Vinyl Chloroacetate: History, Chemistry, and Tomorrow’s Potential
A Walk Through Vinyl Chloroacetate’s Past
Vinyl chloroacetate doesn’t make headlines the way plastics or pharmaceuticals do, yet it’s the backbone of a handful of transformative chemical processes. Its journey began in the early days of polymer chemistry in the twentieth century, when researchers turned their attention from natural rubber and celluloid to building-block molecules that could open new industrial pathways. Developed in labs hungry for new ways to link up small chemical units into bigger, more useful forms, vinyl chloroacetate benefited from the breakthroughs in organic synthesis that brought about entire families of halogenated acetates and vinyl derivatives. Talking to older colleagues and reading historic journals, I found that people saw in this molecule a way to reach compounds not easily made with older acetates or simple vinyl esters.
Breaking Down What It Is
Vinyl chloroacetate is a clear liquid under room temperature, sharp to the nose and tough on skin, yet prized for the reactivity it brings to the benchtop. The backbone carries both a vinyl group—always ready to grab onto other molecules in a chain reaction—and a chloroacetate arm, which shouts for attention with its electronegative chlorine neighbor. It isn’t a household item. Instead, it shows up behind the scenes in research, manufacturing, and specialty synthesis. Known by names like 2-chloroacetic acid vinyl ester, this chemical’s presence in a bottle usually signals that someone’s working on something ambitious—a new polymer, a pharmaceutical building block, or a specialty coating.
Parsing Physical & Chemical Traits
Vinyl chloroacetate doesn’t hide its volatility. Once the cap twists off, vapors waft up, making fume hoods not just a box to check but a necessity. This colorless, flammable liquid will react quickly with nucleophiles, bringing to mind more famous vinyl esters but with a fluoride snap that gives it extra punch. The boiling point sits lower than many related esters, so distilling and storing this compound calls for careful attention to pressure and temperature. Water makes it break down, hydrolyzing the ester linkage quicker than some chemists expect, which means craftspeople who work with it take pains to keep things dry and containers tightly closed.
Going Under the Hood: Specifications & Labeling
In an industry where trace impurities can turn a whole batch sideways, vinyl chloroacetate’s technical benchmarks usually center on purity levels—rarely below 98 percent for fine syntheses, purity confirmed by gas chromatography. A sharp odor alerts the prepared, but proper labeling always includes hazard statements about toxicity, fire risk, and the corrosive nature that comes from its chloroacetate group. Consistent quality from commercial suppliers rests on mass spectrometry and NMR confirmation, so researchers know exactly what they’re pouring into the beaker. Specialized transport—amber bottles, careful sealing—follows guidance stemming from both regulatory statutes and the collective headache of too many ruined samples.
How It Gets Made
The hunt for an efficient synthesis of vinyl chloroacetate had chemists drawing on old-school reactions as well as newer catalytic routes. Most methods still lean on reaction between chloroacetyl chloride and vinyl alcohol. Trouble is, vinyl alcohol doesn’t fancy hanging around as a reagent, so clever chemists often work with the more stable vinyl acetate, swapping acyl groups using transesterification, or they generate vinyl alcohol in situ under the right conditions. Chlorinated solvents, precise temperature control, and strong acid scavengers play their roles in keeping the reaction running clean. Yields can swing depending on how tight a grip the chemist keeps on moisture and on how enthusiastically side reactions jump in.
Waking Up the Molecule: Reactions & Modifications
Few molecules offer two points for action like vinyl chloroacetate. The vinyl group can snap into polymerization reactions, laying the groundwork for new materials with tailored properties. On the other hand, the chloroacetate moiety provides a handle for further substitution: nucleophiles like amines or thiols attack at the alpha carbon, paving the way for specialty intermediates that pharmacists, agricultural chemists, or material scientists need. In practice, this means researchers can take vinyl chloroacetate and push it in several directions—toward reactive polymers for the electronics industry, or use the ester’s lability to build more complex organic frameworks.
What’s in a Name?
Vinyl chloroacetate pops up in lab notebooks as 2-chloroacetic acid vinyl ester, chloroacetoxyethylene, or simply VCA. The International Union of Pure and Applied Chemistry (IUPAC) codified the name as vinyl 2-chloroacetate, and databases keep it searchable under these and the ChEBI and CAS registry entries. Over the years, it picked up trade and research names for short—none ever as sticky in the public consciousness as common solvents or flavors, but easily recognized in synthesis communities.
