Zinc Bis(Dimethyldithiocarbamate): A Closer Look at Utility, Safety, and Progress
Historical Development
Zinc bis(dimethyldithiocarbamate) made its entrance in the chemical world with the expansion of rubber manufacturing in the early 20th century. Pioneers in industrial chemistry saw a need for stronger, faster vulcanization. They found this compound played a reliable role in creating durable, elastic rubber goods. As more industries grew—automotive, footwear, wire coatings—this compound became a mainstay. Its popularity spread, as it helped tire factories boost production, and the chemical found its way into conveyor belts, hoses, and even adhesives. By the 1950s and 60s, as environmental awareness sharpened, regulatory eyes started tracking how manufacturers handled the compound. Safety sheets and standards were drafted, shaping the protocols seen today.
Product Overview
Most people know zinc bis(dimethyldithiocarbamate) by trade names or simple acronyms like Ziram. Its structure puts together zinc with two dimethyldithiocarbamate ligands—a mouthful on paper, but it means a compound that fights fungi and cures rubber. Talk to any person in tire or chemical supply, and they’ll mention Ziram for its dual use in crops and manufacturing. Crop protection products on store shelves often feature it as an active ingredient, fighting mildew and rot on a variety of fruits and vegetables. In rubber plants, it acts as a catalyst, speeding up curing without leaving behind sulfurous odors or residue.
Physical & Chemical Properties
Ziram comes in a fine, pale-yellow powder. It packs a mild sulfur scent, reminiscent of matches or struck flint. Not water-soluble, Ziram disperses best in oil-based mixtures or organic solvents. Its melting point sits high, resisting breakdown until well above the boiling point of water, which allows it to withstand the heat-intensive processes of rubbermaking. Chemically, it remains stable under normal warehouse conditions, but storage near acids or strong bases invites breakdown. Many labs test its stability under various pH and temperature ranges, tracking how soon it decomposes and what byproducts emerge—carbon disulfide and amines tend to show up first.
Technical Specifications & Labeling
Factories bag Ziram in 25-kilogram sacks, sometimes lining with polyethylene to block moisture. Each sack bears a hazard diamond, listing acute toxicity, environmental hazard, and instructions for safe handling. Labels mention the chemical’s purity level, usually exceeding 96% for industrial use and slightly less for agricultural formulations. Specification sheets detail limits for related dithiocarbamates, residual solvents, and heavy metals, though most modern producers keep contaminants low. Agriculture-grade Ziram sometimes carries additional wetting agents or inert fillers for easier field application, and rubber-grade Ziram ships with less dust to cut down on airborne risks.
Preparation Method
A standard process for making Ziram starts with dimethyldithiocarbamate salts, generally mixed from carbon disulfide, dimethylamine, and a base such as sodium hydroxide. Adding zinc sulfate or zinc chloride to this mixture prompts precipitation of Ziram. Factories use filtration, washing, and drying steps to reach desired purity. Each stage influences the finished product—impurities or incomplete drying sap performance and introduce workplace risks. Scaled production lines employ closed systems, trapping fumes and dust before they can leave the building. Technicians rely on in-line sensors to track pH and density, adjusting batch conditions to keep quality steady.
Chemical Reactions & Modifications
Ziram reacts briskly with strong acids, splitting apart to release carbon disulfide and amines with a sharp, toxic vapor. Mixed with oxidizers, it breaks down into sulfates or less volatile dithiocarbamate derivatives. Environmental scientists pay close attention to the degradation in soil and water. In agricultural runoff, sunlight and microbial action can turn Ziram into less toxic fragments, but high concentrations persist in sediment. Chemists sometimes modify Ziram for special uses, attaching different ligands or shuffling the metal center to create compounds with tailored resistance to heat or mold.
Synonyms & Product Names
Ziram goes by several names—Zinc dimethyldithiocarbamate, Zineb, and the simple shorthand ZDMC. Agricultural markets list it under fungicide group codes, and rubber supply houses often use industry catalog numbers. On import-export documents, it might appear under the UN number 3077 or as “environmentally hazardous substance, solid, n.o.s.” This variety reflects how widely Ziram’s been adopted, as well as the need for careful handling in global trade.
