Zinc Arsenate: A Complete Commentary
Historical Development
Zinc arsenate earned attention from chemists during the late 19th century, a period when researchers tackled the complexities of metal arsenates for pigments and pest control. Early trials aimed to replace copper-based compounds in agriculture and art. My own dive into archives revealed that factory use took off in Europe during the push to boost crop yields, not long after the dangers of unregulated arsenic became harder to ignore. The transition from curiosity-driven experiments to focused industrial synthesis tracks with humanity’s appetite for solving hunger and bacterial infections. Zinc arsenate found a niche in pigment production and as an insecticide, though regulatory scrutiny ramped up as arsenic’s toxicity became clear.
Product Overview
You don’t encounter zinc arsenate on store shelves. It’s a specialty chemical used where durable, corrosion-resistant performance trumps mainstream appeal. Laboratories tend to handle it in powder or crystalline form, often white or slightly tinted, packed in tightly sealed drums to stop moisture ingress. Industry uses it as pigment in ceramics, as a wood preservative, and in targeted anti-fungal coatings, especially before safer options took center stage. Looking at global markets, demand for zinc arsenate has shrunk, partly from environmental health regulations but also thanks to innovation in less hazardous alternatives.
Physical & Chemical Properties
Zinc arsenate stands out with its dense, granular or crystalline texture. Its chemical formula Zn3(AsO4)2 shows three zinc atoms locked to two arsenate units. The solid remains stable, insoluble in cold water, but can start breaking down in acids—a useful trait for controlled applications. In dry air, it keeps its structure for years, but exposure to moisture introduces hydrolysis concerns. During my time shadowing a pigment manufacturer, I saw how expert handling and pure raw materials cut down variability in crystal size, which impacts the pigment’s color qualities and resistance to leaching. Many forget just how resistant this compound can be—heat, light, and basic chemicals cause little change, explaining some of its staying power in niche markets.
Technical Specifications & Labeling
Manufacturers state composition as a percentage of active zinc and arsenic oxide, plus trace metals. Labeling surfaces the UN number for hazardous chemicals and displays pictograms for acute toxicity and environmental hazard. Globally harmonized regulations demand hazard statements for carcinogenicity, skin sensitivity, and aquatic risks. In my lab days, we handled every container under a double-glove policy, following clear procedures for spills. Product data sheets spell out solubility metrics and melting temperature—critical for researchers who want consistent results. Labels include CAS registration and batch codes to track every shipment from synthesis to final use.
Preparation Method
Producing zinc arsenate means reacting zinc sulfate with sodium arsenate in water, creating a precipitate that’s filtered, washed, and dried. Battery-grade batches start from high-purity zinc oxide and arsenic acid for controlled impurity levels. Scaling up production requires handling the arsenic precursor inside purpose-built enclosures with negative pressure, a measure that many older chemical plants struggled to achieve. While preparing wet slurries, filtration rates slow down if clumping occurs. Some manufacturers use surfactants or grinding to make a finer, more reactive powder. I saw how regional differences in water quality and filtration technology could nudge the final product toward or away from spec.
Chemical Reactions & Modifications
Zinc arsenate undergoes solid-state reactions under heat, particularly with phosphates or silicates, yielding new pigments or anti-corrosive hybrids. Certain industries modify the raw compound by blending with zinc oxide or calcium salts to reduce water solubility or tweak color profiles. In controlled acid environments, arsenate ions can be released, calling for careful management of effluent streams. While reviewing regulatory filings for wastewater plants, I learned how minor tweaks in waste pH pushed arsenic mobility up or down—an operational risk when compliance fines hurt the bottom line. Direct chemical modification remains rare, as most users apply it in the natural form, but nanotech labs sometimes experiment with zinc arsenate nanoparticles for advanced material science.
Synonyms & Product Names
Chemists know this compound by several names: trizinc diarsenate, zinc(II) arsenate, and zinc orthoarsenate. Old catalogues sometimes list it as C.I. Pigment White 4, and you’ll spot it under names like “arsenious zinc salt” in historical documents. The CAS number 10196-18-6 gives a universal reference, especially for technical patents and regulatory filings. I once tracked a supply shipment where three product names pointed to the same white powder—the vendor switched labels depending on customer sector. These naming quirks led to confusion during inventory checks, and they still crop up in literature reviews today.
