Vinyltriethoxysilane—A Crucial Contributor in Modern Industry
Historical Development
Long before modern materials science picked up pace, chemists began experimenting with organosilanes to close the gap between organic and inorganic molecules. In the mid-20th century, industry pioneers developed vinyltriethoxysilane by identifying vinyl groups' reactive nature and coupling them with hydrolyzable silane ends. These early innovations sparked entire families of products crucial for advancing coatings, sealants, adhesives, and composite manufacturing. Engineering teams realized that a tiny tweak in molecular structure could unlock stronger adhesion between glass fibers and organic polymers. Today, this compound remains a testament to the value of incremental innovation built upon years of cumulative laboratory experience.
Product Overview
Vinyltriethoxysilane draws attention from chemists and manufacturers alike because of its versatility and relatively simple molecular makeup: a silane backbone outfitted with a vinyl group and three ethoxy groups. This enables it to link organic polymers with inorganic surfaces, a quality that sits at the core of many advances in plastics, rubbers, and glass fiber composites. The material stands out in applications where surfaces traditionally resist bonding. For companies hoping to make composite parts lighter or coatings more durable, this compound opens possibilities that other adhesion promoters simply can't match.
Physical & Chemical Properties
Clear and mostly colorless, vinyltriethoxysilane shows a typical faint odor, boiling near 160 °C and evaporating readily under reduced pressure. Its modest viscosity helps it soak into porous material or spread evenly as a coupling agent. Ethoxy groups hydrolyze easily in humid air, releasing ethanol; that byproduct points to the necessity for good ventilation and careful handling. At room temperature, this liquid avoids crystallizing. It dissolves in most common organic solvents but shuns water until hydrolysis starts—an asset for storage and transport but a trigger for reaction once film formation or crosslinking becomes important.
Technical Specifications & Labeling
By regulation and custom, containers carry detailed specifications including purity (often above 98%), specific gravity, refractive index, and acid value. For safe transport, containers must list hazard symbols, UN numbers, and recommended storage temperatures. Product sold in the global market requires careful batch testing, with the certificate of analysis outlining relevant figures like water content, color, and identification via gas chromatography. Labels must show CAS number 78-08-0. From my experience, documentation builds trust between sellers and industrial buyers—clear technical data can make or break the purchase decision.
Preparation Method
Chemists typically prepare vinyltriethoxysilane by reacting vinyltrichlorosilane with ethanol in the presence of an acid scavenger. This direct route replaces the harsh chlorine atoms with gentle ethoxy groups, all under controlled temperature and pressure. The reaction generates hydrogen chloride gas, which demands proper scrubbing and waste treatment. Each batch depends on maintaining strict moisture control; exposure to water too early can ruin weeks of raw material and saddle a plant with costly downtime. My conversations with plant engineers reinforce that repeatable yield and purity hinge on obsessive process controls, often using nitrogen blanketing and specialized glass-lined reactors.
Chemical Reactions & Modifications
The hydrolyzable ethoxy groups on vinyltriethoxysilane drive silanol formation in the presence of water, leading to condensation reactions that anchor the molecule to inorganic substrates like glass or metal oxides. The vinyl group remains reactive after hydrolysis, available for further reactions such as copolymerization with vinyl monomers. As a result, this dual-reactive personality gives product designers a way to construct hybrid materials where the interface would otherwise fail. Because surface chemistry never really gets “solved” permanently, every improvement in application methods or modification routes shapes how the industry approaches the next generation of adhesion challenges. Researchers often experiment with modifying the vinyl function or swapping out the ethoxy arms; every small change can give new life to old materials.
Synonyms & Product Names
Often, companies and researchers refer to this product by varied names, including Triethoxy(vinyl)silane and Vinylethoxysilane. Some trade names house it under lines like A-151, Silquest VTMO, or Dynasylan VTEO. Recognizing synonyms ensures any purchaser can navigate technical sheets, databases, and regulatory filings across markets. Clear communication over identity not only avoids costly errors but also spotlights which formulation best fits a tough technical task.
