Zinc Bromate: Exploring Its Path from Discovery to Possibility
Historical Development
People have known about zinc compounds for hundreds of years. Alchemists and early chemists used zinc ores in smelting and medicine even before the element received a formal name. The story of zinc bromate came later with the rise of halogen chemistry during the 19th century. Scientists noticed how combining bromine with metals created unique salts with strong oxidizing powers. As industrial chemistry matured, zinc bromate gained attention in niche applications, thanks to its distinct properties. Academic research in the twentieth century deepened our understanding, especially as researchers pushed the boundaries of inorganic reagents for analysis and synthesis.
Product Overview
Zinc bromate stands as a white crystalline salt with the formula Zn(BrO3)2. Laboratories purchase it mostly for analytical tests, as its strong oxidizing nature supports a range of experimental setups. It hasn’t found the same fame as zinc chloride or bromide in bulk chemistry, but specialty fields still rely on it for particular procedures. Often, high-purity zinc bromate arrives in closely sealed containers, evidence of digital tracking, and QR-coded for traceability. Laboratories respect its power and treat it much like other potent oxidizing bromates, logging each delivery and use to comply with safety rules.
Physical & Chemical Properties
Zinc bromate forms colorless to white crystals, sometimes showing slight deliquescence when stored under humid conditions. The salt dissolves reasonably well in water, generating solutions with strong oxidative abilities. High temperatures break it down, giving off bromine gas and creating residue that leaves no doubt about the strength stored in such a small volume. It has a molar mass of about 334.11 g/mol, a feature that matters when chemists weigh out just the right amount for quantitative analysis. Handling care keeps dust and skin contact to a minimum, as the oxidizing impact can overwhelm both delicate equipment and careless hands.
Technical Specifications & Labeling
Manufacturers provide certificates of analysis with every batch, noting zinc content, bromate levels, moisture content, and impurity controls. Typical grading focuses on laboratory reagent (LR) or analytical reagent (AR) classification, usually stating zinc bromate levels over 98% by mass, with impurities from chlorine, sulfate, and heavy metals kept comfortably below accepted tolerances. Labeling highlights its oxidizer status with prominent hazard diamonds, global harmonized system (GHS) symbols, and UN numbers for shipping. The inclusion of emergency procedures, storage tips, and shelf life dates keeps users alert and regulatory teams satisfied.
Preparation Method
In most labs, zinc bromate arises from double displacement reactions. Chemists start with zinc sulfate or zinc nitrate solutions, slowly adding sodium bromate while stirring under controlled temperatures. This process avoids local overheating and dangerous bromine liberation. A fine precipitate of zinc bromate forms, collected by filtration and washed several times with cold water to reduce sodium. Drying under vacuum or in a desiccator produces a stable, free-flowing powder. Scaling up demands extra attention—unwanted side products or poor temperature control can leave workers facing not only waste but also hazardous fumes.
Chemical Reactions & Modifications
Zinc bromate acts as a strong oxidant in both organic and inorganic reactions. Researchers use it to break down organic molecules, especially for groups that resist gentler oxidizers. It can transform certain alcohols into carbonyl compounds, though careful monitoring prevents runaway reactions. Zinc bromate decomposes under acid, releasing bromine and shifting the reaction balance. It reacts with reducing agents, making handling protocols vital—the wrong combination can trigger fires or sudden gas releases. Reducing its risk profile inspires ongoing efforts to tweak the salt with inert support matrices, hoping to keep its chemistry available without the same safety headaches.
Synonyms & Product Names
The chemical market features zinc bromate under several names. Beyond “zinc bromate,” it may turn up as “zinc(II) bromate,” or as its systematic name, “dibromic acid, zinc salt.” More obscure catalogs use identifiers like Bromic acid, zinc salt (2:1), or list its product under various supplier codes with identical composition. The widespread adoption of digital chemical registries means companies increasingly prefer CAS numbers and batch identifiers for tracking, reflecting a bigger trend toward precise sourcing and regulatory compliance.