Safety & Operational Standards
Operations involving zinc arsenate demand rigid safety protocols. Acute inhalation and ingestion risks mean proper engineering controls in every workplace. At the pigment facility where I trained, access required respirators and body coverings at all times, with mandatory monitoring for airborne arsenic. My colleagues used high-efficiency air filters, rigorous personal hygiene, and closed-door handling systems. OSHA’s permissible exposure limit for inorganic arsenic is extremely strict, sparking regular air sampling and health surveillance for workers. Material Safety Data Sheets (MSDS) warn against even brief unprotected exposure. Emergency protocols include lime wash showers for skin decontamination and specialized spill kits to manage solid or liquid waste.
Application Area
Use of zinc arsenate clusters around pigments, wood preservation, and select electronics manufacturing. Artisans once prized its colorfast qualities in ceramics; lumber industries valued its resistance against fungi and boring insects. Higher-end paints sometimes contained it to block mold outbreaks. In parts of Asia, old railway ties show the telltale signs of zinc arsenate treatments. Modern restrictions have forced a phase-out in most consumer-facing goods, but archival restoration and specialist ceramics still rely on legacy compounds. Talking with museum conservationists, I found deep concern over arsenic release from antique objects—safe handling matters both to users and to those preserving artifacts for future generations.
Research & Development
Despite shrinking market demand, zinc arsenate features in ongoing research. Material scientists probe its use in semiconductor doping, leveraging unique electrical properties to create new junction types or boost sensor performance. Environmental chemists study how the compound interacts with soils and water, especially in contaminated former industrial sites. Research teams at several universities develop encapsulation strategies to trap arsenic ions and limit environmental fallout, a topic with roots in legacy mining zones. Debate continues over the potential of low-toxicity analogs; some labs pursue hybrid materials that separate utility from toxicity. My own review of recent patents confirms that controlled modifications often yield better results than outright replacements—finessing what’s known often beats reinventing the wheel from scratch.
Toxicity Research
Toxicological studies show zinc arsenate harms multiple body systems with enough exposure. Chronic contact results in increased cancer risk, vascular disorders, and neurological symptoms. Animal studies cite dose-dependent effects, with regulatory agencies widely agreeing on carcinogenic status for arsenic compounds. Medical records from workers in pigment factories and timber yards during the 1900s connect prolonged exposure to heightened mortality, prompting sweeping reforms in workplace safety. More recent studies track how even low-level exposures impact children’s development in contaminated zones. Environmental persistence remains a red flag—arsenates bind to soil, but may leach out under acidic or reducing conditions. I attended a workshop where soil scientists demonstrated new field kits designed to track and neutralize mobile arsenic; field results still need long-term validation.
Future Prospects
Policies prohibiting arsenic-based compounds in consumer goods shape the future of zinc arsenate. Regulatory bodies push industries toward alternatives, yet specialist applications keep the compound relevant in fields emphasizing performance over volume. Research into arsenic capture and recycling technology offers some hope for mitigation. Environmentally responsive approaches favor in-situ immobilization, harnessing iron oxides or engineered bacteria to ‘lock up’ arsenic in place. I see a role for targeted use in applications where exposure remains tightly controlled: sensors, industrial catalysts, and rare pigments, provided disposal practices match modern safety expectations. The next decade will likely highlight safer analogues and recycled arsenic streams, but for now, zinc arsenate keeps its reputation as a high-risk, high-purpose chemical that keeps researchers, regulators, and industries on their toes.
What is Zinc Arsenate used for?
Where People Use Zinc Arsenate
Zinc arsenate pops up most often as a pesticide. Farmers and pest control crews once counted on it to fight off beetles, termites, and grapevine diseases. The promise sounded simple enough: sprinkle or dust it onto wooden structures, soil, or crops, and get rid of what destroys your investment. In my early days working on a vineyard outside Fresno, I saw some old farmhands still talking about these older arsenic-based pesticides, although tighter laws had pushed them to try newer methods. But the history lingers, especially in the stories families share about how arsenic dust wasn’t just tough on bugs; it stuck around long after any harvest.
