Zinc Nitrate: A Deep Dive into Development, Properties, and Future Paths
Tracing the Journey: Historical Development
Zinc nitrate didn’t just pop up during the latest surge in chemical industry growth. It holds a history stretching back to the roots of modern inorganic chemistry. Alchemists in the 16th and 17th centuries pursued zinc compounds, often stumbling onto nitrate forms during experiments with nitric acid and impure zinc ores. Once commercial nitric acid production became reliable in the 19th century, manufacturers recognized the benefits and applications of zinc nitrate, leading to expanded use in laboratories and various industries. War efforts, like in World War I, boosted zinc compound production for explosives and metal surface treatments. Today, zinc nitrate keeps finding new roles as technology, agriculture, and environmental needs evolve.
Product Spotlight: What Zinc Nitrate Really Is
You won’t find zinc nitrate in most kitchen cupboards, but it pops up plenty in labs and factories. This compound, usually supplied as a colorless crystalline solid, comes with a formula Zn(NO3)2. You’ll often see it as a hexahydrate because the hydrated form stays more stable and less prone to abrupt chemical hiccups. Its unique ability to dissolve quickly in water makes it useful in both solution and solid applications. It’s not a household name, but its impact rolls quietly through everything from galvanizing processes to specialty chemical production.
Physical and Chemical Properties Matter in the Real World
The textbook facts have a purpose. Zinc nitrate crystals look colorless and can absorb moisture right out of the air, turning sticky if left open in a humid room. The solid melts at about 36–38°C (hexahydrate), which poses storage challenges in some settings. In water, zinc nitrate breaks up completely, tossing out zinc ions and nitrate ions—simple, but with huge effects in chemical reactions. The compound acts as a strong oxidizing agent, and mixing it with reducing agents or combustibles creates safety risks. In my own work handling chemicals in high school, you learn quickly which materials command more respect, and zinc nitrate definitely does thanks to these reactive traits.
Technical Specs and Labeling Checkpoints
Industry puts a premium on reliable labeling. For zinc nitrate, expected purity grades cover laboratory, analytical, and industrial uses. You'd want something like 98–99% purity for routine work, with careful labeling covering water content (not just "hexahydrate," but the actual percent moisture). Packing typically favors airtight, moisture-resistant containers to counteract the material’s hygroscopic nature. Labels need more than a name—they call out hazards, like oxidizer symbols and precise concentration ranges, so that every user, even those new to chemical management, stays informed at a glance.
Making Zinc Nitrate: A Straightforward Synthesis
Methods for producing zinc nitrate trace back to classic acid-base reactions. You start with metallic zinc or one of its compounds (like zinc oxide), then react it with nitric acid. The reaction fizzes as hydrogen or nitrogen oxides bubble up, and with the right controls, you get a clear, concentrated zinc nitrate solution. Careful evaporation brings out the crystals. Reliable labs filter and purify those crystals to remove traces of iron or other metals caught in the initial reaction. This direct synthesis is simple enough for experienced chemists to scale from flask to drum, provided the equipment handles the acids and vapors safely.
Reactivity and Tuning: Chemical Behavior Under the Microscope
What jumps out about zinc nitrate is its readiness to jump into new reactions. Add heat, and it decomposes, tossing out nitrogen oxides and leaving zinc oxide behind—a reaction used in past generations to make white pigments. Mix with alkalis, and behold, zinc hydroxide quickly drops out of solution. Add organic substrates, and the nitrate program kicks in, pushing toward nitration. This reactivity delivers zinc nitrate into research on green synthesis, catalysis, and materials modification. At the bench, its willingness to break down or transform offers a toolbox for tuning reaction pathways, but workers always need to keep an eye out for unexpected oxidizing effects.
Synonyms and Product Names: Recognizing Zinc Nitrate in the Wild
In catalogs and research papers, you might notice the same compound dressed up under different labels: Zinc dinitrate, nitric acid zinc salt, or, for the hydrate form, zinc nitrate hexahydrate. CAS Number 7779-88-6 unlocks the hexahydrate specifically in regulatory and procurement contexts. Most industrial suppliers standardize on “zinc nitrate hexahydrate,” but switching languages or regions can pull up all kinds of naming variations. These synonyms can cause confusion, so double-checking product descriptions and formula details always pays off, especially for first-time buyers or staff checking compliance in multinational labs.