Safety & Operational Standards
Anyone who has worked in rubber curing or crop dusting learns Ziram brings certain hazards. Inhalation of dust can irritate the lungs, and skin or eye contact triggers mild to moderate reactions. Factories mandate gloves, goggles, and dust masks, reinforced by local exhaust hoods. Storage compliance demands dry, cool rooms with sealed drums, away from food or animal feed. Emergency response plans include quick cleanup for spills, with instructions on neutralizing residues with lime or soda ash. Regulatory bodies—OSHA, EPA, the EU’s REACH system—each post maximum permissible exposure limits, and routine air sampling in plants stays a priority. In agriculture, application guidelines restrict use close to waterways or in high-wind weather, reducing drift and environmental harm.
Application Area
Rubber manufacturers have relied on Ziram for years to normalize curing times and give products their bounce and stretch. In agriculture, orchard managers spray it across apples, grapes, and nuts to keep fungal blight away during damp seasons. Textile makers sometimes use similar compounds to preserve fabrics or aid in waterproofing. Ziram’s effectiveness has even caught the eye of certain paint and adhesive engineers, who use small amounts to guard against rot. There has been recent pushback from organic farming groups, who prefer non-synthetic alternatives, but commercial growers often point to Ziram’s reliability, especially during wet, blight-prone seasons.
Research & Development
Much research centers on environmental fate—scientists study how Ziram degrades in soil and water, try to predict its byproducts, and run bioassays with native species. Some labs experiment with using Ziram in new lower-dose or slow-release formulations, hoping to cut back on run-off while preserving effectiveness. In materials science and polymer design, researchers evaluate alternatives that provide similar curing speed without dithiocarbamate chemistry, but few have matched Ziram’s performance in stress-strain tests. This pushes active collaborations between academia and manufacturers, seeking a compound that combines safety, speed, and affordability.
Toxicity Research
Ziram’s health effects have become a focal point after several studies highlighted links to endocrine disruption, neurotoxicity, and possible carcinogenicity in high-exposure settings. Rodent studies reveal central nervous system effects at elevated doses, with longer-term exposures impacting fertility and liver enzymes. Human health monitoring focuses on long-term exposure for workers and residents close to heavy application areas, and regulatory bodies, such as the EPA, frequently revisit their risk assessments. Some nations have banned specific uses, especially where routine application could leave residues on food, but many places continue to endorse tightly controlled use. Medical recommendations include regular medical surveillance for chronic users and prompt decontamination in case of accidental exposure.
Future Prospects
Demand for Ziram will shift as industries adapt to stricter green chemistry mandates and consumer preferences. Advances in polymer science spark ongoing searches for safer accelerators, backed by regulatory pressure to lessen toxic loading in consumer goods. Agriculture faces similar scrutiny, with organic certifications tightening and retailers demanding fewer chemical residues. Manufacturers continue supporting stewardship programs, teaching proper storage and safe handling, but the push remains for a drop-in replacement with better toxicology. Growing use of precision agriculture and digital monitoring could one day minimize field applications, further reducing environmental footprints. Environmental degradation and recycling remain hot-button issues—industry leaders pilot closed-loop systems, cutting waste and boosting worker safety, a move that could reframe the place of Ziram and similar chemicals in future manufacturing.
What is Zinc Bis(Dimethyldithiocarbamate) used for?
A Closer Look at Zinc Bis(Dimethyldithiocarbamate)
Chemistry shapes much of what we experience in daily life, often in ways we don’t see. Take zinc bis(dimethyldithiocarbamate)—usually known in the rubber industry by its shorthand, ZDBC or Ziram. I came across it years ago during a tour of a local tire plant, and the technician showing me around spoke about it almost as fondly as the rubber itself. After that, I noticed how often chemicals like this play quiet, vital roles far from the spotlight.
The Rubber Industry’s Friend
Most folks walking down a sidewalk won’t give a second thought to how their shoe soles—or car tires—stay tough and flexible instead of crumbling apart. ZDBC steps in as an accelerator for the rubber vulcanization process. Vulcanization transforms sticky, weak latex into the stretchable, sturdy material we need for tires, hoses, belts, and hundreds of everyday products. This accelerator helps sulfur link up those rubber chains quickly and evenly. Skipping this means rubber turns out weaker, wears out fast, and can crack under stress. Tires built on weak rubber put drivers at real risk.
