Xanthine: More Than Just a Biochemical Curiosity
Tracing the Footsteps: Historical Development
Xanthine did not burst onto the scientific scene out of nowhere. Chemists first isolated it from human urine in the early 1800s, scratching their heads over its role in the body. Looking back, these early discoveries might seem simple, but they set the foundation for understanding a whole class of purines. Over the years, scientists realized xanthine runs through the cellular veins of both plants and animals, turning up not just in urine but in tea leaves, coffee, and all sorts of biological systems. This recognition didn’t just add another line to the annals of organic chemistry. It helped frame the connections between metabolism, disease, and daily staples like caffeine.
Getting to Know Xanthine: Product Overview
Sitting at the crossroads of biochemistry and industry, xanthine is a purine base, found in every cell that carries DNA or RNA. Its relatives—theobromine, caffeine, uric acid—make bigger headlines, but xanthine earns respect in its own right as a metabolic springboard. Xanthine’s value isn’t just in its chemistry or that it pops up in lab results; pharmaceutical firms use derivatives as muscle relaxants and bronchodilators, showing how molecules found in dusty lab glass can end up in the rescue inhalers of asthma patients.
Physical & Chemical Properties
Looking at a sample, xanthine appears as a whitish, crystalline powder. No bells and whistles here. You won’t pick up a strong odor, but add some heat and xanthine decomposes, releasing nitrogen-rich fumes that remind you this is no inert dust. In water, xanthine barely dissolves, though it gets more cooperative in hot temperatures. Chemically, it boasts fused rings: a purine skeleton that lets it serve as a building block for more familiar molecules—think caffeine and uric acid. This structure opens doors for reactions with acids, bases, and enzymes. It might not carry the glitz of gold or platinum, but in the right hands, it proves its worth in many synthesis pathways.
Technical Specifications & Labeling Practices
Labs and factories dealing with xanthine focus on purity—usually 98% or higher when destined for research or pharma work. Standard operating practices demand clear labeling, complete with hazard icons and molecular identities. Unless handled carefully, dust can fly, so robust packaging becomes a health and safety measure, not just a marketing prop. Suppliers lean on international norms from groups like IUPAC and regulatory authorities, keeping the paperwork as straightforward as possible. Too often, minor chemicals get lost among ambiguous labels, but oversight bodies remind everyone that clarity isn’t just a bureaucratic tick-box; it keeps people safe and science honest.
Preparation Method: Getting Xanthine from Source to Shelf
Long before high-tech synthesis, folks relied on extraction from natural products—mainly guano and urine. As times moved on, chemists developed more controlled routes, such as oxidizing guanine or hypoxanthine using agents like potassium permanganate. Industrial setups switched to chemical synthesis for reasons of purity, scale, and efficiency. Lab-scale preparations still follow well-documented protocols involving temperature control, pH adjustments, and careful crystallization. Every step has to ward off contamination by related purines, because pharmaceuticals and research assays can’t afford guesswork. Mistakes at this stage often show up as noise in test results, wasted stock, or worse, misleading toxicity data.
Chemical Reactions & Modifications: Tweaking the Backbone
Xanthine’s chemistry proves versatile. It reacts with acids to form crystalline salts and gets methylated to produce compounds like caffeine, theobromine, and theophylline. These reactions stand out not just as academic exercises but as foundational steps for the synthesis of therapeutic drugs. In labs, enzymatic transformations mimic metabolic processes, making xanthine both a tool for teaching and a stepping stone for new medicines. Shifts in functional groups or ring structure expand its utility into new chemical territory, sometimes creating molecules that change lives, not just equations.
What’s in a Name? Synonyms & Product Names
Ask around, and you’ll hear xanthine go by plenty of other names: 2,6-dioxypurine, xanthic acid (a misnomer, but it crops up), or just its simple chemical abbreviation. Check the literature, and you’ll spot xanthine-based drugs—names tweaked by marketers and medicinal chemists chasing precise effects. The crowded field of purines means trade names can muddy the water, so checking chemical structure, CAS number, and context always matters more than the label on the bottle.
