1,1,2-Trichloroethane: Looking Beyond the Formula
Tracing the Path: Historical Insights
Chemistry's shelf holds a lot of stories, and 1,1,2-Trichloroethane is no exception. Its timeline stretches from curiosity in chemical labs during the mid-19th century through industrial-scale production as industry boomed. The need for safe, stable solvents caught the eye of manufacturers and those interested in making things run smoother, cleaner, and faster. Chemists back then looked beyond basic organics and saw chlorinated hydrocarbons like this one unlocking whole new chapters for paints, coatings, and cleaning. Over time, stricter environmental standards and more awareness about toxicity trimmed its use. Yet, it remains relevant in niche sectors, and understanding its road from simple curiosity to careful regulation says a lot about how chemical science adapts.
The Nuts and Bolts: Product Overview
Examining a transparent, colorless liquid usually feels unspectacular, yet 1,1,2-Trichloroethane behaves in ways demanding respect. Most people never see the stuff outside specialized labs or industrial setups. Its sweetish smell hints at the family resemblance to other volatile organochlorines. Originally, folks used it as a solvent for fats, waxes, resins, and oils. Its compatibility with several organic molecules made it appealing for paint removal and degreasing heavy machinery. That seems like small change until one realizes solvents like this helped shape entire industries before environmental scrutiny forced people to reconsider their toolkits.
Peering Closer: Physical & Chemical Properties
Physically, 1,1,2-Trichloroethane stands out because it evaporates quickly, sinking heavier than air due to a density greater than water. It boils at a moderate 113 degrees Celsius and stays liquid at standard temperature. Chemically, it's stable enough to bottle and move by train tankers, but breaks down under heat or bright sunlight, giving off harsh products such as hydrogen chloride and phosgene. Its molecular structure—three chlorines hanging off a two-carbon chain—shapes how it reacts and why its handling challenges even seasoned operators. Mix it with water, and you’ll see barely any solubility, but blend it with most organics and it dissolves right in, opening doors for industrial processes wanting a powerful yet not overly aggressive solvent.
Labeling and Rules: Technical Specifications
It’s hard to ignore thick technical labels once you've seen them on drums of volatile stock. A proper label would carry its CAS number 79-00-5, hazard codes, and detailed handling precautions. Governing bodies established threshold limits due to acute toxicity and environmental risk. The label tells a trained hand what equipment, storage, containment, and emergency kits they might lean on. The SDS becomes a reference point for those needing the smallest details: purity, impurities like 1,2-dichloroethane or trichloroethylene, and pressure compatibility. Few overlook the fact that accidental release needs rapid and effective intervention.
Making the Compound: Preparation of 1,1,2-Trichloroethane
Production usually means chlorinating ethylene or ethylene dichloride. Essentially, chlorine gas performs the heavy lifting, adding across existing carbon bonds. The industry prefers batch reactors—large, tightly controlled chambers where conditions like temperature and catalyst levels push selectivity for the 1,1,2-isomer. This chemical recipe makes it easy to see why safety protocols get serious attention: generation of unwanted byproducts, risk of leaks, and the need for advanced purification often push up costs and complexity. Deliberate engineering, not just textbook chemistry, pulls this compound from raw materials to a usable form.
What It Does: Chemical Reactions & Modifications
Not many chemicals behave quite like 1,1,2-Trichloroethane under a range of conditions. Under strong heat or ultraviolet, it slips into dehydrochlorination, giving vinylidene chloride or triggering degradation to less pleasant by-products. Mix it with alkalis and one gets a different set of results, sometimes leading to the formation of more complex chlorinated products. In the hands of a skilled organic chemist, its utility as a precursor reveals its deeper value—yielding intermediates for pharmaceuticals and specialty polymers. Alterations to the molecule open the door to derivatives useful upstream of bulk plastics, adhesives, or even as intermediates for insecticides, though the environmental burden always circles back into the conversation.
Many Aliases: Synonyms & Product Names
Chemical naming never stays simple. Lab records from different parts of the world might call it Ethene, 1,1,2-trichloro-, or sometimes Vinyltrichloroethane. Trichloroethylene’s similarities in name and use once created market confusion, making it clear why standardized identifiers like the CAS registry matter. ‘Dichloroethyl chloride’ or just ‘Trichloroethane’ show up in old safety manuals. This patchwork of names comes from decades when chemical catalogues expanded faster than international standards kept up.