What caught my attention is this compound’s ability to speed up production. Less time on each batch means lower costs and fewer city-block-long piles of unfinished rubber parts hogging up warehouse space. Factories save energy too, and that matters these days with every electric bill gnawing at budgets.
Protecting Crops and More
But ZDBC’s story goes beyond rubber. In agriculture, it shows up as a fungicide. Farmers turn to it for shielding fruit and vegetable crops from diseases that could wipe out half a season’s work. My own family relies on a garden for part of our food, so I get the urge to find both effective and responsible crop protection. ZDBC brings results, blocking blights on apples, pears, and more, and gives crops a chance to thrive without the farmer drowning fields in more hazardous chemicals.
Handling with Respect
Everything we gain from ZDBC comes with responsibility. Overuse or poor disposal seeps into soil and water supplies. Studies show that workers handling large quantities face exposure risks, especially with dust or fumes, and some regulatory bodies keep a close eye on workplace limits for good reason. I have seen safety posters in plants outlining glove use, air monitoring, and training meant to keep technicians and the environment safe.
Moving Toward Safer, Smarter Uses
Plenty of manufacturers now look at greener chemistry options or try to cut down on chemical residues. Some rubber makers experiment with different accelerators that promise similar results with less environmental baggage. Farmers push for careful timing and mixing with other tools—crop rotations, resistant varieties, smart irrigation—rather than leaning too heavily on any single synthetic chemical.
Paying attention to how we use chemicals like ZDBC keeps us on the right side of progress. It’s easy to ignore what we don’t see behind the scenes. Once you get a glimpse of what helps cars roll, crops grow, and things last, that invisible chemistry feels a lot more real—and worth shaping wisely.
What are the safety precautions when handling Zinc Bis(Dimethyldithiocarbamate)?
Being Mindful With Chemicals at Work
Zinc Bis(Dimethyldithiocarbamate), a name that’s a mouthful, finds its way into factories as a rubber accelerator and fungicide. Lots of folks on shop floors or in research labs recognize this yellowish powder but don’t always think twice before grabbing a scoop or a bag. Years in chemical production have shown me that the hairs on the back of your neck stand up for a reason around compounds like these; a string of tough lessons handed down from coworkers with burnt skin and breathing problems stick with you for good.
Keep Your Skin Safe
Bare hands, shorts, or rolled sleeves—these choices end in red, itchy burns. The compound can irritate or even blister skin. Nitrile gloves form a solid barrier, and thicker lab coats or coveralls stop powder from finding your skin. My old boss always said, “If you wouldn’t rub it on your lunch, don’t let it near your arms.” Splash goggles, not just dollar-store safety glasses, shield eyes from dust clouds that rise every time someone opens a drum.
Do Not Breathe It In
Zinc Bis(Dimethyldithiocarbamate) dust floating in the air settles into the lungs quickly. Not long ago, a coworker coughed up blackish phlegm after an unplanned spill—he skipped his mask thinking the job was quick. Proper respiratory protection blocks the tiny crystals that set off coughing, tight chests, or, over time, even asthma. A P100 or equivalent mask fits snugly and means business. Don’t trust dust collectors or fume hoods unless you see them checked and cleaned; filters clog and exhaust fans sometimes quit.
Work Clean, Work Ordered
One slip with an open bag dumps powder across everything. Wipe down surfaces and tools after each batch, or you spread that mess with every pass of your hand. Scrubbing benches, scooping powder directly into closed containers, and taping up bags all help. Shoes track powder from storage rooms into break lounges unless you check your soles and watch your step.
Label Everything in Sight
Forgotten jars get confused or, worse, misused. People assume a yellow powder in a beaker is harmless. Mark every container—permanent markers, big letters—and record dates. Make sure everyone on shift knows the symbol for toxic and the chemical’s full name.
Know What to Do for Spills and Exposures
A smart workplace runs regular drills. If powder spills, close doors, shut off ventilation if it pushes air, and use a vacuum with a HEPA filter. Shoveling or sweeping only puts more dust into the air. Managers who keep emergency eyewash stations unlocked and fully loaded save sight and nerves. Wash exposed skin with soap and water—don’t use alcohol wipes, they worsen irritation.
