Zinc Bisulfite: From Origins to Future Uses
Historical Development
People started looking at zinc salts with serious scientific curiosity about a century ago. Chemists realized that adding sulfurous acid to zinc compounds produced something entirely new: zinc bisulfite. The focus early on centered mostly on zinc sulfate, but the need for selective reducing agents and stabilizers set the stage for zinc bisulfite’s debut. Researchers in Europe began isolating it in small quantities by the late-1920s, but the chemistry stayed inconsistent. It didn’t stabilize or store well, so most labs kept working with other zinc salts. Toward the 1960s, with industrial researchers searching for specialized water treatment agents, interest in zinc bisulfite picked up again. Better lab techniques brought purer crystals and repeatable yields. Over the years, industries tuned in, especially pulp and textile processors, because they needed agents that could keep chemical baths stable and less corrosive than alternatives.
Product Overview
Zinc bisulfite comes off as a specialty zinc salt. It rarely shows up on hardware store shelves and sits quietly in the background, supporting bigger processes. Its formula—Zn(HSO3)2—places it firmly in the bisulfite family, but not as a rival to plain sodium or potassium bisulfites. Its main use? To act as a mild reducing agent, often in bleaching, desulfurization, and water purification operations. Many people working in pulp mills don’t see it directly, but the compound punches above its weight by scavenging unwanted oxidants and fighting off pesky byproducts. Unlike zinc sulfate or zinc chloride, zinc bisulfite doesn’t carry strong oxidizing or dehydrating behavior, but instead sits right between, offering a safer profile where harshness creates unwanted byproducts.
Physical & Chemical Properties
Zinc bisulfite appears as a white to faintly yellowish powder or crystalline solid, often clumping in damp environments thanks to its hygroscopic nature. The crystals dissolve easily in water, making them handy for quick preparation of dilute solutions. At room temperature, the salt remains stable, but exposure to strong acids releases toxic sulfur dioxide gas, something safety managers never take lightly. Last time I worked with it, the sulfurous odor became noticeable even through a fume hood. Chemically, it keeps a moderate profile, tending toward reducing reactions but shying away from aggressive behavior. Its solubility helps in fast prep work during batch chemical treatments. Sensitivity to excess heat and humidity shows up when storage conditions slip, so sealed drums or glass containers matter quite a bit.
Technical Specifications & Labeling
Every drum or bag of zinc bisulfite ought to carry straightforward labels. Typical commercial grades state purity between 92% and 97%, with zinc content generally falling near 22%. Water content sits low, but enough to affect shelf life in humid warehouses. Official containers normally sport hazard labels mentioning its corrosive and irritant nature, plus warnings about inhaling dust or allowing contact with acids. The European REACH program and U.S. OSHA standards both consider it hazardous for shipping, so MSDS sheets (Material Safety Data Sheets) need to travel with every shipment. I remember one supplier tacking on a color-coded label for quick visual identification, an idea that stuck with me for any lab with multiple similar-looking compounds. Most buyers ask for granular form, but fine powders sometimes slip into sensitive applications.
Preparation Method
Making zinc bisulfite requires more patience than muscle. Most processes start with high-purity zinc oxide or zinc sulfate. Adding excess aqueous sulfurous acid (not sulfuric!) allows bisulfite ions to react directly with zinc in solution. In one pilot plant I toured, they adjusted the pH just enough to favor bisulfite over sulfite or sulfate without forming zinc hydroxide. The trick comes in managing the stream:gently stirring to prevent hot spots and closely watching for bone-white precipitate. Any shortcut risks forming hard-to-separate byproducts or choking the line with clumped crystals. Once formed, filtration and careful drying deliver the finished salt. One batch always tells: too much heat, and the final product browns or cakes, something the market never forgives. Lab-scale techniques sometimes swap sodium bisulfite, metathesis, and zinc nitrate for quicker synthesis when only small quantities make sense.
Chemical Reactions & Modifications
Zinc bisulfite steps up as a mild, selective reducing agent in several industrial settings. I watched it reduce chlorine dioxide in water purification setups and saw operators leaning heavily on its low tendency to over-react or degrade organic matter. The compound reacts easily with standard oxidants but, uniquely, it sometimes complexes with organic molecules, giving it a role in dye and paper pulping. Chemical tweaks often mean substituting standard bisulfite ions or introducing stabilizers to slow hydrolysis when dissolved. Adding gentle chelators stabilizes zinc, holding back unwanted zinc oxide formation. In organic syntheses, researchers sometimes use it to trigger reduction without jumping straight to harsher agents like sodium borohydride. Lab modifications remain simple: mixing with compatible solvents or altering pH, never requiring exotic steps.
