Vinylene Carbonate: Unfolding the Layers
Historical Development
In the late 1960s, chemists took notice of vinylene carbonate during their hunt for new electrolyte additives. Through the decades, the compound's story weaves neatly through the rise of rechargeable battery technologies. Spotlight landed firmly on vinylene carbonate as lithium-ion batteries began powering up laptops and mobile phones in the 1990s. Researchers in Japan and Europe dug deep, exploring its function and performance. By the early 2000s, battery manufacturers in the United States and China began moving from early-stage experiments to larger-scale tests. This chemical's evolution hasn’t followed a straight line; it shows just how much persistence and curiosity drive innovation. People searched for more stable, longer-lasting battery cycles, and vinylene carbonate held out real promise. As patents rolled in and academic papers grew thicker on the subject, industries slowly built robust supply chains and production know-how around this molecule.
Product Overview
Vinylene carbonate steps onto the industrial stage as a colorless to pale yellow liquid. Scientists value its molecular structure—C3H2O3. Not only does it remain quite stable under ordinary storage conditions, but it also blends smoothly with common organic solvents. Companies have refined the purification process, so most supplied material meets demanding purity levels for research and electronics applications. In everyday experience, it doesn’t let off an overpowering smell and clears quality checks for water and metal impurities. Many suppliers sell it in sealed glass or metal containers to avoid contact with moisture during transport.
Physical & Chemical Properties
Vinylene carbonate melts just above room temperature—people in the lab observe it liquid around 21°C and bubbling up near 130°C. As a result, it handles repeat heating and cooling cycles without fuss. Its density stands near 1.4 g/cm³, and it dissolves easily in polar organic solvents like dimethyl carbonate and ethylene carbonate. Unlike related cyclic carbonates, vinylene carbonate can undergo polymerization and participates keenly in organic reactions due to its double bond. This chemical’s reactivity opens up a toolbox of options for surface chemistry and specialty coatings. At elevated temperatures, it doesn’t cling to chemical stability; instead, it might break down to release gases, so chemists approach thermal processes with respect for its limits.
Technical Specifications & Labeling
Most commercial suppliers post purity numbers above 99%. Water content, if too high, disrupts performance inside battery cells; lab techs run Karl Fischer titrations to confirm safe moisture levels below 0.05%. Heavy metal traces sit far below 10 mg/kg. Companies mark every drum with the batch number, production date, and expiration mark. Barcode labels and QR tags track each lot to the source. Safety icons warn workers of its low flash point. MSDS folders back up every shipment, not just for compliance but because the people handling chemicals need real information at a glance.
Preparation Method
On the industrial floor, vinylene carbonate production relies on careful cyclization. The classic synthesis starts by reacting ethylene glycol with phosgene or related carbonylation agents in a controlled reactor. Some labs now lean toward greener methods, swapping out dangerous phosgene for safer carbonates or catalytic CO2 activation. The crude product runs through distillation columns and vacuum dry-purification to yield a clean, market-ready liquid. These methods keep improving—smaller companies bring in better catalyst systems, cutting costs and lowering waste. Every production line tracks yields, solvent recovery, and energy use, because every efficiency tweak counts over thousands of tons.
Chemical Reactions & Modifications
Vinylene carbonate acts like a versatile building block. It jumps into polymerization reactions, giving chemists an entry into polycarbonates and cross-linked polymers for specialty resins. In the battery world, vinylene carbonate shines during electrolyte formation—its double bond and carbonate group let it take part in the solid electrolyte interface (SEI) layer, which protects electrodes and stretches a battery’s lifespan. Under the right conditions, it undergoes nucleophilic additions or opens its ring, so researchers keep tweaking solvent mixtures and voltages to get the most robust battery cycle life. Curious electrochemists continually trial new modifications, adding bulky groups or tweaks to the ring to build safer, less volatile structures.
Synonyms & Product Names
On shipping manifests and catalogs, vinylene carbonate pops up as 4-vinylene-1,3-dioxolan-2-one or 1,3-dioxol-2-one, 4-vinylene. Technical documents rarely agree on one term, so lab teams double check chemical structures before each experiment. Major producers use their own stock numbers or brand designations, but the chemical’s CAS number—872-36-6—anchors it in international commerce.