Safe Handling: Safety & Operational Standards
The list of safety requirements gets long. Wearing chemical-resistant gloves and well-fitted goggles isn’t optional in any operation using 1,1,2-Trichloroethane. Proper ventilation keeps exposure below recommended limits, and even the cleaners handling rags or machine parts after use must follow the same strict rules. Spills call for absorbent compounds and remote ventilation to limit vapor build-up. Fire presents another threat, as heat and open flames don’t mix well with volatile organochlorines. Companies invest in detection systems and health monitoring for workers, responding to a track record of adverse health effects from chronic, even low-level, exposure. Simple mistakes often lead to costly incidents, underlining the need for ongoing training.
Where It Works: Application Area
For decades, its most important role lay in industrial degreasing and cleaning metal parts. Electronic manufacturing appreciated its ability to lift out tough residues without corroding circuit boards. Chemical synthesis application drew from its power to dissolve certain organic building blocks otherwise unwieldy to work with. Its presence has faded from mainstream use in household products due to toxicology concerns, but controlled, specialized applications, backed by strong engineering controls, still find value. Paint and coating removers, lab-scale organic synthesis, and some intermediate manufacturing hold onto its relevance, but regulatory pressure continues to shrink its role.
Looking Ahead: Research & Development
Research into alternatives, safer working environments, and cleaner breakdown products keeps chemists busy. Academic labs and industrial R&D teams regularly publish new catalysts and process tweaks aiming to reduce byproduct formation and environmental impact. Sensing the next big regulatory crackdown, big departments channel funding into greener or less persistent solvent replacements. Study of reaction pathways using advanced computational chemistry tightens predictions about degradation and fate outside industrial settings. On the frontlines, scientists who once relied on 1,1,2-Trichloroethane now look to bio-based or less toxic options, but the old compound lingers in research, where its quirks and reactivity teach future generations chemical safety and clever problem-solving.
Opening the Black Box: Toxicity Research
Decades of work tell a clear story about risk. Short-term inhalation exposure brings headaches, dizziness, and nausea. Symptoms of chronic exposure stack up fast—liver and kidney injury, impacts on the central nervous system, and links to carcinogenicity. Rat studies from the 1970s through 1990s outlined clear threshold values, prompting health authorities to set conservative occupational exposure limits. Drinking water standards now set vanishingly low limits on any trichloroethane content. Environmental persistence raises long-term soil and groundwater questions, sending monitoring teams out to old industrial sites to check for contamination. These efforts underscore the price paid during periods of lax regulation, sharpening the case for both guidance and enforcement moving forward.
Not Just a Relic: Future Prospects
Even with its declining profile, 1,1,2-Trichloroethane gives us a reason to study past practice and improve future chemistry. Industry’s gradual pivot away from high-hazard solvents comes from health, regulation, and plain economic self-interest. Projects developing closed-loop solvent systems or new techniques for solvent recovery and neutralization shape tomorrow’s processes. Education efforts aimed at rising generations of engineers and chemists now feature chemicals like this as cautionary tales, not standard tools. Lessons learned from its lifecycle encourage smarter design and much tighter controls for materials with similar profiles. Technology for real-time vapor detection and remediation now plays a bigger role than ever. Future research will probably zero in on safer substitutes, advanced containment, and continuous reduction of legacy contamination. Chemical stewardship, not broad application, becomes the lasting legacy of 1,1,2-Trichloroethane.
What is 1,1,2-Trichloroethane used for?
Where You’ll Find 1,1,2-Trichloroethane in Industry
Ask anyone who’s worked in an old chemical plant about 1,1,2-Trichloroethane, and you'll hear stories about how it shows up everywhere solvents are king. This clear liquid finds its way into processes that shape the backbone of manufacturing, especially where heavy-duty degreasing and cleaning have been the priority for decades. Its reputation stems from strong solvent power—it can dissolve greases, oils, waxes, and even the sticky residues you find in engine parts or metal machining shops.
Chlorinated solvents like this one get handed a bigger responsibility beyond scrubbing. You might spot it as a building block in the making of other chemicals. Most notably, 1,1,2-Trichloroethane sits at the start of the line for producing vinylidene chloride. That’s the ingredient that, after a few more steps in the plants, ends up as saran wrap or in certain barrier coatings protecting food or keeping fuel tanks from leaking vapors.
