Zinc Fluoroborate: More Than a Specialty Chemical
The Story of Discovery and Progress
Zinc fluoroborate didn’t just appear on the shelves of laboratories; its story goes back to the search for better electrolyte systems in the mid-20th century. Early work sought alternatives to harsh mineral acids for metal finishing and battery applications. The spotlight landed on fluoroborate salts, thanks to their balance of stability and reactivity. As acid zinc plating processes gained traction for their smoother, brighter coatings, zinc fluoroborate stood out for those wanting uniform results without drifting away from environmental and operational controls. Its introduction didn’t just change how parts look. It set new expectations for chemistry in manufacturing settings, moving beyond simple mixtures to finely-tuned reaction systems.
A Look at What Zinc Fluoroborate Is
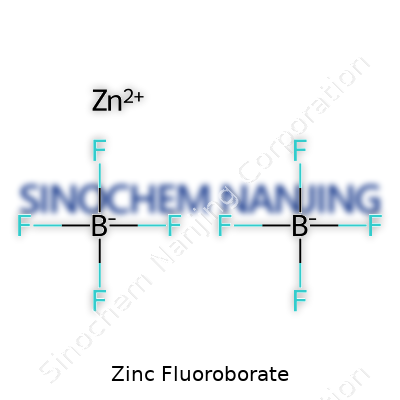
At a glance, zinc fluoroborate may seem like just another white crystalline salt. It takes a back seat to the better-known zinc sulfate or chloride, but to those in the plating industry, its worth is obvious. With a chemical formula of Zn(BF4)2, it brings together the acid strength of fluoroboric acid and the metal cation flexibility of zinc, opening a window to new chemical behavior rarely found in the old standards. Often supplied as a colorless solution or crystalline solid, it contains neither the stinging irritancy of some acids nor the sluggishness of electrolytes with high resistance. The compound’s solubility and ability to carry current between electrodes sets it apart in industries where fast, clean metal deposits make or break quality.
Physical and Chemical Qualities You Can Rely On
One thing people notice about zinc fluoroborate is how it dissolves so easily in water. This property reduces downtime in process tanks and allows for rapid formulation tweaks as work shifts through different product lines or thickness requirements. The solution flows almost like thin syrup, depending on concentration, offering the right viscosity for even metal spread. Chemically, it steers clear of the kind of hazardous reactivity seen with perchlorate-based salts. Its low vapor pressure and stability at room temperature mean operators don’t see as many headaches from fumes or unwanted side reactions. For those dealing with complicated metal pieces, this property brings not just convenience, but also the peace of mind that comes from knowing the chemistry won’t take unexpected turns mid-batch.
The Fine Print: Specs and Labeling Realities
Regulations around labeling and technical specs for specialty chemicals have sharpened over recent years. Zinc fluoroborate products sold today must stick to clear guidelines, listing not just the percent of active ingredient but also moisture, pH limits, and permitted traces of other metals. Precautions on handling reflect decades of hard-learned lessons—this isn’t just ink on paper but the result of real incidents and investigations from across the globe. Companies keep documentation on everything from batch consistency to shipping container integrity, and these practices support safer workplaces, not just compliance for its own sake.
How the Compound Comes Together
Most zinc fluoroborate available on the market results from reacting zinc oxide or zinc carbonate with fluoroboric acid. This is not a process you carry out in a kitchen; it involves careful dosing in closed systems with ventilation controls. As gases bubble off, operators monitor temperature and acidity, working to avoid unwanted side reactions or build-up of impurities. Consistent filtration catches unreacted zinc and keeps solutions free from particles that could compromise a coating tank down the line. From the outside, it might sound routine, but inside chemical plants, small tweaks to timing, mixing speed, or batch size mean the difference between reliable performance and costly rework.
Changes and Chemical Reactions to Watch
Zinc fluoroborate doesn’t sit idly on the shelf. In solution, it dissociates almost completely, offering up zinc ions and the uniquely-structured tetrafluoroborate anion. This kind of ion pairing makes possible smoother coatings than would ever be possible with zinc sulfate, because it staves off hydrogen evolution and pitting. Electroplaters looking to adjust grain size or luster sometimes add specific brighteners or modify current density, knowing that the basic chemistry stays stable. But anyone using this salt must keep tank purity high; stray metals or oxygen can kick off side reactions—depositing unwanted alloys or causing rough surfaces that fail inspection.