Keep Your Mind on the Risks
Zinc Bis(Dimethyldithiocarbamate) can cause headaches and an odd taste in the mouth if inhaled. Chronic exposure causes worse trouble—liver and kidney problems in severe cases, as safety data shows. Ignoring small exposures piles up over years.
To stay safe, talk about hazards with your team. Ask where eye protection is and test the fit of your mask. Don’t cut corners in the last hour of a shift. Respect for the powder keeps you out of the doctor’s office and makes sure everyone clocks out together.
What is the chemical formula of Zinc Bis(Dimethyldithiocarbamate)?
The Value Behind a String of Letters and Numbers
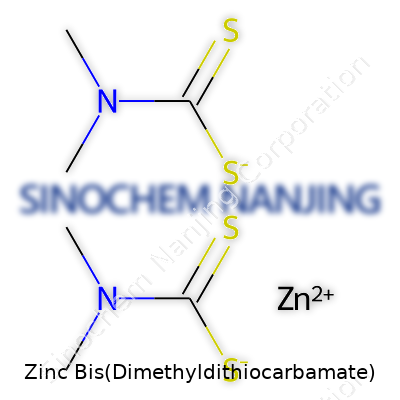
You often see complex names in chemistry that feel overwhelming, but every single one gives real clues about what’s inside. Zinc bis(dimethyldithiocarbamate) sounds complicated, but its formula—Zn(C5H10NS2)2—breaks down into something with true everyday significance. This compound, known commonly as Ziram, comes up often for folks working in materials science, agriculture, and rubber manufacturing.
Why the Formula Matters
Zinc bis(dimethyldithiocarbamate) combines zinc and a special kind of dithiocarbamate ligand. Each molecule holds a zinc atom at its core, then wraps two dimethyldithiocarbamate groups around it. These groups carry five carbon atoms, ten hydrogen atoms, a nitrogen atom, and two sulfur atoms—each, and the formula shows that there are two of these for one zinc. This isn’t just about textbook diagrams. Knowing the right ratio matters for those testing safety or looking for the most effective use in various applications.
Real-World Stakes
I learned just how precise chemistry demands you be during a conversation with a local rubber plant engineer. They measured everything, down to the last atom, seeking not just performance but safety. They relied on Ziram as an accelerator—helping rubber cure faster so tires or seals hold up better under stress. Mistakes in proportions led to waste, failed products, and sometimes environmental controls getting breached.
It’s not just about performance in rubber. Zinc bis(dimethyldithiocarbamate) turns up as a fungicide too, playing a role in how crops get protected from fungal blights. Here, too, the formula dictates how it interacts in soil or on plants, how it breaks down, and how much residue lingers. One farmer told me they checked each label twice, knowing that the tiniest misread could spoil a field or draw a visit from agricultural inspectors.
Getting Safety Right
People working around this compound keep an eye on safety sheets because, like many chemical agents, exposure calls for caution. Misunderstanding the formula or misreading quantities will affect how easily Ziram dissolves or spreads. Overexposure has health effects, so following the precise formula links directly to worker wellness and environmental compliance. My neighbor in occupational health grew tired of lecture tours, but he kept repeating: “Understand your molecules, and you avoid a world of trouble.”
Progress Toward Better Use
Modern labs use spectrometry and chromatography to verify compound structure, making sure Zn(C5H10NS2)2 has exactly what the label claims. This isn’t just academic. Regulators set clear limits because the wrong balance results in run-off problems or product inconsistency. Newer research focuses on breaking down Ziram safely after use, reducing reliance through smarter targeting, and developing safer alternatives for those most at risk of long-term exposure.
Working Smarter With Chemicals
Chemistry has a reputation for rule-following with good reason. With zinc bis(dimethyldithiocarbamate), I’ve learned even a basic formula packs in guidance on how to keep workers safe, crops healthy, and factories running smoothly. Every detail, from the zinc atom to the tangled chain of carbon, sulfur, and nitrogen, plays its part in risks, rewards, and the way forward.
How should Zinc Bis(Dimethyldithiocarbamate) be stored?
Why Storage Matters for Chemicals Like Zinc Bis(Dimethyldithiocarbamate)
Everyday work in a lab or manufacturing facility teaches a person lessons the textbooks sometimes skip. One lesson: careful storage of chemicals can make a big difference in safety, product quality, and even regulatory compliance. Zinc Bis(Dimethyldithiocarbamate) comes with its own storage quirks and knowing how to handle them keeps both people and projects out of trouble.
