Zinc Gluconate: From Discovery to Modern Use
Historical Development
Zinc gluconate emerged from the intersection of nutrition science and pharmaceutical needs in the mid-20th century. Researchers looking to combat widespread zinc deficiency started exploring compounds that offered both high solubility and a mild taste. Early chemists found that gluconic acid, a derivative of glucose, formed a stable complex salt with zinc. By joining the body’s need for trace minerals with this organic acid, zinc gluconate became a widely accepted supplement by the late 1970s. Drug manufacturers favored it for its ease of formulation. As awareness of micronutrient deficiencies grew, zinc gluconate found its way from the pharmacy shelves of North America to global supply chains. Authorities eventually recognized its potential, allowing it into official pharmacopeias and food supplement guidelines. Historic patents tell the story of incremental improvements around purity, particle size, and process scale-up. Regulatory armed with years of safety data, turned zinc gluconate from a nutraceutical curiosity into a trusted everyday aid.
Product Overview
The compound stands as a white to off-white powder with a slightly bitter taste, making it fit for direct tableting or blending in lozenges and syrups. Manufacturers offer it in granular or fine powder forms, with solubility that meets modern beverage and oral care formulation needs. Customers rely on its solid stability against heat and light, giving it a longer shelf life than some other zinc salts. Each batch represents a balance of economic production and rigorous quality testing. Both food and pharmaceutical industry players seek zinc gluconate with tightly controlled levels of lead, arsenic, and cadmium, and they demand records on every lot to satisfy global regulators. Product portfolios typically cater to dietary supplement makers and over-the-counter remedy producers who value ease of flavor-masking and blandness over more metallic salts.
Physical and Chemical Properties
Zinc gluconate has a molecular formula of C12H22O14Zn, blending organic and mineral chemistry. With a molar mass around 455 g/mol, the compound carries a faint odor and fully dissolves in water at room temperature. Its pH in aqueous solution sits between 5.5 and 7.5, matching physiological needs and reducing irritation potential. Density falls just under 1.8 g/cm³. Crystals often appear needle-like under magnification. Thermal decomposition generally starts above 170°C, producing trace zinc oxide and water. Chemists respect its low hygroscopicity, which means the compound doesn’t clump easily. These predictable traits give formulators confidence that each run of product will perform consistently across pharmacy and food supplement products.
Technical Specifications and Labeling
Strict quality standards address purity, heavy metal content, and microbial control—critical considerations for pharmaceutical and food companies. Assays require zinc content above 12% by mass, with loss on drying limited to below 11%. Verified suppliers track lead under 2 ppm and report negligible levels of mercury and cadmium. Particle sizes typically range from 75 to 200 microns for most uses, tailored to the needs of liquid dispersions or compact tablet presses. Labels on consumer packs report net weight, expiry date, and clear instructions on safe storage. Global guidelines from the US Pharmacopeia and European Pharmacopoeia identify minimums and maximums, and compliance means passing regular audits and documentation checks. Formulators know that regulatory clarity on dosage and composition beats uncertainty any day, especially in global trade.
Preparation Method
Production starts with gluconic acid fermentation, using bacterial strains that convert glucose to gluconic acid in large steel tanks. Once purified, gluconic acid reacts with high-purity zinc oxide or zinc carbonate in water under controlled pH. After neutralization, the zinc gluconate crystallizes from solution, and vacuum dryers remove residual moisture. Each step—fermentation, reaction, washing, centrifugation—operates in food-grade conditions. Standard filtration captures impurities and bacteria. Final packaging in air-tight bags prevents contamination. The whole operation relies on lot tracing and sample retention, so if a quality issue surfaces downstream, the source can be pinpointed. Well-trained operators trust their experience more than any manual, knowing how color, odor, and texture forecast quality.
Chemical Reactions and Modifications
The salt’s chemistry centers on zinc binding with gluconate’s carboxyl groups, leading to chelation that increases bioavailability. In pharmaceutical applications, the compound blends with excipients and sweeteners, relying on hydrogen bonding for tableting. Dissolution in water produces free zinc ions and gluconate anions, offering quick absorption through the intestinal lining. Modifications come in particle size reduction, coating with film formers to mask taste, and blending with vitamin C to boost absorption. Complexation with amino acids or other chelates sometimes follows, but gluconate remains a favorite for its safety, predictability, and neutral sensory qualities. Reactions with alkaline or strongly acidic ingredients do cause decomposition, so careful formulation matters, particularly in multi-ingredient products.