Staying Safe: Practical Hazards and Operational Standards
Zinc nitrate doesn’t mess around on the safety front. Left near organic materials, heat, or sparks, it feeds fire with more oxygen, like tossing gasoline on a grill. For years, I worked in lab settings where the rules demanded protective gear, careful segregation of oxidizers, and always, a chemical fume hood when prepping zinc nitrate. The compound irritates skin and eyes, and inhaling dust or mist stings the respiratory tract. Industry standards call for robust storage—dry, away from fuels, in containers with clear hazard coding. Safety Data Sheets outline first-aid for exposure, spill handling, and emergency planning. Teams don’t rely on luck; instead, planning for worst-case scenarios keeps people and facilities safe.
Application Area: Where Zinc Nitrate Proves Its Worth
You find zinc nitrate working quietly in places far from the public eye. In metallurgy, it cleans and prepares surfaces for electroplating or galvanizing, delivering pure metal bonds by stripping away contaminants at the atomic level. Textile operations use solutions to set dyes and improve material finish. In agriculture, some specialized fertilizers call on zinc nitrate to pump up micronutrient content in soil and hydroponic systems. Chemistry research leans on it to make advanced materials, including catalysts and specialty glass. Growing demand for nanoscience and environmental remediation technology also positions zinc nitrate as a useful precursor in pilot projects and emerging commercial applications.
R&D: Current Themes and Smart Directions
Scientists keep coming back to zinc nitrate when designing new functional materials or testing novel catalytic processes. Research groups look for ways to harness its oxidative power for green chemistry, replacing harsher chemical treatments with milder, more sustainable paths. In material synthesis, zinc nitrate solutions help grow metal-organic frameworks (MOFs) with high surface area, good for gas storage or pollutant adsorption. Nanotechnology labs use it as a zinc ion source for preparing nanoparticles that show up in sensors or electronic devices. The breadth of these research pathways shows how even established compounds can find unexpected new career paths as the frontiers of science keep expanding.
Getting a Handle on Toxicity
People ask about safety in the lab, but the bigger picture includes the environment. Acute exposure to zinc nitrate stings—think eye, skin, and breathing irritation. Swallowing enough of it delivers a hefty dose of both zinc and nitrate, which can strain the kidneys and disrupt the body’s delicate balance. Chronic environmental release poses risks to fish and aquatic life, as nitrate ions upset oxygen cycles and zinc builds up in tissues. Toxicologists run tests to understand and set exposure limits, and regulators base workplace standards on that solid data. Responsible waste disposal and spill management sits at the foundation of every serious facility’s environmental program, anchored by the need to protect human and animal health.
Future Prospects: Looking Ahead for Zinc Nitrate
Zinc nitrate faces both opportunity and scrutiny in the years ahead. Demand grows with each new industrial process or high-tech application tapping into zinc’s chemistry. Pushes for cleaner energy, smarter agriculture, and advanced materials draw on its versatile roles, but regulatory oversight regarding nitrate runoff and occupational exposure stands to rise. Researchers focus on inventing safer, greener production methods and tailoring zinc nitrate derivatives for even more specialized uses, like targeted fertilizers that minimize environmental risk. Getting ahead of waste management concerns and aligning product design with evolving environmental policies marks the next challenge—for manufacturers, users, and the communities they serve.
What is Zinc Nitrate used for?
Understanding the Role of Zinc Nitrate
Zinc nitrate doesn’t get much attention outside laboratories or factories, but its influence stretches across more industries than most folks realize. Its clear, almost invisible presence in daily life gives it a kind of quiet power. I came across zinc nitrate years ago, back when I was trying to clean up an old metal fence. I learned about chemical rust removers and was surprised to find this compound listed among the ingredients. Curiosity led me down a rabbit hole, and what I found really changed my appreciation for what seems like a simple chemical.
In Agriculture: Feeding the World’s Crops
Zinc nitrate helps crops grow. Farmers and agronomists use it as a fertilizer additive, especially for plants that tend to show signs of zinc deficiency: faded leaves, stunted growth, low yields. Crops like wheat, rice, and corn get direct benefit from targeted zinc application. Research from the International Zinc Association notes that zinc-deficient soils can cut grain output by up to 40%. Delivering the right nutrients keeps food production ticking along, supports rural livelihoods, and feeds people in countries where every bushel counts.
