Ytterbium Nitrate: A Deep Dive Into A Rare Earth Compound
Historical Development
Discoveries around rare earth elements have always captured the imagination of chemists and engineers alike. Ytterbium nitrate traces its story back to the late nineteenth century, as researchers in Sweden sifted through mineral samples from Ytterby, giving birth to the element ytterbium. Early separation methods relied mostly on labor-intensive precipitation and fractional crystallization. Later, improved analytical techniques offered better methods for isolating ytterbium and preparing its nitrate salt. In those early days, sourcing pure ytterbium nitrate posed a real challenge due to limited access to rare-earth-rich ores and the complicated purification steps. The steady march of technology sharpened each process, trading cumbersome batch chemistry for reliable automation. By the twentieth century, refined extraction and crystallization routines made production more predictable, which supported broader research and industrial adaptation.
Product Overview
Ytterbium nitrate, appearing as a colorless to pale yellow crystalline solid, carries both a hefty molecular mass and a reputation for stability under normal storage conditions. It attracts attention from chemists because of the trivalent ytterbium ion that sits at its center, paired with nitrate groups ready to react under the right conditions. Industrial suppliers now offer it in various grades—commonly as the pentahydrate—catering to research, laser technology, and ceramics. Each batch comes tightly sealed, with purity levels reaching above 99.9% for highly sensitive applications. Rigorous specifics matter, especially in electronics or optics work, where contaminants can create real setbacks. Reliable packaging preserves moisture content and keeps air exposure at bay, which helps protect both the product and its end use.
Physical & Chemical Properties
Ytterbium nitrate’s pentahydrate form ranks as its most familiar face. It features a melting point around 85°C, which unlocks its solubility in water—a trait highly prized in chemical synthesis and solution-phase studies. The resulting aqueous solutions remain clear, but evaporation or gentle heating casts out the delicate crystals. Its chemical structure works well for both reduction and oxidation reactions, giving chemists options for downstream work. The combination of a mildly acidic pH and strong hygroscopic character means careful storage will maintain its integrity over time. Each crystal structure reveals an interesting hydrogen-bonding network between water and nitrate ions—a detail researchers can exploit for coordination chemistry or material science innovation.
Technical Specifications & Labeling
Reliable sourcing means reading the label, not just the headline. Most reputable vendors go beyond CAS numbers and offer precise breakdowns: assay (typically above 99.99%), moisture content (for hydrates), and a rundown of trace metal impurities—like iron, calcium, or aluminum—that can sneak into less refined batches. Technical datasheets also report on bulk density, particle size distribution, and recommended shelf life. Proper labeling extends to hazard statements and materials compatibility notes, giving end users clear instructions to prevent mishaps or cross-contamination. Whether used in a research environment or on the production line, these technical details make it easier for users to predict outcomes and control batch-to-batch variability.
Preparation Method
Lab-prepared ytterbium nitrate usually starts as a late step after hydrometallurgical extraction from ores like xenotime or monazite. Once researchers derive a crude mixture of rare earth chlorides or oxides, selective precipitation and re-dissolution help separate ytterbium from look-alike neighbors. Purified ytterbium oxide takes a bath in dilute nitric acid, producing ytterbium nitrate and excess water. The solution gets filtered and concentrated by gentle evaporation, avoiding high heat that could drive off water too fast or degrade the compound. Careful crystallization from cooled solutions grows the pentahydrate variant. Each cycle stands as a test of patience and attention, because speed or shortcuts often spell losses in yield or purity.
Chemical Reactions & Modifications
Ytterbium nitrate slides into a surprising variety of chemical schemes. As a strong Lewis acid, it plays a role in catalyzing aldol-type reactions or activating carbonyl compounds in organic synthesis. When treated with phosphates or fluorides, the nitrate yields up finely divided ytterbium phosphates or fluorides, tailored for optical ceramics or specialized glass. Complex formation comes easy for ytterbium(III) ions, unlocking pathways to coordination polymers or metal-organic frameworks. In reduction or decomposition reactions—such as those involving hydrogen or even thermal analysis at higher temperatures—the compound sheds its nitrate groups, sometimes with explosive force if handled carelessly. Modified forms, like ytterbium nitrate tetrahydrate or mixed-lanthanide salts, emerge by adjusting crystallization conditions or feedstock blends.