Safety & Operational Standards
Safe handling means serious attention to personal protective equipment: nitrile gloves, splash-proof goggles, and lab coats as essentials. Any plant or lab working with vinyltriethoxysilane must install forced ventilation and monitor for ethanol or silane vapor. The compound’s volatile nature and hydrolysis chemistry mean spills can create slippery conditions and release flammable or irritating byproducts. Thoughtful training in emergency procedures, up-to-date material safety data sheets, and waste protocols limit risks to workers and the community. In my experience, companies that treat safety as an everyday habit—not a checklist item—see fewer accidents and longer employee careers.
Application Area
The most visible impact of vinyltriethoxysilane comes in the production of glass fiber composites for automotive and construction use. Silane coupling agents like this form the invisible bridge between glass fibers and polymeric resin, keeping hoods, bumpers, or window frames lightweight yet tough enough to outlast the elements. Cable insulation and sealants also benefit—either through stronger bonds to mineral fillers or longer service life. Over time, manufacturers have taught themselves to rely on these chemical tools not as a luxury but as a core piece of their competitive edge: without better performance at the molecular level, final products become commodities.
Research & Development
Laboratories continue to probe new uses for vinyltriethoxysilane, turning to it for roles in advanced ceramics, solar panels, and bio-inspired coatings. The molecule’s simple structure offers just enough modularity for chemists to tweak its function for each new challenge. Academic teams chase after fresh catalysts and reaction routes, while industry players track performance over years of outdoor use or electrical stress. Each experiment and field test adds a data point, broadening company and community knowledge.
Toxicity Research
Toxicological studies indicate that pure vinyltriethoxysilane demands respect: vapor inhalation or prolonged skin exposure can irritate tissues, and ask for fast ventilation in case of accidents. Researchers track any signs of chronic toxicity with multi-year animal tests, measuring organ weights and blood chemistry. So far, strict handling and monitoring protocols have reduced health incidents, but regulatory agencies still ask for up-to-date reporting on possible mutagenic or carcinogenic effects. In my time reviewing risk assessments, open collaboration between chemical producers and health authorities made a noticeable difference in safety culture—not just at the plant, but upstream and downstream among suppliers and customers.
Future Prospects
Looking ahead, the pathway for vinyltriethoxysilane points beyond traditional construction and automotive lines. Engineers now explore how organosilane chemistry might drive performance in recycled polymers or energy storage materials. Stricter environmental standards nudge producers toward cleaner reaction routes and lower emissions, potentially opening the door for greener variants and improved process design. Innovation rarely takes a straight road, but the compound’s unique surface-bridging qualities promise continued demand wherever new materials need to function at their best.
What is Vinyltriethoxysilane used for?
Understanding Vinyltriethoxysilane in the Real World
Most folks don’t think twice about what holds together the plastics and fillers in their car’s bumpers, or how electrical wires stay tough through sun and rain. Vinyltriethoxysilane shows up quietly in all sorts of places, though. As a chemical coupling agent, it helps different materials get along—think plastics sticking to glass or metal without falling apart. If you’ve ever watched a phone cable survive years of bending, or a building’s sealant hold strong through crazy weather, there’s a good chance vinyltriethoxysilane played a part.
How Manufacturers Use This Special Ingredient
Companies making plastics, paints, rubber compounding, or adhesives use vinyltriethoxysilane to strengthen finished products. In the plastics world, it helps link fillers like clay or glass fiber into the polymer matrix, so products last longer and don't crack as quickly. Cables and wires get coated with it, which helps the insulation stick snugly to the copper inside. That extra grip matters—you don’t want cables peeling apart or losing their protective layer just from being coiled and bent.
Paint manufacturers also lean on vinyltriethoxysilane for better weather resistance. It can help form a barrier so water doesn’t seep into concrete, stone, or brick. Masonry sealers draw on the waterproof qualities that vinyl groups provide, protecting your patio or driveway from early breakdown. People might not notice the science behind their waterproofed basements, but without additives like this silane, those solutions wouldn’t stick or last.