What’s in a Name?
While “zinc fluoroborate” covers the compound, plenty of literature and supply catalogs refer to it as zinc tetrafluoroborate, zinc borofluoride, or even zinc fluoroborate hexahydrate if water of crystallization is locked in. Some chemists reference its CAS number when specifying it for research or procurement, though this tends to matter more to regulatory affairs or importing clerks. The point is, the chemical itself stays the same, no matter which name pops up on a bag, a barrel, or a journal article.
Doing Things Right: Safety Out in the Real World
Strong acids and metals together create predictable health and safety debates. Zinc fluoroborate, despite its advantages, earns concern for both its toxicity and how it behaves if spilled or overheated. The best-run plating shops rely on closed transfer systems, splash-proof gloves, and ventilation. Wastewater treatment requires a careful plan, since both zinc ions and fluorine-containing byproducts threaten aquatic systems when not held in check. Both government rules and industry best practices call for secondary containment, spill drills, and air monitoring, all backed by proven emergency procedures. Here, I’ve seen that even a small slip—one leaking hose or distracted operator—brings home the need for not just training, but real-world experience.
How Industry Uses It
In all my years touring different finishing shops, few materials create such debate as to their replacement potential and process advantages. Zinc fluoroborate finds its main job in electroplating. Its use in acid-based systems means brighter, even deposits; car parts, computer chassis, and telecom hardware all gain by demanding tight tolerances and uniform protection. Battery manufacturers have also eyed it for electrolyte blends, hoping for higher efficiency and lower self-discharge. You may spot it popping up in laboratory-scale syntheses, where its reactivity fits research chasing novel metal-organic frameworks or catalysts. Any planner thinking beyond cost per kilogram has to weigh these benefits against environmental priorities, knowing the legacy of heavy metals in soil and water can’t be brushed off.
What Lab Work Teaches Us
Research never ends with one generation of products. Universities and industry R&D centers keep poking at zinc fluoroborate—testing new additives for better throwing power, tweaking concentrations for thicker or thinner plating, and modeling ways to cut down on waste. Many research articles focus on grain structure modification or ways to suppress byproduct gas at electrodes. Figuring out the mechanism behind the spectacular leveling action or reducing the environmental load after disposal often starts with trial runs, ultra-pure salts, and painstaking measurement of tank chemistry after hundreds of cycles. Real progress rarely comes from a single breakthrough; it grows from small changes, constant vigilance, and stubborn curiosity.
Toxicity, Health, and the Road Ahead
Toxicology studies on zinc fluoroborate paint a mixed picture. Zinc ions, when carefully managed, do less harm in humans than many other metals—but long exposure, ingestion, or inhalation of dust and mist still carries risks. Fluoroborate breakdown products, particularly boron and fluoride ions, can accumulate and throw off metabolic processes in plants and animals. Occupational health standards enforce strict exposure limits, but disposal concerns linger in communities near plating plants. In the search for less hazardous chemicals, the drive for “green chemistry” often heads for biocompatibility, but major infrastructure changes don’t come overnight. Better filtration, real-time monitoring, and, above all, honest reporting on risk will shape how this salt gets handled in decades to come.
Looking at What’s Next
The role of zinc fluoroborate faces disruption from many angles. As auto makers shift to aluminum parts, and as demand grows for longer-lasting electronics, pressure grows to replace baths that generate heavy metal waste. Some researchers are betting on organometallic complexes or ionic liquids as replacements. Others focus on recovering zinc and fluorine from spent processes, closing the loop and pushing resource efficiency. Whatever direction the market takes, future breakthroughs won’t come from shortcuts or underfunded experiments, but from a sustained push for safer, cleaner chemistry—a lesson obvious to anyone who’s scrubbed a plating tank or handled a spill. Zinc fluoroborate’s journey shows every choice in industrial chemistry echoes far beyond the walls of the plants where it starts.
What is Zinc Fluoroborate used for?
Understanding What Zinc Fluoroborate Actually Does
Zinc fluoroborate draws blank stares from most people. Unless you’ve spent time in a plating shop, chances are it’s never crossed your mind. I first heard about it at an electroplating conference—one of those niche events with more lab coats than suits. Folks there spoke about zinc fluoroborate with the kind of respect that plumbers reserve for a good pipe wrench.