Safety & Operational Standards
Practical experience in the lab quickly teaches respect for compounds like zinc bromate. The oxidizing label warns of fire risks, especially if the salt touches organic matter or combustibles. Breathing in dust leads to irritation, and skin contact brings a risk not just of burns but of poisoning over long exposure. Proper lab practice centers on gloves, goggles, and well-maintained fume hoods. Spill kits for bromates and emergency eyewash stations stand ready. Certain jurisdictions make periodic audits mandatory for oxidizer stocks, and records ensure nothing slips under the radar. Waste protocols ban simple disposal; trained staff collect residues and send them for specialized destruction, often via thermal decomposition managed by professionals.
Application Area
Industrial use for zinc bromate remains rare, eclipsed by more common bromates like those of sodium or potassium. Analytical chemistry textbooks often cite this compound in volumetric titrations or as a reference oxidant for calibrating laboratory methods. In specialized manufacturing, small firms might use it during catalyst preparation or as a short-lived intermediate where its powerful oxidizing potential becomes a benefit not found in mainstream chemicals. Academic research employs zinc bromate for reaction mechanism studies, tracing electron flow with a reliable, predictable oxidizer, especially in research focused on halogen cycles. Hobbyists and casual experimenters rarely encounter it—strict regulations and expense keep this material safely within professional circles.
Research & Development
Recent years have seen efforts to reimagine zinc bromate for safer, friendlier use. Researchers want to harness the oxidizing ability without the safety burden, exploring controlled-release formulations or supporting it on inert beads. Interest grows in pairing zinc bromate with green chemistry approaches, hoping to limit unwanted byproducts and cut toxic waste. Computational modeling helps labs predict reactivity, making experiments safer and more cost-effective. A few projects aim to develop analytical sensors using bromate’s responsive chemistry with colorimetric or electrochemical signals, blending old-school reagents with new materials science. These pockets of innovation keep zinc bromate on the radar for targeted, high-value applications.
Toxicity Research
Zinc bromate’s danger profile raises several flags in toxicological studies. Bromate ions disrupt cellular function in mammals, leading to organ damage, especially in kidneys and the nervous system. Animal testing points to the threat of acute poisoning, with nausea, vomiting, and rapid tissue damage if inhaled, swallowed, or absorbed in even moderate doses. Environmental scientists worry about persistence and bioaccumulation. Zinc may not trigger huge concerns, but bromate ions can resist breakdown and migrate through soil and water. Modern labs run tight monitoring programs, using validated analytical methods to detect even trace spills. Work continues to identify fast, reliable antidotes in the event of accidental exposure and to lower the impact on the natural world, with some early progress on microbial remediation in lab-scale trials.
Future Prospects
Zinc bromate sits at an intersection—valued for specialized reactions, yet pinched by cost, safety, and environmental realities. Its future may not bring massive global markets, but niche fields could still benefit from its unusual properties if research delivers safer, user-friendly forms. Stricter regulations and the push for greener chemistry likely signal a move away from routine use. Still, every so often, a targeted reaction or analytical need will call for just this blend of power and predictability. The next wave of work may look less like mass production and more like custom solutions: miniature cartridges for sensors, supported reagents in flow reactors, or sealed kits for on-demand oxidation. Each development reflects the drive to manage risk—putting knowledge, experience, and trust at the center of chemistry’s progress.
What is Zinc Bromate used for?
The Role of Zinc Bromate in Laboratories and Industry
Most people don’t walk into a store or pharmacy looking for zinc bromate. You won’t find it on a list of common household products. Instead, zinc bromate has a much quieter but important role, especially in labs, specialty chemical processes, and sometimes in academic settings. At its core, zinc bromate works as a strong oxidizer—meaning it encourages certain chemical reactions to occur quickly and completely. This kind of property gets a lot of attention from chemists who need to carry out oxidation reactions with control and precision.
Chemistry students like me used to wrestle with all sorts of reactions in the lab. Zinc bromate would come up in analytical chemistry when learning to measure small amounts of other substances. Because it gives up oxygen easily, it helps identify or separate certain chemicals through oxidation. So, even though its name doesn’t pop up in big headlines, zinc bromate is useful for teaching budding chemists the basics of reaction pathways and stoichiometry.