Synonyms & Product Names
Zinc bisulfite goes by a handful of aliases, confusing buyers new to the field. Common names include zinc hydrogen sulfite, zinc(II) bisulfite, and even less frequent terms like zinc acid sulfite. Some regional suppliers call it “sulfuretted zinc,” but this label crops up mostly in old literature. On international documents, “zinc bisulphite” sometimes appears, reflecting British English spelling. Unique product codes or trademarks don’t pop up often, unlike larger commodity chemicals, but a handful of specialty suppliers tag it with internal product numbers for custom grades.
Safety & Operational Standards
All work with zinc bisulfite demands careful safety practices. The fine powder irritates mucous membranes and, if handled carelessly, can provoke skin rashes or brief respiratory coughs. Spills in open spaces need quick cleanup, because moisture pulls SO2 out of the salt and makes air quality questionable. I always insist on gloves, goggles, and—if working outside a fume hood—a dust mask. The compound reacts fast with acids, evolving toxic sulfur dioxide gas, a well-known danger for anyone unlucky enough to inhale it. Storage cuts risk: dry, sealed drums away from acids and direct sunlight. Industrial operations set out clear SOPs, with regular safety drills and clear evacuation plans for accidental releases. Although not flammable, nearby heat sources should never threaten storage sites. Disposal always lines up with regional hazardous waste codes; never down the drain or in regular trash.
Application Area
Zinc bisulfite plays an understated but crucial role mainly in industrial operations. Paper and pulp manufacturers lean on it to maintain white color during the pulping process, curbing oxidative damage to fibers. Textile plants use it to bleach and treat fabrics without aggressive chemical attack, so their final products hold up better over time. In water treatment, it steps in as a mild reducing agent to tackle trace oxidants, especially chlorine dioxide, sidestepping stronger, more dangerous chemicals. Smaller research labs land on it for redox experiments and select organic syntheses, checking reduction power against other agents. Some niche agricultural trials use dilute zinc bisulfite to study micronutrient delivery, but the main action lives in the big, continuous chemical streams where mild reduction matters more than brute force.
Research & Development
Research teams continue to tinker with zinc bisulfite, searching for better stability and broader applications. Chemists keep testing new stabilizers, hoping to hold the salt’s structure for longer shelf lives. Novel water purification studies try blending zinc bisulfite with novel polymers, checking if composite materials unlock new filtering or neutralizing abilities. Universities run pilot projects, measuring long-term effects of low-dose zinc bisulfite on plant and animal systems. People with analytical chemist backgrounds dig into its potential for use as a gentle, reversible reducing agent in fine chemical synthesis. Challenges persist: keeping the salt stable, reducing dustiness, and lowering the reactivity with atmospheric moisture all show up often in lab notebooks and patent filings. Some researchers chase modifications that might enable more selective reduction, or even let the salt function as a trigger for controlled-release formulations.
Toxicity Research
Toxicology work on zinc bisulfite still hasn’t reached the intensity of studies on zinc sulfate or sulfite, but a few points stand out. Zinc itself counts as an essential trace element, but both zinc ions and bisulfite ions can create toxicity risks above certain thresholds. Inhalation of dust, or direct ingestion, leads to stomach upset, temporary breathing problems, or—in rare, concentrated exposure—neurological symptoms. Animal studies have tracked metabolic pathways, with low doses tolerated well, but high exposures disrupt enzyme systems, largely due to the sulfite content. In water, breakdown to SO2 can create hazards for aquatic species. Safety reviewers recommend keeping exposure well below industry threshold limit values and using personal protective equipment at each handling step. Chronic, low-level exposure remains less studied, a gap universities and regulatory bodies look to close as use widens.
Future Prospects
Looking ahead, interest in zinc bisulfite doesn’t seem set to fade. Cleaner industrial processes push companies to swap toxic or aggressive reagents for gentler options, an area where zinc bisulfite fits neatly. If stabilizing modifications mature, the compound could slip into more advanced water treatment and gentle bleaching applications. Agricultural technology firms eye it for micronutrient delivery in slow-release formats, combining mild reduction with essential trace element support. Bio-based industrial processes, especially those that need non-chlorine bleaching or reduction, might open new doors for usage. Research into safe, long-storage formulations—granulated or coated forms—promises to cut handling risks and broaden possible applications. Big challenges still need tackling: environmental impact of discharge, reliable mass-scale preparation, and in-depth chronic toxicity work all linger on research to-do lists. The appetite for mild, versatile reducing agents, particularly those that help large-scale industries lower their overall environmental bite, should keep zinc bisulfite worth watching in the years to come.
