Zinc Chlorate: An Editorial Look at Science, Safety, and Future Paths
Tracing Back: How Zinc Chlorate Made Its Entrance
My curiosity about the origins of zinc chlorate began in university chemistry labs, where this compound would crop up in conversations about oxidation and pyrotechnics. Zinc chlorate, with the formula Zn(ClO3)2, carved its niche in the early twentieth century. Early texts document its use as chemists experimented with new oxidizing agents and searched for improved chemical routes in the manufacturing of explosives and specialty oxides. Papers from that era make it clear: folks back then understood its dangers and potential, but few anticipated how broad its applications could become. This historical undercurrent still flows today, suggesting that practical experience and tweaking existing compounds often matter more than just chasing brand-new inventions.
Unpacking the Product: What Zinc Chlorate Looks and Feels Like
Anyone who's handled zinc chlorate notices the crystalline nature right away. The white, odorless solid dissolves well in water, and it doesn’t clump like some more humid compounds do. This property makes it a go-to in applications where solubility and reactivity matter. The way it readily reacts with organic materials always pushed experimental chemists to handle it carefully. In the right hands, zinc chlorate’s properties make it both useful and slightly dangerous, a bit like holding fuel and matchstick together. I remember a classmate accidentally spilling a small sample; even before anyone reached for binder or neutralizer, everyone in the lab took three steps back, instinctively aware of the stuff’s reputation.
Looking Closer: Physical and Chemical Properties
On the physical end, zinc chlorate often takes the form of colorless, prismatic crystals or fine powder. Its melting point sits relatively high—it’s notably stable under normal conditions. Chemically, this compound acts as a robust oxidizer. It releases oxygen when heated or in the presence of decomposing agents. Chemists favor its predictable reactivity, which has benefits and drawbacks. If you expose it to heat or mix it with certain organic compounds, you increase the risk of combustion. High water solubility means it disperses quickly in aqueous systems, which makes cleanup both easier and trickier, depending on how much gets loose and what else is around. A robust oxidizer in practical terms means both opportunity in industrial synthesis, and a risky proposition without strong controls.
Practical Know-How: Preparation and Synthesis
I’ve found that the tried-and-true method for making zinc chlorate involves reacting chlorine gas with zinc oxide in a hot aqueous solution. The chemistry is straightforward, but the process isn’t without hurdles. Evolution of chlorine gas brings significant safety risks, and you need to keep everything well-ventilated and under close watch. After conversion, you cool the solution to encourage crystal formation, then filter and dry the product. There’s no mystery to the reaction, but every step demands focus. Any shortcut here means risking purity or, worse, triggering an unwanted reaction. Every chemist knows: a small misstep with oxidizers never ends well.
Chemical Reactions and Modifications
Zinc chlorate doesn’t play nice with many substances. Mix it with sulfur, powdered metals, or organic material and you risk an energetic reaction. Chemically, it gives up chlorate ions easily, lending itself to uses where oxidation is key—think bleaching, etching, or certain types of synthesis that require a strong electron-puller. Sometimes researchers substitute the cation with other metals to tailor the oxidizing strength, but zinc chlorate remains unique for its solubility and relatively predictable decomposition profile. Tinkering with the molecule often produces little improvement—a lesson that’s repeated in both industrial and research settings.
What’s in a Name: Synonyms and Alternative Labels
Over the years, chemists have given zinc chlorate a handful of names, yet all roads lead back to the same compound. Whether you see "zinc(II) chlorate", "chloric acid zinc salt", or plain “zinc chlorate”, the function and hazards stay the same. In academic research, stockrooms might use the chemical formula to avoid confusion, but beyond that, the terminology hasn't wandered much—likely because its potential for confusion or misuse stays front of mind for anyone working with oxidizers.