Why You Won’t Find Zinc Bromate in Every Lab
Not every school or research lab lines the shelves with bottles of this chemical. Handling zinc bromate isn’t simple. It rates as hazardous, because large amounts can explode if mixed with organic materials or exposed to heat. Breathing in the dust or getting it on your skin can cause health problems. Guidelines from groups like the Occupational Safety and Health Administration (OSHA) point to careful storage and strict handling rules. Some safety manuals even tell new lab workers to keep it far from flammable objects or reducing agents. From my early days studying chemistry, I learned to always treat oxidizers with healthy respect—but zinc bromate, you especially don’t take chances.
There’s also the question of whether newer, safer oxidizers can get the job done. In some cases, scientists replace zinc bromate with other chemicals that pose fewer risks. That doesn’t mean zinc bromate is irrelevant, but it limits its use to settings where its unique traits are truly needed.
Potential Environmental and Health Concerns
Here’s another reason people pay attention: zinc bromate can show up in waste streams from chemical plants. Improper disposal threatens both environmental and human health. Zinc and bromate ions, if they reach water supplies, interfere with aquatic life and may create human health risks. It reminds me of stories you hear about chemical spills and the long road to recovery for land and water nearby. Good chemical management means looking past the workbench to think big-picture: how does this affect others beyond the walls of the lab or factory?
Building Better Practices with Education and Innovation
Years ago, chemistry classes emphasized memorizing facts. Now, there’s a shift toward smarter handling and real-world application. Training new scientists to work safely with things like zinc bromate requires resources, smart supervision, and updated procedures. Companies and universities can keep better records, choose greener alternatives when possible, and invest in equipment that contains spills or prevents dust from escaping. Making safe disposal a habit takes time but pays off for everyone downstream—literally and figuratively.
Zinc bromate’s story isn’t flashy, but it highlights the careful balance that comes with all chemicals: using their benefits while managing risks, so science can keep moving forward without leaving problems behind for others to solve.
Is Zinc Bromate toxic or hazardous?
Why Zinc Bromate Calls for Attention
Zinc bromate isn’t a substance most folks think about while going about their day. Still, it sits on shelves in labs and can wind up in workplaces through cleaning agents, chemical research, or industrial byproducts. I remember walking into a university lab and seeing all those bottles lined up—labels with names you’d barely recognize, but behind each is a story of risk and responsibility.
Health Risks Lurk in the Details
The chemistry behind zinc bromate carries real consequences. Based on data published by reputable sources like the CDC and National Library of Medicine, exposure to this compound can trigger irritation in the skin, eyes, and even lungs. Breathe in some of that dust or mist, or get it on your skin, and you might quickly notice redness, itchiness, or worse.
Animal studies have shown organ damage and changes to blood chemistry after swallowing zinc bromate. These effects extend to humans as well. Small accidents in labs have been enough to send people to the hospital for acute symptoms. The bromate component itself gets most of the blame—bromates are considered potential carcinogens and can lead to thyroid and kidney damage after prolonged or severe exposure.
The real sting comes from the fact that even short-term exposure carries risks, meaning any accidental spill, leak, or carelessness in handling could cause serious medical issues. Long-term risks sneak up slowly, but one event can permanently affect your health. The Environmental Protection Agency and agencies worldwide treat bromates with caution, calling them hazardous substances.
Environmental Hazards: Not Just a Human Problem
The risk doesn’t stop at skin. Spilling zinc bromate can mess with aquatic animals and disrupt soil chemistry. Once released into water, bromates harm fish and plankton, crucial links in the food chain. Cleanup after contamination proves complicated and costly. I’ve seen local stories where small chemical spills closed off whole sections of public waterways, hurting both the ecosystem and local fishing economies.
Disposal causes headaches for industry, too. This is not the type of chemical you can casually wash down the drain. Strict environmental controls force companies and labs to treat waste as hazardous, and even a minor slip-up might bring steep penalties. Legal action or costly cleanup follows for those that get careless.
Keeping People Safe: Practical Steps
Knowing the dangers is just the start. Lab workers, manufacturers, and cleaning crews need practical protocols. Personal protective equipment isn’t optional—gloves, goggles, and face masks create barriers that reduce the chance of irritation or inhalation. Training staff to recognize spill risks and respond fast makes a huge difference. Labeling storage clearly and keeping containers sealed prevents accidental contact.