Practical Uses and the Hidden Downside
It’s easy to brush off industrial chemicals as distant concerns, but that changes when you learn about the health and environmental baggage that 1,1,2-Trichloroethane drags along. After years of handling barrels and drums in manufacturing, I came to respect the warnings: inhaling the vapors or letting it touch your skin for too long leads to headaches, dizziness, even liver and kidney problems. Companies that use the chemical have strict rules—ventilation, gloves, goggles, and all the safety gear—because safety lapses stack up quickly.
While nobody wants grease on engine parts, there’s an undeniable mess left behind in water and soil near old sites. The Environmental Protection Agency lists it as a hazardous air pollutant and tracks spills closely. It doesn’t break down quickly, so once it seeps into groundwater, cleanup gets complicated. I’ve come across folks living near contaminated sites who worry about what’s in their wells. Their concerns aren’t abstract—science links it to cancers in lab animals, and evidence in people is alarming enough that regulators stay on alert.
Keeping Risks in Check
Factories that count on 1,1,2-Trichloroethane face tough choices. Old habits linger, and, for a long time, switching to safer alternatives meant retooling machines, retraining workers, and risking the bottom line. These days, there’s mounting pressure both from inside the industry and from watchdogs. Companies minimizing releases get credit for using closed-loop systems—so the chemical doesn’t escape into the air or pour down drains. New solvents that break down more easily or don’t stick around in water offer a way forward, and some shops have made the leap.
For home projects and everyday consumer use, the safer bet is to avoid suspect products old enough to contain this solvent. Labels rarely give the details most people want, so trusted brands and new formulations tend to be a better pick. Strong enforcement helps—we rely on environmental agencies to monitor sites, step in fast during spills, and hold polluters to account. I’ve seen how community voices make a difference when folks band together to demand clean-ups or block dangerous shipments through their neighborhoods.
Living With the Consequences
It boils down to responsibility. The engineering behind industrial solvents like 1,1,2-Trichloroethane moved us forward, but looking back, the cost gets clearer. Keeping the balance between progress and safety requires intervention from industry, oversight from government, and, most of all, honest reporting and real-world health monitoring. As a worker and as a neighbor to one of these legacy sites, I believe in finding safer practices and cleaning up what’s been left behind.
Is 1,1,2-Trichloroethane hazardous to health?
Recognizing the Problem
People rarely talk about chemicals like 1,1,2-Trichloroethane at the dinner table, but maybe they should. Used in industry as a solvent and for making other chemicals, this colorless liquid causes much more than just a bad smell. The health concerns behind exposure don’t just exist on paper. Real folks, whether working in manufacturing or living near old dump sites, have dealt with headaches, drowsiness, and trouble breathing after inhaling its vapors. The U.S. Environmental Protection Agency (EPA) and World Health Organization have flagged it for good reason. Its reputation for nerve, liver, and kidney damage stems from both flooding case reports and decades of animal tests.
How Exposure Happens
I grew up near old chemical plants, so stories of “bad water” showed up in family conversations whenever the smell of chlorine drifted past our porches. If someone’s kids got sick or people muttered about “the well,” suspicion often fell on chemicals leaching out from old drums or pipes. That’s still a real problem. Whether by breathing air at work or drinking contaminated groundwater, exposure isn’t far-fetched. The chemical has a sneaky way of sticking around in soil and water, and industries often struggle to stop leaks from reaching the environment.
What Science Says
Researchers have no shortage of reasons to keep 1,1,2-Trichloroethane on their radar. Repeated animal testing links it to damage in the liver and kidneys. Workers handling the chemical show signs of dizziness, tremors, and even changes in how their muscle reflexes work. Studies pile up about cancer risks, too. The International Agency for Research on Cancer (IARC) classifies it as possibly carcinogenic to humans. Despite the massive word “possibly,” nobody wants to roll the dice with their health.
Turning Concern Into Action
Fact sheets from groups like the Centers for Disease Control and Prevention offer guidance, but these only go so far if safety takes a back seat on the shop floor or in management offices. At the most basic, regular air monitoring and better ventilation protect folks who need to work with this chemical. Industry shifts toward greener alternatives can shrink overall risk. For neighborhoods, real change comes with cleaning up contaminated soil and water—a tough job, but not impossible if regulators and communities push for it.
