Zinc Methylarsonate: Where Chemistry, Risk, and Progress Meet
Historical Development: Lessons from Scientific Curiosity
Zinc methylarsonate didn’t show up overnight. Its story tracks alongside a century filled with changing attitudes about chemicals and their impact, for good and for harm. In the early decades of the 20th century, chemists played with arsenic compounds for everything from agriculture to medicine. Zinc’s inclusion brought new promise, offering pesticidal action and disease management properties. People used to reach for arsenic-based chemicals to tackle blight and pests in the fields. Zinc methylarsonate became one of those "silver bullet" solutions in farming, often celebrated before anyone counted the long-term costs. It serves as a lesson—science can introduce helpful tools, but the wisdom to question and monitor them has to keep pace.
Product Overview: What Sits in the Drum
Ask chemists what makes zinc methylarsonate notable, and the answer goes beyond its formula. You're dealing with an organoarsenic compound—here, both methyl and zinc cations bound to arsenate. In practice, the compound usually appears as a fine, whitish or off-white powder. It dissolves reasonably well in water, making it easy to apply, whether in agricultural rows or fruit orchards decades past. The substance earned attention because it combined arsenic’s power with what seemed like manageable handling properties, unlike some of the more poisonous and volatile arsenic relatives. Yet, arsenic lingers. Its tendency to accumulate brings long-term risks—a warning that can get muffled under the pressure to protect crops or increase yield.
Physical & Chemical Properties: What You See, What You Don’t
Zinc methylarsonate tells its story through its physical nature. Its powder form makes it easy to disperse, but that also means a steady risk of dust inhalation. It absorbs water, and its solubility means ground and surface water can pick up traces where rainfall or runoff carries it. Chemically, it stands as a salt; the methyl group changes arsenic’s typical behavior, sometimes making it more mobile. The addition of zinc tweaks the compound’s toxicity profile, but it never erases the deep-seated concerns over arsenic exposure. Chemists measure its performance as a pesticide by its stability and interaction with soils. Factors like soil type, pH, and organic content influence how fast the compound breaks down, whether it leaches, and how long it sticks around—a detail that matters if you try to restore land for other uses later.
Technical Specifications & Labeling: The Devil in the Details
Back in the day, the technical labeling of zinc methylarsonate products spelled out the arsenic content, instructions for mixing with water, target pests, and warnings about skin or eye irritation. Standards for labeling grew stricter after evidence built up around arsenic’s long-term health risks. Today’s modern chemical stewardship insists on clear hazard identification, emergency procedures, and information on safe disposal. Accurate technical labeling isn’t just a formality; it shapes field practices and provides a line of defense for those who use or may be exposed, especially when the lines blur between "worker" and "neighbor."
Preparation Method: Making a Powerful Yet Risky Tool
Manufacturing zinc methylarsonate means coaxing a series of reactions to yield a material useful enough for pesticides, but hazardous if mishandled. It starts with arsenic acid—reacting it with zinc salts under controlled conditions. Methyl groups get introduced using reagents like methylating agents, sometimes under heat and with specialized equipment to capture any volatile arsenic byproducts. It's a simple recipe in principle, but the toxicity of raw materials requires more than a backyard setup. Each part of the process calls for strict ventilation, waste management, and controls against leaks, and that infrastructure raises the entry bar—at least in theory—keeping use confined to regulated settings. The sector's historic corners might tell another story, with small producers sometimes cutting corners before regulations clamped down.
Chemical Reactions & Modifications: A Double-Edged Sword
Chemically, zinc methylarsonate interacts with the world in ways that can scramble intentions. Exposed to sunlight, water, acids, or biological material, the molecule can break down—not always into harmless fragments. The methyl group can shed off, liberating inorganic arsenic, often more poisonous and persistent. Interactions with soil microbes sometimes speed this transformation. Modifications aiming to tweak its solubility, breakdown rate, or selectivity for pests have appeared in research literature, but the underlying risk echoes through every variant: you can’t simply dial down arsenic’s legacy by swapping molecular add-ons. This tug-of-war between utility and safety shapes every subsequent application and regulation effort.