Even in schools and research centers, integrated safety plans cut the risk of exposure. Kits with absorbent materials, neutralizing agents, and the knowledge to use them make labs safer. Regular inspections, proper ventilation, and responsible chemical storage all add up—small steps that have saved countless people from a trip to the emergency room. Based on experience, having people ready to handle an emergency is just as important as using the right gear.
Looking Ahead: Prevention Over Recovery
Most accidents happen in settings where procedures slip. Creating a culture that prioritizes health and environmental protection can mean the difference between a regular workday and something headline-worthy. Leaders sharing information and following regulations teach respect for these risks. Smaller businesses sometimes cut corners or lack resources—offering outreach, training, and government support helps keep everyone up to speed.
Zinc bromate may not be part of daily conversations, but its hazards remain real for the people who handle it and the world they live in. Vigilance, education, and honest communication close the gap between risk and responsibility.
What is the chemical formula of Zinc Bromate?
Breaking Down the Science
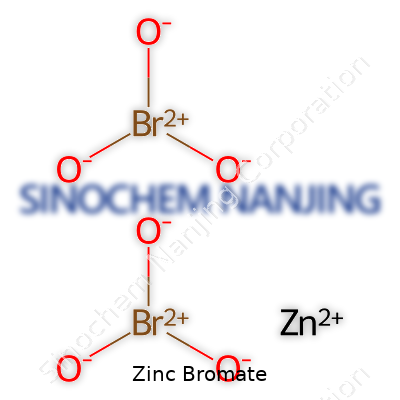
Zinc bromate carries the chemical formula Zn(BrO3)2. Looking at this formula, the curious part to many comes down to the arrangement of atoms and their charges. Zinc stands with a +2 charge. Bromate, a polyatomic ion, offers a -1 charge for each unit. It takes two bromate ions to balance one zinc ion, so you get Zn(BrO3)2. That little ‘2’ tacked onto bromate means both math and chemistry agree—not some random assignment, but a careful balance of electrical charges. In school chemistry labs, these moments stick—the thrill of puzzles solved at the periodic table.
Why Does This Matter Outside the Classroom?
Zinc compounds turn up everywhere—coatings, supplements, batteries, chemical catalysts. Bromate rings alarm bells for folks in water treatment or food safety because certain bromates can cause health issues. The formula isn’t just for dusty textbooks; it tells processors and safety officers what they’re actually working with. If you see Zn(BrO3)2 on a data sheet or hazard label, it’s time to think about handling, disposal, and personal safety. The Environmental Protection Agency in the U.S. keeps a close eye on bromates for good reason. Studies have shown that high bromate exposure links to negative health outcomes, including an increased risk of cancer in certain animal studies. So this isn’t just about trivia at the science fair.
Inside Industry and Daily Life
The clear identification of compounds like zinc bromate helps ensure both safety and precision. Chemical plants, plating shops, even research labs rely on knowing if they’re dealing with zinc bromide, zinc bromate, or some other similar-looking compound. The formula tells everyone just what might react, how to store materials, and which protocols to follow. Back in my own research days, accident reports often traced back to someone mistaking one compound for another based on a lazy read of the label. That’s not just inconvenient—it can turn dangerous quickly. Proper use of chemical formulas prevents these errors.
Supporting Safer Practices
Educators feel frustrated when students memorize chemical formulas without connecting them to real outcomes. Take zinc bromate’s formula: if you understand what each piece stands for, you’re more likely to manage chemicals with respect and care. This mindset has driven policy changes. Industries ramped up staff training and improved labeling partially because long chemical names got mistaken or ignored. Safety drills, standardized containers, and relying on clear formulas all cut down on lab accidents. A lot comes back to education—making sure workers and students both recognize those subscripts matter.
The Path Forward
Looking at chemical formulas isn’t about dry recitation. Recognizing Zn(BrO3)2 involves a habit of careful checking, alertness, and asking questions. More public education on chemical naming helps the next generation avoid preventable mishaps. Companies investing in up-to-date labeling systems and hands-on safety workshops get ahead by lowering accident rates. Those steps hinge on getting the fundamentals right—knowing exactly what the formula says, and what it implies for real-world handling.
How should Zinc Bromate be stored?