This compound plays a big role in surface finishing and electroplating, where a clean, even zinc coating makes or breaks the whole process. The stuff dissolves easily in water and creates a bath where metal objects can pick up a layer of zinc for corrosion resistance. Plenty of metal parts—think nuts and bolts or auto parts—get their start in a tank with zinc fluoroborate in the mix. If someone wants a smooth, fast-depositing zinc layer, this chemical has proven itself time and time again. Big auto suppliers and electronics manufacturers bank on it.
Industry Impact and Health Considerations
The global economy runs on reliable machines, and those machines need reliable parts. Zinc fluoroborate’s legacy shows up in hardware stores, toolboxes, even the phone you might be holding. Without a reliable finish, metal gets eaten up by the elements, and life gets expensive.
My own entry into the metalworking world drove home the safety side. Chemicals like zinc fluoroborate work wonders in the right hands, but they pack a punch. Improper handling, leaks, or poor ventilation can cause skin and respiratory irritation. Chronic exposure at a chemical plant, left unchecked, can cause more serious health headaches. There’s no room for shortcuts here, and that lesson sticks after seeing a colleague break out in a rash from skipping his gloves in a rush. Trust in the science, but trust in the gear too.
Risks, Environmental Management, and Solutions
Beyond worker safety, zinc fluoroborate brings up bigger questions. In the old days, chemical waste often went straight down the drain. Not anymore. Today, laws like the Clean Water Act force industries to treat their wastewater before sending anything downstream. But loopholes and cost cutting still slip through, especially in places that lack tough enforcement. Heavy metals and fluorides won’t just vanish if dumped into rivers. They build up in soil and water, threatening fish, plants, and, eventually, people.
A better way points through strong regulation, regular audits, and serious investment in treatment technology. Engineers have developed ways to recover fluorides from wastewater, and companies committed to responsible production invest in these systems. A plant manager showed me how even simple equipment upgrades cut their discharge of harmful ions to near zero. Sharing these solutions across borders matters, since demand for plated parts won’t fade as construction, automotive, and communications industries evolve.
Final Word
Zinc fluoroborate might never make headlines, but its story intersects with nearly every part of daily life. It brings together chemistry, regulation, livelihoods, and public health. Its power calls for both scientific understanding and common sense. Time spent around those plating tanks drives home that in industry, shortcuts cost more than they save.
What is the chemical formula of Zinc Fluoroborate?
Understanding the Basics
Sometimes, the smallest pieces of scientific information open doors to powerful real-world changes. Take zinc fluoroborate, with its chemical formula Zn(BF4)2. It looks complex at first, but a closer look brings a lot into focus. Zinc stands for an element that helps protect metals from corrosion. Fluoroborate comes in as the partner that makes a big difference in electroplating and other technical processes. This isn’t abstract chemistry—Zn(BF4)2 shows up on shop floors, in batteries, and anywhere surface finishing gets real attention.
Formula Structure and Chemistry
Zn(BF4)2 signals a compound with one zinc ion and two tetrafluoroborate anions. Those tetrafluoroborate groups, made up of boron and four fluorine atoms, remain stable under tough conditions. The structure isn’t something you only see in textbooks. It shows up in labs, in industrial tanks, and sometimes even in high school science classrooms. Students and engineers work with these compounds because they want results that improve performance, reduce waste, and add value.
Real-World Uses
Zinc fluoroborate comes up in conversation for anyone facing electroplating challenges. I remember how many manufacturers in my hometown ran into problems with uneven coatings. Once zinc fluoroborate turned up as part of the electrolyte in their process, results spoke for themselves. People noticed smoother, more durable layers on auto parts and electrical connectors. Cleaner deposits mean less rework and fewer raw material losses.
Researchers aiming for lightweight, reliable batteries also pay close attention to zinc fluoroborate. The same properties that make it ideal for plating—its solubility, conductivity, and chemical stability—also influence battery performance. When reliability matters, you can’t ignore something as tough as this compound. Factories that deal with safety standards turn to it as well, since alternatives sometimes bring higher risks or unpredictable reactions.
Why It Matters
Every detail in a formula can have a big economic impact. A mistake with the chemical makeup could lead to equipment failures, increased costs, or safety incidents. In my own college labs, missing a subscript or swapping one element for another in a formula could mean starting an experiment over from scratch. Zinc fluoroborate’s exact formula isn’t trivia—it’s a checklist item for quality control and environmental compliance.