Synonyms and Product Names
Industry players know zinc gluconate under several names depending on market and manufacturer. These include Zinc D-gluconate, GZF, and E578—its European food additive code. Trade names sometimes play off ‘Nature’s Zinc’ or ‘ZincVital’ for supplements, and generic forms fill the bulk commodity market. Multinational players ensure labeling in multiple languages to follow national guidelines. Ingredient lists simply state “zinc gluconate,” avoiding confusing the end user, but technical sheets share fuller details to satisfy procurement officers and R&D labs.
Safety and Operational Standards
Evidence-based regulatory frameworks shape how the supplement industry and pharma handle zinc gluconate. FDA rules cap elemental zinc levels in over-the-counter tablets, partly to avoid the risk of toxicity with chronic intake. Good manufacturing practice (GMP) standards focus on equipment hygiene, cross-contamination control, and air quality in production areas. Workers handle raw zinc compounds with gloves and masks to limit dust inhalation, as chronic zinc exposure sometimes affects respiratory health. Warehouses set strict humidity controls, as shelf life depends on keeping the powder dry. Labels warn consumers not to exceed the stated dose, and emergency protocols prepare staff for accidental spills or overexposure. Adherence to ISO quality standards often separates reputable suppliers from shortcuts and recalls.
Application Area
Doctors and nutritionists recommend zinc gluconate mostly to address early cold symptoms, as lozenges and syrups deliver zinc where viruses first enter. Chewables join immune support and children’s vitamins, as the mild flavor fits picky palates. Sports nutrition brands tout zinc’s role in muscle repair and testosterone balance, packaging it as a key part of daily multivitamin blends. Animal nutrition and fortification of infant formula add to demand, especially in regions facing stunted growth due to mineral deficiencies. Researchers have studied its role in wound healing, and hospital protocols sometimes introduce it for patients with recovering tissue injuries. Toothpaste and oral rinses include the compound for its action against gum inflammation. Industrial chemists have even explored it as a mild anti-corrosive in coatings, since the compound leaves minimal residue and doesn’t harm water systems.
Research and Development
Scientists continue to test zinc gluconate’s reach, from improved absorption to compatibility with probiotics and natural flavorings. Trials in controlling the severity and duration of respiratory illnesses keep research teams busy, as does work investigating zinc’s effect on taste modulation and the sense of smell. Advances in microencapsulation technology promise cleaner flavor profiles and timed release. Development teams focus now on optimizing granulation behavior for direct compression and reducing dust formation during handling. Some labs collaborate with genetics experts to individualize supplement recommendations based on absorption genes. Environmental researchers look for greener synthesis through biocatalysts in fermentation, which could further cut manufacturing emissions. Intellectual property filings often target these avenues, aiming to offer more effective and appealing zinc supplements than previous generations.
Toxicity Research
Toxicology studies confirm that zinc gluconate has a wide margin of safety at recommended intake levels, with nausea and gastrointestinal upset showing up only with large overdoses. Animal studies pinpoint a no observed adverse effect level (NOAEL) around 100 mg elemental zinc per kg bodyweight per day. Regulatory panels, including WHO and EFSA, land on a tolerable upper limit for daily adult intake near 40 mg as a precaution. Research has noted transient loss of taste—anosmia—in rare cases involving high zinc lozenge use. Laboratory evaluations weigh risks of chronic exposure in industrial settings, with attention given to kidney and immune system markers. Some toxicological inquiries shift toward nanomaterials containing zinc, but the bulk of the literature supports the safety of the established gluconate salt when used as directed. Still, public health voices caution against long-term unsupervised use, as zinc competes with copper metabolism and may tweak cholesterol levels in sensitive populations.
Future Prospects
Looking ahead, demand for zinc gluconate seems set to climb with rising awareness about micronutrient support, immune health, and non-antibiotic remedies for colds. Manufacturers are eyeing innovations that fine-tune particle size and boost palatability. Consumer pushback against high-sugar supplements is spurring sugar-free and natural sweetener formats. Regulatory changes in global markets, especially China, could affect supply dynamics and price. Scientists are tracking bioavailability data from personalized nutrition trials, which may shift how products are marketed and prescribed. Environmental concerns about resource use have driven some producers to adopt renewable energy and greener process chemicals, which could appeal to eco-conscious buyers. As public health campaigns continue to stress the role of essential minerals, zinc gluconate holds its place both on pharmacy counters and in food fortification schemes, promising to adapt as the science and market expectations change.