Safety & Operational Standards: Keeping Vigilant
Personnel handling xanthine keep a close eye on ventilation, gloves, dust masks, and material safety sheets. Regulatory requirements depend on jurisdiction, but responsible organizations rely on the precautionary principle—if inhaled or ingested in large quantities, xanthine can irritate the airways and stomach. Waste disposal follows hazardous chemical guidelines, and lab techs get trained to spot abnormalities. Forgetting these protocols rarely ends well; the risk of cumulative exposure or contaminated workspaces is real, not just paper theory.
Stepping Into the World: Application Area
Xanthine’s immediate fame comes in clinics and research labs. Physicians turn to xanthine derivatives for respiratory therapies, easing bronchospasm and breathing difficulties in chronic illness. Scientists use it as a stable reference compound in assays or to mimic human metabolic processes in vitro. Beyond medicine, agriculture and food research teams track xanthine to check for adulteration in coffee and tea or to better understand livestock metabolism. Its uses aren’t flashy but are deeply woven into technologies that feed, heal, and inform modern life.
Pushing the Envelope: Research & Development
Current research scans the horizon for new xanthine analogs that might lead to gentler asthma therapies or potential treatments for neurodegenerative diseases. Structural tweaks to the xanthine molecule aim to dial down side effects or increase bioavailability. Much of the innovation comes from cross-disciplinary work—a biochemist teams up with a pharmacologist and a computational modeler, all seeing new opportunities in a molecule that past generations might have overlooked. At the same time, researchers use xanthine as a metabolic probe, tracking how organisms break it down or transform it under stress, connecting lab findings to big-picture questions about health, pollution, and adaptation.
Toxicity Research: Lessons from Old and New
Toxicologists mapped out the dangers of xanthine early on, noting that high doses cause tremors, vomiting, and, rarely, seizures in lab animals. Chronic exposure generally raises concerns thanks to xanthine’s role as a metabolic intermediate—overload the system, and uric acid levels spike, hiking the risk of gout and kidney stones. Newer studies explore how xanthine metabolism intersects with genetic differences, drug interactions, and environmental pollutants, reflecting real-world complexity rather than textbook simplicity. This line of work matters for hospitals, regulatory agencies, and anyone charting a safe course for novel drugs.
Looking Forward: Future Prospects
Xanthine doesn’t dazzle, but its future runs parallel to advances in personalized medicine and metabolic research. Machine learning meets traditional bench science to predict how tweaks to the xanthine skeleton might unlock fresh treatments or diagnostic tools. Environmental sciences pay more attention too, testing the xanthine cycle as an indicator of soil and water health. Pharma startups and academic labs keep mining the purine family for new therapeutic angles, and xanthine stays in the conversation, part of a toolkit anyone interested in life, health, or innovation needs to understand. Its story reminds us that even molecules on the margins shape health, industry, and research in ways that demand both curiosity and caution.
What is Xanthine and what is it used for?
Digging Into Xanthine
Walk into a conversation about human biology or pharmaceuticals, and xanthine often doesn’t get much attention. Most people haven’t even heard the name, but it plays a real role in the body and in some corners of medicine. Xanthine is what scientists call a purine base, a building block that slips in the middle of important biological processes, especially those tied to how our bodies make and break down DNA and RNA. It pops up when cells break down purines, which come from both what we eat and what our bodies naturally produce. If you have ever sipped coffee, you’ve brushed up against xanthine’s family tree without knowing.
Where Xanthine Fits in Everyday Life
Xanthine doesn’t headline the story when folks chat about stimulants, but its relatives sure do. Caffeine, the stuff in coffee and many sodas, belongs to the xanthine group, as do theobromine in chocolate and theophylline, which doctors use for certain breathing problems. Every time someone grabs an energy drink or a dark chocolate bar, they’re crossing paths with chemicals that have a similar backbone to xanthine.
Doctors sometimes talk about xanthine when testing for certain health issues. Xanthine itself doesn’t do much in healthy bodies because it gets broken down quickly by an enzyme called xanthine oxidase. Problems come up if that enzyme doesn’t work right. Too much xanthine in the blood and urine can signal rare genetic disorders, particularly xanthinuria, which can show up as kidney stones or other kidney problems.