What is Zinc Bisulfite used for?
What Makes Zinc Bisulfite Stand Out
Zinc bisulfite doesn’t usually pop up in dinner conversations. Still, in the world of industry and environmental management, it actually plays a unique role. With experience working alongside wastewater engineers and talking with factory managers, I’ve seen first-hand how zinc bisulfite provides solutions for problems you don’t want left unsolved.
Where You Find Zinc Bisulfite in Everyday Life
Most folks don’t realize how much effort goes into making water, paper, and other basics as clean and safe as they appear. Take water treatment: facility operators depend on zinc bisulfite as a reducing agent. It helps break down excess chlorine after disinfection, so the water pumped out doesn’t carry dangerous leftovers. The same chemical comes into play in pulp and paper factories. Workers struggle to prevent pulp from yellowing, and this compound cuts down on oxidation, keeping the paper whiter than it would be otherwise.
Folk wisdom might say “let it be,” but with zinc bisulfite, industry professionals know that leaving contaminants or unwanted reactions unchecked can risk both public health and the shelf-life of products. Unsafe drinking water and yellowed paper are issues that are more expensive to fix down the road. The value of this chemical shows up in quiet reliability.
The Science Behind the Applications
People working in water treatment use zinc bisulfite because it interacts with chlorine, neutralizing it so that the water, once released, doesn’t harm fish, crops, or communities downstream. Tests from the Environmental Protection Agency (EPA) support this process, as strict limits on residual chlorine demand a fast and safe way to bring it to zero.
Paper manufacturers notice another perk: improved brightness and quality in finished products. That doesn't just mean nicer-looking notebooks; mills save money by reducing waste and cutting energy use during production.
Balancing Benefits With Safety
Experience teaches that even the most useful things come with risks. Zinc bisulfite exposure can cause irritation or worse if handled carelessly. Anyone who has worn a chemical suit on a humid summer day knows there’s nothing pleasant about emergency rinses after an unexpected splash. Training, proper storage, and strict labeling keep teams protected. OSHA guidelines spell out safety rules, and regular drills make sure no one ignores them.
Opportunities for Greener Choices
Some plants in the chemical industry are searching for formulas that deliver the same cleaning power but create less sludge and fewer byproducts. Researchers investigate alternatives, as industries face tighter pollution limits. The answers might not replace everything overnight, but talking openly about greener technologies keeps pressure on manufacturers to cut down on waste and find safer options.
Looking Ahead
Safe handling and smart use make zinc bisulfite an ally in tackling today’s industrial challenges—turning raw water into something fit for families and keeping everyday products sharp and dependable. In offices, on factory floors, or across research labs, people keep asking how to get the most out of this chemistry without costing the earth. The work to answer that question keeps factories safer, water cleaner, and products better for all of us.
Is Zinc Bisulfite safe for human consumption?
Understanding Zinc Bisulfite
If you’ve spent any time looking through ingredient lists, you might have seen some chemical names that sound a little intimidating. Zinc bisulfite falls right into that category. Used mostly in industry, it acts as a bleaching or reducing agent. Unlike the zinc found in your daily multivitamin, this compound mixes zinc with bisulfite, which comes from sulfur. This isn’t just the zinc you find in oysters or pumpkin seeds.
What Science Says About Its Safety
Zinc is an essential mineral — your body uses it for immune function, wound healing, and metabolism. We know a lack of zinc leaves people more open to infections. Bisulfites, on the other hand, are familiar as food preservatives, keeping dried fruits and wines from going bad. Both have separate safety records in regulated amounts. The trouble starts when the two come together in a chemical compound, like zinc bisulfite. Scientific studies and regulatory assessments don’t cover this exact mix for food or supplement use. The FDA lists zinc sulfate and gluconate as safe sources of added zinc, but not zinc bisulfite. Experience from food safety research tells us unknown chemicals shouldn’t go into food until their effects are well understood.