Treating Metals and Fending Off Corrosion
Walk into a car plant, a steel mill, or an electrical workshop and zinc nitrate is sure to play a part. It’s a staple in the process of “passivation,” a treatment that helps metals avoid rust and other forms of corrosion. If you’ve ever wondered why some nuts and bolts keep their shine longer than others, it’s usually because their surfaces were treated using complex formulations that include zinc nitrate. The longevity of metal infrastructure owes something to this less-known chemical. Fewer breakdowns and replacements also help save money and reduce waste.
Laboratory Work: Producing Specialty Chemicals
In the lab, zinc nitrate acts as a starting point for a range of reactions. It’s a source of pure zinc ions, which are vital for preparing catalysts, pigments, and advanced materials. Researchers often choose it for the consistency it brings to chemical syntheses. For instance, manufacturers rely on its predictable behavior while creating dyes or medicines. My own experience working with university chemistry labs showed me just how important quality and safety are, especially when scaling up from the test tube to something that fills a warehouse.
Environmental Cleanups and Water Treatment
Zinc nitrate plays a part in environmental protection too. It contributes to water purification processes and can help remove unwanted contaminants. Environmental agencies monitor metal ions like lead and cadmium in rivers and drinking supplies; scientists use zinc compounds in some test kits and as part of cleaning reactions that trap and separate out toxins.
Addressing Risks and Handling
With all the benefits, there’s a flip side. Improper handling brings risks—like skin and eye irritation. Factories must use solid protocols for storage and disposal so there’s no release into local ecosystems. Good training, up-to-date safety data, and clear labeling help keep workers healthy. Companies and regulators both share a role in making sure each batch is managed with care.
Building a Safer, Smarter Future with Zinc Nitrate
Every day, the story of zinc nitrate unfolds quietly across farms, factories, and research labs. Smart application, up-to-date practices, and a steady commitment to safe handling ensure that its positive impact outweighs any negatives. Whether you’re growing vegetables, fixing a metal gate, or working in a lab, it’s clear this chemical packs more punch than most of us realize. Future advances in safety and environmental technology will only deepen its role in sustaining our economy and well-being.
Is Zinc Nitrate hazardous or toxic?
Understanding Where Zinc Nitrate Shows Up
Zinc nitrate often pops up in labs, fertilizer production, textile dyeing, and sometimes in chemistry classrooms. Its white, crystalline appearance might look harmless, giving off the impression of something as plain as table salt. Life experience teaches us appearances can fool. The real story sits in its chemical makeup.
Hazardous to Health: What Science Says
Anyone who has dealt with zinc nitrate knows it doesn’t belong anywhere near food. Experts warn that this compound can irritate skin, eyes, and especially the respiratory tract. A spill or a cloud of dust raises more than a little discomfort. Exposure at work, even in small amounts, brings on symptoms most hope to avoid — coughing, headaches, feeling weak. Nitrates add a special concern. The body doesn’t handle them well if ingested or inhaled for too long.
Workplace safety regulations like OSHA treat zinc nitrate with respect and caution. Permissible exposure limits exist for a reason. I once watched a coworker ignore gloves to “just rinse a flask out.” Minutes later, his hands turned red and itchy. He didn’t end up in the emergency room, but he needed medical attention. People develop skin rashes, eye pain, or worse with repeated or careless handling.
Fire and Environmental Impact
Few expect something without a sharp odor to bring serious risk in a fire. Yet, zinc nitrate acts as an oxidizer, feeding fires instead of dousing them. Toss it into a blaze alongside combustible material, and the flames jump higher. Firefighters use full gear and protective masks during industrial accidents for a reason.
Rainwater washing spills into nearby soil raises other problems. Environmental agencies track nitrate runoff closely. Too much can spike toxin levels in water sources, threatening fish and other aquatic life. Local water boards sometimes issue alerts after fertilizer accidents, and cleanups cost thousands.
Managing Risk in Labs and Industry
Good habits make all the difference. Storing the compound far away from heat, organics, or anything that burns, addresses many hazards. Regular leak checks and training reduce the odds of trouble. A former supervisor drilled us on emergency showers and spill kits every quarter. That repetitive training paid off the day a glass container cracked during cleanup. Quick response contained the danger, preventing skin exposure and environmental contamination.