What is zinc gluconate used for?
A Look at Zinc Gluconate’s Purpose
Zinc often gets overlooked, but it has always mattered to our bodies. Zinc gluconate, one of its most common supplements, steps in when the daily diet falls short. Many people see it on pharmacy shelves, tucked inside cold remedies, lozenges, and multivitamins. I remember reaching for these lozenges during the winter, hoping to ward off a lingering scratchy throat. There’s a good reason for its popularity. Studies show zinc impacts the immune system, wound healing, and even the senses of taste and smell.
Zinc deficiency can sneak up. It happens more often in people following restrictive diets or among seniors. The body doesn’t store excess zinc, so it’s easy to run low if each meal lacks variety. In my own family, a loved one struggled with slow wound healing—a visit to their doctor led to a simple blood test and, after a few weeks on a zinc supplement, their immune response improved. As reported by the National Institutes of Health, nearly 10% of Americans don’t meet zinc recommendations, making it relevant for a huge part of the population.
Immune Support, Not a Cure-All
Many discover zinc gluconate because of its starring role in cold remedies. Research published in scientific journals points out that zinc plays a direct part in the development and function of key immune cells. Some studies link early zinc lozenge use to fewer days of annoying nasal symptoms during a cold. This isn’t a miracle cure; results vary, and taking too much in pursuit of a quick fix can upset the digestive system. As with any supplement, moderation matters most.
Zinc gluconate works for more than cold relief. Doctors sometimes recommend it for people undergoing treatments that impact taste, like chemotherapy. In clinics around the country, zinc helps restore the sense of taste, improving life’s small pleasures like morning coffee or a favorite dessert. In the world of skincare, it often features in regimens for acne-prone skin, although more research is underway to sort out the benefits. Everything points to zinc’s importance, but each body is different, and needs change over a lifetime.
Choosing Supplements with Care
Supplements promise quick results, but not all are created equal. The Food and Drug Administration regulates them differently than prescription medicines. Reputable brands anchor their products in quality testing and transparent sourcing. Real experts—registered dietitians or physicians—should always guide decisions about using a zinc supplement. Overdosing can lead to nausea or interfere with other minerals like copper, underscoring the need for balance. Some multivitamins contain enough zinc for daily needs, so most people don’t need extra unless they have a diagnosed deficiency.
Keeping Health Simple and Honest
Zinc gluconate fills the gap when real life makes it hard to eat a balanced, nutrient-rich meal. It isn't a one-size-fits-all fix, but for some, it’s a necessary boost. Focusing on food first—nuts, beans, meats, and dairy—often covers most zinc needs. Supplements support, not replace, a good foundation. Honest advice and simple choices—like checking with a professional before adding new pills to the mix—make all the difference for lasting wellness.
What are the benefits of taking zinc gluconate?
What Draws People to Zinc Gluconate?
I often notice lots of folks reaching for zinc during cold season. That’s not just old wives’ wisdom. Zinc is one of those minerals most diets do not quite provide in full, especially if you go heavy on processed foods. Zinc gluconate lands on shelves because it’s easier on the stomach than other forms of zinc and absorbs pretty well.
The body can’t make zinc, so we get all we need from food or supplements. Shellfish, beef, and lentils bring it in, though not everyone gets enough from their plate. Relying on a supplement like zinc gluconate covers the gap for vegans, vegetarians, and anyone skipping red meat.
Immune Health: More Than Just a Buzzword
Research shows that zinc supports immune response in several ways. Cells rely on zinc when gearing up to fight off bacteria and viruses. Low zinc spells trouble: cuts heal slower, you get sick more easily, sense of smell and taste dips. The Journal of Family Practice shared evidence that taking zinc gluconate at the start of a cold can shorten symptoms by about a third. It won’t erase your sniffles overnight, but it does seem to help most people recover faster.
Doctors and nurses watch zinc levels closely in older adults and people with chronic sickness. The body’s ability to absorb nutrients tends to drop with age or certain stomach issues, leading to hurdles in recovery. Keeping zinc in check can be one part of staying out of the hospital during flu season.
