minimum checklists. Incentives for reducing use, switching to safer alternatives, or investing in recycling programs can drive big change. Regulatory frameworks grow tighter for a reason. Over decades, ignoring chemicals like this already cost public health, trust, and tax money to clean up.
People sometimes ignore chemical risks until the damage spreads. Growing up in a river town downstream from factories, I learned that what’s poured out somewhere rarely stays there. Every person who handles 1,1-Dichloroethane has the power to make sure it gets locked up, shipped out, and destroyed safely — keeping it off the land and out of water. That’s worth the extra effort.
| Names | |
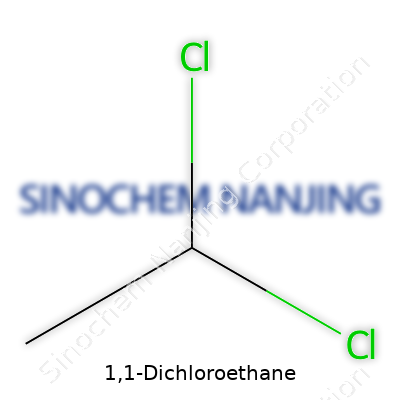
| Preferred IUPAC name | 1,1-Dichloroethane |
| Other names |
Ethylidene chloride 1,1-DCA Alpha,alpha-dichloroethane |
| Pronunciation | /ˌwʌn.wʌn.daɪˌklɔːr.oʊˈɛθ.eɪn/ |
| Identifiers | |
| CAS Number | 75-34-3 |
| 3D model (JSmol) | `3DModel: JSmol='ClCCCl'` |
| Beilstein Reference | 1200072 |
| ChEBI | CHEBI:35921 |
| ChEMBL | CHEMBL39119 |
| ChemSpider | 6197 |
| DrugBank | DB02041 |
| ECHA InfoCard | 03c9d5a4-6abe-4ffe-b014-7314c3f327b8 |
| EC Number | 602-020-00-8 |
| Gmelin Reference | Gmelin Reference: 8277 |
| KEGG | C01743 |
| MeSH | D000919 |
| PubChem CID | 13949 |
| RTECS number | KI0525000 |
| UNII | 7YD5F20CIR |
| UN number | UN 2362 |
| Properties | |
| Chemical formula | C2H4Cl2 |
| Molar mass | 98.96 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet odor |
| Density | 1.17 g/mL at 25 °C (lit.) |
| Solubility in water | 5.5 g/L (20 °C) |
| log P | 1.48 |
| Vapor pressure | 64.4 mmHg (20°C) |
| Acidity (pKa) | pKa ≈ 19.2 |
| Basicity (pKb) | 12.33 |
| Magnetic susceptibility (χ) | -62.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.4449 |
| Viscosity | 0.44 cP (20°C) |
| Dipole moment | 1.91 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 208.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -181.7 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -571.7 kJ/mol |
| Pharmacology | |
| ATC code | D6AX12 |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H225, H319, H336, H351 |
| Precautionary statements | P210, P261, P280, P304+P340, P305+P351+P338, P312, P337+P313, P403+P233 |
| NFPA 704 (fire diamond) | 1-2-0 |
| Flash point | 7 °C (closed cup) |
| Autoignition temperature | 413 °C |
| Explosive limits | Explosive limits: 5.6–14% |
| Lethal dose or concentration | LD50 oral rat 970 mg/kg |
| LD50 (median dose) | LD50 (median dose): 850 mg/kg (rat, oral) |
| NIOSH | NIOSH: *JH9285000* |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of 1,1-Dichloroethane: 100 ppm (350 mg/m³) |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 1000 ppm |
| Related compounds | |
| Related compounds |
Ethylidene dichloride Ethylene dichloride Chloroethane 1,2-Dichloroethane 1,1,1-Trichloroethane |