Risks of Consuming Zinc Bisulfite
Compounds with bisulfite can trigger trouble for some people. Folks with asthma might get respiratory symptoms. There are rare cases where bisulfites cause serious allergic reactions. As for zinc bisulfite, research on how it breaks down in the body—and what byproducts stick around or get absorbed—remains thin. Most experts don’t put their trust in chemicals that haven’t been studied for human consumption. Lab toxicity tests sometimes show irritant effects at higher doses, especially for things not meant to be eaten. No regulatory agency in the US, Europe, or Asia approves zinc bisulfite for food or supplement use. That alone sets off alarm bells for many in food safety.
Recognized Zinc Sources in Nutrition
Over the years, regulators and scientists have settled on a few forms of zinc as safe and effective in foods and pills: zinc sulfate, zinc oxide, and zinc gluconate. Thousands of clinical trials use them in nutrition studies. These forms dissolve safely in stomach acid, don’t interact badly with other food ingredients, and have well-understood absorption rates. The World Health Organization, the US National Institutes of Health, and countless universities have weighed in with solid numbers on recommended intake. People relying on these sources lower their risk of deficiency, with little worry about harmful effects when taken as directed. Using a wild card like zinc bisulfite makes little sense given established, studied choices.
Key Takeaways and Steps Forward
Food safety doesn’t leave much room for guesswork. If a chemical compound hasn’t earned trust through long-term studies and official approval, asking people to eat it doesn’t sit right. Companies develop safer options by sticking to compounds with a track record and proven benefit. The answer here seems clear: stick to what’s known and safe, and demand transparency from labeling. We all eat better when food makers and regulators pick evidence over expediency. Getting safe, well-studied minerals in our diet leaves less room for unpleasant surprises down the line.
What is the chemical formula of Zinc Bisulfite?
Understanding Zinc Bisulfite at Its Core
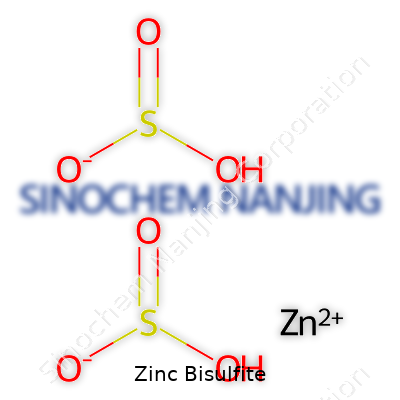
Zinc bisulfite doesn’t show up in conversations the way table salt or baking soda might, but it quietly finds space in chemical supply lists and research papers. Anyone who has handled chemicals for water treatment or pulp bleaching knows the mess of similar-sounding names—there’s zinc sulfate, zinc chloride, and then, less often, we see zinc bisulfite. Its formula shows up as Zn(HSO3)2. That’s one zinc ion paired up with two bisulfite ions, each carrying a mix of sulfur, hydrogen, and oxygen atoms. The subscripts in the formula sound technical, but breaking them down, we’re just counting parts that react together to make the compound.
Why Accuracy in Chemical Formulas Matters
A missing atom in a formula turns a mild irritant into something corrosive. I remember a time during my undergraduate years, a friend mixed up bisulfite and sulfite compounds in a basic analytical lab. One corresponds to HSO3- and the other to SO32-. The results made a mess of our experiment and turned a simple qualitative test into damage control for the afternoon. Nothing drives home the importance of chemical literacy like an unexpected reaction or the smell of burning sulfur. Real risks exist when misinformation about chemical identities makes its way into practice, especially for those less familiar with the lab jargon.
Applications and the Need for Responsible Chemistry
Zinc bisulfite comes into play as a reducing agent, often showing up in water treatment or in specific industrial settings. Each sector depends on accurate chemical sourcing. In my time working with water safety projects, I watched how teams checked and double-checked reagents. If the label read “zinc bisulfite,” anyone in charge needed to know they were getting Zn(HSO3)2, not something else. Getting this wrong could throw off results and potentially contaminate what should be clean water for a whole community. Companies and schools, big or small, count on reliable information at every step—right from scientific supply catalogs to the training manuals in a teaching lab.
Challenges with Naming and Safe Handling
Chemists and students alike run into name confusion, especially from misleading product names or poorly maintained storage areas. You might spot “zinc bisulphite” on one shelf, “zinc bisulfite” on another—small spelling differences sometimes hint at regional Terminology rather than real chemical differences. Getting the formula right, and verifying the chemical inventory, matters much more than the pronunciation or spelling. Every year, news trickles in about accidents made worse because a team trusted a supplier or old database entry without checking against trusted sources.
