Zinc Permanganate: History, Challenges, and Horizons
Historical Development
Zinc permanganate came into focus as chemists in the twentieth century explored ways to expand oxidative chemistry beyond traditional heavy-metal salts. With permanganate’s deep purple allure and undeniable power as an oxidizer, researchers in university labs tinkered with various cation pairings. Zinc, relatively abundant and less hazardous than lead or mercury, soon emerged as an attractive choice. Early reports describe hard-won syntheses and inconsistent yields, but scientific curiosity kept the experiments moving forward. Over years of refinement, synthetic methods improved, leaving behind a trail of detailed notes and gradual shifts in protocol. The road wasn’t paved with instant breakthroughs; more often, it demanded patience, sharp observation, and repeated failures. This steady effort helped secure zinc permanganate’s place both in chemical literature and as a stepping stone toward greener, versatile oxidizers.
Product Overview
Zinc permanganate shows up most often as a solid with a striking, dark color hinting at its manganese content. It belongs to the family of permanganates, all known for their eager acceptance of electrons—they like to take, not give. Compared to potassium or sodium permanganate, zinc’s role brings unique traits to the table, many of which flow directly from zinc’s chemistry in aqueous or solid-state environments. Tinkerers and professionals alike chase these subtleties, trying to harness them for analytical work, specialty oxidations, or materials science. I’ve noticed that suppliers tend to highlight the material’s reactive highlights—sometimes overshadowing practical challenges such as shelf-life or handling quirks, which only come to light through experience.
Physical & Chemical Properties
This compound usually presents as a dark purple crystalline powder, echoing the unmistakable hue of permanganate ions. Its solubility depends heavily on temperature and the presence of competing ions. In water, it will release both manganese-based oxidizers and divalent zinc. As with many oxidizers, zinc permanganate doesn’t prop up well under light, heat, or organic contamination. It decomposes with a tendency to liberate oxygen and manganese oxides, so even casual handling can trigger changes in color and reactivity. That quirk looms large for chemists used to the more indestructible potassium permanganate, and it means material has to be stored and used with extra care. These real-world limitations shape both safety routines and expectations about shelf stability.
Technical Specifications & Labeling
Any bottle sporting the name “zinc permanganate” ought to carry warnings about its strong oxidizing effect. Labels spell out dangers—contact with organic matter, acids, or reducing agents often leads to stubborn stains or even fire risk. Detailed molecular weights and purity percentages look impressive, but real peace of mind comes from small-print notes about storage and packaging. Manufacturers often mention glass or inert liners to keep the material dry and shield it from stray organic vapors. Attention to label details reflects genuine experience rather than marketing flourishes: in my lab days, a missing or ambiguous hazard symbol could mean confusion or, worse, avoidable accidents.
Preparation Method
Typical lab-scale preparation hinges on combining an aqueous solution of permanganate, often with a soluble zinc salt. The chemist’s challenge lies in controlling pH and concentration to keep zinc from hydrolyzing or the permanganate from prematurely decomposing. Slow mixing in the cold, maybe with controlled pH buffering, helps coax out zinc permanganate crystals. Once filtered, the product needs quick drying and isolation from stray reducing contamination. On a larger or industrial scale, the process can look similar but with tighter controls, as the consequences of a runaway oxidizer reaction scale up fast. More than once, I’ve watched small mistakes—such as skipping a final wash—lead to batches so unstable that disposal became the only safe answer.
Chemical Reactions & Modifications
Zinc permanganate acts as a source of reactive oxygen, converting alcohols, alkenes, or other vulnerable substrates. The zinc ion itself, while forming soluble complexes, doesn’t hog the limelight but still plays subtle roles in shifting equilibria or binding with reaction intermediates. Many researchers experiment with partial substitutions or blending, hoping to fine-tune oxidative power or selectivity. In practical reactions, side effects like formation of insoluble manganese dioxide, changes in color, or precipitation of zinc salts are more than cosmetic—they affect yields, filtration work, and sometimes the safety profile of a process. Even simple attempts to modify the salt, such as by adding stabilizers or swapping in different buffer ions, can backfire, leading to rapid decomposition or unpredictable results. Careful incremental changes, rather than sweeping substitutions, usually pay off here.