Synonyms & Product Names: Names Hide Nothing
Walk through a chemical inventory, and you'll spot synonym after synonym for zinc methylarsonate: "Methyl arsonic acid zinc salt," "Zinc methylarsenate," and trade names varying by region or manufacturer. For decades, those names ended up in registries and on purchase orders, sometimes under an alphabet soup of labels intended more for regulatory navigation than transparency. For farms and orchards, any product name marked with "arsenate" or "arsenic" should ring warning bells. The diversity of naming conventions can make tracking environmental impact or historic use a real slog. Researchers and environmental cleanup teams spend too much time decoding old inventory sheets just to identify what’s leaching into groundwater.
Safety & Operational Standards: Hard Lessons Learned
Zinc methylarsonate brings forward stark lessons about workplace safety and environmental stewardship. Arsenic compounds require more than gloves and goggles—a full PPE suite, ventilation, and meticulous storage all reduce immediate risk. Dust management stands out; minute airborne particles can bypass even good intentions. The operational standards set decades ago often fell short, based mostly on acute, short-term effects. Since then, tightening rules demanded engineering controls, exposure monitoring, and ground-level training, particularly in legacy applications where contaminated soils or old stockpiles linger. Emergency protocols keep evolving as research uncovers new long-term impacts, recognizing that accidents or improper disposal haunt communities downwind or downstream. Many regions now outright prohibit manufacture, sale, or use of this compound, underlining the high bar for trust in chemical safety.
Application Area: From Fields to Environmental Concern
Initially, agriculture drove the adoption of zinc methylarsonate—orchards used it to manage insects, diseases, and sometimes rodents. Its solubility and perceived stability looked like a blessing. Only over time did arsenic's persistence in soils and potential to contaminate water supplies add complication. Modern agriculture nearly abandoned these arsenicals, learning the environmental cost far outweighs any short-term win against pests. Some environmental testing labs and soil remediation teams now focus on legacy sites, tackling the long tail of its application with remediation work that can take years. Its use in research as a chemical precursor or reference standard lingers, but never without layers of specialized controls.
Research & Development: Science in Search of Safer Tools
After regulatory bans hit, research on arsenic compounds like zinc methylarsonate shifted gears. Labs switched focus from tweaking the molecule for use in pesticides to finding ways to detect, neutralize, and remove remnants from the environment. Newer analytic methods can spot contamination at trace levels, charting migration through soil and water. Some researchers try to design "greener" molecules that do a similar job against plant pests yet break down quickly and harmlessly—a goal still more aspiration than reality. Remediation science, not product development, drives most new published work: techniques like phytoremediation, soil washing, and microbial treatment are in the spotlight. Ongoing projects also probe sub-lethal exposure effects in humans, animals, and beneficial insects—sometimes uncovering risks that the initial crop protection focus ignored.
Toxicity Research: Unpacking the Damage
Arsenic’s threat isn’t news to anyone who’s followed public health debates. Zinc methylarsonate builds on that legacy, delivering both acute and chronic hazard through every route—skin, lungs, and, far too often, drinking water. Scientists now understand that chronic, low-level exposure underpins cancer, nerve disorders, cardiovascular issues, and developmental harm for children, even at concentrations believed "safe" just a generation ago. Data show that breakdown products in soil can outlast the original application by decades, especially in poorly managed sites. Regulators, health professionals, and environmental groups still grapple with testing protocols, cleanup technologies, and public notification systems robust enough to address sources that keep popping up in legacy farmland or industrial sites.
Future Prospects: Toward Health, Transparency, and Environmental Repair
There’s little appetite anywhere in the world for resurrecting agricultural use of zinc methylarsonate. The focus now points toward vigilance and healing. Governments and environmental agencies prioritize mapping and isolating historic contamination, sometimes with new biological treatment methods that promise more affordable and sustainable remediation. Health professionals push for tighter drinking water standards and for outreach in communities sitting atop tainted groundwater. For scientists, the challenge isn’t in reengineering zinc methylarsonate itself, but in developing truly safe, effective ways to ward off pests and disease without sewing new risks into the soil or water for another generation to deal with. The conversation moves well beyond this single compound—it’s about learning from past missteps and committing to transparency, rigorous science, and the sort of stewardship that keeps both people and ecosystems in mind.
What is Zinc Methylarsonate used for?