Building Better Practices
Anyone working with potentially hazardous substances benefits from clear education. If your school or company hosts chemistry workshops, invite speakers who’ve had hands-on, not just textbook, experience. Companies in the chemical space should make chemical data sheets accessible, readable, and crystal clear—nobody wants to dig through a 30-page document to figure out if they’ve got Zn(HSO3)2 or something different. Keeping updated training materials and periodic audits helps prevent dangerous mistakes—both in industry and in classrooms where tomorrow’s chemists are training.
Answering the question about zinc bisulfite’s chemical formula uncovers a bigger truth: accuracy isn’t just academic pride; it’s practical safety and real-world financial sense. A clear formula—Zn(HSO3)2—means less confusion, less waste, and less risk all around.
How should Zinc Bisulfite be stored?
The Basics Behind Good Storage
Zinc bisulfite, a salt used in water treatment, bleaching, and some specialty chemical processes, doesn’t get much attention until something goes wrong. Poor storage brings real headaches, from wasted product to safety scares. Most people dealing with chemicals recognize the smell of trouble when bags break or liquids spill, but letting zinc bisulfite get damp or contaminated creates quieter problems: lost potency, clumpy residue, or even dangerous reactions.
Practical Lessons from Real Workplaces
I remember a warehouse job where a few careless pallets sat near a leak. Over weeks, the white powder yellowed and formed hard cakes—it became useless, and the boss had to call in hazardous waste pickup, adding cost to embarrassment. This happens far more often than published incident reports show.
Keeping zinc bisulfite dry beats any complicated solution. Store it on pallets or sturdy shelving to keep bags and containers away from floors. A concrete slab may seem fine, but humidity seeps up. Raised racks also help with airflow—less chance of mildew, even during rainy seasons. Always look for a place with a tight roof; one drip can cost hundreds in spoiled material.
Why Temperature and Air Matter
People sometimes laugh about warnings against “extreme temperatures,” but anyone who’s pulled product from a building with little climate control in July or January knows better. If zinc bisulfite shifts past its shelf life due to constant swings—freezing freezes, then sweaty heat—expect it to stick together, degrade, or grow suspiciously chunky. No one wants to toss out half a season’s supply.
Many suppliers recommend staying in the ballpark of ordinary room temperature, keeping windows closed, and using exhaust fans or dehumidifiers in muggy places. Old buildings with cracked windows send moisture right into every open seam. Even the most expensive packaging won’t save a product from careless handling or a humid storage shed.
Guarding Against Contaminants
Zinc bisulfite reacts with acids, oxidizers, or even some cleaning agents. If someone hoses down a nearby spill with bleach, strong fumes or visible “off” reactions may crop up. Label the area with warnings, and train staff never to store any reactive substances close together. It’s all too easy to pile general-purpose cleaning supplies with warehouse stock, and that’s where bad chemistry starts.
Dust from cement, rust from old shelving, and dirt from pallet splinters also sneak in. Workers sometimes don’t realize how often a broom sheds fibers or how much impact comes from a torn bag. Closing containers tightly and sealing all openings goes further than buying expensive new racks.
Better Storage Can Save Money
Lost material hurts the bottom line and adds risk for workers. Clear labeling and dedicated shelves, along with simple safety checks, usually keep things in good shape. Regular inspection—opening a sack or two, feeling for dampness or lumps, sniffing for any odd sour odor—signals problems before they turn into waste. Good training, smart layout, and everyday attention produce safer, cheaper, and more reliable chemical use.
Are there any safety precautions when handling Zinc Bisulfite?
No Room for Shortcuts
Zinc bisulfite crops up in a lot of chemistry labs and industrial setups. No matter the scale, smart handling saves trouble. Strong smells and stinging fumes prove it’s not a substance to treat like table salt. Masks, gloves, and eye protection matter every single time. They’re not window dressing. Even brief, careless contact on skin or in eyes causes burns or lasting irritation.
Breathing Becomes the First Risk
People usually notice the sulfur smell before health effects kick in. Breathing zinc bisulfite dust or mist leads to coughing or even difficulty catching your breath. In my lab days, using a fume hood wasn’t just a box-ticking exercise. Those hoods quickly stripped the air of nasties I didn’t want to inhale. Lack of one, or skipping the exhaust system, multiplies health risks. NIOSH recommends using proper ventilation in any chemistry workspace. Facility managers ignore this and employees get sick or miss work; the numbers back that up.
Skin and Eye Damage
Splashes happen. I’ve seen people reach over an open bench flask in a hurry and pay the price. Zinc bisulfite burns hurt, sometimes leaving marks that last. Quick washing lessens pain, but permanent damage isn’t out of the question without immediate rinsing. The folks who test these chemicals stress goggles and face shields for a reason. No one wants to fumble to the eyewash station, barely able to see, because they forgot their gear.