Synonyms & Product Names
Chemists often refer to this compound as zinc(II) permanganate or manganic acid, zinc salt. Academic literature sometimes leans on systematic nomenclature, but commercial suppliers prefer shorter, catchier names. It pops up under a handful of labels, all referencing the core zinc-manganese pairing. Mislabeling remains a persistent problem, as similar names among permanganate salts can lead to accidental switches. In a cluttered reagent cabinet, I’ve seen more than one mix-up between the zinc and potassium versions—which stresses the value of diligent labeling and double-checking, especially before launching a sensitive reaction.
Safety & Operational Standards
Anyone who’s worked with permanganates knows the importance of gloves, goggles, and good ventilation. Even a small spill on fabric or skin can leave dark, stubborn marks and sometimes even chemical burns. Zinc permanganate, while not volatile, puts out plenty of dust if handled carelessly. If that dust finds its way onto a hot plate or an oily rag, it can cause a fire that burns much hotter and faster than expected. Standard guidance calls for working with limited quantities, cool and dry storage, and cleanup protocols that treat any contaminated material as hazardous waste—not trash. Vent hoods and dedicated disposal containers prevent mishaps, but personal vigilance remains the best defense against accidents in shared spaces. Years of routine practice have taught me that simple habits—never working alone, always labeling transfer containers—save more trouble than the thickest safety manual.
Application Area
Zinc permanganate serves in specialized roles across chemical synthesis, analytical procedures, and sometimes water treatment. Synthetic chemists trust it as an oxidizer for certain delicate transformations where more aggressive agents could cause unwanted side reactions. Environmental labs sometimes reach for it in trace metal determination, taking advantage of its reactivity while avoiding heavier metal residues. Occasionally, discussions arise about wider application in disinfection or soil remediation, but these often stall on questions of cost, handling, and comparative benefit. Every so often, specialty applications pop up—think organic synthesis where selectivity matters or blends with other metals to tune properties. To gain more traction, advocates will have to make a stronger case for advantages over stalwarts like potassium permanganate, especially where regulatory concerns about manganese residues or process safety shape buying decisions.
Research & Development
Most investigation into zinc permanganate focuses on making the compound more stable, scalable, and user-friendly. Lab teams test new synthesis routes, stabilizer additives, and blending techniques, hoping to cut costs or improve performance. On the analytical side, research digs into how zinc permanganate behaves in complex matrices, such as environmental samples or biological fluids, aiming to map out limitations and hidden strengths. Collaboration between academic and industry teams sometimes yields process tweaks or instrument improvements—incremental changes that add up. Looking further, researchers evaluate green chemistry credentials, comparing zinc permanganate’s life-cycle impacts against other oxidizers, mindful that sustainability claims only hold weight with supporting data and careful waste handling protocols. These development efforts require not just inventive chemistry but also open conversation with users who see both the upside and the headaches of daily use.
Toxicity Research
The toxicological record for zinc permanganate centers on both its individual components and the combined effect. Permanganates in general gain notoriety for their oxidizing strength, posing risks on contact, ingestion, or inhalation. Zinc, often pegged as a micronutrient, takes on new character at higher doses or when combined with strong oxidizers—increasing concern about irritation, tissue damage, or longer-term environmental persistence. Most toxicology studies focus on acute effects: skin burns, mucous membrane irritation, and systemic symptoms from accidental ingestion or inhalation. Animal studies, plus occasional industrial incident reports, shape regulators’ recommendations on workplace exposure, disposal, and environmental release. Producers and users alike must monitor developments in this area—experience proves that chemicals often slip from benign status to restricted use when better toxicity data or rare but dramatic incidents come to light.
Future Prospects
Zinc permanganate stands at an interesting crossroads. Modern chemistry seeks greener, safer, and more selective reagents, and zinc’s relatively low toxicity keeps it in the running for sensitive applications. Advances in material science may reveal overlooked uses, such as battery electrodes or targeted water remediation. Efforts to stabilize the salt, package it more safely, and reduce side products could unlock broader industrial uptake. At the same time, industry skepticism about cost, handling risk, and regulatory pressure could check enthusiasm. For broad acceptance, champions of zinc permanganate will need clear evidence of unique benefits—advantages that justify extra effort, training, or spending. As researchers probe its limits and compare it against new generations of oxidizers, practical voices from the lab bench will shape its trajectory. Some will embrace its strengths; others, burned by a costly mishap or disappointing trial, will move to newer alternatives. The next decade will likely see zinc permanganate’s fate decided not just by chemistry but by the complex interplay of economics, regulation, and hard-earned hands-on experience.
What is zinc permanganate used for?