Importance in Everyday Applications
The need for tough, long-lasting products helps keep these silanes in demand. In construction, bridges and buildings take a beating from moisture and temperature swings. By using vinyltriethoxysilane-enhanced sealants or coatings, crews get better results out in the field. As a seasoned DIYer, I’ve seen plenty of caulking crumble within a year or two when a cheaper formulation hits the shelves—dealers who use silane additives often deliver sealants that handle more abuse before showing wear.
In automotive work, bumpers, dashboards, and trim pieces face constant mechanical stress. Fillers like talc or glass fiber, when paired with vinyltriethoxysilane, help plastics flex instead of snapping apart. Fewer replacements mean less plastic waste down the line and fewer broken parts heading to landfill. Looking at wiring in cars or power tools, insulation made with proper coupling agents remains tougher, which helps prevent dangerous shorts or failures, especially in humid or oily environments.
Challenges and Looking Forward
Like a lot of specialty chemicals, vinyltriethoxysilane raises questions about worker safety and environmental impact. Proper handling matters at factories and even on construction sites since volatile organic compounds can escape from uncured products. Industry guidelines recommend good ventilation and personal protection—which I’ve found makes a world of difference for anyone exposed day in, day out.
Scientists keep pushing for greener chemistry. There are ongoing studies to develop silanes that break down harmlessly after use, or that work just as well without hazardous byproducts. As someone watching the push for more sustainable construction, I’d wager the next decade brings safer formulas, along with tighter rules about disposal and worker education. Until then, using vinyltriethoxysilane responsibly will keep our plastics, adhesives, and coatings performing up to real-life expectations.
What are the safety precautions when handling Vinyltriethoxysilane?
Respecting the Risks
Vinyltriethoxysilane turns up in plenty of workplaces. From treating glass fibers to helping plastics grab hold of paint, it offers real benefits. Yet once you pop the drum, the substance inside reminds you why we keep chemical safety fresh in our minds. This clear, colorless liquid quickly gives off its sharp smell, enough to signal its power to cause harm. Inhaling the vapors can irritate your nose and throat, while direct skin contact may lead to redness, dryness, or even nasty burns. Your eyes deserve protection here, too, since stray droplets can sting and damage tissue fast.
Gear Up for Protection
Rolling up your sleeves and grabbing a pair of cotton gloves won't cut it. Butyl rubber, nitrile, or neoprene gloves block vinyltriethoxysilane from seeping through. Splash goggles, not just regular safety glasses, keep your vision clear when pouring or mixing. Coveralls stop liquid from reaching your skin, and long sleeves mean business. Respirator masks with organic vapor cartridges become crucial if good air flow is not guaranteed.
Ventilation Isn't Optional
Anyone handling volatile chemicals knows that fresh air reduces risk. Open windows only do so much. Fume hoods, exhaust fans, and even local air extractors quickly drive off vapors. Relying on natural breeze or thinking the smell isn’t strong enough won’t protect your lungs. OSHA and Europe’s REACH regulation both stress sufficient ventilation, not as a bonus, but as baseline practice around substances like this.
Safe Storage Matters
Vinyltriethoxysilane wants out of its bottle. It reacts with air moisture, giving off ethanol, which is flammable. Store it tightly sealed, away from acids, water, and sources of ignition. Temperature swings in the storage room spell trouble. Steel drums with solid gasket seals do the job, with placement away from direct sunlight, heat, or spark risk. Never keep food or drink in the same area — an accidental spill can turn into a medical emergency fast.
Spills and Cleanups: No Shortcuts
A puddle of this silane means you drop everything and take action. Keep spill kits near work benches. Absorb the liquid with sand or commercial absorbents, not sawdust. Double bag the waste in polyethylene-lined drums, and label them properly. Only trained staff should attempt large spill cleanups, and evacuation makes sense if fumes get thick. Reinforcing regular drills with real-life practice helps everyone snap into gear faster.
Training and Respect Beat Overconfidence
It’s easy to imagine that going through motions meets regulations. If you’ve seen old-timers ignore glove warnings or skip eye protection, the temptation might creep in to do the same. Yet cases of chemical burns and lung damage come from small lapses. Regular training, hazard labels in straightforward language, and a company culture that values speaking up protect everyone's health. Experience reminds us: safety doesn't slow down good work, it keeps it going day after day.