Storage Needs Attention
Open sacks or poorly sealed containers spell trouble. Zinc bisulfite picks up moisture from air and breaks down, giving off sulfur dioxide, which burns the lungs. Dry, sealed, labeled containers save everyone grief. In workspaces with kids or pets around, storage containers end up double-sealed and out of sight. Responsible handling means treating even a leftover pinch with respect.
Waste Disposal and Environmental Harm
Pouring leftovers down the drain leads to headaches—both legal and environmental. Zinc compounds poison water and soil. City water techs track chemical spills, fines hit quick, and repairs aren’t cheap. In industry, waste-hauling companies have rules for collecting dangerous byproducts. At home, small amounts go to hazardous waste days run by the community.
Simple Tools for Protection
No need for fancy gadgets to manage risk. Good gloves—nitrile or neoprene, not the cheap plastic kind—hold up against splashes. Aprons mean fewer ruined shirts and stop wicking to skin. Goggle straps aren’t always comfortable, but they keep eyes in the clear.
Clear Procedures Keep Workers Safe
Routine training sessions keep mistakes rare. Posters and quick guides by the workbench help more than an unread manual stuffed in a drawer. Putting safety data within eyesight changes how people act. I remember a day where the team drilled emergency decontamination, which cut response time in half when an accident actually happened weeks later.
Better Alternatives Exist
Some labs switch to safer chemicals if zinc bisulfite isn’t the only option. For certain uses, replacing it with less reactive alternatives pays off, especially where students or new workers come in. Evaluations and risk assessments point the way forward here.
Safety Means Staying Consistent
Solid habits and simple equipment provide the best protection. Respect for the chemical and clear rules stop almost every mishap before it happens. In the long run, it comes down to discipline and a willingness to put health first—on the job, at home, everywhere this substance appears.
| Names | |
| Preferred IUPAC name | zinc(II) hydrogen sulfite |
| Other names |
Zinc hydrogen sulfite Zinc(II) bisulfite |
| Pronunciation | /ˈzɪŋk baɪˈsʌl.faɪt/ |
| Identifiers | |
| CAS Number | 24305-60-8 |
| Beilstein Reference | 1071704 |
| ChEBI | CHEBI:91258 |
| ChEMBL | CHEMBL3301072 |
| ChemSpider | 15232265 |
| DrugBank | DB15854 |
| ECHA InfoCard | ECHA InfoCard: 100.028.874 |
| EC Number | 231-949-6 |
| Gmelin Reference | 87877 |
| KEGG | C18719 |
| MeSH | D019294 |
| PubChem CID | 22681619 |
| RTECS number | ZH6300000 |
| UNII | 45QC94564K |
| UN number | UN2651 |
| CompTox Dashboard (EPA) | `DTXSID20895898` |
| Properties | |
| Chemical formula | Zn(HSO3)2 |
| Molar mass | 179.54 g/mol |
| Appearance | White crystalline powder |
| Odor | Slight sulfurous |
| Density | 1.86 g/cm³ |
| Solubility in water | Soluble in water |
| log P | -3.7 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 6.0 |
| Basicity (pKb) | 8.7 |
| Magnetic susceptibility (χ) | −2.8×10⁻⁶ cm³/mol |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 167.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -815.7 kJ/mol |
| Pharmacology | |
| ATC code | A12CB08 |
| Hazards | |
| Main hazards | May cause respiratory and skin irritation; harmful if swallowed; may release toxic sulfur oxides upon decomposition. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | Harmful if swallowed. Causes serious eye irritation. |
| Precautionary statements | Keep container tightly closed. Store in a cool, dry, well-ventilated place. Avoid breathing dust, fume, gas, mist, vapors or spray. Wash thoroughly after handling. Wear protective gloves, protective clothing, eye protection, and face protection. |
| NFPA 704 (fire diamond) | 2-0-1 |
| Explosive limits | Not explosive |
| Lethal dose or concentration | LD50 (oral, rat): 2000 mg/kg |
| LD50 (median dose) | LD50 (median dose) = 2125 mg/kg (rat, oral) |
| NIOSH | ZT6698000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Zinc Bisulfite: "Not established |
| REL (Recommended) | 200 mg/kg |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Zinc sulfite Zinc sulfate Zinc sulfide Sodium bisulfite Calcium bisulfite |