Safety & Operational Standards
Working with vinylene carbonate means suiting up with gloves, goggles, and good ventilation. Its low flash point—below 100°C—demands safe handling far from open flames or sparks. Safety teams train crews to store it in tightly sealed containers and label spills at once. Waste fits strict local guidelines for solvents. OSHA and GHS standards guide training modules and emergency drills—old hands know the fine points of cleanup, from absorbent pads to neutralizing agents. Health experts say prolonged exposure may irritate eyes, skin, or lungs, and they flag the need for both sensible PPE and up-to-date ventilation checks.
Application Area
By far, lithium-ion batteries form the main demand driver—for both large-scale EV cells and smaller electronics. The compound’s SEI-forming power directly affects cell life, and everyone from carmakers to solar manufacturers takes this role seriously. In specialized polymer synthesis, it pops up as a cross-linking agent. Some research teams look into its use in advanced electrolytes for supercapacitors and hybrid energy storage. Material scientists also run experiments on coatings, thin films, and even printable electronics. For each of these, end-user concern zeroes in on cost, purity, and long-term performance, not just theoretical scores.
Research & Development
Lab teams push hard to make safer, higher-performing batteries. In Asia, dozens of university groups experiment with new doping methods to tune SEI layers using vinylene carbonate. US and European consortiums fund pilot projects scaling up greener production routes. Computational chemistry, with better simulation tools, now helps predict how modified vinylene carbonate molecules might boost cycle life or suppress dendrite growth. Industry-academic partnerships pay for life-cycle testing in demanding conditions—from cold startups in winter to rapid charging on the highway. Results feed straight into real-world improvements; for every academic paper, commercial research vets the claims in tough quality labs.
Toxicity Research
Early toxicity tests flagged eye and respiratory tract irritation in direct-contact mouse models. Chronic exposure studies have yet to draw final lines on workplace health risk, so manufacturers stick to robust PPE and ventilation. Regulatory agencies in the EU and US keep watch over new findings, especially as greater battery demand drives up the global production. Waste handling guidelines require collection and treatment, not open dumping, especially near water sources. Medical research still explores any lingering impacts, especially for people involved with recycling old lithium-ion cells. Even with the compound’s low overall toxicity, the responsible call always leans toward caution, not shortcuts, until full risk assessments emerge.
Future Prospects
Over the next decade, global shifts toward renewable energy and electric vehicles put pressure on every ingredient in battery supply chains. Vinylene carbonate isn’t immune to these pressures; its supply and price fluctuate with new mines, regulations, and competing solvent technology. Companies invest in scaling up sustainable manufacturing, cutting down on hazardous input chemicals, and shrinking the carbon profile of each batch. As recycling technologies catch up, chemists want to recover and reuse polymer breakdown products, so vinylene carbonate may see a second life in recovery and purification systems. As academic teams publish new molecular tweaks, like hybrid boron or sulfur substituted variants, real-world engineers keep one eye on performance and the other on environmental impact, always weighing cost, longevity, and recyclability against the technical advantages in the race to develop the batteries of tomorrow.
What is Vinylene Carbonate used for?
What Sets Vinylene Carbonate Apart?
Vinylene carbonate isn’t a household name, but its impact reaches millions who rely on rechargeable electronics every day. Electric vehicles, smartphones, laptops—all these depend on lithium-ion batteries. These batteries have transformed how people work, commute, and connect. Vinylene carbonate quietly fuels this progress, improving battery performance in ways that most users never notice.
Making Batteries Better
The main job of vinylene carbonate involves working as an additive in battery electrolytes. Unlike older battery chemistries, lithium-ion designs can lose capacity or even catch fire if exposed to the wrong conditions. Adding vinylene carbonate during production helps solve both problems. It works by creating a protective film on the battery’s anode, a barrier known among engineers as the solid electrolyte interphase. This isn’t just jargon—for consumers, this barrier means a battery that lasts longer and runs safer.
Research continues to show that batteries made with vinylene carbonate keep their charge for more cycles than those without it. Numbers tell the story: some studies credit this additive with increasing battery lifespan by as much as 30 percent. The improvement doesn't stop there. By reducing side reactions that cause swelling or potential short circuits, vinylene carbonate plays a big role in keeping lithium-ion technology from failing on the job.