A Glance at the Compound
Zinc permanganate isn’t exactly a name that you see flashing on billboards or trending in science circles. Many folks outside the world of chemistry might never bump into it, but for those who work with specialty chemicals, it holds a bit of intrigue. Sitting at the intersection of transition metals and prolific oxidizers, it brings together the properties of zinc, so valued for corrosion resistance, with the potent oxidation power of permanganate. That blend makes it stand out among a crowd of more familiar compounds.
Applications Rooted in Chemistry
You can spot zinc permanganate in a lab’s arsenal, thanks to its strong oxidizing ability. Chemists might turn to it when they need to alter organic molecules, especially in small-scale synthesis projects. Here, it turns certain structures into something new—reshaping alcohols or breaking apart double bonds in aromatic rings—actions that form the backbone of research chemistry and industrial applications. It’s useful for processes where gentle touch isn’t the priority, but reliable results matter.
Beyond classic organic chemistry, zinc permanganate finds a niche in water treatment. People worry about what flows from their taps, so oxidizers get used to break down pollutants and organic residues, including those that can slip past standard filtration. While manganese-based oxidizers aren’t always the first choice at big plants, there’s growing interest around smaller or specialized settings—think fieldwork, emergency response, or cleaning up stubborn local contamination—where its particular mix of solubility and reactivity carries advantages. In these situations, clear water isn’t just about taste. It’s about trust, health, and public confidence.
Challenges and Health Concerns
Any talk about permanganates brings up the dangers. Strong oxidizers demand care. Zinc permanganate, like its cousins, can start fires if mixed with the wrong substances and may irritate the eyes, skin, or lungs. Handling safety takes priority wherever it’s stored or shipped. Lab veterans know stories of surprise reactions—or worse—where protocols slipped. Regulations keep a close eye on storage and use, as mishandling can lead to spills, environmental damage, or health risks.
Zinc itself isn’t all bad news. Our bodies need a little bit of it every day, but with permanganate in the mix, things get trickier. Waste management means paying attention to the byproducts. Permanganate breaks down, but intermediates like manganese oxides or unused reagents require targeted disposal. Regulatory agencies like the EPA underline the need to manage all oxidizers, not just the famous ones, and keep waterways free from contamination that puts fish or people at risk.
Deciding Where Zinc Permanganate Fits
Choosing to use it often comes down to a trade-off. Does the application need a soluble oxidizer with a specific strength? Are alternatives like potassium permanganate or sodium hypochlorite fit for purpose? In specialty sectors, those with a focus on research or niche water treatment, the unique mix of zinc and permanganate might offer an edge. The market doesn’t always favor the uncommon, but scientific progress grows from exploring new options.
Moving forward, safer handling and better education on the chemistry and risks of zinc permanganate matter most. Continued research might highlight new benefits or uncover different roles for this compound outside its niche. No single substance solves all problems, but with care, even those off the beaten track can help tackle real-world challenges in labs, water systems, and industry.
What is the chemical formula of zinc permanganate?
Seeing Chemistry Beyond the Classroom
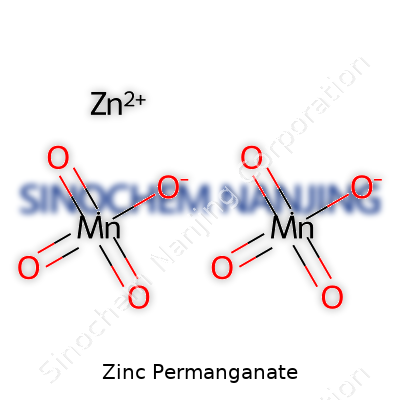
Zinc permanganate sounds like something you run across in a textbook or a dusty corner of a lab, but chemistry does not exist only in the pages of school notes. Getting familiar with its formula, Zn(MnO4)2, points to a skill beyond rote memory—the ability to connect formulas with the logic behind the science.
Why the Formula Sticks with People in the Field
Plenty of folks, even people with hands-on jobs, may wonder why a formula like Zn(MnO4)2 matters. Think about a water treatment plant. Permanganates show up again and again to help purify water. Someone choosing zinc permanganate over potassium permanganate takes into account the reactivity, toxicity, and impact on equipment. That decision starts at the chemical formula. It tells workers what kind of ions will appear, which reactions will follow, and what byproducts to expect.
From Chalkboards to Real Solutions
Growing up, most folks saw chemical equations as chores—something for the next quiz, then out of mind. In manufacturing, those formulas step into the spotlight. Knowing that zinc forms a +2 ion, and permanganate brings along a -1 charge, helps everyone from plant engineers to small batch chemists balance equations—and budget for what they need. A spill, a reaction gone sideways, or even a simple mix-up all feel less risky when the crew on hand knows what Zn(MnO4)2 will do.
