Zinc Arsenite: More Than a Chemical Formula
Historical Development
Chemists in the 18th and 19th centuries first paid attention to compounds like Zinc Arsenite. Early research focused on finding the right methods for isolating and identifying metal arsenites, as curiosity about metallic salts ran high. Practical uses were rare due to toxicity issues, but the quest for pigments and agricultural chemicals drove further exploration. Some factories produced it on a limited scale, with scientists noting both its vivid colors and poisonous nature. Over time, tightening regulations and a deeper understanding of arsenic toxicity limited commercial enthusiasm. Most applications faded, leaving only a handful of specialized research and safety-focused uses behind. Today, knowledge built over centuries helps guide both handling and policy decisions.
Product Overview
Zinc Arsenite appears as a light-colored crystal or powder. In the lab, it draws attention for its unusual structure and unique chemical behavior. Industry sometimes looks at it with suspicion due to its arsenic content, yet a few research circles still give it the spotlight. Manufacturers rarely list it as a go-to compound, but those who work with metal arsenites often keep Zinc Arsenite under lock and key, more as a curiosity or historical reference point than as a standard shop item. Supply chains for it stay short, focusing on specialized labs and regulated environments. When someone brings up this substance, it’s likely in the context of university research, safety training, or regulatory discussion.
Physical & Chemical Properties
In the lab, Zinc Arsenite forms off-white to greyish crystals, sometimes powders, which won’t dissolve in water. The substance breaks down at high heat, releasing arsenic vapors and zinc oxide. Its solid-state structure shows zinc ions sitting among arsenite anions. Handling it often involves gloves and careful ventilation—this stuff gets a strong spot on the “handle with care” list. Its chemical resistance means it rarely reacts with weak acids or bases, but you can set off unpleasant reactions with strong acids or oxidizers. Researchers like to measure its density, melting point, and refractive index for reference, but real-world use rarely goes beyond those basic properties due to the risks involved.
Technical Specifications & Labeling
Chemists, when they order Zinc Arsenite, demand clear information. Labels list chemical formula (ZnAsO2), purity level, CAS number, potential contaminants, and batch number. Manufacturers print warning statements directly on the bottle: “Toxic,” “Handle only in fume hood,” and “Risk of cancer through inhalation.” Most suppliers also attach a safety data sheet, a must for lab compliance. Regulations often flag it for restricted use, especially in schools, hospitals, and places open to the public. Each container ends up in special storage—acid-resistant shelving, strong ventilation systems, and locked doors. Disposal instructions line up with hazardous waste protocols for arsenic-containing compounds.
Preparation Method
Synthesis in the lab often starts with zinc sulfate and sodium arsenite solutions. Mixing these under controlled pH gives you a white precipitate. After filtration and washing, the solid dries in a well-ventilated hood, away from any acidic fumes. Some older texts mention using zinc nitrate with disodium arsenite or suspending zinc oxide in arsenite-rich water. These techniques always run under strict supervision, with everyone geared up in goggles, gloves, and lab coats. Each reaction step gets documentation, as errors cost both time and safety. Disposal of rinse water and solid waste calls for arsenic treatment protocols—a lesson learned from past incidents where careless handling harmed both people and the environment.
Chemical Reactions & Modifications
Zinc Arsenite isn’t very reactive with most common chemicals. Pouring acids like hydrochloric or sulfuric over it causes arsenic trioxide gas to form and escape, raising serious safety alarms. Add strong oxidizers, and you risk forming even nastier arsenic species. Some research groups look at ways to bind Zinc Arsenite into polymers or glass matrices, reducing mobility but not eliminating risk. Others try doping its crystals for unique optical properties, though this stays a niche experiment. Every reaction involving this compound gets careful hazard review before anyone opens a bottle.
Synonyms & Product Names
Over the years, catalogs and textbooks have published various names for Zinc Arsenite. Some call it “Zinc ortho-arsenite,” “Zinc(II) arsenite,” or simply “zinc arsenite.” A handful of outdated product codes—often Local or European—still pop up in safety literature, listing trade names from companies long gone. More modern databases prefer its IUPAC name and chemical formula, helping reduce mistakes in cross-border shipments or research papers. Casual references use the name “white arsenite” in historical contexts, but in today’s world, the scientific community keeps to standardized naming to avoid confusion.