The Risks Few Could Ignore
Scientists have known for a while that zinc arsenate brings real dangers, mostly traced to its arsenic content. Arsenic compounds, including the zinc kind, can collect in crops, soil, and water. Swallowing, touching, or breathing arsenic dust over months or years raises the odds of skin, lung, and bladder cancers. Kids and pregnant women face extra risk. Kidney damage and immune system harm can turn up, too. In parts of Central California hit hard by groundwater contamination, children got sick after playing near fields where old arsenic-based dusts lingered. Lessons from these places drove governments to rethink what’s truly safe for crops and communities.
Fact-Checking the Practical Use
Zinc arsenate hardly makes it onto store shelves today, at least for everyday buyers. Laws in the United States and the European Union started restricting and banning arsenic-based pesticides back in the late 1970s and 1980s. Still, you might come across industrial or institutional settings—think old railroads, lumber mills, or infrastructure repair—where stocks of arsenic compounds still turn up. Historic railroad ties and treated telegraph poles may leach arsenic into their surroundings for decades. It’s important to check local records and cleanup plans, especially if you work or live near these legacy structures.
Real-World Alternatives and Change
After the U.S. Environmental Protection Agency flagged arsenic pesticides, manufacturers started exploring copper and borate compounds, or plant-based controls, for managing pests and treating wood. Growing up, my own family’s garden got battered by insects, but newer methods—like beneficial nematodes, or neem oil—carried less risk to our health and rivers. Some farmers I meet still worry these substitutes lack the brute force of old chemicals, but soil testing and rotation often prove just as powerful for lowering insect pressure. People have to weigh not just the short-term fix, but the decades-long safety for their families.
Looking at Safer Practices
Care for the land and communities runs deeper than stopping a single pest or rot. People want food, clean water, and safe places for their kids to play. Banning or restricting harmful compounds like zinc arsenate matters because families all over the world live with the toxic leftovers from chemical shortcuts of past generations. Detoxing from that legacy means reading labels, asking questions if you see mysterious white dust in old barns, and pushing for honest cleanup of old industrial sites. It’s not just about protecting crops—it’s about not saddling your neighbors or grandkids with a toxic mess.
Is Zinc Arsenate toxic or hazardous?
The Quiet Dangers Behind Zinc Arsenate
Zinc arsenate isn’t a compound you encounter every day, but its reputation for toxicity rings loud in scientific circles. I’ve spent years researching chemicals used in everything from old pesticides to industrial coatings, and every path leads to one truth: anything with arsenic demands caution.
What Makes Zinc Arsenate Worrisome?
Zinc by itself isn’t the villain. Many multivitamins include zinc for immune support. The real trouble comes from arsenic, which has a long history of poisoning crops, water, wildlife, and humans. Zinc arsenate belongs to the family of inorganic arsenic compounds. Studies link these to cancer, nerve damage, and skin lesions from ongoing exposure.
Working near zinc arsenate isn’t just about avoiding a spill on your boots. Inhaling dust, letting it touch your skin, or swallowing even small traces can cause harm. Research from the World Health Organization counts arsenic among the most hazardous substances you can find in a workplace. Arsenic compounds like this persist for years in soil and water. They leach, they linger, and people pay the price.
History Offers a Warning
Years ago, farmers used zinc arsenate to kill insects in orchards. Sometimes, orchards still test positive for arsenic decades later. Old pesticide containers rust in forgotten corners. If left unchecked, the soil in these spots poses risks to kids, pets, and workers who manage the land. Cancer clusters around former arsenic-treated fields aren’t urban legends—they’re unfortunate facts in public health records.
Worker Stories
Talk to chemical plant workers who handled zinc arsenate in the 1980s. Many share stories of persistent coughs, skin rashes, and unexplained fatigue. Long-term exposure raises the risk of respiratory infections and nervous system disorders. It’s not just a statistic. Lab technicians with minimal protection developed chronic illness after handling small amounts, proving low-level exposure adds up fast.
Environmental Cost
Zinc arsenate’s danger extends beyond factory gates. Once released, it binds to sediments in rivers and seeps into groundwater. Drinking wells near contamination sites sometimes show levels of arsenic far above safety limits. Fish and small animals ingest the chemical too, and it moves up the food chain. Data from the US Geological Survey highlights the difficulty in cleaning up these messes. One patch of contaminated dirt can impact families for generations.
Paths Toward Safer Practice
The solution starts with respect for the threat. Label every container. Train every worker. Install decent ventilation and keep protective gloves and masks on hand. Sites using or storing zinc arsenate need regular soil and air checks. Testing for arsenic shouldn’t just be a line in the annual budget—it needs to be enforced with real action and oversight.