Keeping It Safe: Handling and Standards
Safety advice about vinyl chloroacetate comes from lived experience as much as it does from regulatory protocols. Few in the lab forget their first splash—skin goes red fast and the air grows harsh if even a drop escapes the bottle. Direct contact brings real, lasting irritation and inhalation leads to coughing, sometimes nausea. Everyone who works with this reagent fixes their goggles tight, gloves snug, and makes sure the fume hood runs strong. Strict fire rules apply thanks to low flash point. Written rules from OSHA, REACH, or local safety officers may fill pages, but the culture on the ground comes from senior chemists passing on stories of ruined glassware and unexpected reactions. MSDS sheets hammer home both long-term toxicity concerns and acute risks, warning of liver and kidney impact in case of chronic exposure.
Where It Makes a Difference: Applications
Vinyl chloroacetate stays almost invisible to consumers, yet it turns up in the supply chains of medicine, agrochemicals, and specialty materials. I’ve seen it used in the lab to create intermediates for pain relievers, and colleagues in paints talk about it as a backbone for specialty polymers resistant to water and solvents. The molecule wins fans among polymer chemists aiming for unique electrical or adhesive properties—something about that blend of reactivity and modifiability that solves problems no simpler ester can manage. For labs scaling up small molecules, it acts as a versatile step on the path to much larger, more valuable final products.
Digging Into Research & Development
Vinyl chloroacetate finds itself at the center of several research paths. Polymer science looks to tweak the vinyl group’s reactivity to improve coatings or adhesives, while medicinal chemists hunt for derivatives that can slot into active pharmaceutical ingredients or drug delivery systems. I recall some excitement in academic circles about grafting vinyl chloroacetate units onto biodegradable polymers, tweaking water solubility, or tuning release rates for actives in controlled delivery. Green chemistry initiatives try to cut out chlorinated solvents or reduce reliance on harsh acids, aiming for more sustainable syntheses without throwing off the yield or purity standards that drive the industry. Few projects make national news, but persistent grad students and inventive bench chemists keep nudging forward what this molecule can do.
Toxicity Research: Health, Hazards, and Honest Limits
Talk with toxicologists or thumb through the literature, and you’ll hear stories about the risks that chloroacetates introduce. Laboratory exposure often means irritation, but animal studies point to longer-term risks to the liver, kidneys, and nervous system from repeat exposure—mainly by inhalation or accidental ingestion. Regulatory bodies set tight exposure limits, both in air and in workplace contamination, not only because of the molecule itself but because breakdown products can carry their own set of hazards. In legal and industrial circles, liability and waste disposal become real issues, especially in bulk chemical plants. Lab safety training still emphasizes fast cleanup, neutralization, and thorough ventilation as a guardrail against hazard escalation.
Looking Forward: Possibilities on the Horizon
Vinyl chloroacetate rests at an awkward crossroads: indispensable for some cutting-edge chemistry, but hobbled by toxicity concerns and demanding production needs. The future likely includes greener and safer variants, perhaps by swapping out the chlorine for less hazardous groups or engineering catalytic processes to replace harsh reagents. Automation and improved ventilation technology should bring safer workflows. I see the strongest hope in research that links new uses in advanced polymers, drug molecules, or functional coatings to process improvements that keep workers and the environment safer. Students choosing their projects in organic synthesis or materials science won’t escape vinyl chloroacetate’s legacy anytime soon, but with each step forward in safety, utility, and sustainability, the molecule stands less as a hazard and more as a key to next-generation chemistry.
What is Vinyl Chloroacetate used for?
Where Vinyl Chloroacetate Shows Up
Many people never hear about vinyl chloroacetate, even though this compound shapes plenty of the everyday environment. Factories use it mostly to form specialty plastics and resins. These often turn up in coatings, adhesives, or packaging films. In my first summer job at a plastics plant, nobody talked much about the chemistry, but almost everyone knew the ghostly, acrid smell drifting from the reactor when a new batch ran. The heart of it was always an odd clear liquid—vinyl chloroacetate—going in at just the right time.
This stuff acts as a kind of bridge. Chemists blend it with other ingredients to tweak the softness, stickiness, or strength of their finished plastics. For instance, flexible packaging that feels silky smooth on the outside? Some of those films started with a few drops of vinyl chloroacetate in the mix. Paint chemists like it too, especially for acrylic coatings that need a balance between toughness and flexibility. By hooking vinyl chloroacetate onto the polymer backbone, companies fine-tune materials to stand up to wear or spill for longer.