Synonyms & Product Names
Beyond the world of laboratory codes, ytterbium nitrate appears under several monikers. Chemists may see it written out as ytterbium(III) nitrate pentahydrate, Yb(NO3)3·5H2O, or even as nitric acid, ytterbium(3+) salt hydrate. Industry buyers track it under catalogue shorthand, often noting country-specific product registrations. These alternate names show up in customs documents or on international shipments, so clear identification short-circuits confusion and keeps compliance professional. Knowledgeable buyers stay sharp to avoid hitches caused by labeling inconsistencies, especially when juggling regulatory paperwork or cross-border transactions.
Safety & Operational Standards
Every handler of ytterbium nitrate should approach it with respect, and maybe a touch of caution. Though the toxicity risks rank lower than many transition metal salts, the compound can pose health hazards upon ingestion or inhalation. Irritation to eyes, skin, or respiratory tract ranks as a common complaint among careless handlers. Laboratory best practice relies on gloves, goggles, and local exhaust ventilation. Storage calls for cool, dry shelves and away from strong oxidizers or reductants. Emergency protocols cover accidental spills, encouraging dilution with water and absorption by acid-resistant materials. Each batch includes a Safety Data Sheet (SDS) spelling out relevant hazard codes, firefighting advice, and disposal recommendations—good habits that protect not just people but facility reputation as well.
Application Area
Not every material influences as many sectors as ytterbium nitrate. In lasers, its trivalent ion delivers up unique luminescence, supporting advanced solid-state lasers and infrared amplifiers. Materials scientists mix it into specialty glasses and ceramics, chasing after qualities that resist thermal stress or boost refractive index. Recent trends see chemists leveraging its Lewis acidity to catalyze organic reactions in fine-chemical synthesis efforts. Ytterbium nitrate’s reliable solubility paves the way for thin-film deposition and nanomaterial manufacturing, essential in sensors and future electronics. Rare-earth batteries and fuel cells pull further demand, hunting for electrolyte stability and specific ionic conductivities. Each new use case expands its footprint, leaving a legacy that spans from laboratory benchtop to industrial pilot plants.
Research & Development
Ongoing research into ytterbium nitrate favors both basic science and applied technology. Teams across Europe and Asia probe the fine structure of ytterbium complexes for clues in quantum computing hardware. Materials developers chase improved phosphors and upconversion nanoparticles for more efficient LED lighting and medical imaging tools. Chemists continue to experiment with reaction conditions and ligands, seeking out new synthetic pathways or catalytic cycles. Each published study adds another pixel to the global picture, showing just how far modern labs have come since the early days of hand-cranked distillation and backbreaking ore sorting. The push for even purer, more reactive ytterbium nitrate compounds remains relentless, driving new instrumentation and better analytical standards.
Toxicity Research
Most data suggests yttrium-group rare earths like ytterbium nitrate display only moderate toxicity, especially compared to heavy metals such as mercury or lead. Recent experiments using cell cultures and rodent models show minimal bioaccumulation, but raised eyebrows over chronic exposure risks. Researchers keep an eye on lung and kidney effects, particularly in workplace settings with fine powders and high concentrations. Environmental scientists continue to study its breakdown products in soil and water, aiming to catch long-term effects before they spiral into bigger problems. Anyone using this salt should keep updated with regulatory stances and risk assessments—a practical nod to evolving science and potential shifts in workplace standards down the road.
Future Prospects
Materials science fuels an ongoing hunger for rare earths, and ytterbium nitrate refuses to stand still. As quantum computing inched closer to reality, demand for ytterbium compounds seemed likely to climb, since these salts play well with next-generation qubits and photonics. Battery and energy-storage designers also eye it for doping and electrolyte tweaks, where even modest gains can shift entire product lines. Medical research turns to ytterbium-labeled complexes for targeted imaging, unlocking high-contrast and low-impact diagnostic tools. Production shifts toward cleaner, greener synthesis routes, often supporting circular economy strategies by recycling electronics and recovering rare earth content. Tomorrow’s progress will rely on tighter process controls, cross-border cooperation, and new academic-industry partnerships, putting ytterbium-based materials at the center of scientific momentum and innovation.
What is Ytterbium Nitrate used for?
A Rare Element with Real-World Uses
Anyone who’s dipped a toe into the world of rare earth elements has run across some interesting compounds. Ytterbium nitrate stands out among them, not because it’s flashy, but because of the way it often works quietly behind the scenes. Most people don’t often talk about this chemical outside a lab, but its usefulness stretches from medical imaging to energy research.