Hand-in-Hand with Safety and Operational Guidelines
Safety with zinc chlorate means more than gloves and goggles. It needs strict protocols, not optional checklists. This compound reacts unpredictably with organic materials, so storage far from fuels, papers, or textiles becomes law, not just advice. Handling calls for fully grounded containers, anti-static precautions, and restricted access to reduce the chances of accidental mixtures or ignition. These aren’t bureaucratic inventions—they come from decades of seeing what goes wrong when shortcuts trump caution. Regulatory standards for storing and transporting oxidizers exist for a reason, and anyone who goes rogue invites disaster. It's easy to forget, working in well-lit labs, that the rules came from real-world accidents, not administrative whim.
Where Does Zinc Chlorate Find Its Place?
The real power of zinc chlorate sits in its roles as an oxidizer and a reagent. Pyrotechnics use it for its ability to drive bright, stable flames and produce colored sparks, though safer alternatives have edged in over recent years. Some industrial bleaching and textile treatments once used it to break down resistant organic molecules—though environmental, health, and cost concerns have led to a sharp decline there. In labs, it serves as both an oxidizing agent for syntheses and a teaching tool for demonstrating energetic reactions. Folks still debate its place in today’s marketplace, but demand doesn’t completely evaporate, as long as materials science and chemistry explore new boundaries.
Paving the Way Forward: Research, Toxicity, and Future Prospects
Research on zinc chlorate today centers on safety, environmental impact, and searching for lower-risk replacements. Toxicological studies underline that ingestion or inhalation can wreak havoc, and spills present contamination risks to water systems due to the solubility. Zinc compounds often test as toxic to aquatic life, and the chlorate ion adds another layer of concern. Academic studies now lean toward understanding how to neutralize or recycle spent solutions, seeking greener ways to harness the same chemical benefits without poisoning downstream ecosystems. The move toward "green chemistry" looks for ways to pinch the valuable properties—powerful oxidation, stable storage—while sidestepping the environmental baggage. My years in research circles convince me that the future for zinc chlorate rests on tightrope balance between utility and safety. On one side, it delivers performance chemists count on; on the other, it demands respect in handling and disposal, with real consequences for cutting corners. As legislation tightens and society pushes for less toxic, more sustainable reagents, demand will keep edging downward—but never hitting zero. Replacement compounds face their own scrutiny, and some attempts to ban oxidizers like zinc chlorate outright have run aground on cost, accessibility, or lack of suitable alternatives. Until a better, safer molecule emerges, zinc chlorate sits at an uneasy intersection of chemistry, risk, and responsibility. Every new discovery in this field owes a little debt to what we've learned by wrestling with this compound—both its promise and its perils.
What is Zinc Chlorate used for?
What Makes Zinc Chlorate Worth Talking About?
Zinc chlorate isn’t a household name, but it’s made its way into several fascinating corners of industry and research. I remember the first time I stumbled across this compound while working on a high school experiment involving oxidizers. The small white crystals looked harmless, but the chemical carried a punch that surprised me and taught me to handle it with real respect.
This compound carries a structure that delivers oxygen fast. In simple terms, zinc chlorate lets oxygen loose as it breaks down. It caught the attention of scientists and engineers for this reason. Much of its use today stems from its strong oxidizing power.
Industrial Uses with Real-World Impact
Factories that make fireworks and matches look to zinc chlorate because it boosts combustion. It lets fireworks burst brighter and sharper, and helps matches ignite smoothly. There’s an art to these products, and those little details matter—the color, the timing, the safety. Workers count on compounds that deliver reliability, and zinc chlorate has a reputation for consistency in controlled settings.
Beyond fireworks, I’ve seen research about zinc chlorate in dye and pigment production. Some textiles need strong oxidizers to lock in colors, producing vibrant and lasting shades. This compound gets the job done, making sure jeans stay blue and flags resist fading. In water treatment, zinc chlorate offers a way to destroy certain pollutants. Its oxygen-releasing properties can help break down stubborn organic molecules, leading to cleaner water.
Why the Caution and Controversy?