Safety & Operational Standards
Lab managers treat Zinc Arsenite as a high-hazard chemical, categorizing it alongside other arsenic products. Safety protocols draw from lessons learned the hard way—always use a certified fume hood, wear a double layer of gloves, and keep exposure times short. Teams working with this substance go through yearly training, with extra attention on proper waste disposal and spill response. Most facilities keep emergency arsenic antidotes on hand, along with eyewash stations and full-body showers. Air monitoring and personal protective equipment get more scrutiny than for most lab chemicals. Fire departments treat spills like HAZMAT calls, using special respirators and closed-system vacuums, not mops or brooms.
Application Area
People rarely use Zinc Arsenite in big industry applications. Its toxicity blocks use in farming, pigments, or routine industrial chemistry. Academic researchers sometimes use it to study metal-ligand bonding, arsenic behavior in matrices, or as a trace component in mineralogy. Historical texts point to its limited use as a pigment—those days are over, thanks to better knowledge of occupational health risks. Some niche fields, like forensic science and environmental testing, still reference it when tracking arsenic pollution or studying heavy metal interactions. These uses stay restricted to highly controlled situations, not open markets or consumer goods.
Research & Development
Research teams today focus on understanding the environmental mobility and toxicological behavior of compounds like Zinc Arsenite. Studies run simulations on how rain or soil acidity can dissolve arsenites from old industrial sites, tracking contamination pathways. Others experiment with embedding Zinc Arsenite in stable crystal lattices to immobilize arsenic, hoping for safer handling or long-term storage. Analytical chemists develop sensitive methods to detect traces in environmental samples, work that supports cleanup and regulation. Collaborations between material scientists and toxicologists often produce fresh insights, even if the direct market demand for new Zinc Arsenite applications stays low. Funding comes mostly from public health grants and government monitoring programs.
Toxicity Research
Working with Zinc Arsenite means confronting its hazardous nature head-on. Scientists have described severe health effects—chronic exposure leads to nerve damage, cancer risk, and organ failure. The dust proves especially dangerous when inhaled, and even brief skin contact can raise risks. Animal testing has repeatedly confirmed these findings, leading to high-level classification on global safety lists. Poisoning cases from earlier in the 20th century prompted stricter regulations, record-keeping, and emergency response protocols. Anybody working in toxicology learns to respect arsenite compounds, learning proper PPE protocol and spill response before stepping into a lab. This history of harm underlies both training requirements and ongoing research.
Future Prospects
Future use of Zinc Arsenite will likely focus on research, not commercial products. Steps toward advanced environmental remediation, pollution monitoring, and forensic science drive most current interest. Breakthroughs in polymer chemistry or materials science may find safe ways to lock arsenic away, but few expect to see expanded use outside controlled lab settings. Regulatory limits probably won’t loosen, especially as public health studies keep rolling in. Most experts see the future of this compound as an object lesson in chemical safety and historical policy shifts, not as a rising star in industry or technology.
What is Zinc Arsenite used for?
Understanding Zinc Arsenite’s Place
Zinc arsenite stands out as a chemical compound that rarely shows up outside specialized industries. Most people never come close to it, but those who work around heavy metals and pigments have reasons to pay attention. Its greenish-yellow powder has found its way into wood preservatives, pigments, and some specialty glass manufacturing. Older generations used zinc arsenite more freely, especially for wood preservation, before safer and less toxic alternatives appeared. Today, regulators and scientific consensus agree that this compound, made from zinc and arsenic, brings serious health and environmental risks if handled loosely.
Where You Might Find It
Industrial use drove most of the demand for zinc arsenite. Long before stricter standards, it served as a pigment for paints and ceramics, providing a distinct color that lasted. In the lumber industry, wood treated with zinc arsenite resisted insects and rot. Workers believed it to be a miracle solution at the time. Science caught up with those claims, showing that exposure brings hazards, including arsenic poisoning and cancer risks. Even trace levels in environmental runoff, soil, or dust can harm wildlife and contaminate water sources.