There’s a sustainability angle, too. Companies searching for low-toxicity, efficient chemicals keep choosing Zn(BF4)2 for electroplating or as an additive. Regulators like to see clear, accurate product labeling, and workers care about what goes into their mixing tanks. Getting the chemistry right makes everyone’s life easier.
Addressing the Challenges
Production workers and small shop owners sometimes find handling or storing zinc fluoroborate intimidating. Clear guidelines and ongoing training remove barriers. My experience shows that, early on, confusion around chemical names and formulas led to ordering mistakes. Investing time in basic chemistry education shaped safer, more efficient businesses. Accurate information shared at all levels—from procurement to shop floor—brings people together, reduces costly errors, and boosts confidence.
When industries rely on chemistry, accuracy protects people and the environment. Whether in a school science lab or a massive manufacturing plant, knowing that Zn(BF4)2 stands for zinc fluoroborate keeps processes running and standards high. It’s a reminder: in science and industry, details draw the line between progress and setbacks.
Is Zinc Fluoroborate hazardous or toxic?
Getting Real About Its Hazards
Zinc fluoroborate doesn’t show up on grocery store shelves, but it definitely appears in quite a few industrial settings. It’s a colorless or slightly yellowish liquid, often used in electroplating or as a catalyst in certain chemical reactions. People who work in factories or laboratories probably run into it more often than anyone else.
The main concern here lies in its makeup. Zinc itself plays a vital part in daily nutrition—our bodies only need a trace, though. The real trouble comes from the “fluoroborate” end. Any compound containing fluorine or boron tends to warrant a closer look. Fluorides irritate skin, eyes, and lungs. Overexposure to borates has its own harsh effects, like reproductive and developmental issues in animals. While eating a zinc supplement poses little risk, splashing zinc fluoroborate on your hand doesn’t belong in that same category.
Occupational Safety and Real-World Health Effects
Most reputable safety databases—including the European Chemicals Agency and OSHA—flag zinc fluoroborate as hazardous. Strong evidence points to health dangers. The fumes sting if breathed in. Accidents in poorly ventilated plating shops sometimes aggravate asthma or worse, give workers corrosive burns if spilled. Swallowing even a small amount leads to severe nausea, stomach pain, or poisoning.
Back in college, an old chemistry professor always kept a spill kit close by, because a few drops on a glove could eat right through the latex. No one wanted to learn about chemical burns the hard way. I remember him emphasizing how just a tiny splash on skin brings real pain, and a soaked shirt means a trip to the hospital. Regulatory agencies don’t throw that “hazardous” label around for fun.
Long-Term Environmental and Human Impact
Once zinc fluoroborate lands in waterways or soil, it doesn’t just disappear. The boron and fluorine can harm fish and plants, while zinc itself builds up over time. Industrial spills and leaky storage tanks leave a lasting mark. During cleanup at a plating facility, workers have seen dead grass and sick fish downstream. These stories match research: water tests from polluted sites find higher fluoride and zinc readings.
Factories following proper storage and disposal procedures help minimize these risks. Yet mistakes still happen. My own stint as a research lab assistant meant attending safety training each year, learning how to neutralize a spill before it spread. Many industries have responded by improving containment, using double-walled tanks and drain covers. In strong compliance regions, regulators inspect sites unannounced just to catch lazy disposal practices.
Moving Toward Safer Practices
People often forget that chemical safety depends on both the compound and how it’s handled. Mandatory safety data sheets tell workers to wear gloves, goggles, and respirators around zinc fluoroborate. Simple steps—labeling containers, double-checking transfer pumps, installing chemical showers—guard against injuries. Companies also phase out zinc fluoroborate when less toxic alternatives exist. The same wheels of industry that once embraced convenience now keep health and the environment in mind.
Consumers, neighbors, and workers all deserve to know about the risks present right next door. Real accountability means listening to those who face the danger directly and enforcing honest safety standards. Solutions often look obvious on paper: clear rules, sturdy handling tanks, reliable emergency response. Progress won’t come from treating such substances as harmless, only by treating their hazards seriously, based on facts and lived experience.
How should Zinc Fluoroborate be stored?