Lighting Up Medical Research
Back in college, I spent late nights helping run fluorescence tests on biological samples. Ytterbium nitrate showed up time and again in the background protocols. Researchers value it when preparing phosphors for imaging—those compounds that help spot tumors or trace infections in a living body. Ytterbium’s unique fluorescent properties let scientists track what’s happening inside tissues more clearly than traditional dyes. Luminescent markers based on ytterbium—combined with nitrates—have sharpened MRI contrast and even helped with early diagnoses in some cancer research breakthroughs.
Shifting Energy to Where it Matters
Ytterbium nitrate helps in making special materials that convert energy from one form to another. Mix it into glasses or crystals and you can produce lasers that work at specific wavelengths. Fiber lasers using ytterbium form the backbone of metalworking in car factories, powering precision welds that don’t scorch paint or warp panels. Energy transfer studies in advanced solar cells also use compounds like ytterbium nitrate while searching for ways to grab more sunlight for clean electricity. One study reported that ytterbium-doped nanocrystals improved solar cell efficiency by capturing otherwise lost photons.
From Research Tools to High-Tech Gadgets
Lab work involving ytterbium nitrate isn’t all test tubes and theoretical models. Scientists have developed safer, longer-lasting batteries by blending ytterbium nitrate into cathode materials. The rare earth element offers a promising route for battery makers looking to build better electric vehicles or more resilient grid backup systems. There’s also talk of ytterbium nitrate in quantum computing—where it could help make well-behaved qubits for next-gen processors.
Challenges and a Path Forward
Each time I see a story about rare earth shortages, I remember how much modern tech relies on these tiny amounts of metal salts. The catch is that mining and refining rare earths like ytterbium often means harsh chemicals and damage to local communities. Chinese mines have supplied most of the global stock, but negotiators and researchers keep chasing safer recycling and better alternative sources. The European Union and US have both invested in sourcing more rare earths from scrap electronics. A growing number of startups also work on cleaner extraction or even bio-mining projects. Every improvement in recycling or recovery helps ensure scientists and engineers can keep pushing for new tech without burning through the planet’s resources.
Looking at Tomorrow
If history has shown anything, it's that the small stuff can spark big changes. Ytterbium nitrate may not grab headlines, but new discoveries keep pushing its uses into more corners of industry and medicine. Whether you care about cleaner energy, advanced electronics, or lifesaving scans, the demand for smart, sustainable chemical solutions means compounds like ytterbium nitrate matter now more than ever.
What is the chemical formula of Ytterbium Nitrate?
What Makes Ytterbium Nitrate Interesting
Everyday chemistry doesn’t usually turn heads, but ytterbium nitrate can change a conversation, especially if you care about technology or sustainable energy. The chemical formula for ytterbium nitrate, Yb(NO3)3, looks simple on the surface. Behind those numbers and letters sits a substance with roles in laser technology, electronics, and even green energy. In real experience, clarity on a formula like this sometimes means the difference between a working experiment and a ruined batch of material. Confusion over formulas led me, once, to mislabel a sample and spend half a day troubleshooting in the lab. Understanding matters, and detail matters even more.
Breaking Down the Formula
Ytterbium joins the rare earth elements—a phrase tossed around a lot, but rare earths show up in headphones, smartphones, and fiber lasers. In Yb(NO3)3, one ytterbium atom links with three nitrate groups. Each nitrate contains one nitrogen and three oxygens. The three nitrates balance the charge of ytterbium, which goes into compounds as a trivalent ion, meaning it carries a 3+ charge. This structure isn’t just a trivia fact. It shapes how chemists store it, transport it, and react it with other elements. My time in the lab taught me to respect these combinations—any shift in ratios or storage conditions, and you lose time, money, and sometimes even safety.
Importance in Modern Technology
Jumping beyond classrooms, ytterbium nitrate pops up everywhere from research labs to high-efficiency lighting. Fiber lasers, which rely on ytterbium’s properties, power cutting and welding jobs for solar panels and medical tools. The technology inside your phone owes some thanks to rare earths like ytterbium, tucked invisibly inside delicate circuits. Cloud servers, which keep the internet alive, need rare earth-enabled magnets and lasers. A world without ytterbium would look much less connected and productive than the one we see today.