Paying Attention Saves Health
Working with vinyltriethoxysilane requires more than just ticking boxes on a checklist. The risks are real, but so are the tools to control them. Investing in quality protective gear, strict storage routines, and constant respect for good ventilation doesn’t seem optional after a bad spill or a trip to the hospital. These habits build a buffer for everyone—from newcomers to seasoned hands.
How should Vinyltriethoxysilane be stored?
Real-World Storage Challenges
Vinyltriethoxysilane does not belong on a cluttered shelf in a back room. I’ve seen more than a few chemical storerooms in the past dozen years, and each one tells the same story: corners get cut, spills start to stain, and sometimes the warning labels fade into the background. With a compound like vinyltriethoxysilane, that relaxed atmosphere could open the door to bigger problems.
Why People Should Care
Vinyltriethoxysilane serves as a crosslinking agent and adhesion promoter, and it can be a key ingredient in coatings, plastics, and adhesives. At the same time, its flammability and sensitivity to water keep it firmly on the “handle with respect” list. Breathing in its vapors creates health risks. Regular exposure could lead to irritation, coughing, or even longer-term health effects. The Environmental Protection Agency and the European Chemicals Agency both signal caution—safe use matters, safe storage matters more.
Room for Improvement in Many Labs
Basic mistakes still happen, often because people get used to routine. I’ve walked into labs where sealed bottles weren’t truly sealed and where excess humidity quietly started the hydrolysis process. Silanes love to react with water vapor. As the glass gets cloudy and pressure builds, a simple mishap turns expensive. Anyone who has ever cleaned up after a bottle burst knows: sticky, corrosive contamination sticks around.
Best Practices for Storage
Keep vinyltriethoxysilane away from direct sunlight, flames, and heat sources. Store it in a cool, dry, and well-ventilated place—think of a locked chemical cabinet with temperature controls if possible. Original containers remain the safest bet. Manufacturers design them to slow evaporation and reduce exposure to air. Wear gloves and goggles every time the bottle comes out. My own routine always includes a check for crusty or swollen caps, since that means the seal has failed or moisture sneaked in.
Highly reactive chemicals like silanes find friends in the wrong places. Paper, wood, and cotton act like wicks and should never sit nearby. Instead, use a metal or chemically inert containment tray. I’ve heard stories of small leaks soaking into cardboard and quietly generating heat, kicking off an unexpected fire.
Responding to Leaks and Incidents
No one plans for leaks, but everyone should prepare for them. A clean spill kit with absorbent pads (not sawdust) prevents panic. If a bottle tips over, ventilate right away. Any sign of vapor means it’s time to leave and let the air clear. Emergency showers and eyewash stations should not be blocked. Fast access saves time and, sometimes, eyesight.
Moving Toward Better Safety
Regular training changes the culture in a workplace. Short refreshers on chemical handling—quarterly, if not monthly—help even experienced staff. Inventory checks flag outdated containers before trouble starts. Technology has made monitoring easier: simple humidity meters and temperature loggers keep conditions in the right range and send out alerts before real danger builds.
Taking storage seriously protects the health of workers and extends the shelf life of expensive chemicals. In a world where so many things get brushed aside, safe chemical storage deserves action, not just a passing thought.
What is the chemical formula of Vinyltriethoxysilane?
Chemical Formula and Structure
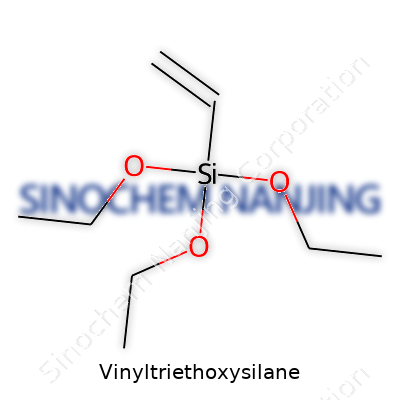
Vinyltriethoxysilane has a simple formula: C8H18O3Si. It brings together a vinyl group with three ethoxy groups attached to a silicon atom. That combination looks simple on paper, but the real power of this molecule comes from how those pieces interact with other materials. The structure helps bridge organic and inorganic materials, making it valuable far beyond what you might expect from the name alone.