Addressing Industry Challenges
The search for dependable, rechargeable power keeps growing with the shift toward electric vehicles and energy storage for solar and wind energy. Car buyers worry about how long a battery will last between charges, and companies face intense pressure to avoid safety incidents while meeting sustainability targets. Vinylene carbonate helps carmakers and battery producers bridge those concerns without needing earth-shattering breakthroughs.
Safety matters, and recent high-profile recalls in the electric vehicle space have highlighted just how important it is to trust what’s under the hood. Some battery fires can be traced back to chemical breakdown at the anode, something vinylene carbonate’s stable film helps prevent. Though it seems like a small part of a complex system, its cost relative to potential losses from faulty batteries makes it an easy investment for manufacturers.
Ethics, Sustainability, and the Future
The world is watching every step of the battery supply chain, from raw materials to recycling. Some additives create new waste problems or increase the cost of safe disposal. Vinylene carbonate stands out here. Its chemical stability means less harmful byproducts, supporting cleaner battery production and recycling. Supply chain transparency remains a challenge, but manufacturers turning toward greener materials see vinylene carbonate as a step in the right direction.
People who depend on long drives, reliable gadgets, or backup power may never read a label mentioning vinylene carbonate. Yet, its influence shows every time that phone holds a charge on a busy day—or an electric car packs in a few extra miles before plugging in. Engineers will keep looking for ways to make energy cleaner and more efficient, and this curious molecule will likely keep showing up in their toolkits.
Is Vinylene Carbonate hazardous or toxic?
Vinylene Carbonate and Everyday Applications
Vinylene carbonate plays a big role in rechargeable battery technology, especially in lithium-ion batteries found in phones, laptops, and electric cars. If you’ve ever searched for the reasons behind longer battery life or stability under tough conditions, chemists often point to additives like this one. Its ability to form a protective film inside battery cells helps prevent degradation and overheating. But, like many chemicals that push technology forward, vinylene carbonate also prompts serious safety questions, especially for those who work with it daily.
Real Hazards in the Lab and Workplace
Many assume that chemicals working behind closed doors in a battery pack stay contained. Reality tells a different story. Production lines, research labs, and battery recycling plants all bring workers into direct contact with these substances. Vinylene carbonate remains a liquid at room temperature and evaporates quickly into the air. In my lab days, even a brief whiff of solvents or reagents meant opening windows and double-checking protective clothing.
Manufacturers list vinylene carbonate as an irritant. Direct skin contact leads to itching, redness, and sometimes burns if left unwashed. Inhaling vapor can sting the nose and throat, and in extreme cases, trigger dizziness or headaches. Data sheets from chemical suppliers caution users to avoid breathing the fumes, to keep the liquid off skin, and to use goggles and gloves. Animal studies, mainly with rodents, point to lung irritation and possible liver stress after big exposures, so safety officers rightly emphasize strong ventilation and protective clothes.
Long-Term Risks and Environmental Concerns
It’s not just about the burns or coughs seen right away. Vinylene carbonate contains reactive chemical bonds that break down under sunlight or heat, sometimes giving off carbon monoxide, a colorless gas that brings its own dangers in closed areas. Factories and labs need regular air quality checks and strict handling protocols to keep long-term risk in check.
Accidental spills can seep into drains or soil, though vinylene carbonate breaks down in water over time. That said, chemical spills rarely end cleanly. Traces left behind can still harm small water life and soil bacteria. Environmental managers monitor runoff closely during manufacturing and disposal. Funded clean-up programs, proper training, and material recovery systems make a real difference here.
Building Safer Labs and Greener Tech
Instead of leaving hazard warnings as fine print, battery companies have started to train workers in hands-on ways: live drills, personal anecdotes, easy-to-read safety charts. During college years spent as a chemistry assistant, watching a more experienced colleague suit up before each shift taught me a lot more than laminated posters ever did. Tracking health outcomes over longer careers also helps researchers and workers understand slow-acting risks, not just what happens after a spill or fire.
Another key step involves swapping out more toxic materials when possible. Some battery researchers aim to design similar protective additives that break down faster in the environment or pose less threat in handling. Government regulations set legal exposure limits, but chemical companies push for even tighter internal standards after learning from safety lapses.