Companies tracking inventory and auditing safety gear show a proactive approach. These efforts echo what health and safety guidelines recommend: handle only what you need, use it for a purpose, then secure it tightly. Waste disposal companies know zinc nitrate goes through neutralization or special incineration, not down the drain.
Looking Forward: Safer Handling and Alternatives
Educating staff, students, and the public about the risks shapes the everyday experience with chemicals like zinc nitrate. Proper labelling helps stop accidents before they start. Some scientists search for safer alternatives in manufacturing and agriculture, aiming to lower health and environmental costs.
Zinc nitrate doesn’t cause issues just by existing. Trouble starts when basic precautions get skipped or misunderstood. The compound serves its purpose across several industries, but earning those benefits means never dropping your guard. Living and working with it safely demands respect, common sense, and ongoing attention—a lesson lived and learned by those who’ve experienced the real-world consequences of carelessness.
How should Zinc Nitrate be stored?
Stepping Into the Lab: Lessons You Don’t Forget
Zinc nitrate seems, at first glance, like just one more crystal-covered container crowding the shelf in a high school lab or commercial storeroom. Plenty of folks who work with chemicals barely notice it beyond the occasional label check. My early days running samples in a municipal water plant changed that perspective fast. One careless mistake or quick shortcut left a strange, spicy smell in the air, a burned countertop, and a lecture burned into memory for good.
Real Risks That Don’t Always Stand Out
Zinc nitrate doesn’t shout danger, but its quiet nature can lull people into a false sense of security. In any work setting, this white, powdery compound can become a problem if left open or exposed to heat. Moisture brings clumping. Any contact with organic material—even a stray paper towel or bit of dust—can start a chemical reaction. More than one warehouse worker has found out the hard way that zinc nitrate will corrode metal shelving or damage wood over time.
Keeping Zinc Nitrate in Its Place
A good rule has always been to treat it like you would sugar—only hungrier and pickier. Airtight containers made of plastic or glass keep humidity away, and they help block the transfer of odors and contaminants into the chemical. Store in a dry spot with a solid shelf, away from the furnace, heat lamps, or even computer towers that might kick out extra warmth. Even the light from sunny windows can warm jars enough to crack seals or start condensation.
Personal experience tells me that labels fade and instructions get lost in storage rooms faster than anyone expects. Use bold, waterproof markers to relabel every container. Keep records of open dates. Train the newest worker as if they’ll need to teach the next. It only takes one shift change for poor habits to spread, and soon you’ll see granules crusting up or find someone left a scoop behind. Don’t let them push “just for now” storage into corners near acids, peroxides, or organic solvents, because old nitrate can turn unstable.
Laws and Guidelines: More Than Red Tape
Official advice may seem like busywork, but it’s built on lessons from accidents and fires. Agencies including OSHA have set clear standards for keeping reactive chemicals safe: keep the lid on tight, lock the door, and stop unauthorized access. Few operators look forward to unannounced inspections, but the rules don’t exist just for paperwork. In places with lots of temperature swings or heavy storm seasons, pay extra attention, since leaking packaging often gets missed until it’s too late.
Connecting Safety to Daily Practice
No clever container or expensive sensor will replace old-fashioned vigilance. If I ask coworkers to store zinc nitrate right, it’s not paranoia—it’s protecting time, money, and health. Awareness spreads fast after a close call, but wouldn’t it be better to prevent the scare altogether? Share practical tips. Drag the chemical out for a demo every few months. Make reminders stick by tying them to real stories instead of routine lectures.
Handling zinc nitrate safely deserves more than just a checked box on a training sheet. Storing it thoughtfully—dry, tightly covered, away from heat, with good labeling—means no one has to scramble when something leaks or a shipment tips on a Friday afternoon. The less often you have to clean up a sticky mess, the sooner you finish the shift. That memory from the water plant never really fades. Let it remind us to set up good habits before we wish we had.
What is the chemical formula of Zinc Nitrate?