Looking at Health and Environmental Impact
Anyone who’s seen manganese stains knows not all chemicals come without a downside. Handling permanganates means understanding how they break down and what hazards may linger. Safety data sheets always name the formula. If a formula like Zn(MnO4)2 pops up on a label, the right safety gear needs to show up too—gloves, goggles, and sometimes special handling for waste. Tossing unknown materials down the drain invites risk to both workers and the water table. Green chemistry leaves its mark by tracking formulas from lab to landfill, not just from blackboard to beaker.
Putting Chemical Knowledge to Work
Anyone investing time in chemistry should see formulas as more than a jumble of symbols. Fact check: the formula for zinc permanganate—Zn(MnO4)2—means two permanganate ions pair with each zinc ion, reflecting how charges must balance. This structure influences solubility, possible reactions, and safe handling practices. Teachers who point this out move students beyond memorization and into reasoning territory.
Building Better Tools for Industry
Industries need workers and leaders with strong chemical sense. Mixing batches, troubleshooting a reaction, or tweaking products all rely on being able to read a formula like Zn(MnO4)2 and grasp the consequences. Good decisions happen when folks can draw the connection from what’s written on drums or datasheets back to actual risks and benefits. Mentors in labs and shops can boost skills simply by encouraging questions about formulas, not just outcomes.
Moving Toward Smarter Use of Chemicals
Understanding what makes up zinc permanganate, down to its formula, gives people more than a trivia answer. It sharpens the approach to safety, guides responsible recycling, and supports clear thinking in emergencies. Naming a chemical fully, with its correct formula, proves the value of precision—and that’s useful whether someone stands at a school desk or a workbench.
Is zinc permanganate hazardous or toxic?
Getting Real About Chemical Hazards
People love answers that are black or white: safe or unsafe, toxic or harmless. Chemistry rarely offers that certainty, and zinc permanganate is a perfect example. I’ve worked in labs where unfamiliar compounds always raised one question—what’s the risk? The answer rarely came from the safety data sheet alone. Instead, it grew from hands-on experience, scientific literature, and talking with people who handle these chemicals daily.
Breaking Down What Zinc Permanganate Is
Zinc permanganate isn’t a household name. You won’t find it under your kitchen sink or sitting in your medicine cabinet. It’s usually talked about in research circles, sometimes as a potential oxidizer or catalyst. Like other permanganate salts, it contains the powerful permanganate ion—those manganese atoms surrounded by lots of oxygen. The presence of zinc makes things interesting but doesn’t neutralize the risks associated with permanganates in general.
Spotting the Hazards
Permanganates are famous for being strong oxidizers. Anyone who’s mixed potassium permanganate with sugar in a lab knows how quickly things can go wrong. They can cause fires if they touch something combustible. It looks like zinc permanganate acts much the same way—it wants to react, sometimes violently, with organic materials. Toss in some heat and you’re looking at a possible explosion, not just a fizz or puff of smoke.
Toxicity is another story. For most permanganate salts, the manganese ion can spell trouble if inhaled or ingested. Chronic exposure sometimes leads to neurological symptoms—memory issues, muscle tremors, even mood swings. Zinc typically earns a gold star as a trace mineral, safe in small doses, but swallowing it as part of a permanganate salt isn’t the same as chewing a multivitamin.
Contact with skin burns, staining, and irritation are documented effects. I have seen what even mild exposure to permanganate solutions causes—slow-healing, brown or purple patches that itch for days. Without gloves and goggles, lab workers notice fast how unpleasant these salts can be.
What the Science Says
There’s little direct toxicology research on zinc permanganate, at least compared to bigger players in the chemical family. The EPA and CDC provide guidance on permanganate salts, especially potassium and sodium versions. Animal studies have shown serious health effects after sustained or high exposure. Given that zinc permanganate is similar in structure, treating it with the same level of caution feels like basic common sense.
Inhaling the dust or getting it in eyes are real risks for workers handling this stuff without protection. The easy solubility in water makes spills a problem for both people and the environment. Permanganate ions don’t play nice with aquatic life, causing long-term harm to waterways. I’ve spoken with environmental safety advisors who treat all permanganate spills as emergencies, not just inconveniences.