Everyday Solutions
Each time industry swaps out 1,1,2-Trichloroethane in manufacturing or chooses smarter storage to prevent leaks, that’s one step closer to safer workplaces and neighborhoods. The more companies take environmental responsibility seriously, the fewer toxic leftovers appear downstream. Regulators and local health departments play a big role by enforcing stricter limits and keeping a close watch on contamination. Families living near old disposal sites deserve more than just a warning sign—they need long-term monitoring and support, including testing and cleanup if pollution is found.
Why It Matters
Stories about industrial chemicals shouldn’t just be trivia for scientists. For anyone living, working, or raising kids near places where substances like 1,1,2-Trichloroethane are stored or used, this risk feels real and immediate. We all breathe the air, drink the water, and count on each other’s vigilance. Tackling the hazards calls for action—at home, in industry, and from leaders able to get regulations enforced. Because health should never play second fiddle to convenience or profit.
How should 1,1,2-Trichloroethane be stored?
Understanding the Risks
1,1,2-Trichloroethane lands on the hazardous chemicals list for good reason. It looks like a simple, clear liquid, but there’s danger beneath the surface. It can catch fire at higher temperatures and produces toxic fumes that threaten both lungs and health, especially in a closed setting without strong airflow. Many stories in chemical manufacturing and lab work revolve around fumes that drift where people least expect, causing dizziness, headaches, and worse if exposure drags on. It thrives in places with little respect for the rules.
Why Proper Containers Build the First Line of Defense
A jug or drum with cracks or a loose lid invites trouble. Vapor sneaks out, and moisture or stray heat slips in, changing how the chemical acts or making it less stable. I remember walking through old storage units and seeing makeshift containers—with handwritten tape holding caps down—leaking sticky residue. That sends chills up anyone’s spine who has read accident reports. Sturdy, corrosion-resistant drums or tightly sealed glass bottles, clearly labelled, work best. The labels matter: when someone else walks in and sees the chemical’s name and skull-and-crossbones, they pause and think twice.
Temperature and Light: The Silent Aggravators
Heat and sunlight stir up chemicals in ways we don’t always see. Store the liquid near a furnace, and the heat raises pressure inside the canister, risking a bulge, crack, or even a ruptured lid. Sunlight sometimes softens plastics, turns liquids yellow, or lets reactions spark. Trichloroethane prefers cool, dark spaces. Anything above ordinary room temperature ramps up volatility. So, basements or insulated sheds with a steady environment get the job done. That’s the difference between a stable bottle and an unexpected spill or toxic cloud.
Keep It Dry, Keep It Safe
Water and corrosive vapors bring a slow, creeping threat. They break down containers made from the wrong materials and interact with the liquid inside, making it unpredictable. I’ve seen supply closets where someone left water pipes dripping over storage shelves—ruining containers and leaving everything sticky and suspect over time. Shelves kept dry and well off the ground hold up best. A spill tray underneath, ready to catch stray drops, can stop small accidents from turning massive.
Separation From Incompatibles
Trichloroethane doesn’t play nice with reactive chemicals. Acids, alkalis, and oxidizers set off reactions you don’t want near people or equipment. Old habits sometimes put everything on one shelf, but years in safety training taught me that dedicated cabinets for hazardous solvents make a real difference. By keeping trichloroethane far from anything energetic or unstable, risk falls dramatically.
Simple Steps Build Strong Habits
Most problems come from shortcuts—leaving lids loose, ignoring labels, or stacking heavy drums up high where they might tumble. Routine checks on the shelf and the containers, along with regular ventilation to sweep out stray vapors, help everyone breathe easier. Spills call for a quick response: absorbent pads, gloves, and a scoop for any broken glass. If you stick with strict inventory records, you catch containers before they degrade. It’s these small, almost boring routines that build up true safety.
Training and Transparency Save Lives
Each worker must know what’s in the bottle, what it looks like, and what to do if something breaks open. Clear instructions on the wall and hands-on drills—rolling up sleeves and practicing—build muscle memory. No one wants to be caught fumbling in an emergency. Armed with honest, current information, teams stay safer together.
What are the safety precautions when handling 1,1,2-Trichloroethane?
Why Respecting This Chemical Matters
The first time I came across 1,1,2-Trichloroethane in an industrial setting, I saw seasoned workers act with an extra level of caution. This liquid isn’t the sort you can bluff your way around. Highly volatile, harmful to the body, and not a substance you can simply wash off in case of contact. Lax practices in the lab or on the factory floor have led to hospital trips and environmental scares before. From headaches and nausea to liver damage and even cancer risks, getting careless with this chemical means signing up for unnecessary trouble.
