Vanillin: Beyond a Popular Flavor
Historical Development
Vanillin came onto the scene much earlier than most people might imagine. Long before lab coats and high-tech equipment, vanilla beans provided the prized essence essential to desserts, perfumes, and even medicinal remedies. People spent years extracting this subtle flavor the slow way—curing beans under the sun, sweating, and fermenting them. In the late 1800s, a break arrived when chemists discovered how to isolate vanillin crystals from vanilla pods. Scientists soon started synthesizing vanillin in the lab, leading to commercial production. Over time, demand far outpaced supply from natural sources. As production methods improved, synthetic vanillin replaced natural extraction for most of the world’s vanilla-flavored products, bridging the gap between affordability and accessibility.
Product Overview
Look at the label on most baked goods, chocolates, or beverages and vanillin shows up. Vanillin, a compound with a warm, sweet, creamy aroma, gives vanilla its characteristic fragrance and taste. Although it can be found in natural vanilla bean pods, the majority available today comes from synthetic processes. In industry, vanillin appears as white to pale yellow needles or powder, packing a concentrated punch in tiny amounts. This material moves in bulk between food companies, cosmetic labs, perfumeries, and even pharmaceutical plants. Its reliability, consistency, and low cost have made it a mainstay for flavoring and scent anywhere a hint of vanilla makes a difference.
Physical & Chemical Properties
Vanillin’s physical profile helps explain why companies lean on it. The compound forms needle-shaped crystals that dissolve well in alcohol, ether, or hot water, though they struggle in cold water. With a melting point around 81–83°C, vanillin stays solid at room temperature, making it easy to ship and store. Chemically, it’s a phenolic aldehyde, with the formula C8H8O3, and includes a distinctive methoxy group that brings its aroma to life. This structure allows for predictable reactions with other ingredients—important in foods and fragrances alike—and helps maintain stability over time. Such robustness keeps the flavor or scent from fading too quickly, saving products from losing their appeal on shelves.
Technical Specifications & Labeling
Manufacturers must meet specific purity standards to market vanillin for food or cosmetic use. Food-grade vanillin usually tests above 99% purity and shows only trace amounts of related impurities. Each batch undergoes checks for melting point, solubility, appearance, and sometimes residual solvent content if solvents appear during processing. Labeling rules differ globally but nearly always require stating the nature of the vanillin—natural, synthetic, or biotechnological. Natural vanillin, extracted from vanilla pods, gets a much higher price tag and may carry “natural flavor” status, while the synthetic kind must state its artificial origin. Some producers now turn to biotechnological fermentation to answer the call for “natural” vanillin, but regulatory agencies scrutinize source materials and processing methods before granting the coveted label.
Preparation Method
Producers follow a few main paths to vanillin. Extraction from vanilla beans remains incredibly costly and yields only tiny amounts. Most commercial vanillin starts its journey from guaiacol (a petrochemical derivative) or lignin (a byproduct from paper production). The guaiacol method starts by methylating catechol, then oxidizing the resulting product to form vanillin. The lignin route depends on alkaline oxidation, breaking the complex lignin molecule down to the vanillin structure. Fermentation methods use engineered microbes to transform ferulic acid into vanillin. Each route has its merits and challenges—petrochemical sources raise sustainability concerns, while lignin and fermentation need clever engineering to scale affordably without quality variation.
Chemical Reactions & Modifications
Vanillin’s chemical backbone offers opportunities for creating derivatives and modified products. Common reactions include methylation to make ethyl vanillin—another potent flavor agent—or condensation with ammonia to craft Schiff bases for research. In perfumery, companies sometimes manipulate vanillin’s aldehyde group, extending its sensory profile or pairing it with other fragrance chemicals. The presence of both an aldehyde and a phenolic group encourages a long list of organic reactions, supporting both flavor innovation and the development of specialty chemicals. This chemical flexibility allows research teams to build novel blends or functional additives from a familiar base, often without needing to redesign whole production lines.
Synonyms & Product Names
Vanillin moves through the marketplace under many names. Some call it 4-hydroxy-3-methoxybenzaldehyde or methyl vanillin. In commercial catalogs, it often appears with identifiers like “artificial vanilla flavor,” or less often, “vanilla crystal.” Regulatory documents refer to it by CAS Number 121-33-5. These names help connect buyers in food, pharma, or fragrance sectors to the same core compound, although precise branding may highlight natural or synthetic origins. Some suppliers clarify with descriptors such as “fermentation vanillin” to attract customers seeking products aligned with current consumer preferences or labeling rules.