Handling It Safely
For those in research, strict protocols matter. That means real personal protective equipment—lab coats, goggles, nitrile gloves—and proper ventilation. Storing zinc permanganate away from flammable material makes a big difference. Training goes farther than paperwork; seeing examples, learning from others’ mistakes, and getting hands-on instruction stick longer than a list of do’s and don’ts.
Disposal also demands respect. Never wash extra or waste down the drain. Collection as hazardous waste protects both water systems and people downstream. Where I worked, we always ran permanganates through reduction steps—turning them into less reactive products—before final disposal.
What This Means for Everyday Folks
Most people outside of chemical labs or industry never run into zinc permanganate. For users, it pays to ask suppliers for full safety documentation and use the compound in ways that minimize exposure. For communities near production or disposal sites, local authorities should demand transparency about storage, handling, and waste practices.
It’s tempting to think obscure chemicals like this live only on the margins. Reality check: the safe handling of dangerous substances always comes down to respect, education, and preparedness. This lesson holds up whether you’re mixing up a new catalyst in a high-tech lab or treating a chemical spill in an old storeroom.
How should zinc permanganate be stored and handled?
People Think About Safety For a Reason
Anyone who works with chemicals in a lab, plant, or classroom knows the rules for a reason. I remember early on in my chemistry career, a supervisor hammered this point: shortcuts with strong chemicals often bring bigger headaches. Zinc permanganate falls into the family of strong oxidizers. This isn't just a buzzword. In daily practice, you learn that "oxidizer" means "can start a fire fast if mixed wrong." Fires in labs are scary, but so are toxic fumes and reactions that go out of control.
Zinc Permanganate Wants to React
Zinc permanganate is purple, powdery, and builds up energy in its bonds. Contact with organics like paper, cloth, or even a dusty benchtop can trigger reactions you definitely don’t want. In dry air, the powder might cake up but stays risky if any spark or heating happens. That's why folks in the lab work with clean tools, avoid loose paper, and keep containers properly capped after each use. I’ve seen what a spill can do — one messup turned into a cleanup job no one envied because oxidizers stain everything, even equipment thought to be “safe.”
Facts You Can’t Ignore
OSHA and European chemical guidelines both point out the risk of fire and toxic manganese emissions if things go wrong. Exposure can irritate the lungs and skin, especially when powder gets airborne. I remember labs where a whiff of permanganate meant someone probably made a mistake. You don’t need a health and safety poster to remember that – one breath drives it home.
Real Storage Practices
Safe handling starts with the container. Use a tightly sealed, non-reactive bottle or drum — glass or certain plastics work, while metal can corrode. Keep everything in a cool, dry storage room, away from sunlight and any flame. In my experience, storing next to acids or flammable solvents creates a real risk, even if the shelf feels “organized.” One splash or vapor leak can turn a quiet day into trouble. If I ever saw containers near organic chemicals, my first move was to separate them — a small thing, but it saves lives.
Training Makes a Difference
You can buy MSDS sheets and print out rules, but actions matter more. Labs I respected always gave hands-on training for staff and students, not just in using gloves and goggles, but in what to do if things go wrong. Neutralizing spills with sand or a specific neutralizer, not water, prevents extra heat or splashing. Fast evacuation drills aren’t just a box to check; people remember them when it matters.
Solutions — Practical and Possible
Instead of locking up chemicals and hoping nobody touches them, workplaces should rethink layout, label everything in plain language, and run regular checkups for leaks and expired stock. If storage space is short, it’s safer to order in smaller batches. I’ve seen places cut costs by buying big, only to waste cash tossing degraded powder later. Reporting close calls helps everyone learn, not just the person who slipped up.
Responsible management of zinc permanganate means accepting its power, building habits rooted in respect, and making safety a daily habit. Anyone who handles it owes their co-workers more than just a quick glance — they owe them attention and care, every shift, every day.
Where can I buy zinc permanganate?
The Search for Zinc Permanganate
Zinc permanganate rarely ends up on typical shopping lists. Most people outside the lab do not even know it exists. It stands out in industrial chemistry as a powerful oxidizer. Interest in this compound comes up in specialized circles—professional chemists, institutions, and curious science hobbyists might want to buy it. The big question—where can you safely get it?
Why You Will Not Find Zinc Permanganate at Big Retailers
Chain retailers do not carry zinc permanganate. Walk into a hardware or gardening shop, and you will not see it on shelves. Big online marketplaces like Amazon and eBay usually avoid permitting sales of niche, reactive chemicals. That is not because people do not ask—it is about regulation, potential misuse, and strict shipping rules.