Nobody wants battery breakthroughs to come at the cost of burning hands or trouble breathing. Clear labeling, dedicated safety training, and new chemical design come together to lower risks for everyone involved—from the factory floor to the recycling plant. Vinylene carbonate may let your phone last longer between charges, but its safe use depends on hard-earned habits, not shortcuts.
What is the chemical formula of Vinylene Carbonate?
Understanding Its Formula and Relevance
Vinylene carbonate draws attention because its molecular structure unlocks performance improvements for lithium-ion batteries. Its chemical formula is C3H2O3. That’s three carbon atoms, two hydrogens, and three oxygens, bonded in a unique ring. People working with rechargeable batteries or researching electrolytes spot this molecule because it solves real-world problems—no theoretical appeal here.
How Vinylene Carbonate Makes a Difference
Electrolyte additives rarely get the fame they deserve, but they make batteries safer and more efficient. With the world shifting to all things electric—cars, bikes, phones—it creates huge demand for smarter, longer-lasting batteries. Scientists turn to molecules like vinylene carbonate to tweak performance, then watch the results play out in labs and factories. Its role centers on improving the charging cycle, reducing harmful side reactions, and increasing battery lifespan.
Through direct experience in lab environments, deliberately testing batteries with vinylene carbonate against those without shows a marked improvement in performance. The molecule works by helping to form a stable and protective solid electrolyte interphase on the electrode, which minimizes the breakdown and extends useful life. Without this protection layer, batteries lose capacity much faster and generate more waste.
Risks and Environmental Concerns
Every chemical brings trade-offs. I’ve talked to engineers who find vinylene carbonate essential, yet they never lose sight of the health and safety warnings stamped on its datasheets. Proper ventilation, careful handling, and protective gear all matter. Mishandling could cause harm, especially in industrial setups where large volumes get stored and used. Those risks require clear company policies, regular staff training, and rigorous review of procedures.
Waste management remains another question. While vinylene carbonate itself doesn’t rank among the most hazardous industrial chemicals, battery recycling must get done properly to avoid environmental contamination. This ties directly into the wider challenge of electronic waste piling up as demand for smart devices keeps rising. Cities and corporations both scramble to fix the recycling gap, but progress still feels uneven. Managed right, the impact stays small; handled carelessly, it grows into a community headache.
Pushing for Solutions
Too often folks focus on only one part of the equation. Inventors develop formulas with promise, yet adoption stalls if regulations can’t keep up or if costs spiral. Based on industry reports and conversations with analysts, the safest way forward happens when researchers, manufacturers, and policy-makers all share responsibility for handling, disposal, and innovation. Tighter cooperation would mean faster feedback for improving processes and safer working environments for everyone involved.
Education also makes a real difference. No one expects everyday consumers to memorize chemical formulas, but empowering workers and students with clear science brings better decisions and less risk down the line. It always helps to know where things come from, how they’re used, and what happens after their job is done.
So, C3H2O3 may not roll off the tongue or sparkle in headlines, yet its role inside batteries points to bigger trends. Advanced chemistry, smart safety, and shared responsibility shape whether new solutions hit the mark for people and planet alike.
How should Vinylene Carbonate be stored?
Understanding Real Risks in the Lab and on the Floor
Vinylene carbonate pops up often in battery research and as a useful chemical in organic synthesis, but it doesn't get much attention until something goes wrong with storage. Plenty of folks underestimate how easily a chemical like this can go off and how critical a stable environment becomes, especially if you’ve ever opened an old bottle only to find the contents have turned yellow or started to crust along the cap.
I learned quickly that this kind of chemical, clear and almost innocent-looking, carries real hazards if basic care gets skipped. Vinylene carbonate reacts to moisture in the air. It starts to break down, sometimes building up pressure inside containers or forming polymers that can clog up dispensing equipment. Toss a bottle in a warm, humid storeroom, and you’ll find yourself dealing with sticky gunk and unreliable chemistry.
Temperature and Humidity: The Practical Realities
From firsthand messes and plenty of secondhand stories, keeping vinylene carbonate away from heat sources changes everything. At higher temperatures, the substance can decompose and become a safety headache for lab workers. It's not about fancy climate controls in every case but about sticking with the basics. Low room temperature works. Keep it away from direct sunlight, heating vents, and places known for temperature swings.