Safety & Operational Standards
Food producers and chemical processors who handle vanillin put safety at the forefront. Vanillin represents a low-exposure risk to humans, approved by food authorities globally at standard usage rates. Safety data sheets warn against inhaling vanillin powders or allowing it into the eyes, as irritation may occur, just as with many fine chemical dusts. Processing plants rely on ventilation, personal protective equipment, and safe handling protocols to avoid dust buildup and accidental spills. Facilities storing or transporting vanillin keep it in cool, dry conditions away from strong oxidizers and acids, maintaining stable quality while protecting workers. Quality control labs routinely analyze vanillin concentrations in finished goods, verifying compliance with food safety regulations and customer requirements.
Application Area
Everywhere people want the taste or aroma of vanilla, vanillin finds a foothold. Bakeries scale up the volume in cakes and cookies, creating the creamy note familiar to anyone who’s ever had a scoop of vanilla ice cream. Soft drink and dairy companies depend on vanillin for consistent flavor across global markets. Perfume houses rely on vanillin’s sweet, inviting scent to add depth to both high-end and mass-market fragrances. Pharmaceuticals sometimes feature vanillin as a masking agent, helping bitter pills go down easier. Even in the world of animal feed, small doses of vanillin increase palatability for livestock, driving improved nutritional outcomes. This versatility reflects not just vanillin’s sensory impact, but also its stability during production, making it trusted not only by chefs but also by chemical engineers.
Research & Development
The search for greener, more sustainable vanillin anchors much of the current research. Biotechnologists develop new microbial strains capable of transforming low-value agricultural byproducts into vanillin, aiming to both boost availability and lower environmental impact compared to petrochemical routes. Chemists explore new catalysts and reaction conditions for improved yields in synthetic processes, targeting reduced waste or minimal use of hazardous reagents. Sensory scientists work to maximize the flavor fidelity of bio-vanillin, trying to match not just chemical purity but also the nuanced bouquet of natural vanilla. Collaborations between academia and industry deliver advances in extraction, purification, and process safety, giving rise to standards that drive continuous improvement—not only in cost but also in sustainability, traceability, and trace impurity profiling.
Toxicity Research
Vanillin holds an established reputation for safety among food regulators. Past and ongoing studies assess short- and long-term toxicity in both humans and animals, documenting metabolic pathways and potential allergens. Some research points to mild cytotoxicity at massive concentrations, but the quantities required far exceed what anyone finds in a normal diet or perfumed environment. Allergenicity remains rare, though occasional reports describe contact dermatitis from high concentrations or industrial exposure. Food authorities maintain recommended limits, balancing public health and industrial utility. Constant review of new data ensures that any emerging risks, particularly for sensitive populations, receive fresh scrutiny.
Future Prospects
A changing regulatory landscape and shifting public values continue to shape vanillin’s market and research priorities. The food world asks for clearer labeling and increasingly pushes for “natural” ingredients, responding to shoppers who read every label. Bio-based vanillin brings opportunities not just for marketing but also for cost and environmental advantages as processes scale up. Synthetic vanillin will probably continue to dominate industrial uses for years, thanks to proven processes and low economics, but green chemistry and traceable supply chains stand to differentiate premium products. Research into novel applications—such as in pharmaceuticals, personal care, or even as a functional additive for food preservation—will set the stage for future growth. Teams that bridge chemistry, biology, sensory evaluation, and sustainable business models will likely lead the way as the next chapter for vanillin unfolds.
What is Vanillin used for?
The Familiar Scent of Vanilla
Most folks recognize vanillin as the flavor that makes vanilla ice cream, cakes, and cookies taste the way they do. My own earliest memory of this came from baking with my mom—she’d add a splash of extract and the whole kitchen filled with this sweet, almost creamy aroma. Yet, only a tiny bit of the vanilla flavor we find in food actually comes from vanilla beans. In reality, vanillin, the key element, almost always comes from a lab these days. That change didn’t happen for no reason.