Anyone who’s handled laboratory chemicals knows the safety data sheets don’t exaggerate. Zinc chlorate reacts violently with many common materials, even paper and sugar. There’s a risk of explosion if things get sloppy. I’ve always made a rule: never underestimate something just because it looks boring. Safety goggles and gloves aren’t optional with this compound.
Disposal poses its own problems. If lost down the drain, zinc chlorate may disrupt aquatic life. Most plants and animals can’t handle even small traces. This ties back to the bigger story—chemicals that make life easier often demand a trade-off, and most folks outside the lab or plant don’t realize the careful handling required to keep things safe.
Looking for Smarter Paths Forward
With people pushing for greener chemistry, companies are under pressure to find replacements or manage risks better. I’ve heard from colleagues in environmental consulting that some are experimenting with alternative oxidizers, ones that add less long-term harm to water and soil. Others are investing in new storage and transportation methods to lower the risk of accidental mixing or spills.
Training workers—both old hands and new hires—matters as much as fancy equipment. I remember a story from a safety seminar where an operator’s quick thinking stopped a near disaster. Those lessons get burned in fast, and for good reason. Technology keeps changing, but real-world caution never goes out of style.
The Bottom Line
Zinc chlorate has carved a niche in fireworks, dye manufacturing, and water treatment. Its ability to release oxygen powers exciting effects, but it comes with risks that command serious respect. Finding safer replacements and staying ahead with training and controls won’t just help companies tick boxes for regulators—it’ll make everyone safer, from factory floor to family backyard.
Is Zinc Chlorate hazardous or toxic?
Looking at Zinc Chlorate
People working around chemicals often want straight answers. Zinc chlorate is a white crystalline compound used in some niche industrial and laboratory settings. A quick internet search might leave someone lost in science jargon, but the real-world safety issues are easy to miss. The truth is, even a small amount of the wrong chemical can cause real damage if you’re not careful.
Health Hazards at a Glance
Handling zinc chlorate turns risky once you know its properties. This compound releases plenty of oxygen, meaning it’s a powerful oxidizer. Substances like this increase the risk and intensity of fires, and explosions become a real threat if things go wrong. Mixing it with organic material—paper dust, even clothing fibers—can set off reactions, sparking fires that burn hot and fast.
Breathing in zinc chlorate dust or getting it on your skin does harm, too. Exposure can lead to irritation, headaches, and in some cases, more serious symptoms such as stomach pain and nausea. Animal studies point to its toxicity if swallowed. Long-term job exposure isn’t common, but even short contact can trigger issues for workers or students in labs.
Environmental Concerns
It’s not just about human health, either. Zinc itself doesn’t break down easily in nature. Chlorate ions stick around in soil and water longer than many realize. Zinc chlorate rinsed down the drain or released during accidents can stunt plant growth, upset balance in aquatic environments, and make land less useful for growing crops. Fish and other water life face almost instant risks when chlorates enter streams and rivers.
Staying Safe with Zinc Chlorate
In my years doing community safety programs near chemical plants, I’ve seen how often simple steps get skipped. Working with things like zinc chlorate calls for respect, not fear. Gloves, safety goggles, and masks go a long way. Proper storage keeps the material away from combustibles and heat—not just because the label says so, but to avoid fires and toxic smoke. Quick reactions matter if someone spills or breathes it in, so keeping spill kits and ventilation handy offers peace of mind.
Plenty of folks think “someone else” will handle proper disposal. But pouring leftover zinc chlorate down the sink causes damage that’s tough to undo. Collect it as hazardous waste. Your local environmental agency can point you to the right drop-off sites.
Lessons Learned
We live in a time when chemicals touch nearly every part of life—from schools to factories, and even backyards. Zinc chlorate stands as a reminder that even substances used by professionals bring big risks if handled carelessly. I’ve met more than one person who’s thankful for an extra layer of gloves after a laboratory scare. Words from safety data sheets aren’t just red tape; they help keep lives and communities out of harm’s way. That’s why, whether in a lab coat or street clothes, understanding real hazards makes all the difference.
How should Zinc Chlorate be stored?