Pipework, Plant Smells, and Protective Equipment
Factory workers who deal with compounds like this keep safety at the front of their minds. Vinyl chloroacetate does not belong in the open air. Breathing it in, even for a short while, stings the nose and throat. Chronic exposure causes much bigger trouble. I remember scrubbing valves with gloves so thick I could hardly grip a wrench, still feeling nervous about pinhole leaks. Good air flow and serious maintenance cut down risks, but one slip-up could put the whole team on edge.
Supply Chain Grows in Complexity
Vinyl chloroacetate’s usefulness in specialty chemicals has made it a steady item for many chemical plants across the globe. Yet, anything tied to the plastics pipeline faces change. Tighter environmental rules in Europe and North America have shaken up old business routines. Factories now need to track emissions and waste flows down to the last kilogram. Plant managers scramble for processes that cut down fumes, recycle leftovers, or switch out toxic chemicals for cleaner ones. Major customers—like car or electronics companies—ask tough questions about every ingredient. They want proof a raw material does not hide big social or environmental risks.
Why Oversight and Innovation Matter
People building the future of chemicals keep pushing for smarter synthesis routes. New lab research looks at swapping some chlorine chemistry out for less hazardous building blocks, sometimes using plant-based sources. As countries add stricter workplace air rules and green chemistry grants, labs get closer to scaling up alternative ingredients. I’ve seen lab techs spend months banging their heads over how to lessen waste. Every new success—in less wasteful synthesis or better recycling—comes from stubborn day-to-day effort that rarely makes the headlines.
Companies and regulators stay busy checking that materials like vinyl chloroacetate don’t end up causing silent harm. In a fast-evolving market, the safest and most responsible producers will keep the trust of major buyers. The industry’s past left societies cleaning up soil and water long after the plants closed. The future, built on practical chemistry and sound oversight, can offer all of us healthier materials and a safer workplace.
What are the safety precautions when handling Vinyl Chloroacetate?
Working with a Substance You Can’t Treat Casually
I remember my first experience stepping into a chemical storage area. The sharp scent of solvents came right through the door and the warning symbols plastered everywhere. It became obvious that some substances demand a lot of respect—vinyl chloroacetate lands squarely in that category. In industrial labs and manufacturing, taking shortcuts with safety can go wrong quickly, especially with reactive chemicals. Vinyl chloroacetate isn’t something you just uncap and pour without thought. It has the potential for skin burns, serious inhalation risks, and can ignite if handled carelessly.
Personal Protective Gear Isn't Optional
Labs and plants dealing with vinyl chloroacetate don’t leave PPE to chance. Direct skin contact burns and leaves long-lasting damage. I’ve seen hospital records from chemical incidents—gloves, splash goggles, even full face shields are non-negotiable. Nitrile gloves, not latex, help keep this stuff from getting through. Long-sleeve lab coats and flame-resistant coveralls form a basic barrier. Respirators matter as well, given that fumes off this liquid can irritate the respiratory system and even cause more severe reactions. Spending on good-quality protection beats any medical bill or cleanup job after an accident.
Good Ventilation Cuts Down Risks
One of the obvious things about vinyl chloroacetate is its volatility. In spaces with poor ventilation, vapors start to build up, forming an invisible danger zone. This isn’t only a health hazard—there’s a real fire risk, since vinyl chloroacetate lights up easier than you might expect. Good air movement, capture hoods, and local exhaust take the threat down several notches. I’ve toured older facilities where vents whir constantly near storage drums, where any chemical fume gets swept away before someone even notices it. Skimping here just increases the long-term cost and stress.
Spill Response: You Can’t Wing It
Maybe the worst thing I’ve seen is the panic that follows a chemical spill in an unprepared shop. Spills of vinyl chloroacetate release noxious fumes and can stick around in places you wouldn’t predict. Fast action with absorbent pads, the right neutralizers, and non-sparking cleanup tools heads off bigger trouble. Workers need clear training—what to grab, where to move waste, how to protect anyone in the spill zone. Emergency showers and eyewash stations right by the work area help, especially if someone gets a splash. Every cleanup should end with a double-check: skin, clothes, gear—nobody wants this stuff hitchhiking home.
Storage and Handling Make the Difference
It doesn’t take long for careless storage to catch up with a shop. Vinyl chloroacetate degrades in sunlight and reacts with a few common substances. Sealed containers, out of direct light, in temperature-controlled spaces, keep it stable. Clear labeling, strong shelving, and keeping strong acids and bases far from vinyl chloroacetate keep accidents off the books. Storing less than you actually need for work lowers risk. In some places, only trained and trusted operators get access, to cut down the odds of a rookie mistake.