Glass producers also experimented with zinc arsenite as a clarifying agent, seeking clearer, more durable products. These industries face the tough reality that innovation often leaves a mark, sometimes dangerous, and demands greater responsibility over time. Today, laws across the world restrict and monitor its use tightly. The cost of ignoring these rules goes beyond fines—communities and ecosystems pay a heavier toll.
Health and Environmental Costs
Direct contact with zinc arsenite can bring short-term symptoms like headaches, breathing trouble, and skin irritation. Inhaling dust or swallowing material is far more dangerous. Chronic exposure adds up, with risks ranging from long-term organ damage to cancer. Children living near old rail yards or lumber sites can absorb arsenic from contaminated soil. Wildlife exposed to run-off show high death rates and birth defects.
During my time volunteering in community health, families living near industrial sites would ask about strange illnesses and unexplained symptoms. Most felt in the dark about possible connections to abandoned factories or treated lumber piles. Local soil tests often traced contamination back to heavy metals including arsenic compounds. These personal stories reveal the real-world impact that dry chemical labels never show.
Moving Toward Safer Alternatives
Modern industries minimize or avoid using zinc arsenite. Copper-based preservatives, borates, and less toxic synthetic chemicals now take the place of old arsenic mixtures for wood protection and pigments. Strict guidelines, better ventilation, and personal protective equipment reduce exposure for workers. Watching environmental agencies enforce cleanup of old industrial zones gives hope that these lessons stick.
Communities benefit when companies commit to transparency, post clear chemical records, and fund site remediation. Doctors and public health workers need strong support for ongoing medical surveillance near high-risk areas. Schools and families deserve clear education about local environmental history. When regulators, scientists, and industry leaders share responsibility, the damage from chemicals like zinc arsenite gets easier to recognize and fix. For those of us who’ve seen what happens without these safeguards, the lesson holds: prevention always works better than regret.
Is Zinc Arsenite hazardous to health?
A Close Look at Zinc Arsenite
Zinc arsenite doesn't pop up in conversation often, yet folks working in certain fields might run into it. It’s a chemical compound containing zinc and arsenic, both of which have drawn plenty of attention in labs and factories. My first exposure to the topic came during a college seminar on workplace safety, and I remember how quickly everyone’s mood changed once the professor said "arsenite." The word alone set off alarms. And for good reason.
What Makes Arsenic Dangerous?
Arsenic, a well-known poison, lands on the list of elements that’ve caused trouble for centuries. History books highlight its use for everything from wood preservation to pesticide manufacturing, sometimes with little regard for health outcomes. Scientists don’t even debate arsenic’s toxicity — chronic exposure to arsenic compounds can harm just about every major organ. According to the Agency for Toxic Substances and Disease Registry (ATSDR), breathing or swallowing arsenic compounds has been linked to cancers of the skin, bladder, and lungs. The World Health Organization points out that millions of people worldwide face arsenic in their environment or workplace, and the risks aren’t minor.
Zinc’s Role Complicates Things
Zinc on its own doesn’t have a reputation for causing harm in moderate amounts. It’s even an essential nutrient, and many take it in pill form to prevent colds. But mixing it with arsenite changes the picture dramatically. The compound formed — zinc arsenite — takes on the dangerous traits of arsenic. Exposure can happen in several ways: inhalation of dust, swallowing contaminated particles, or even through skin contact. Like most hazards, the body’s response comes down to dose and duration. Symptoms can sneak up slowly, from headaches and nausea to more severe neurological and respiratory problems. If exposure keeps up, long-term risks grow, including higher odds of developing cancer.
Industry Practices Raise the Risk
Workers who handle chemicals or metals might face zinc arsenite without fully realizing it. Historically, manufacturing plants cut corners on protective measures or left staff underinformed. It reminds me of talking to a friend whose father worked in metal smelting — he came home covered in dust and never wore a mask. Years later, health issues crept in. The Centers for Disease Control and Prevention warns that arsenic dust travels far and lingers long, especially inside just-ventilated buildings. One study from the National Institute for Occupational Safety and Health highlighted higher rates of lung issues and skin lesions among people exposed to arsenic compounds on the job.