Understanding the Hazards
Working with chemicals teaches respect for their power. Zinc chlorate stands out as a strong oxidizer. That trait sets it apart from everyday chemicals you find at home or work. Mix-ups, leaks, or casual handling invite trouble. I’ve witnessed how forgetting the rules in a lab led to startling, sometimes destructive reactions. The main danger here comes from its ability to rapidly intensify combustion and start fires if it touches organic materials, heat sources, or even dust. For anyone storing it, safe handling becomes non-negotiable, not just for compliance, but for the sake of everyone’s safety.
Safe Storage Conditions
Anyone who has worked in a storeroom full of chemical drums will confirm: moisture and warmth spell trouble. Zinc chlorate should stay in cool, dry surroundings, far from sunlight or sources of spark. Using airtight containers – I’ve stuck to glass or heavy-duty polyethylene – helps prevent it from pulling in humidity, which could lead to accidental decomposition or even an intense reaction. In my experience, steel or metal containers increase risks because they corrode or might trigger a reaction, so sticking with plastic or glass reduces headaches.
Organizing shelves by chemical family also saves time and worry. I never keep oxidizers and organic substances anywhere close to each other. Even a little spilled sugar or paper can turn an accident into a disaster. Specialized cabinets with fire-resistant walls give an added sense of security. Many storage mishaps originate from laziness or ignorance about segregation. In one case I remember, a badly capped bottle of oxidizer ruined equipment – and it could have been worse had it mixed with cleaning chemicals nearby.
Labeling and Record Keeping
Accurate labels play as big a role as sturdy containers. I’ve seen labs where hand-written, faded labels hid the real contents. That led to near misses and wrong assumptions. Labels should always state the chemical name, concentration, and date received. A logbook close to the storage area helps track usage and keeps everyone on the same page about what’s coming in or going out. Someone who checks the expiration dates and any sign of container damage can spot problems early. This attention to detail isn’t about bureaucracy; it prevents emergencies or even legal issues later on.
Training and Emergency Readiness
No storage method stands up if people aren’t prepared. Every new staffer, lab tech, or student should get training specific to chemicals like zinc chlorate. I’ve run drills where we practiced quick evacuations or used spill kits under pressure. Anyone can freeze up during an actual emergency. Regular practice builds familiarity with the location of neutralizing agents, first aid kits, and fire extinguishers. After seeing how a single spark almost cost a colleague dearly, I never skip safety briefings – that repetition saves lives when seconds count.
Regulations and Best Practices
Regulatory agencies keep updating guidelines on storage, handling, and disposal. These aren’t just boxes to check off. Inspectors have shut down labs with sloppy storage, and insurance companies deny claims if basic protocols get skipped. Zinc chlorate’s oxidizing nature brings extra scrutiny from fire marshals and environmental officials alike. Storing only as much as needed, checking SDS sheets, and sending unused or expired stock to certified disposal ensures no one inherits a dangerous legacy down the line.
At the end of the day, safe storage springs from respect and vigilance. Forgetting those lessons, even once, leaves you open to costly, irreversible consequences. The rules are written in the experiences of people who learned them the hard way. Following them keeps workplaces running and people unharmed.
What is the chemical formula of Zinc Chlorate?
Straight Talk on Chemistry
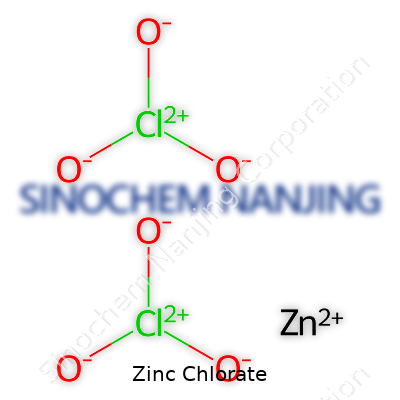
In labs and classrooms, formulas help unlock the secrets of how elements join together. Zinc chlorate stands as a good example, often popping up in high school assignments and some industry processes. The chemical formula for zinc chlorate is Zn(ClO3)2. Seeing the “Zn” at the front points to zinc, and the “ClO3” chunk marks it as a chlorate compound. With two chlorate ions bonded to every zinc atom, this compound looks simple on paper—though its chemistry gets more intricate.