Safer alternatives now exist for almost every use once dominated by zinc arsenate. Farmers no longer need to rely on it for pest control. Older construction sites and industrial buildings benefit from thorough assessments before redevelopment. Community groups push for public maps of contaminated zones so people can protect themselves and their kids.
Why Vigilance Pays Off
Ignoring the risks means endangering health and futures—especially for workers and children exposed to invisible poisons. By learning from past mistakes and investing in modern protections, society can reduce the threat this hazardous material still poses. Zinc arsenate’s dangers are real, and taking them seriously isn’t optional—it’s the only path forward.
What are the storage and handling guidelines for Zinc Arsenate?
Why Attention to Zinc Arsenate Storage Pays Off
Zinc arsenate shows up on lists of hazardous materials for a reason. With years spent working in facilities handling industrial chemicals, I’ve picked up a keen understanding of what it means to safeguard both people and the environment. Zinc arsenate’s toxicity adds a unique layer of risk. Inhaling its dust or getting it on your skin can spell trouble fast, and improper storage leads to risks nobody wants to face. The substance contains arsenic, a heavy metal tied to cancer and nerve damage, so extra vigilance isn’t optional—it’s a duty.
Storage Guidelines Based on Long-Term Health and Safety
Secure, climate-controlled rooms keep zinc arsenate stable. Dry areas remain the gold standard here. Moisture triggers chemical reactions, and for this compound, that leads to slow breakdown and contamination of nearby surfaces, possibly releasing toxic arsenic compounds into the air. My early mistake of storing chemicals close to a utility sink taught me the hard way that accidental leaks don’t just wash away. Dedicated shelving made of corrosion-resistant material reduces incident risk.
Lockable storage keeps unauthorized hands away. Zinc arsenate carries high potential for deliberate misuse and accidental exposure alike. During facility audits, I’ve witnessed plenty of occasions where someone simply forgot to lock a cabinet, which turns a safe environment into a hazard zone. Keep detailed logs of access and regular inventory checks to limit loss and promote accountability. Clear label systems using standardized hazard warnings make a meaningful difference when a crisis hits. A lost few seconds locating or identifying a chemical can become life-altering.
Handling Practices Rooted in Real-Life Incidents
All it takes is a bit of dust and poor personal protection to earn an emergency room visit. Use heavy-duty gloves (nitrile holds up the best), chemical-resistant aprons, and face shields or safety goggles. Even the most experienced worker slips up. I’ve seen seasoned techs wipe sweat from their face absentmindedly, only to end up with a painful rash. Protective gear is a non-negotiable reality where zinc arsenate is concerned.
Open work should take place in ventilated hoods or fume cupboards. Dust control stays high on the list, since fine particles travel further than you’d think. I’ve watched employees try to take shortcuts and transfer powders on open workbenches. These shortcuts end with costly cleanup, regulatory headaches, and health scares. Wet methods, such as using damp cloths for spills (never sweeping dry), work well for minimizing airborne dust.
Waste Disposal and Regulatory Focus
Improper disposal doesn’t just break regulations—it poisons groundwater for years. EPA guidelines require collection of all residues and waste in sealed, clearly marked containers. Many facilities work hand-in-hand with specialist hazardous waste firms for collection and off-site treatment. One supervisor taught me that documentation for traceability can be a lifesaver if inspectors show up or a spill occurs.
Training stays at the core. No matter the efficiency of procedures, an untrained worker can tip the risks back into disaster zone in seconds. From onboarding through ongoing refreshers, hands-on safety demos and periodic drills engrain best practices and foster a sense of real ownership. Zinc arsenate remains a powerful industrial tool, but never without careful handling, thorough training, and rock-solid commitment to safety and environmental care.
What form does Zinc Arsenate come in?
The Dirt Under the Fingernails of Chemistry
Walk into a chemical storeroom and you won’t find jars of zinc arsenate in every color of the rainbow. Most times, what you’ll see is a chalky white powder that looks a lot like the stuff that finds its way into garden soil amendments or an old painter’s palette. Zinc arsenate owes its appearance to its basic chemistry—layered like many other inorganic crystals, it refuses to dissolve easily in water and won’t melt unless things get extremely hot.