Wound Repair and Skin Health
People with cuts or burns heal slower without adequate zinc. Growing up, my grandmother used calamine for bug bites—turns out, that white layer is zinc carbonate, a cousin of zinc gluconate. Skin cells need zinc to grow and clot. Moderate supplement use helps with acne, eczema, and sometimes diaper rash in babies. The American Academy of Dermatology singles out zinc as key in skin barrier function, especially for people battling persistent acne outbreaks.
Supporting Brain and Hormone Function
Zinc supports enzymes tied to brain health and memory. A study in the journal “Nutrients” links zinc deficiency to mood swings and even depression. For men, zinc has a role in testosterone balance as well. I’ve talked with coaches at the gym who recommend zinc for muscle recovery and energy, especially after intense training.
Plant-based eaters often don’t get enough zinc, since phytates in grains make absorption tougher. Zinc gluconate comes into play for anyone missing out on animal foods or following a heavily restricted diet. It’s a safety net for those not getting zinc from meals.
How Much Is Enough?
Standard adult dose sits around 8-11 mg daily, though some folks take more under guidance for specific health concerns. Too much creates new problems: nausea, stomach pain, or copper deficiency over time. Always best to get a blood test and talk to your doctor before starting, especially if you already take other supplements or medications.
Zinc gluconate stands up as a practical choice for many, backing up the science most of the time. For anyone always fighting colds, struggling with slow-healing skin, or eating a limited diet, it isn’t just hype. It delivers real results, as long as you use it wisely and stay within recommended doses.
Are there any side effects of zinc gluconate?
Why People Turn to Zinc Gluconate
Cold and flu season sets off the yearly hunt for something—anything—that might keep symptoms away. Some folks turn to zinc gluconate, hoping for a shortcut to fewer sick days. It’s easy to find in lozenges and supplements, promising to knock a few days off the sniffles. The real question stares back once you’ve bought that bottle: Is this stuff completely safe? Or does it come with a string attached?
Common Side Effects: Not Always Just Stomach Grumbles
Stomach discomfort tends to show up first after popping a zinc gluconate supplement. For me, a chalky aftertaste and mild nausea felt like rude company while fighting a sore throat. A closer look at research lines up with my experience. Upset stomach, heartburn, and even vomiting have all been reported, even at standard doses. Large studies, like one in the Journal of the American Pharmacists Association, cite these complaints again and again.
Taking more than the recommended dose doesn't help. Instead, it can spark headaches or leave your sense of taste out of sorts. I’ve heard from friends that metallic taste can linger after using zinc lozenges, sometimes for hours. High doses—over 40 mg a day—can eventually block your ability to absorb copper, setting the stage for other health problems like anemia or even nerve trouble. These problems sneak up, so shifting from occasional lozenge to daily supplement should be a thoughtful move, not a knee-jerk reaction.
Zinc and Immunity: Chasing a Quick Fix
Zinc plays a role in immune function, no doubt about it. Studies point to some benefit in cutting cold durations, but going overboard rarely turns out well. A 2020 review in BMJ Open highlighted that consistent use above suggested doses can cause gastrointestinal side effects and copper deficiency.
The Food and Nutrition Board keeps the “tolerable upper intake level” at 40 mg per day for adults. The body’s delicate balance can tip quickly—too little zinc and infection risk jumps, too much and trouble brews elsewhere.
Drug Interactions: One Pill Can Bump Another
Medications can collide inside the body. Zinc, for instance, can disrupt absorption of antibiotics such as tetracyclines or quinolones. That means you could walk away from the pharmacy thinking you’re helping, but actually dragging out an infection instead.
For folks with kidney disease, any supplement needs careful thought. The kidneys clear zinc, so a compromised system lets it pile up. Checking with a doctor becomes non-negotiable here.
Navigating Supplements: More Than Just Reading the Label
Supplements don’t need the same FDA scrutiny as prescription meds. You often rely on a good reputation or word of mouth. Quality issues and dose inconsistencies pop up from time to time. Sticking with brands reviewed or certified by third parties helps limit surprises in the bottle.
Balanced eating still beats any supplement. Shellfish, beans, and dairy all deliver zinc the body absorbs with less fuss. Instead of jumping straight to pills, an honest look at your diet might answer the deficiency question before it even starts.