Breaking Down Its Formation Steps
Growing up, teachers drilled in the basics of building formulas: find the charges, balance them, and you’re set. Zinc carries a charge of +2. Chlorate, a cluster of chlorine and oxygen, carries a charge of -1. It takes two chlorate ions to balance one zinc ion, creating a neutral compound, which gives us Zn(ClO3)2. Having that solid logic in mind helps avoid slip-ups, especially in test settings or lab calculations.
Why Zinc Chlorate Matters
Seeing those blue-and-white crystals on a lab table might not move many folks, but zinc chlorate has found roles that stretch into science projects and research efforts. In pyrotechnics, some experimenters value its ability to release oxygen during chemical reactions, which can help drive combustion processes. The compound also pops up in certain analytical chemistry procedures. I remember a botched experiment in college when we handled different chlorates—mislabeling made for a mess, and it taught me how critical precise formulas are in the lab. Getting that formula wrong brings real risks, from ruined data to safety issues.
Health and Safety Concerns
People don’t usually bump into zinc chlorate at home, but its chemical relatives teach us to take it seriously. Contact with skin or inhalation can lead to health problems—chlorates have a reputation for being harmful if mishandled. Handling any chlorate, including zinc chlorate, means following all safety procedures: gloves, goggles, good ventilation. For schools and research labs, this isn’t just about crossing items off a checklist. It’s about building trust and making sure nobody walks out with burns, headaches, or worse.
Solutions for Safe Handling and Learning
Every school and lab needs strong practices for dealing with reactive compounds. Teachers and lab managers can hold short safety checks before starting experiments. Posters showing the right protective gear, guides on how to dispose of unwanted materials, or reminders to check labels twice all play their part. Digital apps now track chemical inventories, flagging when certain items need special measures. Raising awareness matters more than any written guideline. Students pick up on cues from seasoned staff, and those habits carry over long after graduation.
Learning the Formula—and the Larger Lesson
On its face, Zn(ClO3)2 might just look like another blend of letters and numbers. But for anyone who’s ever mixed chemicals or corrected a classmate’s formula, it means paying attention and caring about details. Chemistry comes alive not just through facts, but in the careful actions of people who use its lessons safely and responsibly.
What are the safety precautions when handling Zinc Chlorate?
Understanding the Risks
Zinc chlorate isn’t especially famous outside labs, but anyone who’s handled chemicals knows how critical it is to take the right safety precautions. Zinc chlorate packs a punch—considered both an oxidizer and a toxin, it can upset your skin, lungs, or worse if handled without care. Lab techs and science teachers talk often about safety glasses and gloves, but there’s more people ought to know before even opening a bottle.
Personal Protective Gear: It Matters
Goggles aren’t just eyewear fashion in the lab. Even a tiny splash of zinc chlorate solution can burn your skin or eyes. Respirators turn out to be another common-sense must-have. Fumes or dust from this stuff can sneak up your nose and do real harm. Gloves, especially those rated for chemical protection, create a reliable barrier for hands that can easily slip and wipe your face or touch food without thinking. Lab coats or aprons help prevent accidental spills from soaking into your clothes and making contact with skin. Every layer counts since zinc chlorate doesn’t ask for permission before causing trouble.
Safe Storage: Out of Sight, Out of Harm
Years working with chemicals taught me to respect the ways things can go sideways during storage. Zinc chlorate doesn’t just sit on a shelf; it reacts strongly with organic materials, acids, and other combustibles. Leaving it near paper towels, cleaning rags, or alcohol can end in disaster. Sealed, labeled containers cut down on confusion or accidental mixing. These containers need to live on sturdy, dedicated shelves well away from heat or sunlight. Safety data sheets sit close by, not buried in a drawer, so nobody has to guess what’s in the bottle or how to respond if things spill.