Through my own years in chemistry, specialty chemicals like this always meant working with authorized suppliers. It is not a casual purchase. Forget your average chemical shop; reliable sources are found through reputable laboratory supply companies. Sometimes, I would need to show credentials or institutional affiliation. Shipping comes with even more strings attached: regulations treat oxidizers with care, for good reason.
Regulations and Ethical Sourcing
Zinc permanganate's strong oxidizing properties mean it falls under regional or federal hazardous materials laws. In the US, the Environmental Protection Agency (EPA) and Department of Transportation (DOT) enforce rules on how you can order, store, and transport such substances. In Europe, REACH regulates chemical access. Sellers can only ship to verified professionals for legitimate use. That means most private buyers hit a wall.
Some online chemical marketplaces cater to research institutions and universities. Examples include Sigma-Aldrich, Alfa Aesar, and Lab Alley. Most suppliers will require documented intent—researchers must explain the application and prove their qualifications. Even if you meet the rules, shipments might get delayed or blocked without proper paperwork. It reflects real risks: improper handling can trigger fires, health incidents, or environmental harm.
What Responsible Sourcing Looks Like
Trying to order zinc permanganate from overseas web shops or anonymous “rare chemical” sites leads to trouble. Poorly labeled shipments, risky storage, or outright scams pose a real threat. I have seen colleagues lose money and even receive substances that were not what was advertised. More importantly, handling oxidizers in a non-lab setting can be downright dangerous.
In my lab experience, the right steps start with documenting the reason for use. Licensed facilities keep Material Safety Data Sheets on hand, post safety warnings, and store oxidizers with protective measures. For the general public, DIY chemistry crosses into legal gray areas or worse. Sites offering easy access rarely follow the rules, and authorities track purchases through customs and postal services.
Much More Than a Simple Purchase
Access to a controlled chemical like zinc permanganate should always run through approved channels. Staying within legal boundaries means respecting public safety and environmental health. For every person who asks where to get it, the better question might be “what purpose justifies the risk?” Professionals follow rules not just because regulators say so but because the outcomes of a mistake can be irreversible. Responsible sourcing—through licensed suppliers, with clear proof of purpose—remains the only credible path. Anything else runs a risk far greater than inconvenience.

| Names | |
| Preferred IUPAC name | zinc(II) manganate(VII) |
| Other names |
Permanganic acid, zinc salt Zinc(II) permanganate |
| Pronunciation | /ˌzɪŋk pərˈmæŋ.ɡə.neɪt/ |
| Identifiers | |
| CAS Number | 10196-23-9 |
| Beilstein Reference | Beilstein Reference 3588224 |
| ChEBI | CHEBI:86359 |
| ChEMBL | CHEMBL38212 |
| ChemSpider | 16268807 |
| DrugBank | DB15860 |
| ECHA InfoCard | 03a63add-4008-4e21-9e7a-1430df2cceb7 |
| EC Number | 243-258-5 |
| Gmelin Reference | 69457 |
| KEGG | C18751 |
| MeSH | D000402 |
| PubChem CID | 159410 |
| RTECS number | ZS6475000 |
| UNII | 7R8M7D910A |
| UN number | UN1479 |
| Properties | |
| Chemical formula | Zn(MnO4)2 |
| Molar mass | 285.33 g/mol |
| Appearance | Violet crystalline powder |
| Odor | Odorless |
| Density | 3.44 g/cm³ |
| Solubility in water | Soluble |
| log P | -3.2 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 11.3 |
| Magnetic susceptibility (χ) | +10800.0e-6 cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 254.2 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -813.2 kJ/mol |
| Pharmacology | |
| ATC code | Zinc Permanganate does not have an ATC code. |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes severe skin burns and eye damage, may intensify fire. |
| GHS labelling | GHS02, GHS05, GHS06, GHS08, GHS09 |
| Pictograms | GHS05,GHS07,GHS09 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H410 |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P370+P378, P501 |
| NFPA 704 (fire diamond) | Health: 2, Flammability: 0, Instability: 1, Special: OX |
| LD50 (median dose) | LD50 (median dose) of Zinc Permanganate: **600 mg/kg (oral, rat)** |
| NIOSH | ZNM |
| PEL (Permissible) | Not established |
| REL (Recommended) | 0.1 mg/m³ |
| Related compounds | |
| Related compounds |
Potassium permanganate Sodium permanganate Calcium permanganate Zinc sulfate Zinc nitrate |