Uses in Medicine
Pharmaceutical companies don’t use pure xanthine much, but its relatives have shaped thousands of lives. Theophylline stands out as an asthma medicine. It works by opening up airways and relaxing muscles in the lungs, helping people caught in the grip of an asthma attack breathe easier. This drug, along with caffeine and theobromine, has been part of the standard toolkit for diseases involving the lungs or even babies with apnea, which is a dangerous pause in breathing.
Xanthine’s chemistry also offers a pathway for researchers to experiment with ways to block or change enzyme activities linked to gout or certain kidney diseases. Drugs like allopurinol block xanthine oxidase. By blocking this enzyme, doctors prevent the buildup of uric acid crystals in joints, which cause gout. This isn’t just an academic point—millions depend on this every year to manage pain and avoid serious complications.
Potential Solutions to Health Problems Connected to Xanthine
A big challenge sits with rare enzyme deficiencies linked to problems handling xanthine and its relatives. Better screening, especially for genetic disorders in families with histories of kidney stones, can help catch problems before they turn severe. Labs could look for smarter, faster ways to identify xanthine in blood and urine, helping to catch trouble early.
Outside rare inherited problems, the best defense remains a healthy mix of food and drink, since balance in the diet keeps the body from being overloaded. For patients already on medicines affecting xanthine pathways, regular blood checks and close contact with a healthcare team help avoid side effects.
Xanthine may not have the name recognition of caffeine or the everyday presence of chocolate, but what happens to it inside our bodies touches a surprising corner of health. A little knowledge helps bring hidden processes out into the open, making it easier to spot trouble and improve lives.
Are there any side effects of taking Xanthine?
Understanding Xanthine and Its Uses
Xanthine compounds show up in a lot of places—like coffee, tea, and even some medicines prescribed for asthma. Caffeine and theobromine, which people get from chocolate and cola, sit in this family of chemicals. Doctors sometimes turn to certain forms of xanthine, like theophylline, to help relax airways for people with breathing trouble. Most people have a little xanthine in their bodies just from food, and for many, it doesn’t cause any big problems. It’s only when people take concentrated doses, especially through medication, that some real side effects can kick in.
Short-Term Side Effects and Personal Experience
Think about the rush you feel after guzzling too much coffee. Restlessness, a thumping heart, fingers tapping across the table—these mark some of the mild side effects that come from too much xanthine. In my college days, staying up late with strong espresso would keep my mind spinning, but headaches and jumpy nerves always reminded me I’d gone too far. When someone uses these compounds in a medical setting, things can get more serious. High doses, especially from medicines like theophylline, can trigger nausea, vomiting, cramps, and even irregular heartbeats.
Why These Effects Matter
The body processes xanthine through the liver. Differences in genetics or other medicines can slow this process and allow more of the active compound to build up than expected. That buildup can tip someone from a coffee buzz into real health trouble—especially in older adults. I’ve seen relatives on prescribed theophylline who found daily life disrupted by tremors and constant stomach upset. Researchers and pharmacists stress the danger when combining xanthine drugs with antibiotics or medicines for heart rhythm problems, since this combination can raise levels and lead to toxicity.
Evidence from Research
Southampton General Hospital ran studies on patients using theophylline for lung disease and uncovered a clear pattern: as blood levels of the drug climbed above safe zones, patients often ended up with arrhythmias and seizures. The U.S. National Institutes of Health still lists these rare but severe side effects for all methylxanthine drugs. Even the FDA keeps warnings on some prescription leaflets, with instructions for regular blood tests to track how much is in the system. For most healthy people, drinking a cup of coffee or tea rarely creates risk, but prescribed xanthine drugs need extra caution.
What People Can Do
For those drinking coffee or tea, moderation keeps most side effects at bay. Problems usually only show up from supplements or prescription medicines. Doctors suggest routine blood tests for anyone on theophylline, especially those over sixty or with liver issues. Pharmacists stand ready to walk folks through possible drug interactions and help spot overdose signs, like confusion or shaking hands. Patients should share their full medication lists, including over-the-counter items, with their healthcare team.
Moving Toward Safer Use
Clearer education and warning labels for prescription xanthine drugs could make a difference. Regular communication between patients, doctors, and pharmacists helps stop dangerous levels before things get out of hand. Better research on genetic differences in liver metabolism might open doors for dose adjustments, so more people could get benefits without the risks. From my own view, nothing beats simple honesty about habits and symptoms in every doctor’s visit.