Ventilation: Don’t Breathe It In
Proper airflow in workspaces does more than keep the air fresh. With zinc chlorate, the risk of inhaling dust or vapor remains a real threat. Fume hoods and chemical workstations with real exhaust systems move dangerous particles out of breathing range. Cracked windows or cheap fans won’t do the trick. Respirators help, but good lab design takes the pressure off workers to play catch-up with airborne risks.
Cautious Handling: No Room for Rushing
Zinc chlorate deserves steady hands and a clear head. I saw once how even cracking a container open too quickly sent a puff of dust into the air. Pouring, weighing, or mixing needs to happen slowly and thoughtfully—spills or splashes happen fast. Never use metal tools that might spark; plastic or wooden spatulas make safer choices. Clean every surface right after using zinc chlorate, and keep rags soaked in water, not dry, so they won’t ignite unexpectedly.
Emergency Response: Better Safe Than Sorry
Bottles of neutralizing agents and boxes of baking soda have bailed me out of more than one mistake. Nearby eyewash stations and showers save precious seconds if an accident occurs. Anyone working with zinc chlorate should know exit routes and emergency contacts. Ingesting or touching this chemical needs immediate attention from medical professionals, so a good first aid kit isn’t just backup—it’s a necessary part of the workspace.
Working Together
No one stays safe working with zinc chlorate alone. Clear communication and frequent safety drills kept my teammates and me sharp and ready. Even short training sessions helped newcomers avoid rookie mistakes. Mistakes happen when people get too comfortable or ignore small risks. Keeping those habits, that respect for the risks, has saved more than a few afternoons in the lab from turning into disasters.
| Names | |
| Preferred IUPAC name | zinc dichlorate |
| Other names |
Chloric acid, zinc salt Zinc(II) chlorate |
| Pronunciation | /ˈzɪŋk ˈklɔː.reɪt/ |
| Identifiers | |
| CAS Number | 13520-79-7 |
| Beilstein Reference | 1206826 |
| ChEBI | CHEBI:91256 |
| ChEMBL | CHEMBL1232096 |
| ChemSpider | 55744 |
| DrugBank | DB15678 |
| ECHA InfoCard | 100.016.477 |
| EC Number | 231-942-8 |
| Gmelin Reference | 126769 |
| KEGG | C18734 |
| MeSH | D015837 |
| PubChem CID | 24563 |
| RTECS number | ZH2980000 |
| UNII | 4K1O8V5E61 |
| UN number | UN1436 |
| Properties | |
| Chemical formula | Zn(ClO3)2 |
| Molar mass | 247.18 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | DENSITY: 2.2 g/cm3 |
| Solubility in water | Very soluble |
| log P | -2.2 |
| Vapor pressure | Negligible |
| Magnetic susceptibility (χ) | Paramagnetic |
| Refractive index (nD) | 1.398 |
| Viscosity | 10 cP (20 °C) |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 207.1 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -732.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -367.3 kcal/mol |
| Pharmacology | |
| ATC code | A12CB05 |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS09 |
| Pictograms | GHS03,GHS05 |
| Signal word | Danger |
| Hazard statements | Hazard statements: Harmful if swallowed. Causes serious eye irritation. May cause respiratory irritation. |
| Precautionary statements | P210, P220, P221, P280, P305+P351+P338, P306+P360, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Autoignition temperature | 410°C |
| Explosive limits | Not explosive as a solid |
| Lethal dose or concentration | LD50 (oral, rat): 500 mg/kg |
| LD50 (median dose) | 106 mg/kg (Rat, oral) |
| NIOSH | WH3575000 |
| PEL (Permissible) | PEL = Not established |
| REL (Recommended) | 1 mg/m³ |
| IDLH (Immediate danger) | IDHL: 10 mg/m3 |
| Related compounds | |
| Related compounds |
Zinc Chlorite Zinc Chloride Magnesium Chlorate Calcium Chlorate |