How to Protect Against Zinc Arsenite
Nothing beats awareness and proper protection. Facilities owe their workers not just masks but also reliable ventilation and regular air quality checks. Some companies invest in closed systems that limit any contact with dust or fumes. Education plays a huge part as well — safety meetings and clear guidelines hammer home the risks, especially for newcomers. Outside the industrial world, public agencies keep an eye on soil and water contamination to catch any spillovers from factories or illegal dumping. I’ve seen community campaigns push local officials for clearer safety standards, and those efforts help drag companies toward better habits.
Final Thoughts
In short, zinc arsenite brings real health dangers, mainly due to its arsenic content. The risks aren’t a relic of past industry — they live on wherever the compound gets produced or handled carelessly. Prioritizing worker safety, upgrading equipment, and holding companies accountable could spare plenty of trouble. Lessons learned from earlier decades still apply, and every new case of exposure should serve as a signal that more can be done.
What are the physical and chemical properties of Zinc Arsenite?
Introduction to Zinc Arsenite
Zinc arsenite is not something most people encounter in everyday life. Its name alone hints at two traits: zinc, which finds its way into everything from sunscreen to batteries, and arsenic, the infamous element that brings more risk than benefit in most settings. This compound forms a pale green powder, and anyone who’s worked in a chemistry lab knows to treat it with respect. Unlike the familiar rust of iron, zinc arsenite never sneaks up on you—its toxicity demands caution from the start.
Physical Characteristics
This compound won’t catch your eye like copper or gold. Its appearance is largely unremarkable. The powder sits finely divided, often light yellowish-green. There’s no distinct smell wafting up to warn the unwary. It will not dissolve easily in water, making it a stubborn presence compared to salts like sodium chloride. Its lack of solubility often limits environmental spread, but don’t let that distract from the health hazards.
Zinc arsenite doesn’t burn. At high temperatures, it decomposes rather than turning to vapor. That can lead to some nasty byproducts. Heat zinc arsenite and you’ll find yourself dealing with arsenic oxides in the air, and no one should breathe those in.
Chemical Behavior
A compound like this speaks louder in its chemistry than in its looks. It combines zinc ions (Zn2+) and arsenite ions (AsO2-), setting up a bond that’s hard to break apart with water alone. Give it an acid bath, and it breaks down, releasing both zinc and arsenic ions. Anyone who’s ever accidentally opened the wrong bottle in a storeroom can appreciate the mess this makes—both for the lab and for anyone’s lungs who’s standing nearby.
In the wild, this compound knows how to grab onto surfaces and stick around, especially in soils rich in minerals. Its environmental stay means even small spills leave a mark long after the initial event fades from memory. Once released, its slow movement can easily fool people into thinking the risk has passed. In reality, its leftovers can build up in local ecosystems and end up in food chains.
Health Hazards and Real-World Impact
No one wants their water supply tainted by zinc arsenite. As someone who’s seen the fallout of well water laced with arsenic—a metallic taste, stomach pain, skin trouble—I don’t have to guess the danger this compound brings. Chronic exposure can lead to cancer, skin lesions, and nerve trouble. Acute exposure can knock a person down fast, leaving headaches, vomiting, and worse. Zinc by itself offers benefits in tiny doses, but alongside arsenite the risks dwarf those benefits.
There’s a reason modern industrial practices avoid producing, transporting, or storing this compound unless there’s no choice. Accidents in older mining sites and industrial dump grounds still leave scars in nearby towns. People living near these places talk about crops that won’t grow right and streams where fish have disappeared. Arsenic as a contaminant has been studied by the World Health Organization, and regulators worldwide have set strict limits for exposure. Zinc arsenite falls under this umbrella, bringing stricter scrutiny every year.
Toward Safer Handling and Remediation
Dealing with zinc arsenite means going beyond gloves and masks. Sealed processes, good ventilation, and rigid protocols form the backbone of modern safety strategies. Cleanup of contaminated land isn’t a simple job; it takes trained teams, secure disposal, and sometimes even removal of entire layers of topsoil. Plants known for "phytoremediation"—like some species of ferns—can help, slowly drawing out the arsenic from soil, but this work takes patience and funding.
Even as science advances, old stocks and abandoned sites demand vigilance. Lessons from past industrial recklessness make it clear: compounds as risky as zinc arsenite call for long memories and careful hands.
How should Zinc Arsenite be stored and handled safely?