An Old Solution with New Questions
Zinc methylarsonate isn’t a word that pops up at the local hardware store. Still, many in agriculture and plant science recognize it for its history and weight. This chemical found its way into the fields as a pesticide, especially in fruit crops like apples and pears. It’s a blend of two elements that both impact plant health and pest control: zinc, which is an essential micronutrient, and arsenic, a toxin for many forms of life. The combination delivers a punch to certain pests that can damage crops, helping farmers avoid big losses.
Why Zinc Methylarsonate Mattered
Farmers have always looked for practical ways to keep their crops healthy. Zinc methylarsonate promised reliable pest management. For decades, orchardists sprayed and dusted this compound on trees to fight off troublesome insects. According to data from the mid-20th century, these arsenical pesticides reduced the loss of apple and pear harvests to codling moth larvae—a major agricultural pest. That kind of impact isn’t something to take lightly. One bad cycle with the wrong bug can wipe out a year’s work and investment.
Serious Risks and Shifting Priorities
Using something that contains arsenic brings risks. Science didn’t always have the tools to study what went into the air and soil. In the last forty years, we gained a stronger understanding of how arsenic contaminates land, water, and living things. Long-term exposure links to several cancers, nerve damage, and developmental issues in children. Older orchards sprayed with arsenical pesticides showed raised soil arsenic levels decades later, putting farm workers and nearby residents at risk.
I’ve walked through some of those older fields myself and questioned the history under my boots. If the ground keeps old chemical residues, that changes how we think about food safety, groundwater protection, and property values. The EPA finally banned most arsenical pesticides for food crops in the 1980s. Zinc methylarsonate is now tightly restricted or off the market altogether across many countries. Farmers must consider both short-term pests and the long-term health of their land.
The Challenge of Chemical Legacy
The problems didn’t vanish with the ban. Old orchards may still contain soil with arsenic levels high enough to trigger concern. Some universities and extension offices have published guidance for testing and remediation. Techniques like removing topsoil, planting grasses, or growing crops that don’t readily absorb arsenic have become part of the toolbox. These efforts come with real cost, but ignoring old chemical residues is not an option. It’s about protecting families, farmworkers, and water supplies.
Better Solutions Going Forward
The story of zinc methylarsonate reminds us that one generation’s solution can be the next generation’s headache. Integrated pest management, which uses a mix of biological, cultural, and limited chemical controls, has stepped in. Instead of relying on harsh compounds, farmers shift to safer pesticides, beneficial insects, and farming techniques that keep pests in check with less risk to health or the land. Regulations have grown stronger, and both buyers and growers push for greater transparency about what ends up in our food system.
Zinc methylarsonate played a big role in controlling pests, but its risks led to its phase-out. Today, the conversation moves beyond quick fixes, putting a spotlight on stewardship and sustainability. Those lessons shape what gets sprayed, planted, and consumed on farms everywhere.
Is Zinc Methylarsonate hazardous or toxic?
Understanding What Zinc Methylarsonate Is
Zinc methylarsonate doesn’t show up on the shelf at your local hardware store, and most folks have never heard of it. This compound’s roots trace to agricultural use, where it has popped up as a pesticide. Arsenic catches attention straight off—the word raises red flags, and with good reason. For decades, scientists have warned the public that many arsenic-containing products come with big risks. This isn’t just theory; these risks have been proven in many studies around the world.
Health Concerns Tied to Arsenic Compounds
Experience speaking: Growing up in a farming community, the stories about old arsenical pesticides made people uneasy. The old timers knew something was off when neighbors started getting rashes, coughing, or even worse illnesses after spraying these chemicals around the orchard. That skepticism got backed up by science. The World Health Organization highlights that arsenic exposure, even at low levels over time, can trigger a wide range of health problems—skin damage, cancer, blood pressure shifts, and both lung and nervous system issues.
Zinc methylarsonate is no different in this respect. It contains arsenic. Ingesting or regularly inhaling dust or droplets containing this compound puts people at real risk. It won’t show its effects overnight, but months or years of exposure can chip away at organ health—especially for kids or anyone with compromised health to begin with.
Environmental Muck Left Behind
Walking through old orchards, it’s still possible to spot patches where nothing seems to grow quite right. These spots mark where arsenic compounds—zinc methylarsonate among them—got dumped or lingered in the soil long after use. The residue doesn’t stay put, either; heavy rain carries it into ponds and streams. Fish, birds, pets, and people all get exposed, sometimes without knowing. Soil and water testing confirm it: arsenical compounds stick around, and their impact lingers for years.