Looking at Safety from Experience
People often check the label, shrug, and toss chemicals on the shelf. I’ve seen plenty of labs put storage on autopilot—even for tricky compounds like zinc bromate. From days in an undergraduate lab, to stops in small manufacturing, there’s one lesson that sticks: treating strong oxidizers as an afterthought doesn’t end well. Zinc bromate, with its sharp oxidizing power, fits that reality.
Risks on the Table
Forget a technical lecture—zinc bromate’s dangers show up fast with poor storage. It can trigger fires or mix violently with common materials. This isn’t academic paranoia. A spilled container ended with scorched countertops in my old chemistry club. After that, everyone respected storage protocols.
Experience Speaks on Proper Containers
Grab a cheap plastic lid or a corroded tin, and you’re asking for trouble. Zinc bromate calls for sturdy glass or specialized plastic. Any hint of metal prompts corrosion, and flakes end up contaminating the compound. In my own home setup, the container earns a weekly check—cracking lids or accidental mixing stops before it begins. Simple habits, but with big payoffs in long-term safety.
Cool, Dry, and Out of the Sun
Humidity loves to mess with storage. A damp shelf leads to clumping and worsens reaction risks. In one summer, a forgotten bag absorbed enough moisture to form a slushy paste—a ticking time bomb. Keeping containers tightly closed really isn’t just a suggestion. High school storerooms and university stockrooms that kept oxidizers low on shelves and away from vents rarely had problems. Never near a heating pipe, always in a stable, dry place. Even sunlight speeds up degradation—so away from the window goes the jar.
No Neighbors Allowed
Zinc bromate acts up with a surprisingly wide set of chemicals. Tossing it near old solvents or acetone courts disaster. Mixing oxidizers with anything organic or flammable has led more than one lab to swap stories about minor explosions. Many institutions run separate cabinets—for oxidizers only. It always felt a little over the top in my early days, but over the years, those bright signs saved people from expensive (and scary) cleanup.
Labels Matter—Really
No matter how small the sample, a missing or unclear label invites someone to treat it like table salt. Stories of mislabeled bottles are everywhere. Include the chemical name, a hazard warning, and a record of when it was stored. Even a Sharpie date solves confusion. In my time, we kept a log next to the storage area—no science fiction, just old-fashioned common sense.
Practice Makes Habit
Nobody remembers every guideline from training. What sticks is a mindset—treating harsh chemicals with consistent respect. Regular self-audits, twice-yearly reminders, and visible safety sheets build the habits that protect hands and labs. Labs with a bit of pride in their storage rarely find themselves on the receiving end of a crisis.
Looking Forward: Simple Steps, Real Results
Zinc bromate poses clear hazards but solid storage steps—good containers, dry shelves, sensible separation—manage most threats. Labs, schools, and even home hobbyists relying on tested routines avoid emergencies and keep their teams safe. It’s boring, sometimes tedious work, but it preserves livelihoods and reputations.
What are the safety precautions for handling Zinc Bromate?
Working With a Tough Chemical
Zinc bromate isn’t the kind of compound you stumble across doing routine lab work. It brings both oxidizing power and toxicity to the table, so nobody can afford to treat it like table salt. Splash some on your skin or in your eyes, and you’ll remember that lesson for life. Reports from the National Institute for Occupational Safety and Health (NIOSH) stress regular injuries tied to haphazard handling of strong oxidizers. Chemistry classrooms and labs carry spill scars as reminders.
Understanding the Risks
Breathe in zinc bromate dust, and your lungs might feel the sting. Get it on your hands, and you’ll get itchy red skin or even burns. If it ever touches your eyes, damage follows fast—sometimes permanent. That’s not all. Bromate ions can kick up risks of kidney damage and even cancer if exposure happens often enough.
Because it oxidizes so easily, a spark, a bit of friction, or even mixing with flammable material can start a fire or a miniature explosion. Over the years, the U.S. Department of Transportation flagged containers of zinc bromate because of its readiness to fuel dangerous reactions.
Precautions That Make a Difference
I learned from long days spent in teaching labs that following proper procedures saved time and skin. Gloves matter—choose ones tested for chemicals. Most nitrile gloves keep liquid from soaking through, and they hold up longer than latex. Always put on safety goggles. If there’s ever a spill, splash, or even a speck that floats up during weighing, those goggles prove their worth. For bigger jobs or any chance of a splash, face shields and lab coats give more coverage.