Real-World Significance in Manufacturing
In industries, Vinyltriethoxysilane plays a key role as a coupling agent. That means it helps things stick together that wouldn’t naturally want to. Take fiberglass and plastics. You mix these materials using silanes, and you get composites that are stronger and more durable. Without silanes, connectors between glass fibers and polymer resins grow weak, leading to products that break down sooner, whether it's in pipes, car parts, or construction materials. From personal experience in the construction field, relying on well-bonded composites has been the difference between repairs that last and projects that need costly revisits within just a few years.
Performance Benefits Rooted in Chemistry
The ethoxy groups in Vinyltriethoxysilane react with water to form silanol groups. These bond strongly with minerals like glass and concrete. On the other side, the vinyl group can chemically link with organic materials, such as the resins found in plastics. It’s this dual nature that makes the compound such a workhorse in adhesives, paints, and protective coatings. In my own work with industrial coatings, adding this silane translates into products that can handle moisture and temperature shifts better than untreated materials. Paint peels less, adhesives last longer, and coatings just hold up better under stress.
Risks and Safe Handling
With valuable properties come some handling risks. Exposure to moisture starts the ethoxy group reaction, which leads to ethanol release. That’s something to keep in mind on a busy factory floor, since ethanol vapors can ignite and bring health issues if people breathe them in over time. Extended exposure to the liquid also causes skin and eye irritation. Using proper ventilation, gloves, and safety goggles helps keep these risks down in any workplace that handles silanes.
Environmental Concerns and Solutions
Like many organosilanes, if Vinyltriethoxysilane spills or leaks, it doesn’t last long in the environment, breaking down to simpler forms. Still, if not managed correctly, its byproducts, especially ethanol, can add to air pollution. Setting up containment and responsible storage makes a big difference. I’ve seen that taking a proactive approach—regular monitoring, fast cleanup, and proper disposal channels—makes for safer plants and less risk to waterways or air quality. Chemical manufacturers are improving on this front, focusing on process efficiency and less waste. There’s always a trade-off between advanced material performance and what that does to the environment, but choosing innovations in recycling and safer handling can tip the balance in our favor.
Conclusion
Vinyltriethoxysilane may look like a simple molecule at first glance, yet its chemical formula C8H18O3Si powers many of the breakthroughs we count on in materials engineering. From stronger buildings to reliable electronics, the chemistry under the surface shapes our lives—and so do the choices we make about safety and environmental care while working with it.
Is Vinyltriethoxysilane compatible with other silanes or resins?
How Vinyltriethoxysilane Works in Real Industry Settings
I’ve seen a lot of talk about whether vinyltriethoxysilane can get along with other silanes or if it’s better to stick to one type. Out in the field, product formulators often aim to squeeze more performance out of their materials. Vinyltriethoxysilane brings alkoxy groups into the mix, along with a vinyl function. This means it can react or bond, depending on the partners present.
Getting Technical, but Not Out of Touch
Vinyltriethoxysilane stands out for its double life. Its vinyl group forms links with unsaturated resins—think polyester or epoxy. The ethoxy parts latch onto surfaces rich in hydroxyl groups like glass, minerals, or even some metals exposed to air. This dual nature helps build tight bridges between materials that often want to pull away from each other, such as fillers in a plastic matrix.
The chemistry is flexible enough that it gels well with plenty of silanes, especially those packing similar hydrolyzable groups. I’ve seen labs blend vinyl and amino silanes to boost adhesion for high-performance composites, especially in automotive panels or fishing rods meant to take a beating. These mixtures help control both internal strength and bonding to whatever comes next in a layer or laminate.
