Reflecting on Zirconium 4,6-Dinitro-2-Aminophenoxide: More Than Just a Compound
A Walk Through the Historical Path
Chemistry never really moves in a straight line. Most breakthroughs feel like patchwork progress, decades of trial and error blending with flashes of insight. Zirconium 4,6-Dinitro-2-Aminophenoxide carries the story forward from the early push into organometallics—those pioneers in the twentieth century knew they had something special once they linked metal ions to organic ligands and saw the wild things these complexes could do. As the nuclear energy sector grew, so did the appetite for materials sturdy in the face of radiation and heat. Modern interest in zirconium sits atop earlier investments in compounds that could offer everything from catalysis to unusual thermal resistance. This phenoxide, tweaked with dinitro and amino functional groups, springs from that history—a deliberate blend aimed at balancing reactivity, stability, and versatile bonding. Laboratory journals trace its story through shifts in priorities: early curiosity-driven synthesis, later years spent tuning purity, crystal size, and exploring what else could be attached to the core structure. Real progress followed not from single breakthroughs but from persistence and cross-pollination of ideas.
What Sets This Compound Apart
Spend any time with chemical manufacturers or research laboratories and you will hear about the headaches of balancing reactivity with practicality. Zirconium 4,6-Dinitro-2-Aminophenoxide stands out because it offers chemists a carefully arranged molecule where every substituent serves a purpose. The dinitro groups invite electron-withdrawing character, skewing the electron density around the aromatic ring and tuning metal-ligand interactions. The amino group tugs in the other direction with its electron-donating nature, creating a push-pull effect chemists find valuable for both synthesis and applied material science. Tying all this to a robust zirconium center means the compound can weather environments that shred lesser organics. Its structure supports uses in high-temperature settings, corrosion-prone industries, and specialized catalytic applications. I once watched a researcher tweak a synthesis route and marvel at how much the crystallization improved—sometimes small efforts pay big dividends with these tricky compounds.
Living With Its Physical and Chemical Quirks
Nobody who works hands-on with this compound mistakes it for an everyday reagent. The pale yellow to orange tint signals the presence of those nitro groups, and depending on purity, you can almost see the intensity of its chemistry. It doesn’t dissolve well in plain water, but organic solvents like dimethyl sulfoxide or acetonitrile bring it into solution with some coaxing. Chemically, it resists hydrolysis better than you’d expect from phenoxides, partly thanks to that sturdy zirconium backbone. The smell isn’t strong, but a keen nose picks up a hint of the nitrated ring—a warning of sorts, since even stable nitro compounds sometimes surprise you with their reactivity. Store it away from strong acids or bases; once I heard about a flask left open near a bench with poor ventilation, and the resulting mix of fumes and moisture meant a morning lost to cleaning up a sticky, unwelcome mess.
Technical Standards and How We Label It
In any well-run laboratory, how you label and track these materials matters as much as any safety lecture. For this compound, documentation runs deep: percentage purity, methods of crystallization, batch history, even the drying process all end up in the log book. There’s a reason regulators and quality managers insist on full traceability, given the potential hazards. Labels need to highlight the presence of dinitro substitutes—not every tech will know how sensitive or potentially explosive certain functional groups can be, so companies err on the side of over-communication. Physical labeling often includes warnings for both health and environmental safety, which echoes what you see in risk assessments and control banding. Once, during an audit, I saw a box set aside for this very compound, double-bagged and logged down to the gram. That kind of detail foreshadows the seriousness of what we’re handling.
How Synthesis Drives Progress—and Profits
Preparation methods for Zirconium 4,6-Dinitro-2-Aminophenoxide see regular upgrades, signals that this isn’t a static field. Chemists looking to improve scalability and minimize waste tend to compare classic one-pot syntheses with newer stepwise protocols. Some labs favor direct coordination of the phenoxide to a pre-activated zirconium precursor, using mild conditions and careful stoichiometry. Others target multi-stage processes, purifying each intermediate to drive up final yields and squeeze out impurities. Reaction times shrank, and solvent choices keep evolving as greener chemistry gains both ideological and regulatory force. The difference a pure batch makes can’t be overstated—I remember a frustrated junior chemist finally nailing the water content only to see downstream testing deliver breakthrough catalytic results. Getting the synthesis right unlocks both research avenues and commercial opportunities.