Key Risks and Challenges
Zinc Bis(Dimethyldithiocarbamate) reacts pretty fast to moisture and light. Left exposed, it’s likely to break down or clump up, which turns what should be a reliable compound into something unpredictable. Humidity sneaks into open bags or poorly sealed drums and next thing you know, the powder becomes lumpy and less dependable. Uncontrolled light and heat speed up degradation, reducing performance in rubber production or agriculture.
In workplaces I’ve seen, misplaced confidence in worn-out packaging or improper labeling creates accidents and regulatory headaches. Nobody wants a storage room full of unknowns or risk health issues because fumes leaked from a cracked container.
Good Storage Habits for Zinc Bis(Dimethyldithiocarbamate)
The best practice starts with a tightly sealed, high-quality container—think a heavy-duty drum with a gasketed lid rather than a flimsy bag. Metal or rigid plastics give better protection from moisture and accidental knocks. Labels need to stay readable, showing the compound’s identity and any hazard warnings. It seems basic, but rushed jobs or old labels lead to confusion quickly.
A well-ventilated, dry space gives this compound a longer shelf life. Humidity stays low in a dedicated storage room with clear rules: no eating, no open flames, and no unauthorized visitors. I’ve seen places establish humidity monitoring as part of daily rounds, which proves handy during rainy seasons or in poorly insulated warehouses.
Direct sunlight doesn’t belong in the chemical storeroom. Blocking out windows and using solid doors keeps UV rays off sensitive powders. Organized shelving means containers never touch the ground and nothing blocks aisles. Instead of putting everything on one or two packed shelves, spreading the load prevents spills.
Health and Environmental Considerations
Anyone who handles Zinc Bis(Dimethyldithiocarbamate) should suit up—gloves, protective eyewear, and a dust mask if there’s any risk of powder. Cleaning up after spills needs dedicated kits and a clear protocol. From experience, the best labs schedule regular refresher training to keep safety front of mind.
Disposal gets attention too. Simply tossing excess compound with regular trash goes against both common sense and regulation. Used material or contaminated packaging belongs in a hazardous waste stream, handled by certified disposal vendors. Regulatory agencies like OSHA and the EPA in the US set rules for a reason—protecting both workers and local environments.
Simple Solutions Go a Long Way
It seems obvious until it isn’t: proper storage supports safer workplaces and reliable results. Taking time for good practices—strong containers, dry and dark rooms, clear labeling—pays off by protecting people and avoiding regulatory trouble. Management often sees the cost of better storage as an investment rather than an expense, which matches what I’ve observed in facilities where accidents get rare and workflow stays smooth.
For anyone working with Zinc Bis(Dimethyldithiocarbamate), the best storage strategies come down to vigilance, training, and a commitment to getting the details right every single day.
Is Zinc Bis(Dimethyldithiocarbamate) hazardous to health or the environment?
Understanding the Chemical
Zinc Bis(Dimethyldithiocarbamate), known in industrial circles as Ziram, shows up in agriculture, rubber manufacturing, and even paints. Anyone who’s spent time around farms or old workshops knows chemicals like these form a big part of modern industry. They help prevent fungal growth in crops and lengthen the life of certain rubber goods. On paper, it looks like a workhorse. Pull back the curtain, though, and the story grows more complicated.
Health Concerns for Workers and Consumers
Direct experience on farms and in greenhouses makes it clear: handling Ziram cropland dust isn’t like digging in the vegetable patch. People exposed to the powder or solution often report headaches, skin irritation, and breathing troubles. Inhaling fine particles isn’t good for anyone, but exposure over a long time could bring more serious issues, including shaking hands and trouble with coordination. This happens because Ziram blocks an enzyme called aldehyde dehydrogenase in the body—a key bit in how our nerves function. Based on evidence in chemical safety documents, Ziram doesn’t belong anywhere near unprotected skin or lungs.
Farmers using fungicides sometimes struggle with insufficient protective equipment, especially during harvest season. Chemical companies issue thorough instructions, but tight budgets and fast-paced schedules too often push safety down the list. Absorbed through the skin, Ziram can sneak past gloves not meant for true chemical resistance. As someone who’s rinsed off after long field days, the risk is real—clothes get tossed, but skin exposure stays in mind.