Regulation and Alternatives
Many governments have cracked down on these compounds. The Environmental Protection Agency flagged these kinds of substances as unsafe, and the phase-out of arsenic pesticides in the U.S. started rolling long ago. The bans rely on studies that measured arsenic levels in food, water, and even children’s hair near treated areas. Zinc methylarsonate does not get a free pass, since its molecular structure lets arsenic easily break free in the environment.
Plenty of options exist for folks needing pest solutions without the fallout: neem oil, insecticidal soap, and integrated pest management all steer clear of arsenic. Nobody wants to trade a short-term win in pest control for a long-term hit to health or clean water. The science points to precaution as the clear approach. That’s what E-E-A-T means here—listening to the experts, learning from the past, and passing that knowledge to the next grower, gardener, or policymaker.
Moving Forward With Caution
Honest experience and data show that taking chemicals with “arsenate” in the name too lightly carries risks not worth the gamble. Families deserve peace of mind about what’s in their soil and water. Any product with a toxic track record should stay out of reach, no matter how tough the pest problem appears.
What is the chemical formula of Zinc Methylarsonate?
Understanding Zinc Methylarsonate
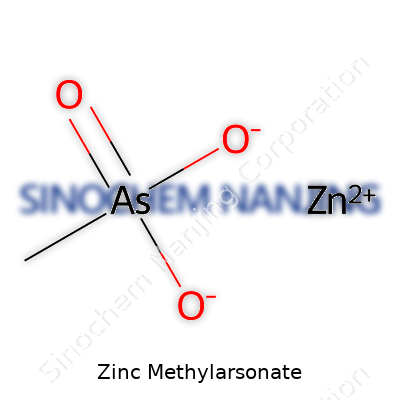
Zinc methylarsonate stands out in both agricultural and chemical circles. The formula, Zn(CH3AsO3)2, tells a story about combining zinc with methylarsonic acid. This compound has drawn attention because parts of its chemistry—arsenic and heavy metals—raise real-world concerns. Zinc, a common element in many supplements and industrial products, partners here with a methylated arsenic group. You don’t run across these hybrid compounds in daily life, but their reach extends into soil science and environmental safety.
How the Formula Shapes Its Use
Knowing the formula breaks down the compound into its building blocks: one zinc atom, two methyl groups, and two arsenic atoms paired with oxygen. For farmers and researchers, this kind of chemical structure shaped controversial uses in pesticides and herbicides during past decades. Because arsenic carries toxicity risks, officials and scientists flagged these arsenicals even as they found them effective in tackling pests once hard to control. In the field, methylated forms of arsenic can act differently than inorganic arsenic species. As a chemist, I’ve studied similar transformations in lab soil columns and noticed how complex the breakdown pathways get—especially after repeated exposure or weather changes.
Risks That Come With This Formula
The formula itself explains why people worry. Arsenic, even in an organic methylated form, can move through soil and water. Research from the US Environmental Protection Agency shows that methylarsonates can convert to more mobile and toxic inorganic arsenic forms under certain microbial or acidic conditions. Years ago, the shift to organoarsenic compounds seemed like a step up from older arsenic-based chemicals, but the long-term story shows lingering contamination in old orchards and farmlands. Neighbors who live near old application sites often ask if their garden crops are safe to eat—it all traces back to this chemistry and the legacy of how these molecules travel and change.
Digging Into Regulation and Safer Alternatives
Today, you won’t see new approvals for zinc methylarsonate-based products. Most regulatory agencies pulled support after recognizing the environmental and health impacts. The European Union and US banned or phased out these formulations, but residues remain across regions once considered prime for fruit production or cotton farming. In my community, we've faced challenges cleaning up soil that tests positive for arsenic weeks after record rainfall. Cooperative extension offices often hand out booklets, but people want real answers—remediation isn’t as simple as removing the topsoil, not with water tables and deep roots involved. Recent research looks to phytoremediation and bioremediation to reduce arsenic mobility, relying on plants and microbes to break down or isolate what’s in the ground.