Never work with zinc bromate near open flames or hot surfaces. It only takes one lapse for an accident to jump from a minor scare to an emergency. University safety officers know these stories too well: someone uses a heat gun or careless burner, and suddenly the reaction goes wild. Store the chemical in dry, cool cabinets made of non-reactive materials, far from flammables or anything organic. Separate oxidizers from fuels the same way professionals keep matchsticks and gasoline apart.
Vents and fume hoods play a crucial role. Most modern teaching and research labs use them by default, and for good reason. One whiff of concentrated dust triggers coughing and hurts your lungs. Fume extraction keeps lab air safe to breathe and drastically cuts down the risk of chronic exposure. Don’t skip routine air monitoring—it’s a game changer for peace of mind.
Cleaning Up and Training
Spills do happen, and reaction speed is everything. I’ve seen students freeze during a spill. Drills help. Lay out an absorbent material that works against oxidizers, sweep with special brooms or pads, and never just toss the waste in the usual bin. Specialized hazardous waste containers keep the danger contained until qualified disposal contractors pick them up, following local environmental guidelines.
Good safety comes from planning and experience, not just reading the manual. Hold regular training sessions, keep emergency showers accessible, post contact details for poison control next to the work area, and run drills until everybody feels confident. No shortcut replaces knowing each step and practicing safety as part of every lab session.
Turning Lessons Into Action
Zinc bromate doesn’t offer second chances. Safe handling boils down to staying protected, paying attention, and treating every session as high-stakes. Labs with good track records put people above shortcuts, invest in real training, and never assume everyone will just “know what to do.” That’s how you preserve both people and progress.
| Names | |
| Preferred IUPAC name | zinc dibromate |
| Other names |
Zinc dibromate Zinc(II) bromate |
| Pronunciation | /ˈzɪŋk ˈbroʊ.meɪt/ |
| Identifiers | |
| CAS Number | 13520-92-8 |
| Beilstein Reference | 14607 |
| ChEBI | CHEBI:86361 |
| ChEMBL | CHEMBL3300892 |
| ChemSpider | 61740 |
| DrugBank | DB15974 |
| ECHA InfoCard | ECHA InfoCard: 100.022.881 |
| EC Number | 236-389-1 |
| Gmelin Reference | 4125 |
| KEGG | C18768 |
| MeSH | D015853 |
| PubChem CID | 24418 |
| RTECS number | EZ9600000 |
| UNII | YV7M436C8N |
| UN number | UN1474 |
| Properties | |
| Chemical formula | Zn(BrO3)2 |
| Molar mass | 333.2 g/mol |
| Appearance | White crystalline solid |
| Odor | Odorless |
| Density | 4.2 g/cm³ |
| Solubility in water | Soluble |
| log P | -0.85 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 4.07 |
| Magnetic susceptibility (χ) | +63.0e-6 cm³/mol |
| Refractive index (nD) | 1.781 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | S⦵298 = 197.9 J∙mol⁻¹∙K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -732.1 kJ/mol |
| Pharmacology | |
| ATC code | V07BB |
| Hazards | |
| Main hazards | Oxidizer, harmful if swallowed, causes severe skin burns and eye damage. |
| GHS labelling | GHS labelling: Danger; H272, H315, H319, H335, H410; P210, P220, P261, P273, P305+P351+P338, P337+P313, P370+P378, P391, P501; Oxidizer, Irritant, Harmful to aquatic life |
| Pictograms | GHS05,GHS07 |
| Signal word | Danger |
| Hazard statements | H272, H302, H314, H410 |
| Precautionary statements | P220, P221, P261, P264, P270, P273, P280, P301+P312, P305+P351+P338, P330, P337+P313, P370+P378, P391, P404, P405, P501 |
| NFPA 704 (fire diamond) | 2-0-2-OX |
| Lethal dose or concentration | LD50 oral rat 100 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 780 mg/kg |
| NIOSH | DH8225000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Zinc Bromate: Not established. |
| REL (Recommended) | Minimize |
| Related compounds | |
| Related compounds |
Zinc sulfate Zinc chloride Zinc nitrate Zinc carbonate Zinc phosphate Zinc bromide Potassium bromate Sodium bromate |