Why the Chemical Formula Matters in Everyday Chemistry
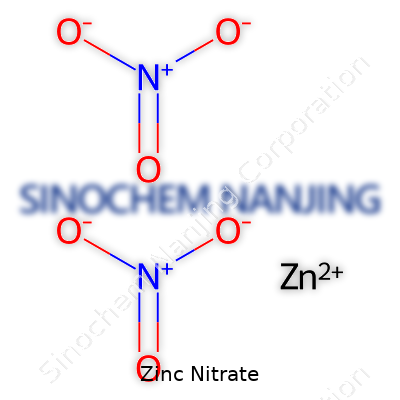
Zinc nitrate often shows up in school labs, garden centers, and across various industries. Its chemical formula—Zn(NO3)2—looks simple, but those few elements packed together play a role in countless reactions, from classroom experiments with crystals to processes that help feed the world. Knowing the correct chemical formula isn’t just about passing an exam. It means you actually understand what’s happening during a reaction, whether you’re mixing chemicals in a beaker or treating plants in the field.
The Makeup of Zinc Nitrate
Take zinc, the metal you find in sunscreen or pennies. Pure zinc shows resilience but doesn’t dissolve in water easily. Pair it with nitrate, a group that comes from nitric acid, and you get a new compound that dissolves like sugar in water. The formula Zn(NO3)2 tells us that for every zinc ion, two nitrate ions complete the molecule. Nitrate’s nitrogen and three oxygens balance out zinc’s positive charge, forming a salt that travels well in solution. This ability doesn’t just impact test tubes—it’s what lets farmers deliver zinc to the roots through liquid sprays on their crops.
Real-World Uses and Safety Concerns
Growing up on a small farm, I often wondered why certain patches of our field grew stronger than others. Sometimes, the soil gets tired and simply can’t deliver enough zinc, even though it’s crucial for plant health. Adding zinc nitrate turned out to be a reliable fix because the compound dissolves fast, making zinc available to the plants right away. The formula Zn(NO3)2 gives you exactly what you need to figure out how much zinc to apply. Too much and you risk harming the soil and water supply; too little and you miss the benefit.
Quick solubility comes with a downside. The nitrate part can run off into waterways if handled carelessly. Nitrate pollution triggers algae blooms and saps oxygen from lakes and rivers, hurting fish and the people who depend on clean water. Proper handling and precise calculations based on the exact formula go a long way in preventing environmental headaches.
Supporting Responsible Use With Knowledge
Using zinc nitrate responsibly means knowing exactly what’s in your hands. Its chemical formula, Zn(NO3)2, doesn’t just guide people in labs but supports sustainable practices in agriculture and industry. Careful storage, clearly labeled containers, and proper training lower the chance of dangerous mistakes. The Environmental Protection Agency and local agricultural experts set guidelines for how much nitrate gets released into the environment. Following those rules protects both crops and communities.
Small steps make a difference. Wear gloves. Check the label twice. Double-check the math for your mixture based on the real formula. Education starting in classrooms can shift habits once those same students begin managing orchards or city parks. Accuracy isn't just a science requirement—it’s the foundation for any safe use of chemicals.
Solutions for Balanced Application
Precise measuring tools help prevent overuse, and ongoing soil tests catch problems early. Digital tracking can alert users to potential run-off risks, based firmly on the actual molecular formula in use. Community outreach shapes a safety-first culture from the ground up, rooted in basic knowledge—like understanding that Zn(NO3)2 stands for more than a label on a bottle. It stands for a commitment to safety, responsibility, and healthy growth on every level.
How do you safely handle and dispose of Zinc Nitrate?
Recognizing the Hazards
Zinc nitrate ends up in a lot of labs, high school classrooms, and certain industrial spots. Looking at a small bottle, it doesn’t seem very impressive, but appearances can mislead. It’s an oxidizing salt, it likes water, and you don’t want skin or eyes exposed. I’ve watched classmates ignore the “wear gloves” sign and end up with red, irritated skin. Vapors feel slightly irritating in a stuffy lab room, especially if someone heats it above room temperature. Breathing in any chemical dust is never ideal, and this one can trigger a scratchy throat or more.
Handling Zinc Nitrate Safely
Start with basic personal protective gear: snug-fitting chemical splash goggles, proper lab gloves like nitrile, and a lab coat. Never open a container of zinc nitrate unless a functioning eyewash station and emergency shower stand nearby. One day, our neighboring chemist got some on his hand while weighing out powder, which led to a scramble for the sink; water quickly diluted it, but it got everyone’s attention. If you ever spill a little, sweep it up only if you’re suited up. Never get complacent, even with routine tasks.