Training and Communication Save Lives
The strongest safety systems start with people. In shops or labs that handled vinyl chloroacetate safely, I saw briefings before every shift, yellowed safety sheets pinned up next to supplies, and story-swapping about past close calls. Safety culture isn’t a cliché—people remember lessons when they hear about real incidents. Regular drills and spot inspections help everyone keep their guard up. It’s worth repeating that listening to workers who spot odd smells or old leaks keeps everyone a few steps ahead of danger.
What is the chemical formula of Vinyl Chloroacetate?
The Chemical Formula: C4H5ClO2
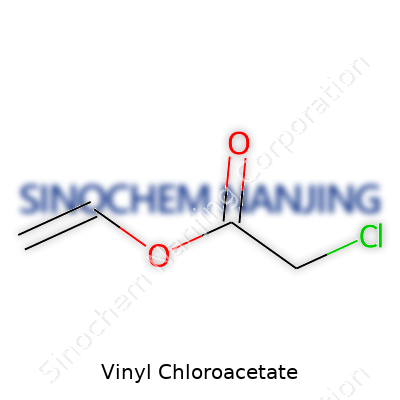
Vinyl chloroacetate belongs to a group of chemicals often used as building blocks in organic synthesis. Its chemical formula, C4H5ClO2, spells out a story most folks never see. You’ve got four carbon atoms, five hydrogen atoms, a single chlorine, and two oxygens in every molecule. A trained eye will spot the vinyl group (–CH=CH2) attached to a chloroacetate group, drawing interest not just from chemists but from industries that depend on unique monomers and intermediates.
The Real-World Uses That Drive Demand
Vinyl chloroacetate rarely gets the splashy headlines you see with more common chemicals, though it plays a quiet but important role. Used in specialty polymer production, its impact reaches paints, coatings, adhesives, and, sometimes, fine-tuned pharmaceuticals. Many years in the chemical industry have shown that every monomer like this serves as a pivot point: molecules built with care, destined for applications that call for just the right kick of reactivity. For example, producers searching for improved flexibility or chemical resistance in their polymers keep an eye out for raw materials like vinyl chloroacetate, which provide a way to adjust key performance properties.
The importance of this chemical stretches beyond recipes in a lab. Producers choosing between raw materials weigh costs, health and safety features, and downstream risks. Vinyl chloroacetate brings a touch of chlorine, making it more reactive and opening doors for other functional group substitutions. At the same time, this added chemical muscle brings the need for greater safety, as chloro derivatives often come with toxicity warnings and tighter handling protocols.
Health, Safety, and Responsible Manufacturing
Handling vinyl chloroacetate tells a story about respect for safety. A slip-up in ventilation or an oversight in protective equipment could have serious consequences. Chlorine in this molecule, combined with the acetate group, means routine exposure isn’t something anyone wants to risk. Acute risks range from respiratory irritation to organ toxicity. Long-term or repeated exposures force health and environmental officials to pay even closer attention.
Workers on the plant floor must rely on robust training, material safety data sheets, and real-time exposure monitoring. It’s not just about dodging immediate accidents. Environmental stewardship kicks in, since improper disposal can send chlorinated compounds into groundwater or rivers, setting off ripple effects down the line. Prevention matters more than cleanup ever will.
Future Outlook and Safer Practices
Safer chemical handling and responsible sourcing make a difference. Researchers have put years into finding safer analogues or greener production methods. The job never quite finishes. Each safety incident, no matter how minor, prompts an internal review at most responsible companies, nudging teams to rethink processes or invest in better containment and air handling. Companies that want to stay competitive and ethical have learned that investing in engineering controls and continuous education pays off.
Vinyl chloroacetate will keep finding its way into high-value manufacturing, but so will attention to workplace health and environmental protection. These priorities lift up everyone involved—workers, neighbors, and customers—by reducing risk and boosting trust in chemical technologies that help shape modern life.
How should Vinyl Chloroacetate be stored?
Understanding the Concerns
Vinyl chloroacetate looks like a simple chemical on paper, but shelves lined with safety bulletins tell a different story. A lot of people overlook the hazards until something goes wrong. My time around research labs taught me lessons best learned from someone else's mistakes, not your own. Vinyl chloroacetate brings real dangers, including toxic fumes if it escapes and risky reactions if it finds the wrong companion. A chemical like that won’t forgive carelessness.