Resin Relationships: What Has Worked
Vinyltriethoxysilane plays nice with polyester and epoxy systems. I’ve watched batches mixed in as an additive or even pre-treated onto glass fibers, with obvious jumps in strength and toughness. Surface energy shifts, so the resin wets out the filler better and fewer weak spots show up under testing. Water resistance improves, shrinking the risk of creeping failure from humidity.
Epoxy shops have added this silane side by side with other coupling agents in a single blend, and the mix doesn’t foam up or settle out as long as the ratios aren’t pushed to extremes. Too much can lead to over-crosslinking, making the end product brittle, so experience counts when deciding on dosage.
Missteps and What Matters Most
Sometimes, overengineering leads to headaches. I’ve seen crews try to layer too many different silanes, chasing every property in the book—fire resistance, flexibility, and everything else. Results get muddy, and it’s hard to troubleshoot when something cracks or delaminates.
The big trick is understanding your raw materials and using data, not just wish lists. Before stacking up six chemistries, it pays to work with technical teams and scale up slowly. Just one silane—like vinyltriethoxysilane—often gets the job done if selected for real needs, not hypothetical scenarios.
Looking Ahead
Industry wants lighter cars, more durable wind turbines, and coatings that laugh at weather. Blending vinyltriethoxysilane with compatible agents—amino, epoxy, or mercapto silanes—delivers some of the answers. These combos aren’t magic bullets, but they bring a steady stream of gains for strength and durability.
Moving forward, smart material choices and real-world testing will keep unlocking value. There’s no need to reinvent the wheel with every batch, just a commitment to learning and sharing honest results from the line or the lab.
| Names | |
| Preferred IUPAC name | triethoxy(ethenyl)silane |
| Other names |
Triethoxyvinylsilane Vinyltriethoxy-silane Silane, triethoxyvinyl- Ethenyltriethoxysilane |
| Pronunciation | /ˌvaɪ.nəl.traɪˌiːθ.ɒk.si.saɪˈleɪn/ |
| Identifiers | |
| CAS Number | 78-08-0 |
| Beilstein Reference | 1721440 |
| ChEBI | CHEBI:78063 |
| ChEMBL | CHEMBL1375981 |
| ChemSpider | 21420 |
| DrugBank | DB14096 |
| ECHA InfoCard | 03d755fc-e2e9-4bfc-9e57-ac8b826e2a0d |
| EC Number | 220-941-2 |
| Gmelin Reference | 84967 |
| KEGG | C12045 |
| MeSH | C07-399-850-875-770 |
| PubChem CID | 15260 |
| RTECS number | YX9625000 |
| UNII | N9U2N4H0G7 |
| UN number | UN1993 |
| CompTox Dashboard (EPA) | DTXSID2020183 |
| Properties | |
| Chemical formula | C8H18O3Si |
| Molar mass | 190.32 g/mol |
| Appearance | Colorless transparent liquid |
| Odor | Sweet |
| Density | 0.88 g/cm3 |
| Solubility in water | Reacts with water |
| log P | 0.5 |
| Vapor pressure | 0.3 hPa (20°C) |
| Acidity (pKa) | 11.4 |
| Basicity (pKb) | 13.5 |
| Magnetic susceptibility (χ) | -6.72×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.3960 |
| Viscosity | 1 mPa.s (25°C) |
| Dipole moment | 4.32 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 286.6 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | –606.1 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3629.7 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H226, H315, H319, H332 |
| Precautionary statements | P210, P261, P280, P301+P312, P303+P361+P353, P305+P351+P338, P337+P313, P370+P378, P403+P235, P501 |
| NFPA 704 (fire diamond) | 1-2-0-健康-炎性-反応 |
| Flash point | 62 °C |
| Autoignition temperature | 287 °C |
| Explosive limits | Explosive limits: 1.3% (LEL), 9.5% (UEL) |
| Lethal dose or concentration | LD50 Oral Rat 2,460 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral, rat: 4.3 g/kg |
| NIOSH | GGG |
| PEL (Permissible) | PEL (Permissible) for Vinyltriethoxysilane: Not established |
| REL (Recommended) | 50–200 mg/kg |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Vinyltrimethoxysilane Vinyltriacetoxysilane |