Zinc nitrate never goes down the sink. Water plus zinc nitrate, especially in quantity, eats through many metals and pipes by releasing nitric acid. I’ve seen corroded stainless drains after just a few weeks of careless rinsing. Always have dedicated, labeled containers for waste, and keep solutions far from sinks and drains. I store any excess powder in airtight, compatible bottles, labeled with both the content and date. Make sure it’s out of direct sunlight and away from combustibles, because zinc nitrate feeds fire if the right fuel appears.
Environmental and Health Risks
Zinc compounds crop up in environmental tests after factory runoff, and high nitrate levels pollute rivers downstream. Neither fish nor people do well when exposed to these pollutants over time. My town’s water board monitors nitrate because it triggers blue baby syndrome, and spills of compounds like zinc nitrate add to the total nitrate load. Fines for accidental runoff reflect the serious consequences—local industry learned this lesson the hard way ten years ago, spending thousands remediating a spill site.
Ongoing exposure in the workplace links to irritation of lungs, skin, and eyes. Over longer periods or higher doses, toxic metals strain the body’s organs. Writing safety audits, I noticed the most consistent issue: people underestimating long-term effects of “just a little exposure.”
Disposal: Doing It Right
Never treat chemical disposal as an afterthought. Solid zinc nitrate and liquid waste belong in sealed, properly labeled hazardous waste containers. At my university, only a licensed disposal company picked them up, carrying official paperwork and special storage bins in case of spills on the road. If your local rules allow, some treatment facilities neutralize small amounts in controlled conditions, adding strong base to precipitate zinc hydroxide for easier containment—but this step only works with training and proper gear.
Always check the local environmental rules—every state or country holds different expectations on toxic waste. I call the regional waste management agency for up-to-date directions, because regulations shift yearly. Don’t guess or follow online forums; fines and environmental damage cost far more in the end. Training everyone who handles chemicals saves headaches. Clear labeling and communication prevent mix-ups, whether in a classroom or in industry.
Making Safety a Habit
Chemical safety turns into second nature over time, but never let routine dull vigilance. Shortcuts invite spills and injuries. Gloves, goggles, and a little patience make all the difference for zinc nitrate and any other hazardous compound. Sharing these habits with each new student or hire makes labs and workplaces a little safer for everyone.
| Names | |
| Preferred IUPAC name | zinc dinitrate |
| Other names |
Zinc dinitrate Celloxan Zinc(II) nitrate |
| Pronunciation | /ˈzɪŋk ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 7779-88-6 |
| Beilstein Reference | 14608 |
| ChEBI | CHEBI:78067 |
| ChEMBL | CHEMBL1440524 |
| ChemSpider | 33146 |
| DrugBank | DB14544 |
| ECHA InfoCard | 100.028.294 |
| EC Number | 231-943-8 |
| Gmelin Reference | 604687 |
| KEGG | C01763 |
| MeSH | D015922 |
| PubChem CID | 24597 |
| RTECS number | ZS5820000 |
| UNII | N0IHT5XY8D |
| UN number | 1436 |
| Properties | |
| Chemical formula | Zn(NO3)2 |
| Molar mass | 189.41 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 2.065 g/cm³ |
| Solubility in water | 118 g/100 mL (20 °C) |
| log P | -0.2 |
| Acidity (pKa) | ~4.0 |
| Basicity (pKb) | 7.73 |
| Magnetic susceptibility (χ) | `+142 × 10⁻⁶ cm³/mol` |
| Refractive index (nD) | 1.557 |
| Viscosity | 2.15 cP (25 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 146.4 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -348.0 kJ/mol |
| Pharmacology | |
| ATC code | V06DA02 |
| Hazards | |
| Main hazards | Oxidizing, harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS02, GHS05, GHS07 |
| Pictograms | GHS03,GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H319, H410 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Autoignition temperature | 410 °C (770 °F; 683 K) |
| Explosive limits | Non-explosive |
| Lethal dose or concentration | LD₅₀ oral (rat): 1190 mg/kg |
| LD50 (median dose) | LD50 oral rat: 1190 mg/kg |
| NIOSH | ZS5425000 |
| PEL (Permissible) | PEL: 0.1 mg/m³ |
| REL (Recommended) | 1 mg/L |
| IDLH (Immediate danger) | 50 mg/m³ |
| Related compounds | |
| Related compounds |
Zinc sulfate Zinc chloride Zinc acetate Zinc carbonate Nickel nitrate |