Humidity sneaks up on you. It might seem harmless, but a cap not screwed tight will let just enough moisture in to trigger slow degradation. I prefer a dedicated desiccator for small quantities or, failing that, a tightly sealed bag with a few silica gel packets tossed in. For bigger, industrial containers, using specialized air-tight drums with gasketed lids makes a real difference.
Choosing the Right Container
Not all bottles are built the same. Avoid using containers with metallic caps. There’s a chance of an unwanted reaction. Polyethylene and glass work better for vinylene carbonate. Never reuse bottles that have held other chemicals, because trace contamination changes the whole chemistry—I've watched batches ruined from small oversights like this.
Use the original packaging if possible since manufacturers usually test compatibility. Only transfer to a new bottle for a good reason—and always label the new container with the chemical name and the date.
Ventilation and Segregation
Store vinylene carbonate in a space with good airflow, kept away from acids, bases, and incompatible solvents. Cross-contamination causes more headaches than most people expect. Stashing this chemical near oxidizers, or letting it sit next to strong acids, is asking for trouble. A segregated shelf with clear chemical groupings pays off every time someone avoids an accident by grabbing the right bottle.
Eyes on Expiry, Ears Open for Hazards
Check containers routinely. Look for changes in color, consistency, or smell. Document what you see. Manufacturers give expiration dates for a reason: the shelf life isn’t a sales gimmick. Degraded vinylene carbonate makes experiments unpredictable, and it ups the risk of pressure buildup or leaks.
If disposal becomes necessary, don’t just dump leftovers down the drain. Many localities classify vinylene carbonate as hazardous waste, and for good reasons tied to its chemical behavior in water. Follow local or institutional protocols, and call trained staff for questions, as shortcuts only come back to haunt you and those who work with you.
Knowledge and A Little Discipline Save Headaches
No big secret hides behind the safe storage of vinylene carbonate. Attention to detail, regular checks, and respect for chemical reactivity go a long way. The same goes with any lab or process—the smallest bit of discipline with storage saves time, money, and sometimes keeps people safe who don’t always know what’s in that unmarked bottle at the back of the shelf.
Where can I purchase Vinylene Carbonate?
The Allure and the Catch
Vinylene carbonate attracts plenty of interest these days. Whether you work in a research lab studying next-generation batteries or keep an eye on new tech in the energy storage space, you have probably heard its name get tossed around in meetings and papers. With lithium-ion battery chemistry advancing, a small molecule like vinylene carbonate suddenly becomes a big deal. It boosts performance, stabilizes electrolytes, and supports the durability found in modern power banks and electric vehicles. That draws attention, but it also comes with some real speed bumps for anyone just looking to snag a bottle.
Not a Shelf Staple
Most folks won’t spot a bottle of vinylene carbonate at the local hardware store or on the shelves of big-box chemical supply stores. This isn’t isopropanol or bleach. It falls under a different category, both because of its uses and its risks. Research-grade compounds often mean special paperwork, proof of a legitimate project, and compliance checks that triple-check who is handling them and why. Rules around chemical distribution have tightened after well-known incidents with harmful materials landing in the wrong hands. Real, professional buyers show a record of buying similar reagents and can quickly answer safety questions when suppliers ask.
Trusted Suppliers Only
For most scientists or tech startups, major chemical suppliers form the first stop. Sigma-Aldrich, Alfa Aesar, TCI, and Merck each carry vinylene carbonate, clearly listing catalog numbers and prices only for registered accounts. These suppliers read like a Who’s Who list for anyone who keeps a glovebox or fume hood running. Orders usually need approval from a team leader or university official, identifying information, established lab credentials, and a shipping address tied to recognized organizations. Package delivery includes tracking, sometimes special handling for temperature and spill risk. Priority always falls to research institutions, battery manufacturers, or businesses directly tied to electronics research.
Regulatory Gatekeeping
Why these hurdles? Authorities face a balancing act: keep innovation alive, block misuse. Vinylene carbonate is classified as a reagent with strong chemical reactivity. Under the wrong conditions, it becomes hazardous. Safety reviews look at purchaser track record and purpose. In the United States, the Chemical Facility Anti-Terrorism Standards (CFATS) and, for other countries, equivalents in Europe or East Asia, have become familiar roadblocks. Transport regulations make sure this chemical only hits the hands of those who can store, use, and dispose of it safely.