Supply and Sustainability Challenges
Rare earths sound rare, but mining and refining them presents a bigger headache. Digging up ytterbium throws off hazardous waste and eats up energy. At a past mining site visit in southeast Asia, I saw first-hand the environmental scars left behind. The chemical process for extracting ytterbium nitrate uses acids, generates byproducts, and sometimes leaves toxic leftovers sitting in open pits.
Recycling is picking up steam. Big companies and universities are trying to get yttrium, europium, and ytterbium back out of old electronics. Smarter design could mean less waste, less new mining, and more secure access for the tech industry. Honest reporting and transparent supply chains from mine to factory shelf deserve far more attention. It doesn’t take much to shift from convenience to carelessness in these supply chains. Everybody using modern electronics has a piece of responsibility, whether choosing a gadget, reading up on how it’s made, or pitching in to recycle old devices.
What Can Change the Game
Switching tech to use fewer or more abundant elements might ease some of the stress. Labs look for replacement materials, but those don’t always match the efficiency or low energy demands of ytterbium-based technology. Smarter regulations and consumer choices can nudge producers in the right direction. No matter how good a compound looks on paper, real progress happens when the chemistry, the economy, and the environment can all work in step.
Is Ytterbium Nitrate hazardous or toxic?
Why People Talk About Chemical Safety
Ask anyone who has worked in a laboratory or dealt with industrial chemicals: safety questions never go away, no matter how routine the material might seem. Ytterbium Nitrate doesn’t show up in every workplace, but it matters plenty in electronics manufacturing, catalysts, lasers, and research. If you’ve ever gotten a new smartphone or marvelled at advances in fiber optics, you’ve probably brushed against the edge of a supply chain involving rare earth compounds like this one.
Is Ytterbium Nitrate Harmful?
So what about the risks? Most people don’t have Ytterbium Nitrate in their kitchen cabinet, but it’s not outlandish for a high-schooler or chemistry student to run into it. The safety data sheets flag it as an irritant. It can cause problems if breathed in, gets on skin, or enters eyes. Swallowing the powder would send you to the doctor for good reason. European and US regulations ask folks to handle rare earth metal nitrates with gloves, safety glasses, and a fume hood. I’ve seen what happens when people ignore those rules: nose bleeds, skin burns, more than one ruined week.
Having spent years in higher education labs, I watched colleagues react to even minor spills of nitrates with urgency. Though most cases of exposure don’t lead to lasting harm, repeated contact ups the risk. The nitrate part brings extra fire hazard, too, since nitrates can fuel quick and smoky combustion when mixed with the wrong stuff. That’s real-world knowledge, handed down by teachers who saw fires as students.
Understanding the Science Behind Risks
The chemistry world doesn’t rate Ytterbium Nitrate as the most toxic of rare earth salts. It doesn’t pack the brute strength of a strong acid, nor does it readily cross the skin barrier in vast amounts. But research in toxicology journals points out that rare earth elements can build up if exposure happens day after day. Long-term, animal studies show that rare earth nitrates can irritate organs and mess with biological systems. Even though few reports document accidents compared to heavy metals like lead or mercury, the science still recommends caution.
Reducing Exposure and Staying Safe
Lab veterans always repeat one lesson: respect all powdered chemicals, even ones you think you’ve handled a hundred times. I wore gloves and goggles, and I doubled up if there was any chance of dust or splashing. Good habits — washing hands, labeling containers, storing things away from acids or combustibles — fend off most risks. I’ve heard stories of careless disposal leading to waste build-ups or improper ventilation causing headaches or dizziness. Those are easy mistakes to make, but discipline wins out in a well-managed lab.
Industry-wide, waste protocols help protect both workers and the environment. Neutralizing nitrates before disposal, using sealed containers, and providing ventilation do more than check off compliance boxes. They preserve the health of everyone who enters the building. If anyone in your community works with rare earth compounds, encouraging transparency and training cuts down on accidents and misunderstandings fast.
Learning from Experience
I measure chemical safety advice in scars and stories. Every compound brings its own lesson, and Ytterbium Nitrate is no exception. It’s not the most dangerous chemical I’ve seen, but taking it lightly causes problems. With the right habits and knowledge, it stays just another tool; without respect, it becomes one more story told at a safety seminar.