The Real Hazards Behind the Bottle
Zinc fluoroborate doesn’t usually get talked about outside of plating shops and industrial chemistry circles, but the way it’s handled tells us a lot about why chemical safety needs constant attention. I’ve seen careless storage lead to injuries and ruined equipment. This compound isn’t just another chemical in a long lineup; it carries risks that deserve respect. It’s highly corrosive, forms toxic fumes if things go wrong, and can cause environmental trouble if it leaks out uncontrolled. Combine those hazards with the very real chance for human error, and storing it properly becomes more than just ticking boxes for compliance. It’s basic responsibility, plain and simple.
Why Moisture Ruins Everything
Humidity and chemicals usually don’t mix well, but zinc fluoroborate has a way of reacting to even minor exposure. The compound breaks down and starts eating through packaging, turning into hydrofluoric acid and boron trifluoride gas—both dangerous on their own. Chemistry aside, I’ve seen warehouse workers puzzled as containers corrode from the inside out after just a week in a damp storeroom. That’s why dry conditions aren’t optional. A dedicated, moisture-controlled storage area keeps these reactions at bay. Even a small oversight, like an open window or leaky roof, lets in enough moisture to start a chain reaction nobody wants to clean up after.
Keeping Containers Tight and Materials Right
This is one place where cost-cutting bites back. That warehouse manager I mentioned? She tried using bargain-bin barrels and ended up with expensive repairs and hospital time for two employees. Zinc fluoroborate chews through a surprising range of materials. Polyethylene or certain high-grade plastics hold up where metals and regular plastics fail. Containers need real seals that don’t degrade if a little bit of mist or vapor hits them. I’m a big believer in checking seals regularly and never reusing containers that have seen even minor leaks. A chemical-resistant label with clear hazard information means someone picking it up weeks later won’t get caught by surprise.
No Mixing Zones—Simple and Strict
When a chemical reacts with water or other acids, the resulting fumes choke, burn, and cause panic. In factories I’ve visited, the worst near-misses always happen at crowded loading bays where nobody’s watching what gets stacked together. Zinc fluoroborate belongs away from oxidizers, alkaline substances, and strong acids. Putting it on a separate, marked shelf—even dedicating an entire cabinet if you have to—means a truck delivery gone wrong or an earthquake doesn’t create a disaster zone. Good storage starts by actually respecting those separation rules every time, not just on inspection days.
Why Experience Matters in Chemical Storage
Storage manuals can’t prepare someone for real-life emergencies. I’ve seen seasoned pros catch a sweating drum before it turns into a crisis, just because they stayed alert to chemical odors and surface changes. Training every worker who handles zinc fluoroborate with live demonstrations, spill scenarios, and regular safety briefings makes all the difference. Proper record-keeping, double-checking inventories, and never ignoring the odd sound or smell in the chemical room all add up. An ounce of prevention here spares seasoned vets and new hires from learning safety lessons the hard way.
What industries commonly use Zinc Fluoroborate?
Metal Finishing
Factories have used zinc fluoroborate for decades to deliver tough, bright coatings, mostly through the process of electroplating. Picture a line of metal parts—auto fasteners, hardware, and electronics—rolling past a line of tanks, each dipped in specialized solutions. In the tanks with zinc fluoroborate, the bath coats the parts with a layer of zinc. This helps protect metal from rust, keeps it smooth, and even adds a bit of shine. Engineers and shop operators like it because fluoroborate-based baths usually work faster and more reliably than some older choices.
In my experience watching plating lines run, the difference shows up in how fewer touch-ups folks need on parts after one round. Occasional flashes of safety yellow vests walk the aisles, checking gauges and watching for leaks, since safety matters. Environmental controls hang close by, since zinc salts in general can be hazardous if handled carelessly. From industrial zones in North America to factories in Asia, the need to keep cars and machines working drives steady demand for this compound.
Battery and Energy Storage
People don’t always realize that some specialty battery makers use zinc fluoroborate in their electrolyte formulas. Zinc-based batteries, especially those aimed at large-scale grid storage or backup power, use the compound for its reliable chemical behavior and ability to support efficient electricity flow. Zinc batteries look appealing in the energy world because they don’t rely heavily on rare metals. Using a cleaner, repeatable chemistry like zinc fluoroborate can lower waste and improve power output, two factors every engineer weighs.