Real Vanilla—Rare and Pricey
Vanilla beans come from orchids that only grow in specific climates and need pollination by hand. Farmers in Madagascar and a few regions in Indonesia do the heavy lifting, but every bad season, the prices shoot through the roof. Vanilla now ranks among the world’s priciest spices by weight. That crunch on supply made folks look for a steady alternative. Scientists figured out how to make vanillin from wood pulp, guaiacol, or even recycled rice bran. It may not sound romantic, but it keeps vanilla goods in reach for everyone, not just the fancy bakeries.
Beyond Desserts
It’s not just about cookies or chocolate bars. Vanillin shows up in places you probably wouldn’t expect. Some pharmaceuticals—like cough syrups or chewable tablets—use it to mask bitter or metallic flavors. Fragrance companies add it to perfumes for warmth and sweetness. Even in animal feed, vanillin helps animals eat more, which boosts growth rates. In the world of chemicals, vanillin serves as a starting material for making some specialty drugs and other fine chemicals.
Safety and Sustainability
Vanillin’s safety record remains strong. The U.S. Food and Drug Administration considers both natural and synthetic vanillin as safe for use in food. Many companies that make synthetic vanillin now use sustainable sources, sometimes converting waste from rice or wood into this flavor molecule. That ticks two boxes—lower costs and less need to chop down rare orchids or threaten biodiversity in the tropics.
Some Challenges Remain
Still, there’s debate about how much synthetic vanillin can truly match the complex bouquet from real vanilla beans. Beans hold hundreds of trace compounds, not just vanillin. Give someone real vanilla yogurt next to a synthetic one, and the difference usually jumps out. Food manufacturers keep tinkering with blends of natural and synthetic to bring that signature flavor closer to “the real thing.”
What’s Next?
As a food lover and occasional home cook, I try to read labels, especially if vanilla is a central ingredient. Pure vanilla extract costs more, but you taste the difference in custards and simple recipes. Most folks buying snacks at the store look for a consistent flavor, and vanillin delivers exactly that.
In the end, vanillin has freed desserts and countless products from the unpredictability of real vanilla farming. As demand rises, pressure grows to find greener ways to make it and, maybe someday, to engineer even richer vanilla profiles using sustainable biotech. For now, without vanillin, most kitchen shelves and supermarket aisles would look—and smell—a lot less inviting.
Is Vanillin natural or synthetic?
Understanding Where Vanillin Comes From
Walk down any grocery store aisle and you’ll spot vanilla flavor everywhere—ice cream, cookies, even protein bars. Most of that sweet smell isn’t from vanilla beans. It’s vanillin, the main compound responsible for that familiar aroma. People often ask if vanillin is natural or synthetic, and the answer isn’t black and white.
The Vanilla Bean Reality
Real vanilla comes from the seed pods of the orchid Vanilla planifolia. Growing and harvesting these beans takes a lot of work. The vines need tropical climates, pollination has to be done by hand, and each pod gets cured for months. No one rushes this process, which is why vanilla beans fetch some of the highest prices among spices in the grocery store. Pure vanilla extract—made by soaking sliced beans in alcohol—captures all those deep, subtle flavors. It tastes less sharp and slightly smokier than what’s in mass-market vanilla treats. Extract made this way earns its “natural” label.
Making Vanillin in the Lab
The food industry didn’t set out to fool anyone with synthetic vanillin. They needed to keep up with demand. About 99% of vanillin on the market gets made in labs. The original source for lab-made vanillin was actually a byproduct of the paper industry—lignin from wood pulp. More commonly today, vanillin’s built from guaiacol, a compound derived from petrochemicals. Chemists map out the identical structure, so the molecule matches what’s in a vanilla bean. It’s precise, efficient, and a whole lot cheaper.
Natural, Synthetic, or Something In-Between?
Here’s where things turn fuzzy. Some vanillin is labeled “natural,” thanks to creative sourcing. One example is making vanillin from ferulic acid, which lives in rice bran or wheat. Enzymes convert this acid into vanillin. The product doesn’t touch a vanilla bean, yet because it starts with a recognizable plant source, the law allows companies to call it “natural.” In Europe and the United States, these technicalities matter, but the result smells and tastes the same to most people.
The Big Picture for Health and Flavor
As someone who bakes on weekends, the difference jumps out in a homemade custard. Natural vanilla extract brings out round and earthy flavors, while synthetic vanillin delivers that familiar top note that people connect with milkshakes and cereal. Safety isn’t much of a concern—synthetic vanillin has been used safely for decades, with no evidence of harm at the low doses found in foods. But pure vanilla packs over 200 aromatic compounds, defining “depth of flavor.” Vanillin alone is a shortcut.