The Path Forward
Understanding the formula ties straight to practical impacts. Farmers and scientists push for better risk communication, more affordable soil testing, and long-term support for affected areas. Open dialogue, combined with clear, science-backed policies, gives people more control over their environment. As conversations shift toward sustainability and regenerative agriculture, the lessons from zinc methylarsonate remind us how the periodic table still affects what ends up in our food and water.
How should Zinc Methylarsonate be stored and handled?
Why Precautions Matter
Zinc methylarsonate often pops up in labs and in specialized chemical settings. This compound carries some heavy risks for people and wildlife. I’ve spent time in facilities where even a minor mistake with toxins led to serious headaches. One slip—unlabeled containers, a leaky cap, a room that’s too warm—can threaten everyone.
Safe Storage Isn’t Just Box Checking
Storing zinc methylarsonate means thinking about more than just shelves and cabinets. Small choices build up to real safety. I always start with a well-ventilated space. This keeps unexpected fumes from gathering and creates a line of defense against accidental inhalation.
Every bottle and drum needs tight, reliable seals. Strong containers cut down on leaks and slow any spill that does happen. Using glass or HDPE plastic bottles means fewer surprises, since some plastics break down if exposed to fouler chemicals over time. Labels must be clear and permanent. Faded marker or torn tape invites confusion—someone working a double shift might grab the wrong bottle and land in trouble.
Temperature and light control play their part
Heat can change chemical properties and turn a routine job into an emergency. I never store zinc methylarsonate near boilers, heaters, or sunny windows. Cool, dry spots, especially behind lock and key, keep everything stable. Dryness cuts the chance of accidental mixing with water—wet conditions can set off unwanted reactions or corrode the packaging.
Protective Gear: More than a Suggestion
Handling this stuff, even for a minute, means putting on the right protection. No one should approach zinc methylarsonate without gloves, eye shields, and a solid lab coat. I’ve seen raw skin burns and eye injuries happen fast—just from skipping a step because “it’s just a quick transfer.” If a spill happens, a well-fitted mask and extra gloves mean a faster, safer cleanup.
Waste and Spill Response
Steps for dealing with waste make a difference. Specialized disposal bins, not regular trash, give assurance that this compound won’t sit in a landfill leaking poisons. Labeling waste bins and keeping them at hand lowers the risk of someone dumping things down the drain. I’ve learned to keep emergency cleanup supplies nearby: absorbent powders, neutralizing agents, and a documented spill plan. Proper training goes a long way. Knowing how to grab the right kit and where to find it stops small accidents from growing large.
Training and Responsibility
Taking shortcuts is easy when you’ve handled something dozens of times. Regular training sessions for every team member—including temps and night shift—help reinforce good habits. Reviewing procedures and sharing updates from trusted health agencies like the CDC or OSHA strengthens a safety mindset. If a visitor or new worker steps in, I walk them through every control and safety rule, instead of assuming they already know.
Building a Culture of Care
True safety with zinc methylarsonate doesn’t come from rules alone—it grows from experience and mutual care. Looking out for each other, double-checking lockups, and replacing worn labels or seals all reduce the odds of trouble. Nobody wants to pay the price for a preventable mistake, least of all with a substance as potent as this one.
Turning Lessons Into Solutions
Better records, smart storage, personal shields, and open conversations build stronger routines. Relying on cheap supplies or skipping training costs more in the long run. Treating every hazardous chemical with respect and focus, day in and day out, creates a safer workspace for everyone involved.
Where can I purchase Zinc Methylarsonate?
Understanding the Search for Zinc Methylarsonate
Interest in zinc methylarsonate often comes up from farmers, researchers, and, sometimes, collectors studying historical pesticides. My experience working with specialty chemical distributors taught me that not every compound, even with a long history, ends up easily sourced. That’s especially true for something like zinc methylarsonate, which carries significant baggage because of its chemistry.
Tracing the Path: Safety Over Convenience
Zinc methylarsonate served as a pesticide decades ago, mainly for treating certain plant diseases. Governments and science communities eventually recognized how arsenic compounds posed huge risks—not just to those applying them, but also to groundwater and anyone eating food out of previously treated soil. If someone asks where to get this chemical, they're likely to run into a wall of regulations before even discussing a price.