Staying Safe with Zinc
Always compare supplement facts with your needs and current health. The National Institutes of Health offers guidelines that cover all age groups. If symptoms persist after a few days, or if there’s a rash, confusion, or persistent nausea, contact a healthcare professional quickly. Staying safe often just means not ignoring small warning signs.
How should I take zinc gluconate and what is the recommended dosage?
Understanding Zinc Gluconate in Daily Life
Our bodies use zinc for many jobs. It fuels our immune system, helps wounds heal, and keeps our senses sharp. Zinc gluconate stands out as a type of zinc supplement you pick up in pharmacies and health food stores. The reasons for choosing supplements can vary, but too much or too little doesn’t help anyone. Many folks wonder what’s truly the right dose and how to take it for best results.
How Much Zinc is Enough?
My own mistake was thinking more meant better. Years ago, a bit of tiredness and the newsletter ads led me to take zinc every morning without checking how much I actually needed. My stomach got upset, and nothing about my health improved. Honest discussions with my family doctor revealed most adults actually only require about 8 to 11 milligrams of zinc from all sources daily. The National Institutes of Health backs this up — exceeding that 40-milligram-per-day line can spark nausea, headaches, and risk of messing up copper absorption.
Kids and teens get different recommendations, narrowing down to around 2 to 8 milligrams for the younger set. Special stages like pregnancy nudge the suggested daily intake a little higher but always under professional guidance. It’s easy to remember: a single tablet isn’t a magic fix, and the label always matters.
Best Way to Take Zinc Gluconate
The absorption of zinc fights with certain foods and other supplements. Calcium supplements, big dairy servings, or a bowl of cereal rich in iron can limit how well the body absorbs zinc. Experts usually suggest swallowing zinc gluconate with water, apart from meals or at least two hours after eating, especially if the diet leans heavy on those absorption-blockers.
Some people try lozenges to ease throat symptoms during colds. The Cochrane Database points out that lozenges with zinc gluconate sometimes shave a bit off the length of colds, but timing and dosage influence this benefit. If cold symptoms drag on, nonstop dosing doesn’t solve anything and leads to the same stomach issues as regular supplements.
Risks and Red Flags
Just because a product claims to be natural or over-the-counter doesn’t mean it’s safe for everyone. Too much zinc over weeks or months can blunt your immune system instead of supporting it, and throw off mineral balance. Side effects like metallic taste, stomach cramps, or changes in bowel habits usually mean it’s time to pause and talk with a doctor.
Smart Solutions for Using Zinc Safely
Start with a food journal to see if you already get enough zinc from chicken, beans, seeds, and whole grains. Supplements step in mostly for folks with diagnosed deficiencies, special diets, or digestive conditions that block normal zinc uptake. Trustworthy brands help, because not all supplements pack what they promise on the label.
If you think you might need extra zinc, health experts always encourage a open chat with a provider. Testing blood or diet beats guessing and avoids doubling up with multivitamins by accident. Clear goals — not habits built on internet trends — keep your supplement routine safe, targeted, and effective.
Can zinc gluconate be taken with other medications or supplements?
Understanding Interactions in Everyday Life
People often go out to buy zinc gluconate for a cold, a sense of fatigue, or just to support the immune system. It gets tempting to mix it with other over-the-counter vitamins or prescriptions. Once I had a sinus infection and grabbed zinc, thinking more was better, but I didn’t check what was already in my multivitamin. That decision led to a stomach ache and a lingering bad taste that lasted all morning. It reminded me that combining supplements without some know-how can trip up even the most health-conscious among us.
Zinc plays a part in immune response and wound healing. At the same time, too much zinc interferes with the absorption of copper and iron. Many people do not realize that taking high doses of zinc for weeks might mess with these nutrients, leading to new health issues. Research from the National Institutes of Health shows that daily zinc intake above 40 mg for an extended time brings a risk of copper deficiency. If you already take iron for anemia, zinc may block its absorption in the gut if taken together. Doctors often suggest separating the two supplements by at least two hours.
Mixing Zinc Gluconate with Prescription Medications
Antibiotics, especially tetracyclines and quinolones, don’t mix well with zinc. I learned this the hard way while taking an antibiotic for a stubborn cough, only to find out from my pharmacist that zinc binds to these antibiotics in the digestive tract. The result: a weakened effect of the medication, possibly leading to treatment failure. According to the U.S. Food and Drug Administration, people should separate zinc and these antibiotics by at least two hours to keep both working as they should.