Why Extra Care Matters With Zinc Arsenite
Zinc arsenite sounds like a chemical you only find in labs and textbooks, but it shows up in real-world operations, especially in manufacturing and research. The main problem boils down to its arsenic content, which has a notorious track record for being toxic. Thinking about public health and worker safety, it doesn't make sense to take shortcuts or guesswork with something this dangerous. Both OSHA and NIOSH underscore the need for strict control with arsenic compounds, and they aren't exaggerating.
Storage: Keep It Simple, Keep It Secure
The best place for zinc arsenite is a locked, labeled cabinet, away from common work areas or lunchrooms. Nobody should mistake this for a harmless powder. Cabinet material needs to be resistant to corrosion: polyethylene or coated metal give you good durability without the risk of slow chemical breakdown. If someone tries using a rusty cabinet, they’re not just asking for trouble—they’re risking lives. Don’t put it near acids, alkalis, or materials that promote chemical reactions. If you’ve spent time cleaning up after a minor lab accident, you know a tiny, avoidable mistake can spiral fast.
Humidity shouldn’t sneak into the storage space. Moisture lets arsenic get airborne or leak into the rest of the building. Silica gel packets or dedicated dehumidifiers work better than just relying on the standard building HVAC. Regular checks for leaks or powder around the seal save headaches longterm. Labels must be bold, waterproof, and clear—“Hazard: Contains Arsenic”—so nobody turns their back for a minute and forgets what’s inside. There’s a reason trained chemical handlers check these basics each shift.
Handling: No Room for Carelessness
PPE is the rule, not a suggestion. We’re talking nitrile or butyl gloves, snug-fitting goggles, and long-sleeve lab coats fastened properly every single time. Dust masks won’t cut it; if there’s a chance for airborne exposure, certified respirators step up as the only real choice. Washing hands before stepping out of the lab or storage room, even after wearing gloves, just makes sense. You’d be surprised how many veterans forget, distracted by a phone call or tight deadline.
Training shouldn’t fade into routine. Anyone authorized to touch, move, or measure zinc arsenite needs constant reminders about safe transfer and spill management. Dropping a beaker or opening a jar with wet hands wasn’t in the safety manual for a reason—it only leads to regret. Emergency kits with spill absorbent, disposable suits, and detailed steps for cleanup should be within arm’s reach. Practicing a mock clean-up once a quarter drives home what to do before a real accident happens.
Safer By Design: Preventing Long-Term Exposure
Ventilation stands as the unsung hero in shops and labs. Fume hoods take arsenic dust and fumes out of breathing zones. Asking staff to work without one just isn’t fair. Tracking who uses the cabinet and how much they take prevents confusion about where mistakes could happen. Digital access logs or simple sign-in sheets can close gaps in accountability.
In my years around chemical storerooms, the teams that made time for real training and didn’t fudge on standards never had ugly surprises. Those who rushed, skipped steps, or tried to save a few bucks on containers too often ended up dealing with poison control or regulatory headaches. Zinc arsenite isn’t just another bottle on the shelf; treat it with respect, and trouble stays at bay.
What is the chemical formula of Zinc Arsenite?
Getting Down to the Atoms: Zinc Arsenite Unpacked
In the world of inorganic chemistry, names like “zinc arsenite” can easily confuse. Over my years studying compounds, I’ve seen how students fumble over naming conventions, multiply oxidation states in their heads, and still wind up uncertain. The question keeps popping up in classrooms and labs: What is the chemical formula of zinc arsenite?
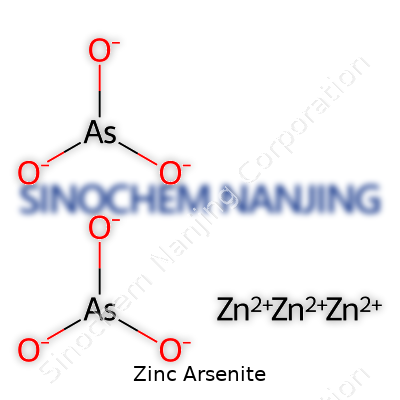
Zinc itself shows a +2 charge, while the arsenite ion is written as AsO33–. To achieve a neutral compound with these ions, two zinc ions match up with three arsenite ions, giving the formula Zn3(AsO3)2.