Black Market Warnings
Sketchy marketplaces or online sources that dodge regulations tend to show up with anything valuable. The urge to cut corners, to get reactivity on the cheap, lures in desperate buyers. Yet, underground purchases almost always trade lower standards for higher risks. No certificate of analysis means nobody knows what actually sits in your bottle. No one inspects for shipment integrity. Most unsafe shipments get caught in customs anyway, followed by legal headaches for the buyer.
Practical Steps Forward
Legitimate buyers do best approaching official vendors—those that know their chemicals, have dedicated safety data for each batch, and offer technical support for applications like battery research. If you only want this compound for a hobby, consider safer, more accessible alternatives. As rules around new battery chemistries evolve, suppliers and researchers can work together to streamline purchase and verification—using digital tracking and improved transparency. Education, outreach, and clear safety protocols back responsible use. This balances progress and protection, which keeps the door open for breakthroughs without inviting preventable accidents or misuse.
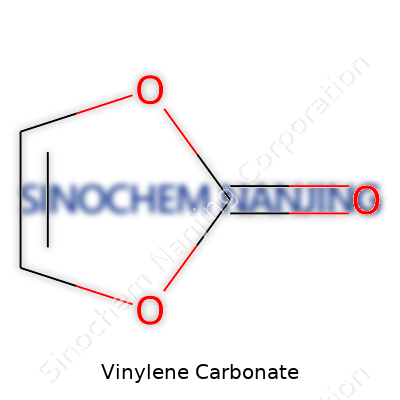
| Names | |
| Preferred IUPAC name | 4H-1,3-dioxol-2-one |
| Other names |
Vinylene carbonate 1,3-Dioxol-2-one VC Ethylene carbonate, unsaturated |
| Pronunciation | /vaɪˈnɪliːn ˈkɑːbəneɪt/ |
| Identifiers | |
| CAS Number | 872-36-6 |
| Beilstein Reference | 1208731 |
| ChEBI | CHEBI:139713 |
| ChEMBL | CHEMBL2106079 |
| ChemSpider | 80866 |
| DrugBank | DB14156 |
| ECHA InfoCard | 03b1c26a-44c1-4413-bf5c-bd0eb76dbb5e |
| EC Number | 214-317-9 |
| Gmelin Reference | Gmelin Reference: 81668 |
| KEGG | C12041 |
| MeSH | D018154 |
| PubChem CID | 72987 |
| RTECS number | FG0525000 |
| UNII | T11X3H6F4A |
| UN number | UN2478 |
| Properties | |
| Chemical formula | C3H2O3 |
| Molar mass | 86.04 g/mol |
| Appearance | White crystalline powder |
| Odor | Odorless |
| Density | 1.41 g/cm3 |
| Solubility in water | Soluble |
| log P | -0.3 |
| Vapor pressure | 0.3 mmHg (20 °C) |
| Acidity (pKa) | 22.15 |
| Basicity (pKb) | 1.42 |
| Magnetic susceptibility (χ) | -53.0 x 10^-6 cm³/mol |
| Refractive index (nD) | 1.424 |
| Viscosity | 2.164 mPa·s (25 °C) |
| Dipole moment | 2.56 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 123.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -473.8 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -1514 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS02,GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H319, H335 |
| Precautionary statements | P210, P261, P280, P305+P351+P338, P370+P378, P403+P235 |
| NFPA 704 (fire diamond) | 2-2-2-☠️ |
| Flash point | 90°C |
| Autoignition temperature | 455 °C |
| Explosive limits | Explosive limits: 4.4–16% |
| Lethal dose or concentration | LD50 (oral, rat) 5,100 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat LD50 = 500 mg/kg |
| NIOSH | VZ1290000 |
| PEL (Permissible) | PEL: Not established |
| REL (Recommended) | 0.03 ppm |
| IDLH (Immediate danger) | Not established |
| Related compounds | |
| Related compounds |
Ethylene carbonate Propylene carbonate Dimethyl carbonate Diethyl carbonate Vinylene urea |