How should Ytterbium Nitrate be stored?
Why Proper Storage Matters
Storing chemicals isn't just a checklist item—it affects lab safety, research reliability, and even the environment around us. Ytterbium nitrate seems pretty harmless sitting on a shelf, but improper handling can cause fires, expensive losses, and health problems. News about chemical leaks or lab accidents grabs headlines for a reason. I’ve talked to fellow researchers who once ignored safety sheets, only for an air-reactive salt to ruin an entire batch of samples—and almost their careers. Respect the material, save yourself a lot of grief.
Environmental Conditions Make a Difference
Ytterbium nitrate pulls in moisture from the air. Leave an open bottle out, and it’ll cake up or start decomposing. Humidity creeps into everything, especially in warmer climates or poorly vented spaces. Set up a drying cabinet or a desiccator to control this; I’ve seen labs enlist large silica gel packs or vacuum desiccators, both effective for keeping the powder stable. Temperature swings accelerate breakdown, so controlled climate storage offers peace of mind. Aim for cool, dry shelving—no tossing it in a back room next to the dish rack.
Choose Containers Carefully
Glass bottles with tight PTFE-lined caps stop both moisture and unwanted reactions. Polyethylene containers work in a pinch, but some plastics allow slow leaks. I’ve seen students decant reagents into old soda bottles—don’t cut corners. Make sure that every container stays clearly labeled, with hazard symbols in plain sight. Mixing up labels or letting markers fade leads to confusion and mistakes.
Away from Heat and Incompatibles
Keep ytterbium nitrate far from anything flammable. Sparks, naked lights, and even sunlight shining through a window can spell trouble. Fires break out where dry chemicals and fuel source share a shelf. Once, a forgotten hot plate sent an entire cabinet into emergency lockdown, ruining samples and costing thousands to clean up. Segregate compounds according to established guidelines—don’t rely on memory or habit.
Access and Monitoring
Handle storage like you’d treat controlled substances: only trained personnel get to touch it. I remember hearing about a visiting intern who, through well-meaning curiosity, left a whole jar open overnight in shared storage. That mistake set the lab back weeks. Regular check-ins, inventory logs, and clear rules reduce the risk of lost or degraded material.
Disposal and Spills
Chemical spills don’t just stain the bench; they become expensive headaches for facilities, and health risks for anyone nearby. Any time a spill happens, use recommended PPE—gloves, goggles, lab coats—and follow cleanup procedures. Disposal requires attention too. Don’t pour anything down the drain or toss it with regular trash. Hazardous waste collection protects both people and the ecosystem.
Building Safe Habits
Safety culture grows from paying attention, not just ticking boxes. Training, periodic reviews, and transparent communication make the difference. Incidents drop when storage isn’t an afterthought. Plenty of labs only get serious after a near-miss. Don’t wait for an accident. Start with clear systems, honest discussion, and respectful handling of ytterbium nitrate—or any specialty chemical.
What are the physical properties of Ytterbium Nitrate?
Understanding Its Appearance and Nature
Ytterbium nitrate, known by chemists as Yb(NO3)3, brings a mix of predictability and curiosity to the table. It shows up as a white, crystalline powder that’s quick to attract moisture from the air. Anyone who’s handled it in the lab knows: leave it out, and it turns clumpy fast. Some might call it hygroscopic, but it really just acts thirsty, drawing in water until even sealed jars sometimes get damp around the rim. This property matters if people intend to weigh out exact doses or want sample consistency, as moisture can mess up both purity and measurements.
Density and Solubility in the Real World
Walk into a lab and drop Ytterbium nitrate into water—no drama, just easy mixing and dissolving. It has a solid density hovering around 2.65 g/cm3, sitting right between lighter nitrates and the densest heavy metal compounds. Since it’s so soluble in water, it shows up in liquid form pretty quickly. Most techs appreciate this because it spares time and cleanup, but it also means storage has to stay dry at all times.
Melting and Decomposition
Push Ytterbium nitrate to its limits, and it doesn't just melt. Once temperatures reach about 85 °C, it leaves its hydration water behind with a hiss. Raise the heat to something closer to 200 °C, and the nitrate decomposes, spewing out nitrogen oxides. In academic circles or industrial processes, this trait comes in handy, especially when folks want to recover ytterbium oxide or create tailored materials for electronics or catalysts.