The Road Ahead
Consumers are more aware and care about transparency. Some companies turn to biotechnology, using yeast or bacteria to “brew” vanillin from sugar. This approach sidesteps petrochemicals, using fermentation to keep things closer to nature. The flavor and scent match the vanilla orchid, but with less strain on forests in Madagascar.
Cooks and eaters decide what matters—cost, sustainability, or depth of flavor. Food labels matter, but so does honesty. Knowing what lands in your food lets you make better choices, whether you crave pure vanilla or reach for something affordable and consistent. The story of vanillin is a reminder that behind every sweet treat, science shapes the flavor as much as the farmer.
Is Vanillin safe to consume?
The Story Behind the Sweet Scent
Open an ice cream carton, bake a batch of cookies, or pour a bottle of soda, and chances are you’ll run into vanillin. Most people recognize the warm aroma of vanilla—even if they rarely ask where the flavor comes from. The truth: natural vanilla extract comes from a tropical orchid. What lines grocery shelves far more often is vanillin, the lab-made cousin.
The Safety Debate
I remember touring a food science lab years ago, standing beside a white-coated technician as he explained how vanillin gets synthesized. “It’s the same molecule that’s in the vanilla bean,” he said. “The only thing different is how we make it.” Hearing that, my first reaction mirrored what most people wonder: If it’s lab-made, does that mean it’s safe?
Over a century of use answers part of that question. Food safety agencies worldwide—including the FDA in the U.S. and EFSA in Europe—review vanillin’s safety based on real-world evidence, not just short-term animal tests. These regulators let manufacturers use vanillin as a flavor because, at normal levels found in food, it hasn’t shown any serious risk. Studies have checked everything from allergic reactions to cancer risk. At the amounts used for flavoring, toxicity concerns never materialized.
The Synthetic vs. Natural Conversation
Some get nervous about synthetic flavors. There’s a belief that natural always trumps manmade. After hearing a neighbor’s strong opinions at a community potluck, I dug into the research for myself. Chemically, vanillin from a vanilla bean and vanillin from a factory share the same structure. To my surprise, the body can’t tell the difference. There’s no hidden chemical marker that tips off your digestive system. What matters is purity and contamination risk. Food-grade synthetic vanillin must meet safety standards that keep impurities in check.
Risks and Overuse
No food ingredient earns a perfect scorecard. Anything in large quantities can become harmful. For vanillin, the numbers back this up: extremely high amounts (well above anything you’d ever eat from flavored food) can irritate the stomach, prompt headaches, and in rare cases, set off mild allergic responses. As a food lover, I find peace in moderation. It wouldn’t make sense to fear cookies or yogurt, but it makes sense to avoid eating factory-sized portions.
Labeling and Consumer Choice
Reading ingredients lists gives people power over what goes into their bodies. Labels in many countries call out real vanilla extract versus artificial flavors. This lets folks who value plant-derived ingredients steer clear of synthetics, and it helps provide transparency for people with allergies or sensitivities. In my house, both products show up. For a treat with a special story or for holiday baking, I’ll reach for real vanilla. For a weekday dessert, vanillin does the trick.
What Makes the Conversation Important?
At the root, people want to trust what’s on their plates. Earning that trust means keeping food science transparent, making data public, and listening to people’s worries. The safest answer isn’t always the one with the prettiest marketing story—it’s the one supported by science and shared openly. Vanillin in moderation fits that description, at least for the people who aren’t allergic.
Learning and Making Better Choices
I tell friends and family: look up what you’re eating, check the labels, and ask questions if something seems off. The long history of vanillin’s use, combined with clear labeling and solid research, helps people make confident choices about what ends up in their cupboards. That’s what matters in the end—having the facts to make decisions in a world full of flavor.
What is the difference between Vanillin and vanilla extract?
Understanding the Roots of the Flavors
Everyone who bakes probably runs into these two little bottles: vanillin and vanilla extract. They both claim some connection to the familiar scent you get from a tray of sugar cookies or a scoop of classic ice cream. Thing is, not all flavorings come from the same source, and not all deliver the same experience.