Temperature, Ventilation, and Proper Containers
Safe storage practices never just mean “putting it somewhere.” Temperature plays a big role. The best choice is a cool, well-ventilated spot, away from direct sunlight or any heat source. That keeps decomposition in check and limits the risk of the vapors building up. Regular room temperature can work, though some experts go for lower temps if the facility allows.
Containers matter just as much as the room climate. Old soda bottles or scratched-up plastic jugs don’t belong in the same sentence as vinyl chloroacetate. Thick amber glass bottles with firm, chemical-resistant seals have done the job for generations. They don’t react with the compound, they block out UV rays, and tight lids stop leaks. I’ve watched lab techs double up with a secondary container, just in case. It’s worth the extra effort to box it up, set it in a tray, and label it so nobody mixes up the bottles.
Avoiding Unsafe Combos
Storing vinyl chloroacetate next to acids, bases, or anything reactive never brought good results. Separation helps prevent accidental mixtures that can kick up fumes or worse. In one facility, I saw color-coded cabinets: red for flammables, yellow for corrosives, blue for toxics and volatiles like vinyl chloroacetate. It sounds old-school, but those simple rules kept hundreds of bottles in the right place year after year.
Clear Labelling and Record Keeping
Labeling saves more lives than people admit. If it’s not crystal clear what's inside and where it should go, mistakes happen. A bold label detailing what’s in the container, when it was received, and when to dispose of it tells newcomers or the next shift what they’re dealing with. Logs or digital tracking add an extra layer, so no batch overstays or gets forgotten during lab moves.
Access and Training
Not everyone should have unlimited access. Only folks who understand the material and have been trained in safe handling should hold the key. Some companies use sign-in sheets to know who took what, when. I remember getting grilled in orientations and annual refreshers—sometimes annoying, but the reminders stick. It’s even better if everyone uses gloves, goggles, and lab coats without even thinking twice. Small routines keep small accidents from turning into headlines.
Disposal Practices
Holding onto outdated stock or leaks for too long invites more trouble. Regular audits and responsible disposal schedules cut down on risks. Partnering with professional hazardous waste handlers keeps any leftover vinyl chloroacetate out of landfills and waterways. Recycling isn’t an option here, so nobody tries to play DIY hero. Following the local and national rules isn’t just for paperwork; it shuts down the worst hazards before they start.
Building a Culture of Safety
From what I’ve learned on the ground, storing vinyl chloroacetate safely calls for more than locked doors. Listening to the stories of veterans and paying attention to the small print in safety data sheets pays off. A routine of careful storage, separation, labeling, regular disposal, and constant vigilance builds trust in every hand that opens that door. Some lessons feel obvious until someone skips a step, and nobody wants to be that name in the safety report.
What are the physical properties of Vinyl Chloroacetate?
Touch and Smell: What Sets It Apart
Vinyl chloroacetate often ends up as a pale yellow to colorless liquid, and to anyone who encounters it, there’s a sharp, pungent odor. The scent alone gives away its reactive nature. Workers in the chemical industry quickly learn to respect its power—vinyl chloroacetate lingers in the air and signals that proper handling gear needs to come out before moving forward. At room temperature, it remains a liquid, a feature that impacts storage, transport, and the way professionals handle it day-to-day.
How It Behaves: Boiling, Melting, and Stability
The boiling point of vinyl chloroacetate hovers around 118-120°C. This isn’t all that high, so it can vaporize faster than many might expect, especially in warm or poorly ventilated areas. I remember seeing how quickly it could disappear from an open beaker if left unchecked. On the cold side, it freezes up at -36°C, so unless you’re working in subzero conditions, you’re dealing with a liquid.
I’ve always found it striking how some chemicals seem to “want” to react, and vinyl chloroacetate fits this mold. It reacts with water—slowly at first, but unless a chemist watches over every drop, hydrolysis produces dark, pungent byproducts. Stability is always under threat if exposure to moisture creeps in. Anyone around vinyl chloroacetate respects the hazards, and that awareness goes beyond the textbook; I saw a fume hood corrode around the edges due to persistent contact. Simple mistakes can turn daily work into a clean-up headache.
Weight and Density: The Numbers Matter
With a density of around 1.22–1.25 grams per cubic centimeter, it sinks in water, which forces you to rethink spill clean-ups if an accident ever happens. Water floats over it, and that increases the risk of spreading contamination across benches and floors. I learned to keep absorbent pads and spill kits within arm’s reach because a simple leak could spread fast and far.