Light and Ytterbium’s Colorlessness
Many rare earth compounds display vivid color, but not this one. Even in concentrated solution, ytterbium nitrate stays colorless—sometimes with just the slightest yellowish tint if impurities sneak in during synthesis. This neutrality plays a part in how scientists use it, as they can easily spot contamination or measure color changes when reacting it with other compounds, a real bonus in spectroscopic analysis.
Sensible Handling and Safety Concerns
Ytterbium nitrate isn’t out to cause trouble, but it can be hazardous. At room temperature, it doesn’t throw off fumes or burn skin, but being a nitrate, it holds oxidation power and can irritate if it touches skin or eyes. Those of us working with it don gloves, lab jackets, and goggles, not because it’s the worst out there, but common sense says it pays to respect any substance that reacts strongly with heat or gets sticky when just a little humid. Facilities handling it store it in airtight bottles, and material safety sheets always sit nearby.
Where Does It Lead?
With its ease of dilution and relatively safe profile, it finds plenty of use in specialty glass, lasers, and advanced electronics. Still, its thirst for water means anyone working with it has to pay attention to storage and keep work spaces well-ventilated during high-temperature work. Compared to some heavy metal nitrates, it brings less toxicity, but treating any rare earth compound with care reflects best practices. Limiting exposure, labeling every container, and balancing respect for chemical properties with efficiency turns routine lab work into a safer, more reliable process.
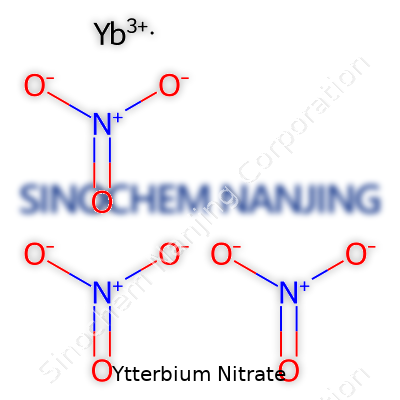
| Names | |
| Preferred IUPAC name | Ytterbium(3+) nitrate |
| Other names |
Ytterbium trinitrate Ytterbium(III) nitrate |
| Pronunciation | /ɪˈtɜːr.bi.əm ˈnaɪ.treɪt/ |
| Identifiers | |
| CAS Number | 35725-34-9 |
| 3D model (JSmol) | `Yb(NO3)3` |
| Beilstein Reference | 358916 |
| ChEBI | CHEBI:86561 |
| ChEMBL | CHEMBL3307236 |
| ChemSpider | 21241907 |
| DrugBank | DB11338 |
| ECHA InfoCard | ECHA InfoCard: 100.033.548 |
| EC Number | 231-799-1 |
| Gmelin Reference | 106093 |
| KEGG | C18667 |
| MeSH | D015857 |
| PubChem CID | 159053 |
| RTECS number | ZH8560000 |
| UNII | SYR8NV3W2U |
| UN number | UN2815 |
| CompTox Dashboard (EPA) | DTXSID5044282 |
| Properties | |
| Chemical formula | Yb(NO3)3 |
| Molar mass | 394.06 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 2.391 g/cm³ |
| Solubility in water | Soluble |
| log P | -4.01 |
| Basicity (pKb) | 8.02 |
| Magnetic susceptibility (χ) | −24.0·10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.6 |
| Dipole moment | 0 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 221.5 J K⁻¹ mol⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | −1625 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | There is no standard enthalpy of combustion (ΔcH⦵298) for Ytterbium Nitrate. |
| Hazards | |
| Main hazards | Harmful if swallowed, causes skin and serious eye irritation, may cause respiratory irritation. |
| GHS labelling | GHS07, GHS09 |
| Pictograms | GHS05,GHS07 |
| Signal word | Warning |
| Hazard statements | H272, H315, H319, H335 |
| Precautionary statements | P264, P280, P305+P351+P338, P337+P313 |
| NFPA 704 (fire diamond) | 1-0-0-OX |
| Lethal dose or concentration | LD50 oral rat 3,245 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 4,550 mg/kg |
| NIOSH | WA3400000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Ytterbium Nitrate: "PEL not established |
| REL (Recommended) | 2 mg/m3 |
| Related compounds | |
| Related compounds |
Ytterbium(III) chloride Ytterbium(III) fluoride Ytterbium(III) oxide Lanthanum nitrate |