Diuretics, a medicine class prescribed for high blood pressure, can also complicate things. They increase zinc loss through urine, causing subtle, long-term deficiency if you’re not careful. People with chronic heart or kidney conditions who use diuretics may want to ask their doctor about checking zinc levels during regular checkups. On the flip side, a simple blood test will show if extra zinc is necessary or if a diet change will do the job.
Combining Zinc with Other Supplements
Zinc lives in a crowded supplement aisle. It competes with magnesium, calcium, and copper for absorption in the body. Loading up on a handful of pills without a plan rarely helps and often causes nausea. Healthline reports that more than 30% of Americans take multiple supplements daily, but few discuss the stack with a professional. Multivitamins already contain zinc, so doubling up can tip the scales toward unwanted effects.
How to Stay Safe
Double-check the label on your medications and other supplements. If unsure, speak to a pharmacist or healthcare professional. I often call my pharmacist before mixing new supplements with my daily meds; that short phone call beats sorting out side effects later. The best approach starts with getting as many nutrients as possible from real food. When supplements become necessary, aim for moderate doses and keep your healthcare provider in the loop. Supplements fill gaps, but knowledge keeps us healthy.
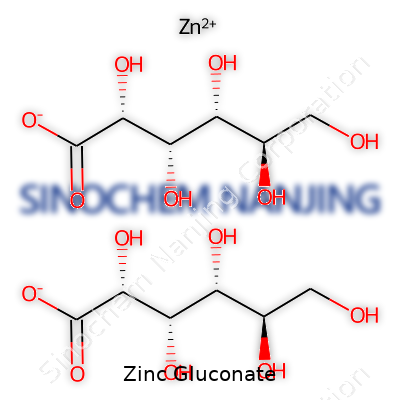
| Names | |
| Preferred IUPAC name | zinc bis[(2R,3S,4R,5R)-2,3,4,5,6-pentahydroxyhexanoate] |
| Other names |
Zincum gluconicum Gluconic acid zinc salt Zinc(II) gluconate Zinc bis(D-gluconate) Zinc digluconate |
| Pronunciation | /ˈzɪŋk ˈɡluː.kə.neɪt/ |
| Identifiers | |
| CAS Number | 4468-02-4 |
| Beilstein Reference | 85368 |
| ChEBI | CHEBI:132948 |
| ChEMBL | CHEMBL1201560 |
| ChemSpider | 21566021 |
| DrugBank | DB04545 |
| ECHA InfoCard | 03ac52b2-d23b-4177-a22d-846c9746b4ab |
| EC Number | Zinc Gluconate EC Number: 231-072-3 |
| Gmelin Reference | 83430 |
| KEGG | C01799 |
| MeSH | D015699 |
| PubChem CID | 24839721 |
| RTECS number | ZH4490000 |
| UNII | J41CSQ5QKH |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C12H22O14Zn |
| Molar mass | 455.686 g/mol |
| Appearance | White to off-white crystalline powder |
| Odor | Odorless |
| Density | Density: 0.7 g/cm³ |
| Solubility in water | Freely soluble in water |
| log P | -4.4 |
| Vapor pressure | Negligible |
| Acidity (pKa) | Acidity (pKa) of Zinc Gluconate: "3.7 |
| Basicity (pKb) | 11.8 |
| Magnetic susceptibility (χ) | '−1.2×10⁻⁵ cm³/mol' |
| Refractive index (nD) | 1.45 |
| Dipole moment | 3.51 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 437.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1568.96 kJ/mol |
| Pharmacology | |
| ATC code | A12CB01 |
| Hazards | |
| Main hazards | May cause eye, skin, and respiratory tract irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | No hazard statements. |
| Precautionary statements | Keep container tightly closed. Store in a cool, dry place. Avoid breathing dust. Wash thoroughly after handling. Use with adequate ventilation. Avoid contact with eyes, skin, and clothing. |
| NFPA 704 (fire diamond) | 1-0-0 |
| Lethal dose or concentration | LD50 oral rat 3500 mg/kg |
| LD50 (median dose) | 3500 mg/kg (rat, oral) |
| PEL (Permissible) | 15 mg Zn |
| REL (Recommended) | 15 mg (as zinc) daily |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Gluconic acid Glucono delta-lactone Calcium gluconate Iron(II) gluconate Potassium gluconate |