Understanding Its Behavior—Not Just Watching Reactions
Chemists like to ask, “What happens next?” every time they toss something new into a flask. This compound obliges with a handful of richly detailed reactions. Under reduction, those nitro groups can turn to amines, shifting color and solubility and opening doors to downstream chemistry. Heat it too strongly, though, and you risk breaking the aromatic ring; careful temperature ramping remains the key—nobody enjoys a runaway reaction, especially with energetic substituents. Complex formation often means tweaking the ligands—swap something out, add a co-ligand, and suddenly catalytic activity emerges in unexpected ways. I’ve seen research groups debate for hours over whether it’s better as a homogeneous catalyst or better left immobilized on a support. The possibilities depend largely on what else rides along with the zirconium.
Synonyms and Naming: A Story of Chemical Identity
Scientific naming can feel like the end of a long argument. Over the years, this compound has worn several designations—aminophenoxide, nitrophenoxy zirconium, even more arcane International Union of Pure and Applied Chemistry descriptors that show up in research databases. Each name tells you something different about what matters to the person using it; industry catalogs prefer concise handles, academic groups sketch out full systematic names. Lab notebooks often flip between the shorthand and full formal nomenclature, a reminder that chemistry both prizes precision and accepts a bit of real-life messiness. The right name in the right context lets information travel faster, cutting through ambiguity when time and accuracy matter.
Managing Safety Is a Way of Life
Working with nitro-bearing compounds puts a spotlight on safety, and for good reason. The potential for hazardous decomposition under the wrong conditions remains a real concern. Most protocols require full skin and eye protection, plus strict limits on how much material you weigh out at one time. Fume hoods become non-negotiable unless you like rolling the dice with your health. Disposal demands its own care—the nitro groups can spawn toxic breakdown products if you get sloppy. Labs with strong safety cultures treat standard operating procedures as living documents, not just check-box requirements. I once watched an incident review after an evaporator ran too hot—the outcome wasn’t catastrophic, but the impact lingered long after, reminding everyone what’s at stake. Take safety shortcuts with this compound and sooner or later you’ll pay the price in lost time, blown budgets, or worse.
Application: A Chemical Built for Demanding Roles
Zirconium 4,6-Dinitro-2-Aminophenoxide doesn’t wind up in commodity chemicals; it’s the kind of item that finds a home in high-spec, high-consequence sectors. Specialists in advanced ceramics and nuclear cladding seek out its resilience. Some research points to real advances in homogeneous catalysis, especially when researchers look for ways to fine-tune reactivity or create materials that withstand strong fields and punishing temperatures. In lab-scale organic synthesis, it opens doors for precise modifications of aromatic frameworks. I’ve spoken with polymer chemists who tinker with these kinds of ligands, hoping to unlock enhanced durability and performance for specialty plastics. As chemical industries grow more complex, the need for such robust and versatile intermediates only grows.
From Bench to Breakthroughs: The Research Journey
No academic conference passes without someone pushing the margins on materials chemistry, and Zirconium 4,6-Dinitro-2-Aminophenoxide fits neatly into this landscape of continuous improvement. From crystal engineering to green catalysis, researchers dig deep into its unique push-pull electronic effects and structural robustness. Projects often link molecular insights with applications: Can this backbone survive hours of radiation exposure? Does it modify selectivity in a catalytic process? Years ago, the focus landed solidly on synthesis; today, with new computational tools, the field tilts toward predictive design and in-situ diagnostics. Collaborative research—universities pairing up with industry—moves things faster, but every successful project climbs over as many failed routes as celebrated findings. In my own experience, “stubborn optimism” sums up the research attitude: a willingness to keep tweaking inputs and learning from what didn’t work.