How should Xanthine be taken or dosed?
Understanding Xanthine: Not Just a Stuffy Chemical
Xanthine sounds technical, but it comes from real daily life. It’s the old backbone of substances like caffeine and theobromine—what most folks get in their coffee, tea, and chocolate. These compounds show up in all sorts of health products and medications, especially for things like breathing trouble (think asthma) and certain cardiovascular problems.
Dosing Xanthine—Not a One-Size-Fits-All Game
Ask any doctor, pharmacist, or nurse, and they’ll tell you: there’s no single right dose for everyone. Body weight, underlying health conditions, and age all play their part. For example, theophylline, a well-known xanthine derivative, needs careful attention. Too little and it doesn’t work. Too much and the system gets jittery or worse, with dangerous heart rhythms or even seizures.
Doctors used to dose drugs like theophylline using “one-size-fits-all” suggestions. Over time, they learned that blood levels can swing a lot from person to person. So, blood tests and close monitoring became part of the process. A lot of folks don’t realize that even what you eat and how much water you gulp down can change how your body handles these compounds.
Why Xanthine Isn’t DIY Medicine
Plenty of people figure if a little helps, more must be better. That’s a risky bet with xanthine drugs. Side effects don’t always show up right away. Faster heartbeats, nausea, headaches, and sleep trouble are just the tip of the iceberg. In my own clinic years ago, a teenager showed up shaky and wired. He was using a caffeine-laden energy supplement on top of an over-the-counter asthma inhaler. The mix pushed his heart rate up so high it scared both of us.
The U.S. Food and Drug Administration puts strict guidelines on dosing. Professional reference guides set the base dose by age and body weight. For theophylline, for instance, most adults start at around 200 mg twice per day. Blood levels guide any dose increase—not guesswork or advice found in an online forum.
What Matters Beyond the Label
People deal with different realities. A person with kidney problems processes xanthine slower. Someone who smokes breaks it down faster. Kids, the elderly, and those with liver disease need special adjustments. Grapefruit juice and some antibiotics change how long xanthine sticks around. In my conversations with pharmacists, they say many folks forget about these interactions until problems happen.
Every person brings a unique mix of habits, genetics, and health details to the table. There’s no “just take two and call me in the morning” answer for xanthines.
Working Toward Safer Use
Good care means communication. People should let their healthcare team know about supplements and medicines they use—no shame in asking questions. It helps to keep the lines open, check bloodwork when asked, and never adjust the dose without input from a qualified professional.
Companies need to keep clear labeling, honest communication, and proper education tools on the shelf. Health professionals stay sharp with new research and real-time patient stories, putting both experience and evidence into every piece of advice.
These straightforward steps keep people safer, reduce side effects, and make sure xanthine products support health rather than create surprise emergencies.
Is Xanthine safe for everyone to use?
Understanding Xanthine in Everyday Life
Most people don’t recognize xanthine by name, but many turn to products containing it every morning. Xanthine is a naturally occurring compound found in coffee, tea, and chocolate. It acts as a stimulant to the central nervous system, so it keeps people alert and focused. Caffeine and theobromine—the parts of xanthine everyone’s familiar with—show up in the drinks and snacks that help people power through their day.
Coffee drinkers benefit from xanthine’s alertness boost but can also feel wired or jittery. Some will even suffer headaches or digestive trouble after too much. There’s a big difference in how each person processes xanthines, partly because of genes and partly because of an individual’s tolerance or health.
Who Should Think Twice?
Kids feel xanthine’s effects quicker than adults, and their smaller bodies handle caffeine differently. Pediatricians tell parents to limit sodas and energy drinks for a reason. Pregnant women also run into trouble if they take in too much xanthine. Studies link high caffeine intake with lower birth weights and sometimes with miscarriage.
People with heart conditions, including high blood pressure, can find their symptoms triggered by xanthine compounds. Stimulants force the heart to work harder, increasing heartbeat and blood pressure. Even a healthy adult who drinks several strong coffees a day may notice chest discomfort or an irregular pulse. That’s something I’ve experienced myself after one too many espressos.