The molecular weight lands near 122.5 g/mol. That doesn’t look like much at a glance, but it shapes how vinyl chloroacetate acts in the body and in the environment. Breathing the vapors, even in trace amounts, means introducing something with real punch into your system. Industry guidelines and OSHA rules reflect the tricky balance between handling necessary materials and protecting people. None of this is theoretical—my own experience with minor exposure brought out headaches and dizziness, making safety feel personal instead of procedural.
Volatility and Solubility: Effects on Use and Safety
Vinyl chloroacetate is volatile. Spillage sends it into the air quickly, and you can pick up that bite in the nostrils long before you see the puddle. In water, vinyl chloroacetate proves only slightly soluble, which explains puddles separating out and needing surfactants or solvents to mop up. With organic solvents like acetone or ether, it mixes much better, making them the go-to choices for cleanup or reaction work. Violating best practices often leads to more than regulatory trouble; I’ve seen how minor carelessness can mean evacuations or ruined equipment.
Being aware of these traits influences every step of storage and use. Companies invest in sealed containers, regular sensor checks, and robust ventilation not out of bureaucratic fear but from hard-learned lessons. The risks aren’t exaggerated. Meeting safety standards means respecting both the invisible and visible sides of vinyl chloroacetate’s physical properties.
| Names | |
| Preferred IUPAC name | 2-chloroacrylic acid 2-oxoethyl ester |
| Other names |
Chloroacetic acid vinyl ester Vinyl 2-chloroacetate |
| Pronunciation | /ˌvaɪ.nɪl klɔːr.oʊ.əˈsiː.teɪt/ |
| Identifiers | |
| CAS Number | ```string 5274-97-3 ``` |
| 3D model (JSmol) | `load =C(=O)OCCCl` |
| Beilstein Reference | 1718736 |
| ChEBI | CHEBI:132962 |
| ChEMBL | CHEMBL2104067 |
| ChemSpider | 17725 |
| DrugBank | DB08707 |
| ECHA InfoCard | 14ebdcb7-301c-4e9d-9a9e-978369fd3219 |
| EC Number | 209-617-9 |
| Gmelin Reference | Gmelin 7926 |
| KEGG | C18606 |
| MeSH | D014765 |
| PubChem CID | 11192 |
| RTECS number | AB8925000 |
| UNII | F49K5G2666 |
| UN number | UN1304 |
| CompTox Dashboard (EPA) | DTXSID4032909 |
| Properties | |
| Chemical formula | C4H5ClO2 |
| Molar mass | 150.54 g/mol |
| Appearance | Colorless liquid |
| Odor | Pleasant odor |
| Density | 1.193 g/cm³ |
| Solubility in water | slightly soluble |
| log P | 0.8 |
| Vapor pressure | 7 mmHg (20 °C) |
| Acidity (pKa) | 13.05 |
| Magnetic susceptibility (χ) | -56.8×10⁻⁶ cm³/mol |
| Refractive index (nD) | 1.445 |
| Viscosity | 12 mPa·s (20 °C) |
| Dipole moment | 2.45 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 325.7 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -186.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -947 kJ/mol |
| Hazards | |
| GHS labelling | GHS02, GHS07, GHS08 |
| Pictograms | GHS02,GHS06 |
| Signal word | Danger |
| Hazard statements | H226, H301, H311, H315, H319, H331, H335, H341, H351 |
| Precautionary statements | P210, P261, P280, P301+P310, P303+P361+P353, P305+P351+P338, P405, P501 |
| NFPA 704 (fire diamond) | 2-3-2-W |
| Flash point | 29 °C (closed cup) |
| Autoignition temperature | 285 °C |
| Explosive limits | Explosive limits: 5.6–15.6% |
| Lethal dose or concentration | LD50 (oral, rat): 88 mg/kg |
| LD50 (median dose) | LD50 (oral, rat): 610 mg/kg |
| NIOSH | DA 6460000 |
| PEL (Permissible) | PEL (Permissible Exposure Limit) for Vinyl Chloroacetate: 1 ppm (4 mg/m³) as an 8-hour TWA (OSHA) |
| REL (Recommended) | 1 ppm |
| IDLH (Immediate danger) | IDLH: 5 ppm |
| Related compounds | |
| Related compounds |
Chloroacetic acid Vinyl acetate Methyl chloroacetate Ethyl chloroacetate Chloroacetyl chloride Dichloroacetic acid Vinyl chloride |