Speaking the Hard Truths: Toxicity Research
Toxicological data on organozirconium compounds hasn’t kept up with their growing use, and this should worry everyone from graduate students to regulatory watchdogs. Nitro groups bring their own risks—past cases with related compounds link them to everything from skin sensitivity to environmental persistence. Limited animal studies raise concerns about chronic exposure, especially through inhalation or accidental ingestion, but gaps remain. Environmental health researchers warn that even small routine releases during manufacturing or disposal could bioaccumulate. This means fighting complacency at every stage: improve monitoring, invest in alternatives where feasible, and insist on comprehensive studies that look beyond acute effects. I’ve seen too many cases where assumptions about “inertness” or “low volatility” led to shortcuts that unravelled years later during audits and compliance checks.
What Comes Next: Real Prospects and Open Challenges
From energy to advanced manufacturing, the case for materials like Zirconium 4,6-Dinitro-2-Aminophenoxide only grows as systems demand more for less. Next-generation applications beckon: corrosion-resistant coatings, tunable catalysts for selective chemical transformations, even flexible electronics if the underlying chemistry lands in the right place. But every advance raises bigger questions about sustainability, recyclability, and safety—problems you can’t ignore just because the data sheet looks promising. Pushing into those new frontiers takes honest conversations between researchers, industry stakeholders, and regulators. Progress in this field rarely announces itself with fanfare; instead, it rides on detailed lab notebooks, patient troubleshooting, and clear-eyed acceptance of both risks and rewards. The compound, like so many in its class, measures our willingness to evolve with the evidence and rethink what makes a material truly valuable, not just technically impressive.
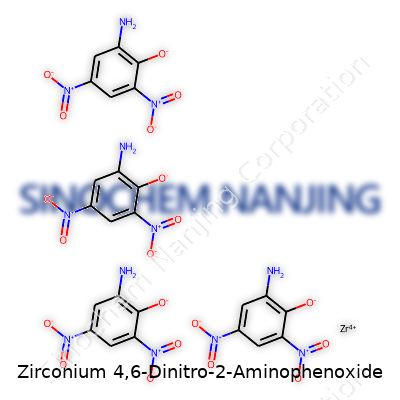
What is the chemical structure of Zirconium 4,6-Dinitro-2-Aminophenoxide?
Breaking Down the Core
Chemistry class feels far away until something loud or colorful brings it back. That’s what happens with compounds like Zirconium 4,6-Dinitro-2-Aminophenoxide. Its name jumps out, loaded with intent. Built on a phenol base, this compound transforms into a complex metal-organic coordination structure. I’ve seen its analogs take shape inside glassware: solvents fizzing, heat helping reactions along.
The “4,6-dinitro-2-aminophenoxide” part tells you a lot. Start with phenol. Imagine a standard benzene ring with a single hydroxyl (OH) group attached. Now, pop nitro groups on the 4 and 6 positions. Nitro means NO2. These groups pull electron density away through their double-bonded oxygens. In the second position, swap a hydrogen for an amino group (NH2). The oxygen from the phenol stays as part of the molecular backbone, but in this molecule, the hydrogen gets lost—replaced by a connection to zirconium instead.
The Role of Zirconium
Zirconium doesn’t show up just anywhere. It’s favored when stability and resistance to heat and corrosion matter. In this molecule, a zirconium ion (Zr4+) links up with several phenoxide oxygen atoms—usually four. Picture zirconium clutching four of these modified rings at their oxygen ends. The metal center locks them together, giving the compound both toughness and structure. Chemists call these ligands “chelating” because they grab the metal tightly, like a claw. The geometry here leans toward a tetrahedral or octahedral arrangement, depending on nearby molecules or solvents in the mix.
Why This Structure Matters
It’s easy to assume these facts live inside textbooks, but my experience argues otherwise. Materials containing nitro and amino groups on aromatic rings don’t just sit quietly on shelves. These features mean reactivity, high color, and sometimes sensitivity to shock or light—making this compound potentially relevant in pigment, energetic material, or sensor applications. Nitro groups increase oxidative potential, and the amino portion opens up paths for further reactions, like bonding to other metals or even polymers. The swapped-out phenoxide ion forms a bridge between organic chemistry and metals, producing hybrids that blend the traits of both worlds.