Why It’s Hard to Find a Supplier
Market demand completely dried up once safer options hit the shelves. These days, legitimate chemical suppliers rarely stock things like zinc methylarsonate for everyday purchase. Many companies see the risk far outweighing any potential profit. Federal agencies, like the EPA in the US, flagged products containing arsenic decades ago. Even ordering it for a legitimate purpose feels like wading through bureaucratic red tape—licenses, background checks, proved intent, specialized storage—all come into play.
Ethics and Responsibility in Hazardous Chemicals
I remember consulting for a lab that had to dispose of inherited old stockpiles. We discovered barrels of legacy chemicals, including arsenic compounds. The disposal costs, on top of the paperwork, doubled because improper handling had already caused some minor soil contamination. This is a wake-up call for anyone thinking about tracking down banned or controlled substances—danger doesn’t fade just because a law got passed. Real people and real communities still carry the burden years later.
Considering Alternatives Makes More Sense
Farmers and gardeners looking to control pests should talk to agricultural extension services or crop specialists. They have up-to-date solutions that offer better results without risking public health or falling foul of the law. Research labs, especially those with historical interests, work directly with established chemical repositories or government institutions—places structured to handle licensing and documentation.
The Path Forward Isn’t DIY
Trying to buy zinc methylarsonate by searching popular chemical shops or overseas online vendors isn’t just a questionable move—it puts one at legal risk and, potentially, ethical trouble too. Smuggling dangerous compounds creates a chain of problems, from shipping hazards to exposure risks along every step.
Instead, approach acknowledged outlets linked to universities or industrial chemical suppliers, not fly-by-night web shops. If a project can only proceed with this compound, the next call should be to the country’s regulatory authority. In the US, that’s likely state-level departments of agriculture or the Environmental Protection Agency. It takes effort and clear documentation, but that’s for good reason—the safety net helps more than it delays.
| Names | |
| Preferred IUPAC name | zinc;[(OS(=O)(=O)[O-])=As]C |
| Other names |
Methylarsonic acid, zinc salt Zinc methylarsonate (Zn(CH3AsO3)2) Zinc methylarsenate |
| Pronunciation | /ˈzɪŋk ˌmɛθ.i.ˈlɑːr.sə.neɪt/ |
| Identifiers | |
| CAS Number | 12007-89-5 |
| Beilstein Reference | 527873 |
| ChEBI | CHEBI:86464 |
| ChEMBL | CHEMBL1233547 |
| ChemSpider | 27243 |
| DrugBank | DB14606 |
| ECHA InfoCard | 100.033.777 |
| EC Number | 222-695-6 |
| Gmelin Reference | 29811 |
| KEGG | C18607 |
| MeSH | D015837 |
| PubChem CID | 24846947 |
| RTECS number | ZH6475000 |
| UNII | 9A37X2053O |
| UN number | UN1437 |
| Properties | |
| Chemical formula | C2H6As2O8Zn |
| Molar mass | 323.301 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | Density: 3.4 g/cm³ |
| Solubility in water | Soluble |
| log P | -1.4 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 3.6 |
| Basicity (pKb) | 7.75 |
| Magnetic susceptibility (χ) | -53.0e-6 cm³/mol |
| Refractive index (nD) | 1.682 |
| Viscosity | Viscous liquid |
| Dipole moment | 2.42 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 200.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -821.9 kJ/mol |
| Pharmacology | |
| ATC code | V03AB06 |
| Hazards | |
| Main hazards | Toxic if swallowed, harmful if inhaled, causes skin and eye irritation. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06, GHS08 |
| Signal word | Danger |
| Hazard statements | H300 + H330: Fatal if swallowed or inhaled. |
| Precautionary statements | P202, P261, P264, P270, P272, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P321, P330, P362+P364, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 2-2-3-W |
| Lethal dose or concentration | LD50 (oral, rat): 900 mg/kg |
| LD50 (median dose) | LD50: 15 mg/kg (rat, oral) |
| NIOSH | ZE6550000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Zinc Methylarsonate: 0.5 mg/m³ (as As) |
| REL (Recommended) | 5-10°C |
| IDLH (Immediate danger) | 5 mg As/m3 |
| Related compounds | |
| Related compounds |
Cacodylic acid Disodium methyl arsenate Sodium arsenite Zinc arsenate |