Sleep gets disrupted by xanthine. Insomnia, restlessness, and anxiety come out strong in people who drink late in the afternoon or grab high-caffeine sodas close to bedtime. Some may believe they are immune, but their sleep trackers or partners would notice the tossing and turning.
Learning from the Data
The U.S. Food and Drug Administration gives a general guideline of 400 mg of caffeine per day for healthy adults. That covers about four standard cups of coffee. Exceeding this bumps up risk for heart palpitations, digestive upset, and anxious moods. The European Food Safety Authority found similar thresholds. Rare people, such as those with certain genetic traits, can develop toxic reactions at lower doses.
Medication is another factor. Certain antibiotics, antidepressants, and heart drugs interact negatively with xanthine, making side effects more serious. Sometimes, the medications slow the breakdown of caffeine in the body, dialing up the effects or making them last longer.
Smart Ways to Use Xanthine
For those with no medical problems, moderate use generally stays safe. Pay attention to how the body responds: trembling hands, headaches, stomach upset, or difficulty sleeping signal it’s time to cut back. Doctors recommend pregnant women switch to decaf or limit themselves to a single small cup of coffee or tea. For families, dropping energy drinks from kids’ diets can help with emotional ups and downs, plus school focus.
Individuals with heart issues, anxiety, or insomnia often see big improvements once they trim caffeine. Decaf options and herbal teas let people enjoy warm drinks without the effects. Everyone benefits from reading labels—energy drinks and supplements sometimes hide high levels of xanthine compounds.
Making an Informed Choice
Xanthine’s everywhere in the modern diet, but not everyone responds the same. Listen to personal experience, ask doctors about risks, read up on new research. People with health conditions should talk with their doctor before grabbing that next cup. Being informed leads to better decisions for both body and mind.
What are the differences between Xanthine and caffeine?
Daily Encounters: Coffee, Tea, and Stimulants
Many people start their morning with a coffee ritual, trusting the caffeine in their mug to brush away sleepiness and sharpen their mind. Caffeine gets plenty of attention, especially for its energy-boosting perks, but fewer folks talk about xanthine, even though it’s a close chemical cousin. Both show up in daily life, tucked inside not only coffee but also chocolate, tea, and some sodas. Knowing the real difference between xanthine and caffeine matters both for those keeping their energy levels balanced and for anyone paying attention to health over the long haul.
Xanthine: The Chemical Foundation
Xanthine stands as an organic compound found in nearly every living thing. It shows up naturally in human bodies as part of how cells recycle bits of DNA and RNA. It’s also the backbone for a group of molecules called xanthine alkaloids, which count caffeine, theobromine, and theophylline among their members. On its own, xanthine rarely attracts notice in the world of food or drink, because it doesn’t pack a stimulant punch like its derivatives.
Caffeine: Stimulant Powerhouse
Caffeine, shaped by roasting coffee beans and brewing tea leaves, works by blocking adenosine receptors in the brain. Adenosine brings on feelings of tiredness; caffeine waves those signals away. Blood pressure edges up, awareness sharpens, bodies feel more awake. Across the world, about 85% of people enjoy some form of caffeine every day, with safe regular limits falling around 400 milligrams—a bit more than you’d find in three strong cups of coffee. Still, too much can stir up jitters, mess with sleep, or spike heart rate. Each body processes it differently, depending on genetics, age, liver health, and daily habits.
Biological Roles and Health Questions
The body handles xanthine as a middleman during metabolism. Cells break it down into uric acid, which the kidneys flush out. High levels of uric acid can lead to gout, a painful joint problem, while rare genetic disorders can disrupt xanthine breakdown and upset this balance. Caffeine, compared to xanthine, doesn’t build up in the same way. Instead, it swings its effects through the nervous system, prompting alertness and, in bigger doses, possibly anxiety or disrupted sleep cycles. Studies over the years have shown some protective effects from caffeine against Parkinson’s disease and some types of cancer. Still, those benefits hinge on moderate doses.