Personal Protective Equipment—Non-Negotiable
Most real-world accidents trace back to either skimping on protective gear or not using it right. A reliable pair of chemical-resistant gloves gives a layer of defense when pouring or transferring this liquid. Splash goggles guard your eyes from sudden sprays or vapors. Laboratory coats stop spills from soaking into skin or regular clothes. Respirators cut the risk of inhaling fumes, which build up quickly in closed rooms.
Ventilation: Keep the Air Moving
Every chemical storage room or process involving 1,1,2-Trichloroethane should have ample airflow. I once worked in a space where vents ran all day, pulling fumes out and bringing fresh air in. Even with fans on full blast, monitoring air quality levels using sensors or badges added peace of mind. Without moving air, vapors hang around, and exposure goes up before anyone realizes it.
Storage Practices That Actually Work
Store this compound in tightly sealed containers, away from heat or sparks. Lock everything up in a flame-resistant cabinet. Storing near acids, bases, or oxidizers only brings drama if leaks or spills happen. Labels need to be fresh and clear so there’s no mistaking what’s inside. I’ve seen people try to reuse containers meant for other chemicals—that shortcut never ends well, usually causing confusion or dangerous reactions down the line.
Spill Response Calls for Quick Action
A spill shouldn’t spiral into panic, but speed and order matter. Any surface that’s been hit calls for absorbent pads or proper neutralizing powder, not rags or paper towels. Discarded matter goes straight into hazardous waste bags. Afterward, hands, benches, and tools get washed down with fresh water. I always encourage colleagues to know their spill kit’s location and ingredients ahead of time.
Medical and Environmental Realities
Exposure symptoms range from dizziness to chemical burns, and the longer someone waits before washing off or seeking fresh air, the worse it gets. Medical attention isn’t optional if someone swallows or inhales a large dose. On top of personal risk, spills seep into drains and soil, polluting ground and water supplies—another reason to keep materials out of regular trash or sinks.
Learning From Hard-Won Experience
Training sessions help everyone remember best practices, but real vigilance comes from watching what happens when things go wrong. In my years in the field, sharing those “close call” stories stuck better than any handbook. Supervisors have a duty to lead by example, modeling safety—even on busy days—so new folks pick up the right habits from the start.
Steps That Make a Difference
It’s easy to think accidents only happen to someone else, or that skipping a step now and then won’t matter. My time with 1,1,2-Trichloroethane convinced me that taking shortcuts invites problems you never saw coming. Sticking to proven habits—protective gear, ventilation, proper storage, spill controls—keeps people healthy and workplaces open. Big results come from steady, careful steps every single day.
What are the environmental risks of 1,1,2-Trichloroethane?
Understanding a Persistent Pollutant
Most people probably haven’t heard of 1,1,2-Trichloroethane. It’s a chemical many factories rely on to make vinylidene chloride, adhesives, and various solvents. In real life, once this compound escapes into air, soil, or water, it tends to linger. Decades ago, chemical spills or leaky storage tanks sent it underground. Today, anyone living near a factory, waste site, or even old junkyards could be exposed.
A Closer Look at Water Contamination
Whenever I visit a rural community, I notice folks rely on groundwater for drinking and cooking. The trouble is, this chemical slides through soil and drifts into wells. Unlike many other pollutants, it doesn’t break down easily. After years underground, it surfaces in tap water. According to research from the U.S. Environmental Protection Agency, 1,1,2-Trichloroethane remains a top concern for groundwater in some industrial regions. People don’t have to taste chemicals in their water to get sick; the health risks run quiet but deep, affecting the liver, kidneys, and possibly the nervous system.
Damage to Wildlife
Fish and frogs breathe through skin or gills, which means they absorb chemicals from water faster than humans do. In streams and wetlands near industrial zones, this solvent disrupts the food chain. Birds and mammals, especially those living off contaminated fish, build up toxins in their bodies. Some studies from the Agency for Toxic Substances and Disease Registry have shown how this chemical stunts growth and reproduction in small animals, tipping the scale for entire ecosystems.