Why Chemical Formulas Do Not Stay on the Chalkboard
Some may shrug at the idea of memorizing Zn3(AsO3)2. Knowing the right formula saves time and keeps people safe. Take water analysis near mining sites: both zinc and arsenic turn up in waste streams. Environmental chemists need to distinguish between zinc arsenite and other, sometimes deadlier, arsenic compounds. One wrong assumption about a formula changes how a cleanup gets handled—and whether a community faces risk from contaminated wells. In my old neighborhood, a public health scare involved confusion between arsenite and arsenate ions; officials nearly made the mistake of treating for the wrong chemical.
A Lesson from Industry and the Lab
Zinc arsenite, in itself, never played a major role in manufacturing, but related arsenic compounds turn up everywhere—from insecticides to semiconductors. When researchers crack open compound names and formulas, they cut through marketing jargon, analyze environmental hazards, and choose safer substitutes. A basic formula like Zn3(AsO3)2 isn’t just an answer on a test; it’s a map for whether an ingredient belongs in a process or if it’s better left out.
Health Risks Unmasked by Chemistry
Safety data sheets tell no lies: exposure to arsenic compounds brings toxic effects, from skin ulcers to organ damage. In my teaching days, students often wondered why the periodic table even included elements like arsenic. But with the right information, such as formulas and structures, we track exposure and label hazards. Workers and environmental scientists make smarter calls on personal protection. Years ago, a small battery factory near my town learned the difference between arsenite and arsenate after employees started complaining of nausea—deep analysis of workplace dust, correlating precise formulas, allowed a rapid and targeted cleanup.
Steps Toward Knowledge and Safety
Building chemical literacy means empowering people, not just lab techs. Zinc arsenite’s formula, Zn3(AsO3)2, unfolds a story of charge balance, environmental duty, and critical thinking. More open training for first responders and school staff helps. Instead of memorizing facts, connect formulas to real life: water quality, workplace safety, and product transparency. Accurate chemistry education, plain language discussions, and public access to chemical databases help cut through confusion, keeping everyone a bit safer while we go about our daily routines.
| Names | |
| Preferred IUPAC name | Trizinc diarsenite |
| Other names |
Zinc orthoarsenite Zinc(II) arsenite |
| Pronunciation | /ˈzɪŋk ˈɑː.səˌnaɪt/ |
| Identifiers | |
| CAS Number | 12044-64-7 |
| Beilstein Reference | 0943757 |
| ChEBI | CHEBI:86315 |
| ChEMBL | CHEMBL3987853 |
| ChemSpider | 22246 |
| DrugBank | DB14604 |
| ECHA InfoCard | 100.034.254 |
| EC Number | 215-116-9 |
| Gmelin Reference | 86434 |
| KEGG | C16587 |
| MeSH | D015832 |
| PubChem CID | 24413 |
| RTECS number | ZK9810000 |
| UNII | 127M55C6CB |
| UN number | UN1436 |
| Properties | |
| Chemical formula | Zn(AsO2)2 |
| Molar mass | 221.29 g/mol |
| Appearance | White solid |
| Odor | Odorless |
| Density | 4.1 g/cm3 |
| Solubility in water | Insoluble |
| log P | -0.3 |
| Vapor pressure | Negligible |
| Basicity (pKb) | 8.99 |
| Magnetic susceptibility (χ) | Diamagnetic |
| Refractive index (nD) | 1.75 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 137.8 J/(mol·K) |
| Std enthalpy of formation (ΔfH⦵298) | -583.8 kJ/mol |
| Pharmacology | |
| ATC code | A06AX02 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; may cause cancer; environmental hazard. |
| GHS labelling | GHS05, GHS06, GHS08 |
| Pictograms | GHS06,GHS08 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P308+P313, P312, P330, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 3-2-2-POISON |
| Lethal dose or concentration | LD50 oral rat 30 mg/kg |
| LD50 (median dose) | 40 mg/kg (rat, oral) |
| NIOSH | ZE1350000 |
| PEL (Permissible) | PEL: 0.01 mg/m3 |
| REL (Recommended) | 0.1 mg/m3 |
| IDLH (Immediate danger) | 3 mg As/m3 |
| Related compounds | |
| Related compounds |
Sodium arsenite Zinc arsenate Zinc arsenide |