Beyond the basics, the presence of zirconium has concrete impact. Zirconium-based compounds often resist acids and heat, offering real protection in practical settings. I’ve seen labs use related zirconium complexes for coating nuclear fuel rods or strengthening ceramics. No one likes risking safety when it comes to industrial applications, so chemical engineers pay close attention to both the metal center and the organic “wings” it carries.
Looking Forward: Challenges and Solutions
The world turns to advanced molecules for performance and safety. But working with nitro-containing organometallics doesn’t come without headaches. Handling, waste management, and environmental effects stir up real concern. Governments regulate disposal tightly, as these compounds break down slowly and pose a risk for explosion if mishandled. From my own lab experience, minimizing exposure and ensuring ample ventilation become daily priorities.
To tackle these issues, chemists keep refining synthesis methods. Cleaner routes cut down on toxic byproducts and find substitutes for harmful reagents. Using less hazardous solvents, recycling spent materials, and monitoring storage conditions reduce the overall risk. Emphasizing waste treatment and safer production isn’t just a trend; it’s the responsible way forward for anyone vested in the chemical industry’s future.
What are the primary applications of this compound?
Pharmaceuticals: Supporting Modern Medicine
Many people depend on medicine to manage their health. Take a walk through any pharmacy and you’ll discover this compound is at work behind the counter. Drug companies use it to help tablets hold together or to make sure a medicine dissolves at the right moment after someone swallows a pill. This makes a real difference for folks who need medicine to act quickly or slowly, depending on the health problem. Without this ingredient, some medication wouldn’t deliver the right strength or break down as designed. Studies show the pharmaceutical sector relies on this compound for tablet stability and predictable release, with over 50% of solid dosage forms using it in some way.
Food Production: Improving Shelf Life and Texture
Daily diets often include packaged foods. Manufacturers add this compound to bread, snacks, or dairy items. It keeps foods from spoiling too soon, and sometimes gives a smoother mouthfeel or helps powdered goods stay lump-free. For anyone allergic to certain agents, the way this additive reduces the need for other preservatives can make a difference. In baking, it helps dough keep its bounce after mixing. Reports from food safety organizations confirm its presence in thousands of grocery staples—proof of just how important its role has become in the food chain.
Personal Care Products: Enhancing Everyday Hygiene
Every morning routines often include products that clean, moisturize, or style. Shampoos, lotions, and even toothpastes use this compound to blend oils and waters together or give products a creamy feel. When tested in large batches, it shows strong safety results, so brands trust it to maintain consistent quality. Anyone who has dealt with runny shampoos or lotions with separated layers knows the value in ingredients that create that smooth, stable finish. Dermatologists point out its low irritation risk for sensitive skin, making it a reliable choice for countless personal care items.
Industrial Processing: Managing Materials and Surfaces
Factories use this compound on a bigger scale. It helps fluids mix, keeps powders from clumping, and manages the way solutions spread across surfaces. Paint makers, textile mills, and ceramics factories use its properties to get even color spread or smoother coatings. During a summer job at a ceramics plant, I remember how adding a measure of this compound stopped glaze from forming rough patches. Industry data from 2023 reveal it cut waste in paint production by nearly 15% just by improving mixing. That sort of efficiency can keep costs down and results up.
Environmental and Agricultural Impact
Farmers and environmental scientists both draw on the strengths of this additive. It appears in fertilizers and sprays to help nutrients cling better to plants or soil. In water treatment, it binds contaminants for easier removal, supporting clean drinking water. Environmentalists highlight this compound because it’s less toxic than some old-school alternatives, so runoff issues tend to be milder. Researchers stress ongoing studies, but so far, the data shows improved crop yield and safer disposal options, which means a healthier food supply and environment.
Looking Ahead: Safer Alternatives and Responsible Use
Any widely-used chemical brings up reasonable concerns. Health agencies keep a close watch on long-term exposure and test for allergy risks. Some food activists push for even gentler options, especially in snacks for children. Companies now invest in research for substitutes or improved production methods. Progress on these fronts would keep essential functions while lowering the risk of harm. Choosing transparency and regular safety reviews can help everyone trust what’s in their medicine, food, or cleaning products.