Threat to the Air We Breathe
Factories vent 1,1,2-Trichloroethane into the air to get rid of waste gases. I grew up near a cleaning products plant—on foggy mornings, an odd chemical smell drifted over the fields. Over time, these vapors can travel far from their source. The National Institutes of Health has flagged evaporation as a key way this pollutant escapes into the neighborhood, contaminating more places than folks realize. Breathing in low levels can bring headaches, nausea, and longer-term problems, like liver damage.
Cleaning Up the Mess
Turning off the tap isn’t always possible. Once this solvent soaks into groundwater, the fix takes muscle and planning. Engineers sometimes pump polluted water to the surface, filter it, and inject cleaner water back into the soil. That approach costs millions, and tax dollars often fund cleanup projects nobody notices. Some scientists are working on bioremediation—using bacteria to eat away at the pollutant. Efforts like these hold promise, but they need strong oversight and enough funding to work.
A Path Forward
Stronger rules for chemical storage and waste disposal can limit spill risks. Factories could use safer substitutes or better containment tech, but many only change under real pressure—from lawsuits, community advocates, and watchdog groups. Local governments and neighbors play a part, too. They can request water testing and demand transparency from manufacturers. Real progress means accountability, not quick fixes.
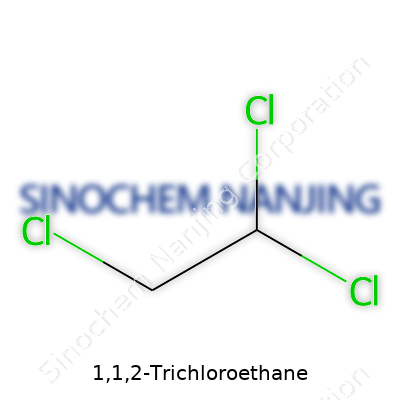
| Names | |
| Preferred IUPAC name | 1,1,2-Trichloroethane |
| Other names |
Ethane, 1,1,2-trichloro- 1,2,2-Trichloroethane Vinyl trichloride beta,beta,Dichloroethyl chloride |
| Pronunciation | /ˌwʌn wʌn tuː traɪˌklɔːroʊˈɛθeɪn/ |
| Identifiers | |
| CAS Number | 79-00-5 |
| 3D model (JSmol) | `/n8b5yoTePSKFGPVmDPi0AJXlOARxJmT6Qk9HgrEUMkVVUcESH9SSnF/AA==` |
| Beilstein Reference | 1206773 |
| ChEBI | CHEBI:35711 |
| ChEMBL | CHEMBL45058 |
| ChemSpider | 6823 |
| DrugBank | DB00244 |
| ECHA InfoCard | 100.003.255 |
| EC Number | 203-458-1 |
| Gmelin Reference | 8229 |
| KEGG | C14078 |
| MeSH | D014258 |
| PubChem CID | 6579 |
| RTECS number | KJ3900000 |
| UNII | 6F6RZH6K3W |
| UN number | UN2831 |
| CompTox Dashboard (EPA) | DTXSID2020955 |
| Properties | |
| Chemical formula | C2H3Cl3 |
| Molar mass | 133.40 g/mol |
| Appearance | Colorless liquid |
| Odor | Sweet chloroform-like odor |
| Density | 1.44 g/mL at 25 °C |
| Solubility in water | 5.97 g/L (20 °C) |
| log P | 2.41 |
| Vapor pressure | 23.8 mmHg (20°C) |
| Acidity (pKa) | ~15.1 |
| Basicity (pKb) | 7.0 |
| Magnetic susceptibility (χ) | -49.4·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.444 |
| Viscosity | 0.813 cP (20 °C) |
| Dipole moment | 2.04 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 206.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -211.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -712.1 kJ/mol |
| Pharmacology | |
| ATC code | JDXA |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H332, H335, H336, H351, H411 |
| Precautionary statements | P260, P261, P271, P280, P301+P310, P303+P361+P353, P304+P340, P305+P351+P338, P312, P321, P330, P337+P313, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-0 |
| Flash point | 54 °C (closed cup) |
| Autoignition temperature | 413°C |
| Explosive limits | 0.9–8% |
| Lethal dose or concentration | LD50 oral rat 1,870 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral (rat) 1230 mg/kg |
| NIOSH | TX9275000 |
| PEL (Permissible) | 10 ppm |
| REL (Recommended) | 10 ppm |
| IDLH (Immediate danger) | 300 ppm |
| Related compounds | |
| Related compounds |
Chloroethane 1,1-Dichloroethane 1,2-Dichloroethane Ethylene dichloride |