I remember the first time I crossed paths with this compound. A box of “insecticide dust” used in old textbooks. Back in those days, zinc arsenate showed up as a fine powder, nothing glossy or fancy. There’s good reason chemistry teachers handle it with care—its toxicity has an earned reputation. The powdered form means it can spread in the air, stick on hands, and linger on surfaces, making safety more than just a checklist item.
Why Its Form Matters
Zinc arsenate stands out because of how people have used it over the decades. Crop protectants, wood preservatives—industries wanted a material that didn’t dissolve and get washed away by rain. This quality leads to its classic powder or sometimes granular state. Think of flour compared to small grit sand. The powder settles into nooks and crannies. The granules spill slowly from a scoop. Each type comes from different ways of manufacturing and grinding, shaped by what the next user needs.
Dispersing a powder gives wide coverage, but it floats around. It gets into lungs and onto work surfaces, which brings its own dangers. Granules avoid airborne spread to some degree, so workplaces go for them when they can. Still, no matter if it’s fine or coarse, it’s toxic through both skin and inhalation. The heavy, earthy effect of zinc arsenate comes not from a sleek manufacturing line but from guys in protective masks filling barrels in facilities with proper ventilation.
Facts, Dangers, and Ways Forward
This stuff isn’t trivia. The production and form determine environmental impact and workplace risks. Fact: most commercial zinc arsenate comes as tetrahydrate crystals. These push its use as solid powders or granules, rather than in a liquid or dissolved state. The EPA has taken a hard stance. They phased out use in residential settings and most agriculture after recognizing arsenic’s cancer risk and long-term environmental consequences.
The old approach—blanketing fields with arsenic-based powders—fills groundwater, stays in wood, and lingers in soil for decades. Alternatives like organic fungicides took zinc arsenate’s place. For legacy cleanup, recognizing where it sits in the landscape matters just as much as knowing what it looks like.
Solutions Require Honest ExaminingHazmat teams now test soil and water for traces. Professionals handle historical stocks with routines that focus on containment, using PPE every step along the way. Governments enforce bans instead of leaving it to personal responsibility. Innovations in agriculture and wood preservation push away from toxic metal salts.
Zinc arsenate’s powder and granule forms keep it both an engineering curiosity and a regulatory headache. From dusty research labs to environmental cleanup teams, knowing its texture and hazards pushes smarter, safer decisions. This compound teaches one lesson: physical form always ties back to health, safety, and history.
What are the safety precautions when using Zinc Arsenate?
Why Proper Handling Matters
Zinc arsenate carries a reputation that prompts a second look at the label. In college, I spent a little time in a dusty old storeroom, where glass jars with faded stickers sat behind three layers of locked doors. Seeing arsenate in the name, you know this isn’t table salt. The stuff brings real risks, even for someone just passing through.
Recognizing the Hazards
Contact with zinc arsenate can lead down a bad road. Skin absorbs hazardous materials faster than many expect. Flights of fancy about dust masks won’t do the job—arsenate poisons through breath and skin. Chronic exposure links to cancer, nerve problems, and issues with nearly every vital organ. In some workplaces, trace contamination could poison shared microwaves, writing utensils, or even chairs. Stories float in every chemistry department about those who ignored warnings; they rarely end well.
PPE Use—Not a Corner to Cut
I’ve seen people grab the wrong gloves or goggles just to get finished sooner, but that choice has consequences. Zinc arsenate requires specialized gloves, not the loose-fitting kind you find on a shelf in a janitor’s closet. Think nitrile, neoprene, or butyl—standard latex breaks down. Cover every bit of exposed skin, lab coat tucked, and real eye protection, not barely-there reading glasses. Respirators rated for toxic dust provide a barrier against invisible harm. Simple surgical masks just give a false sense of security.
Keeping the Workspace Safe
Working only in a certified fume hood keeps poison dust away from the air everyone breathes. A hood draws fumes and particles away from your face and stops toxins from circulating through a building. Double-check the airflow before opening the jar. Keeping every surface clean matters more than with most tasks—dust sits in corners waiting for a careless hand. Never use a broom or dry dust cloth, since these scatter arsenic into the air. Wet cleaning methods or HEPA-filter vacuums become essential tools.