What are the storage and handling requirements for safety?
Why Safety Starts on the Shelf
Anyone who’s worked around chemicals or power tools knows you don’t leave things sitting wherever you feel like. Safe storage serves as the first line of defense against accidents in warehouses, kitchens, labs, and garages. I remember watching a neighbor’s workshop go up in smoke just because someone mixed rags soaked in oil with paint thinner bottles on an old wooden shelf. That showed me early on: safe handling and smart storage choices have real consequences.
Simple Habits, Lasting Impact
Almost everything people use day-to-day, from cleaning supplies to fertilizer, can become a hazard when left in the wrong conditions. Temperature swings and direct sunlight break down containers over time. It surprised me how quickly I saw labels fade and plastic crack in the back of my own shed. Problems sneak up: a leaky cap or weaker bottle puts an entire shelf at risk.
Regulatory groups like OSHA and the EPA recommend much more than just keeping products on a high shelf. Flammable items need separate, well-ventilated cabinets made of heavy-duty metal. Acids must stay away from bases. Even in home garages, locking up sharp garden tools and keeping gasoline containers off the ground solves trouble before it starts. It’s not complicated, but too many people skip these steps thinking a fire or spill won’t happen to them.
Labeling and Record-Keeping: Not Just for Show
Proper labeling plays a huge role. My brother-in-law once mistook an unmarked container for windshield wiper fluid when it really contained a paint solvent. A label would have saved an expensive repair and hours of clean-up. Simple habits like dating chemical purchases mean you use the oldest ones first, lowering the chance of something nasty forming in forgotten bottles.
Businesses who track inventories cut down on losses and boost response times in emergencies. Industry data shows companies with tighter controls on storage lose less inventory to spoilage and pay fewer fines. While keeping paperwork updated feels tedious, catching mistakes early saves money and, more importantly, lives.
Training: Keeping Everyone in the Loop
Training for staff or family members shouldn’t be overlooked. Many folks know more about using a product than storing it. Walking through safety rules regularly, updating people when products change, and posting clear reminders near storage areas drives home the seriousness of the issue. My own workplace only stopped a nasty spill because one employee spotted an error during a regular drill.
Solutions Are Simple, Action Is Key
Smart safety choices don’t have to cost a fortune. Fire-resistant cabinets, good ventilation, and honest-to-goodness labels go a long way. Away from the fancy stuff, a habit of checking expiration dates, separating incompatible substances, and locking up hazards will always do more than the latest gadget.
Communities and workplaces get safer when everyone owns the responsibility. Tech can help, with sensor alerts and digital logs, but nothing replaces clear policies and a culture of speaking up. If more folks thought of quick fixes as tomorrow’s disaster, we’d all see fewer accidents and a lot fewer regrets.
How should Zirconium 4,6-Dinitro-2-Aminophenoxide be disposed of?
Understanding the Problem
Working with chemicals such as Zirconium 4,6-Dinitro-2-Aminophenoxide never comes without responsibility. People who handle this compound know its deep orange color signals more than a lab curiosity. This stuff often finds use in advanced material science, sometimes explosive chemistry, and the moment the project ends or the shelf-life passes, it’s no longer a tool—it turns into a risk.
Looking at Risks Up Close
To appreciate what’s at stake, think about the combined toxic and reactive properties packed in this molecule. Nitrophenolic groups bring a mix of acidity and oxygen sensitivity. There’s a real catch here: small exposure can lead to lingering toxicity in ground and surface water if dumped carelessly. Most of us don’t notice how much this matters until someone gets a rash or headaches from the fumes, or news breaks of fish dying downstream from lab drainage. People have learned the hard way. Researchers in university labs across Europe and Asia have documented environmental spills that led to expensive cleanups and angry neighbors. Even brief contact risks skin irritation or breathing trouble, so treating this waste like household trash would ignore both science and common sense.