Choosing Wisely: Moderation and Awareness
Having spent years finishing workdays with too much black coffee, I’ve learned that caffeine’s helpful jolt can turn on a dime, leaving energy levels whipsawed if I’m not careful. Getting familiar with both xanthine and caffeine helped me cut down headaches and late-night tossing. For anyone sensitive to caffeine, herbal teas or drinks with theobromine—another xanthine derivative, found in chocolate—bring gentle uplift without so much insomnia risk. The lesson here is simple: read up, listen to your body, and remember there’s more to the world of stimulants than one famous name. By understanding the family tree, including xanthine, caffeine, and their relatives, you gain more choices for steady focus and better sleep.
Looking Ahead: Supporting Smarter Choices
Research continues to map the health impacts of caffeine and its chemical family. For anyone confronting health concerns—high blood pressure, kidney troubles, or sleep difficulties—keeping track of both caffeine and total xanthine intake can make a real difference. Labels on energy drinks, coffee blends, and even supplements deserve a closer look. Talking with a doctor or dietitian, especially before making big changes, beats guessing games every time. New products and trends will keep showing up on store shelves, but building knowledge about what’s really inside the cup gives everyone stronger footing for daily decisions.
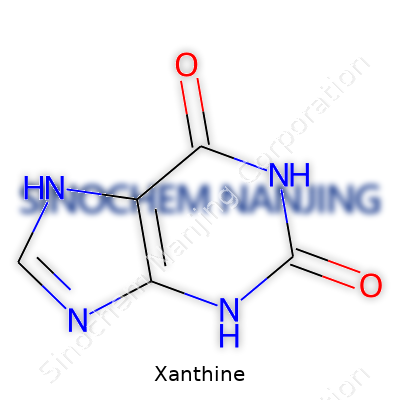
| Names | |
| Preferred IUPAC name | 3,7-dihydro-1H-purine-2,6-dione |
| Other names |
2,6-Dihydroxypurine 3,7-Dihydropurine-2,6-dione Theophylline base 4H-Purine-2,6-dione |
| Pronunciation | /ˈzænθiːn/ |
| Identifiers | |
| CAS Number | 69-89-6 |
| Beilstein Reference | 120927 |
| ChEBI | CHEBI:17712 |
| ChEMBL | CHEMBL267 |
| ChemSpider | 5959 |
| DrugBank | DB01351 |
| ECHA InfoCard | ECHA InfoCard: 100.003.118 |
| EC Number | 3.5.2.2 |
| Gmelin Reference | 47375 |
| KEGG | C00362 |
| MeSH | D027863 |
| PubChem CID | 978 |
| RTECS number | XZ3150000 |
| UNII | 0P7R9AnX5W |
| UN number | UN3077 |
| Properties | |
| Chemical formula | C5H4N4O2 |
| Molar mass | 152.11 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.7 g/cm³ |
| Solubility in water | slightly soluble |
| log P | -0.75 |
| Vapor pressure | 4.21E-10 mmHg at 25°C |
| Acidity (pKa) | pKa = 7.7 |
| Basicity (pKb) | pKb = 10.0 |
| Magnetic susceptibility (χ) | -54.0e-6 cm³/mol |
| Refractive index (nD) | 1.825 |
| Dipole moment | 2.60 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 229.0 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -441.2 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -2934 kJ mol⁻¹ |
| Pharmacology | |
| ATC code | R03DA02 |
| Hazards | |
| Main hazards | Harmful if swallowed, inhaled, or absorbed through skin; may cause irritation to skin, eyes, and respiratory tract. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302: Harmful if swallowed. |
| Precautionary statements | Precautionary statements: P261, P264, P271, P272, P280, P301+P312, P302+P352, P304+P340, P305+P351+P338, P312, P321, P330, P332+P313, P337+P313, P362+P364, P405, P501 |
| NFPA 704 (fire diamond) | 1-0-0-☐ |
| Autoignition temperature | 385 °C |
| Lethal dose or concentration | LD50 oral rat 1230 mg/kg |
| LD50 (median dose) | LD50: 200 mg/kg (rat, oral) |
| NIOSH | RN3679 |
| PEL (Permissible) | PEL: 5 mg/m3 |
| REL (Recommended) | Xanthine has no REL (Recommended Exposure Limit) established. |
| Related compounds | |
| Related compounds |
Caffeine Theobromine Theophylline Paraxanthine Uric acid Hypoxanthine 1-Methylxanthine 3-Methylxanthine 7-Methylxanthine |