Disposal Demands Respect
Dumping zinc arsenate down the drain endangers whole communities. I remember hearing about a worker who once ignored a “Hazardous Waste Only” sign, thinking the sewer would swallow anything. Later, town testing found arsenic downstream—tracing the trail didn’t take long. Every residual or contaminated object—pipettes, wipes, gloves, even disposable cuffs—goes in clearly labeled hazardous containers. Only licensed professionals should handle final disposal.
Preparedness Pays Off
Accidents don’t announce themselves. An eyewash station and safety shower within two strides can save a life. Everyone working near this chemical should know emergency contacts and procedures by heart. Chemical spill kits tailored for arsenic belong close at hand, and every label needs legibility. Working with zinc arsenate brings responsibility for everyone nearby, not just the one mixing chemicals at the bench.
Solutions and Responsibility
Training stands at the core of safety with high-risk chemicals. Regular reviews and hands-on drills keep knowledge fresh. Employers should keep detailed safety data sheets available, encourage a culture of honest reporting, and push for less hazardous alternatives whenever possible. With strict rules, reliable equipment, and a habit of taking the extra step, the dangers of zinc arsenate shrink. Ignoring the risks has real, lasting consequences—for the worker, the environment, and anyone nearby. Thinking ahead and staying careful makes all the difference when handling something that seriously bites back.
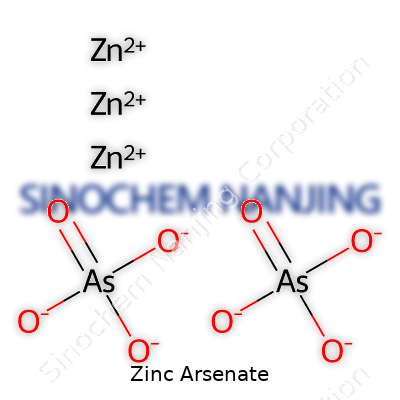
| Names | |
| Preferred IUPAC name | trizinc bis(arsenate) |
| Other names |
Trizinc arsenate Zinc(II) arsenate Zinc orthoarsenate Zinc arsenic oxide |
| Pronunciation | /ˈzɪŋk ɑːˈsəneɪt/ |
| Identifiers | |
| CAS Number | [10194-36-0] |
| Beilstein Reference | 1207045 |
| ChEBI | CHEBI:33313 |
| ChEMBL | CHEMBL38210 |
| ChemSpider | 21569698 |
| DrugBank | DB16006 |
| ECHA InfoCard | 03e02457-2d7e-4fec-9645-c347b0546101 |
| EC Number | 234-994-3 |
| Gmelin Reference | 774612 |
| KEGG | C15941 |
| MeSH | D015531 |
| PubChem CID | 24415 |
| RTECS number | ZK9810000 |
| UNII | 1C5V5087DC |
| UN number | UN1436 |
| Properties | |
| Chemical formula | Zn3(AsO4)2 |
| Molar mass | 397.06 g/mol |
| Appearance | White powder |
| Odor | Odorless |
| Density | 4.39 g/cm³ |
| Solubility in water | Insoluble |
| log P | -35.6 |
| Vapor pressure | Negligible |
| Acidity (pKa) | 12.1 |
| Basicity (pKb) | ~11.85 |
| Magnetic susceptibility (χ) | -24.0e-6 |
| Dipole moment | 0 |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 110.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -1867.5 kJ/mol |
| Pharmacology | |
| ATC code | V07AY30 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or absorbed through skin; may cause cancer; causes damage to organs; environmentally hazardous. |
| GHS labelling | GHS06, GHS08 |
| Pictograms | GHS07,GHS09 |
| Signal word | Warning |
| Hazard statements | H300 + H330: Fatal if swallowed or if inhaled. |
| Precautionary statements | P261, P264, P270, P271, P272, P273, P280, P301+P310, P302+P352, P304+P340, P305+P351+P338, P308+P313, P314, P330, P362+P364, P391, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-3-OX |
| Lethal dose or concentration | LD₅₀ (oral, rat): 20 mg/kg |
| LD50 (median dose) | 105 mg/kg (rat, oral) |
| NIOSH | AW8575000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) of Zinc Arsenate: "0.01 mg/m3 (as As) |
| REL (Recommended) | REL (Recommended): "0.1 mg/m³ TWA (as Zn) |
| IDLH (Immediate danger) | 40 mg As/m3 |
| Related compounds | |
| Related compounds |
Sodium arsenate Silver arsenate Calcium arsenate Lead arsenate Zinc phosphate |