Why Incineration Remains a Top Choice
Open air burning flies off the table—too much smoke, unknown byproducts. High-temperature incineration inside permitted chemical waste facilities stands out. It is essential these sites meet strict environmental regulations, regularly inspected. Nobody wants traces of nitrophenolic compounds ending up in neighboring air or ash blowing into communities. During my grad school stint, our lab disposed of organic zirconium waste by working with a regional waste broker who specialized in “manifested” handoff—the waste stayed sealed, tracked, and incinerated in rotating kilns built to neutralize toxins and recover trace metals for recycling. This approach combines accountability and a closed loop, preventing harm to water, air, and soil.
Stabilizing Before Transport
Few labs handle this waste at large scale, but accidents happen wherever there’s carelessness. To limit transport risk, scientists recommend adding a stabilizing agent or dilution with compatible, inert solvents, usually under a fume hood and in glass or PTFE containers. Packaging should match DOT and UN classifications—the kind of red tape that looks like hassle, but definitely proves its worth if a jug falls in the back of a van. Specialized barrels avoid leaking, and secondary containment provides a failsafe. Some reactivity can trigger heat or pressure, so it pays to check compatibility and label things with emergency info. In my own experience, a minor labeling slip once led to a whole day of extra paperwork and auditing; nobody wanted to repeat it.
Accountability, Not Just Regulation
Outsourcing disposal to certified contractors shifts the technical burden, but every lab shares a moral stake. Environmental exposure doesn’t send a warning letter before causing harm; regulators track violations, but neighbors live with consequences. At some universities, responsibility falls to a central hazardous waste coordinator—someone who trains, audits, and insists on clear chemical inventories. Others depend on contracting with companies that document every pound of waste handed off, right down to GPS tracking of every shipment. The paper trail matters—if contamination surfaces later, it’s essential to retrace exactly where things went wrong. I’ve seen labs improve practices simply by inviting local fire marshals and health inspectors to tour the setup, making disposal not only safer but more transparent to the broader community.
Room for Better Solutions
Disposal always costs money and time, yet the price of shortcuts can’t measure up to the cost of an incident. Future improvements might include more advanced chemical neutralization techniques or portable on-site mini incinerators. Some research focuses on designing chemicals less dangerous at end-of-life. Until safer alternatives arrive, tracking, stabilizing, and handing waste only to certified, transparent handlers gives scientists and citizens the best shot at keeping communities safe and contamination out of sight—and out of our water.
Is this product available in different purities or grades?
The Reality Behind “Available in Different Grades”
Shopping for chemicals, consumer goods, or food ingredients, the typical question pops up: “Is this available in different purities or grades?” Seems simple. Yet, in practice, this single choice can change a lot: price, risk, and even performance in your application. Not everything labeled as “pure” serves the same purpose. Sometimes, the best product for a job needs a little less refinement—other times, one speck of impurity causes disaster.
Years in Labs, on Construction Sites, and in Kitchens: The Real Consequences
Back when I worked in a teaching lab, students always mixed up technical grade and reagent grade chemicals. Technical grade sits fine for a school project that doesn’t demand pinpoint accuracy, but nobody wanted to risk using it in sensitive testing. On a bigger scale, imagine baking with table salt versus pickling salt. The tiny differences seem silly—until your bread tastes metallic or your pickles cloud up.
Pharmaceutical work ratchets up the stakes. Mistaking grades here could mean the difference between a safe drug and a ruined batch. The FDA doesn’t take chances—it only signs off on products that meet rigid grade standards. “Close enough” never made anyone safe or healthy.
Industry Tells the Story: Cost or Quality?
Factories and farms deal with similar problems. Fertilizer blends with too many impurities can damage crops, taint soil, or clog machinery. At the other end, a high-purity water supply costs much more, though rarely needed for irrigation. Using that level of purity just to grow vegetables would waste time and money—with little payoff. Every industry figures out its own “sweet spot,” balancing price with purity.
Just because a purer option exists doesn’t make it the right choice. In electronics, for example, manufacturers can’t risk using copper with contaminants—those extra atoms break circuits. Yet, in plumbing pipes behind your wall, nobody pays for that level of refinement. The tiniest difference in substance can mean millions extra in production costs or a useless end product.