Environmental Fallout
Pollution from Ziram lingers after it does its job. Waterways next to treated fields show traces of the compound. Fish and amphibians get the short end, with reports of slower growth and higher death rates. Non-target insects like bees also take a hit, especially if they visit fields too soon after spraying. Anyone who’s watched tadpoles in a pond knows how easily chemicals upset that balance.
Soil health also shifts over time. The compound sticks around in sediment, much longer than many people realize. Microbial communities may change, shifting how nutrients cycle through the ecosystem. Modern farming tries to manage this, but history shows that “out of sight, out of mind” thinking piles up problems down the road. Looking back at pesticide regulation over the decades, many products seemed safe until someone finally looked hard at long-term effects.
Possible Paths Forward
Avoiding harm calls for a more honest approach. Any chemical used on a large scale deserves regular review. Farmers share valuable insight—those who work the land often see subtle changes first. Integrating their feedback into regulations makes sense and builds public trust. Several countries already impose limits on Ziram use, restricting application and demanding higher levels of worker protection. These steps make a difference.
Growing food without synthetic fungicides still challenges many growers, but promising alternatives show up more often as research moves forward. Biological controls and crop rotation lower chemical loads. The rubber industry also experiments with substitutes for old chemical recipes, especially where workers face daily exposure.
Improved education and equipment stand as the most reliable ways to keep people safe while alternatives catch up. Clear labels, hands-on training, and better protective gear help keep incidents down. In my own family, working safer didn’t take fancy tech—it worked once we understood the risks and stuck to the rules every single day.
Transparent Conversation Matters
No chemical solution solves every problem risk-free. People with dirt under their nails and those steering public health both know that much. Listening to workers, recognizing unfiltered research, and updating policies keeps communities healthier. Real hands-on experiences shape how society decides what belongs in the field and what stays locked away on the shelf.
| Names | |
| Preferred IUPAC name | zinc bis(N,N-dimethyldithiocarbamate) |
| Other names |
Ziram Zinc dimethyldithiocarbamate Methamzine Metazir Carbamix Dimethyl dithiocarbamate zinc salt |
| Pronunciation | /ˌzɪŋk bɪsˌdɪˌmɛθɪldaɪˌθaɪoʊˈkɑːrbəmeɪt/ |
| Identifiers | |
| CAS Number | 137-30-4 |
| Beilstein Reference | 1718736 |
| ChEBI | CHEBI:134188 |
| ChEMBL | CHEMBL2364642 |
| ChemSpider | 21172345 |
| DrugBank | DB14016 |
| ECHA InfoCard | 100.014.273 |
| EC Number | 205-786-1 |
| Gmelin Reference | 81665 |
| KEGG | C18611 |
| MeSH | D002983 |
| PubChem CID | 24899720 |
| RTECS number | ZG6470000 |
| UNII | J9J4409Y5B |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C10H20N2S4Zn |
| Molar mass | 351.93 g/mol |
| Appearance | Light yellow powder |
| Odor | slight amine odor |
| Density | 1.41 g/cm³ |
| Solubility in water | insoluble |
| log P | 1.56 |
| Vapor pressure | <0.1 mm Hg (20 °C) |
| Acidity (pKa) | 13.2 |
| Basicity (pKb) | 2.9 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.760 |
| Viscosity | Viscous liquid |
| Dipole moment | 0.00 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 322.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -217.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -563.5 kcal/mol |
| Pharmacology | |
| ATC code | A01AD11 |
| Hazards | |
| Main hazards | May cause allergic skin reaction; harmful if swallowed; very toxic to aquatic life with long lasting effects |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H302, H317, H400, H410 |
| Precautionary statements | Precautionary statements: P261, P273, P280, P302+P352, P321, P333+P313, P362+P364, P501 |
| Lethal dose or concentration | LD50 (Rat, oral): 2400 mg/kg |
| LD50 (median dose) | Rat oral LD50: 2,950 mg/kg |
| NIOSH | DH9275000 |
| PEL (Permissible) | PEL: 1 mg/m3 |
| REL (Recommended) | 0.5 mg/m3 |
| IDLH (Immediate danger) | Unknown |
| Related compounds | |
| Related compounds |
Ferbam Thiram Maneb Zineb |