There’s growing pressure to cut down reliance on lithium and cobalt, both for cost and security reasons. If you’re working in a power plant or a data center, a backup battery you can trust can make or break your operation. Scientists still keep working to refine how these batteries are built so factories can turn them out on a schedule without running into stuck valves or corroded parts. Zinc fluoroborate’s ability to deliver consistent results helps here.
Chemical Manufacturing
Some chemical plants use zinc fluoroborate as a catalyst to produce other chemicals and coatings. The compound plays a part in synthesis and can aid in the preparation of specialty pigments, lubricants, and even pharmaceuticals. Some paint companies tap it for the production of specific additives, seeking its solubility and strong reactivity. This means more efficient mixing and fewer clumps or sticky residues in the tanks, which is a big deal if you’ve ever had to clean up a clogged production line.
In my time talking with chemical engineers, they often point out that handling costs—wasted product, extra maintenance—can eat into the bottom line fast. Using a material with predictable behavior, like zinc fluoroborate, can mean fewer headaches and cleaner production cycles. Keeping the worksite safe and reducing emissions tie back to company reputation and regulatory compliance.
Challenges and Solutions
No industry fixates on a chemical like zinc fluoroborate without thinking about storage, risk, and disposal. The material carries hazards if inhaled or spilled. Facilities that depend on it juggle employee training, updated ventilation, and robust waste capture. Finding recycling options or alternative chemistries with lower toxicity would ease pressure on health and safety departments, particularly in regions with tighter environmental rules. Switching to automated handling and investing in better containment cuts accidents and keeps workplaces safer.
As the world looks for greener processes and more reliable batteries, zinc fluoroborate’s clear track record in these fields keeps it in demand. Safer, smarter ways to use and replace it deserve more attention. Workers and communities benefit when industry focuses on these practical steps.
| Names | |
| Preferred IUPAC name | zinc tetrafluoroborate |
| Other names |
Zinc tetrafluoroborate Zinc(II) fluoroborate Zinc borofluoride Zinc fluoborate |
| Pronunciation | /ˈzɪŋk fluːˈrɒbəˌreɪt/ |
| Identifiers | |
| CAS Number | 13826-87-6 |
| Beilstein Reference | 1906967 |
| ChEBI | CHEBI:91251 |
| ChEMBL | CHEMBL4296612 |
| ChemSpider | 22718650 |
| DrugBank | DB14545 |
| ECHA InfoCard | 14f5cc6d-cdfb-4b99-9395-86342b660d59 |
| EC Number | '215-677-5' |
| Gmelin Reference | 12029 |
| KEGG | C07116 |
| MeSH | D015224 |
| PubChem CID | 24866344 |
| RTECS number | TT2975000 |
| UNII | 33G6V279WZ |
| UN number | UN3264 |
| CompTox Dashboard (EPA) | DTXSID4075020 |
| Properties | |
| Chemical formula | Zn(BF4)2 |
| Molar mass | 238.01 g/mol |
| Appearance | Colorless or slightly yellowish transparent liquid |
| Odor | Odorless |
| Density | 1.54 g/cm³ |
| Solubility in water | Miscible |
| log P | -4.2 |
| Vapor pressure | Negligible |
| Acidity (pKa) | ~3.6 |
| Basicity (pKb) | 3.74 |
| Magnetic susceptibility (χ) | '-8.1 × 10⁻⁶ cm³/mol' |
| Refractive index (nD) | 1.567 |
| Viscosity | 10-15 cP |
| Dipole moment | 2.14 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 176.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1122 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V07AY |
| Hazards | |
| Main hazards | Causes severe skin burns and eye damage. Harmful if swallowed. |
| GHS labelling | **"GHS02, GHS05, GHS07"** |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | Harmful if swallowed. Causes severe skin burns and eye damage. Causes serious eye damage. May cause respiratory irritation. |
| Precautionary statements | P264, P280, P301+P312, P330, P305+P351+P338, P310, P303+P361+P353, P304+P340, P312, P363 |
| NFPA 704 (fire diamond) | 1-0-0 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 oral rat 110 mg/kg |
| LD50 (median dose) | 200 mg/kg (rat, oral) |
| NIOSH | RN7874 |
| PEL (Permissible) | Not established |
| REL (Recommended) | 2 mg Zn/m³ |
| Related compounds | |
| Related compounds |
Aluminium fluoroborate Nickel fluoroborate Copper fluoroborate Magnesium fluoroborate Tin(II) fluoroborate |