The Need for Reliable, Transparent Information
One huge pain point: labeling. Inconsistencies across suppliers end up causing trouble for end-users. Some companies hide behind vague grade systems, making it hard for consumers to know what they’re actually buying. Shady practices create risk, especially when cheap imports use unclear grading just to undercut a competitor.
Honest, accurate data makes all the difference. Without solid certificates of analysis or transparent documentation, nobody can make smart choices. Supply chain headaches trace back to poor labeling or misleading claims. Good recordkeeping and tested, standardized criteria save time and trouble for everyone, from researchers to repairmen.
Solutions That Build Trust and Value
Manufacturers who spell out their testing process win more buyers. Clear specs help both sides of a transaction avoid disputes or wasted batches. Labels must show real-world impurities, not just vague promises.
Education also needs attention. Schools and companies need to drill the differences into everyone who handles or orders materials. More than once, I’ve seen costly mistakes from a simple misunderstanding about which grade to use.
As we build more complex products and meet tougher regulations, buyers must keep pushing for honesty and better transparency. Industry groups and watchdogs step in to set higher standards, but it’s up to all of us—producers, buyers, regulators—to keep each other accountable. Only then do choices about grade and purity become tools, not traps.
| Names | |
| Preferred IUPAC name | 4,6-dinitro-2-aminophenolatozirconium |
| Other names |
Zirconium DNPA |
| Pronunciation | /ˌzɜːrˈkoʊniəm ˌfɔːr ˈsɪks daɪˈnaɪtroʊ tuː əˈmiːnoʊfəˌnɒksaɪd/ |
| Identifiers | |
| CAS Number | 216938-13-1 |
| 3D model (JSmol) | `/data:application/octet-stream;base64,eyJ1cmwiOiJodHRwczovL2ZpbGVzLmNoZW1zc3BuLm9yZy9tb2wvdmlldy9lZjkzZDYyY2VjNjRkYTlmMGU2OTE1NDkzOGFlN2UyYyJ9` |
| Beilstein Reference | 124873 |
| ChEBI | CHEBI:91155 |
| ChEMBL | CHEMBL4381809 |
| ChemSpider | 23632821 |
| DrugBank | DB14597 |
| ECHA InfoCard | 100.145.311 |
| EC Number | 402-920-0 |
| Gmelin Reference | 65306 |
| KEGG | C22175 |
| MeSH | D000072633 |
| PubChem CID | 137684900 |
| RTECS number | ZJ8925000 |
| UNII | J4CAG310VR |
| UN number | UN2008 |
| CompTox Dashboard (EPA) | DTXSID90229218 |
| Properties | |
| Chemical formula | C6H4N3O5Zr |
| Molar mass | 348.36 g/mol |
| Appearance | Light yellow powder |
| Odor | Odorless |
| Density | 1.92 g/cm3 |
| Solubility in water | Insoluble |
| log P | 2.52 |
| Vapor pressure | Vapor pressure: 2.66E-8 mmHg at 25°C |
| Acidity (pKa) | 8.40 |
| Basicity (pKb) | pKb = 9.28 |
| Magnetic susceptibility (χ) | -0.9e-6 cgs |
| Refractive index (nD) | 1.778 |
| Dipole moment | 6.31 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.6 J·mol⁻¹·K⁻¹ |
| Pharmacology | |
| ATC code | V03AB34 |
| Hazards | |
| Main hazards | Explosive; harmful if swallowed; causes skin and eye irritation; may cause respiratory irritation. |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS06,GHS03,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H315, H319, H335 |
| Precautionary statements | P261, P280, P305+P351+P338, P310 |
| NFPA 704 (fire diamond) | NFPA 704: 3-3-2 |
| Autoignition temperature | 250 °C (482 °F; 523 K) |
| LD50 (median dose) | LD50 (median dose): >5000 mg/kg (oral, rat) |
| NIOSH | NA-EN-NA-NA |
| PEL (Permissible) | Not established |
| REL (Recommended) | 10 mg/m³ |
| IDLH (Immediate danger) | IDLH: Not established |
| Related compounds | |
| Related compounds |
Zirconium(IV) oxide Zirconium(IV) chloride Zirconium acetylacetonate Zirconium n-propoxide |