Vanillin’s Origin and Limitations
Vanillin has one job—it brings a sweet, creamy note that most folks recognize right away as “vanilla.” It comes as a crystalline powder, often made in a lab using materials like wood pulp or even petrochemicals. The science says vanillin is the primary molecule responsible for the vanilla scent. Over 90% of food products labeled “vanilla flavored” actually use synthetic vanillin, so it’s everywhere. It keeps costs down and provides a clean, one-note flavor. But just a single molecule can’t replace the dozens of compounds in a vanilla bean.
The Rich World of Vanilla Extract
Pure vanilla extract gets its complexity from the vanilla bean, specifically cured pods of the vanilla orchid. This process takes months, sometimes even years, to draw out the full medley of flavors and aromas living in those pods. Alcohol and water soak the beans to pull out vanillin and about 200 other flavor molecules. Each batch tells a story—soil, weather, and curing all stamp their personality into the bottle. True vanilla extract needs to have at least 35% alcohol and 100 grams of vanilla beans per liter in the U.S., according to FDA standards.
What’s At Stake in the Kitchen
If you’ve ever tried swapping vanillin for vanilla extract in a homemade custard, you probably noticed something off. Synthetics might trick your nose, but your taste buds pick up the difference right away. Vanilla extract delivers a round, warm depth that’s irreplaceable, especially in recipes where vanilla stands out. Vanillin alone feels flat, sometimes even harsh. Chefs and home cooks with a few years under their aprons will tell you: those little brown flecks from real extract can transform a dish.
Health and Environmental Perspective
Natural vanilla extract has a clean track record for health. No allergen issues, no petroleum surprises. Synthetic vanillin, on the other hand, sometimes uses processes involving chemicals that not everyone feels comfortable running through their food. Growing vanilla beans supports farmers across Madagascar, Mexico, and Tahiti. Those communities rely on fair prices for harvests, and when consumers pick the real thing, they’re supporting rural economies. The flip side—synthetic vanillin helps keep prices low in a world where demand sometimes far outpaces bean supply.
Looking Ahead: Making Better Choices
As someone who grew up baking with both kinds, I’ve seen how frugal choices affect results. Using a splash of real vanilla in a slow-cooked custard wakes up all the hidden sweetness. Synthetic shortcuts do a decent job in big-batch, everyday treats, but for foods with just a handful of ingredients, real extract makes a world of difference. Cooking shows and recipe developers constantly remind us: flavor from real food always wins. When vanilla takes center stage, splurging on pure extract brings more joy to the table. For folks on a tight budget, it’s better to use real extract in small quantities than settle for artificial every time.
Does Vanillin have any allergens or side effects?
The Role of Vanillin in Everyday Food
Step inside any kitchen and there’s a fair chance you’ll spot a bottle of vanilla extract on the shelf. That familiar, warm aroma often comes from vanillin, the main compound in vanilla beans. These days, vanillin shows up in everything from ice cream to protein bars, not just desserts. Food makers turn to vanillin to give familiar comfort even in plant-based or sugar-free foods. Most of the vanillin used doesn’t come from vanilla pods, but from synthetic production, usually made from lignin or guaiacol. Synthetic or not, it keeps popping up in sweets, perfumes, candles, and medications.
Looking at Allergens in Vanillin
People worry about allergens in food and for good reason. The FDA keeps a list of top food allergens. Vanillin isn’t one of them. Most folks eat foods flavored with vanillin and feel just fine. Very rarely, some notice rashes or a mild reaction, usually from contact on the skin. As someone who’s baked for friends with allergies, I’ve always double-checked labels. Pure vanillin almost never causes immune system reactions. Problems often come from other ingredients mixed in with artificial flavors.
Synthetic Versus Natural: Any Real Difference?
I’ve met parents who ask if artificial flavors cause more issues than natural ones. Allergic reactions to vanillin, whether natural or synthetic, stay uncommon. A few studies, including a report found in “Contact Dermatitis,” show that workers in flavor factories sometimes get rashes from heavy vanillin exposure, but these cases are rare outside factories. Animal research using amounts far beyond normal food levels spotted mild stomach upset, but in real life, it would take eating a mountain of cookies to even come close.
Possible Side Effects and Their Likelihood
Here’s the thing: vanillin’s considered safe by agencies like the FDA and the European Food Safety Authority. Evidence shows you’d need to consume hundreds of times more vanillin than you’ll find in a slice of cake to risk harm. Every now and then, individuals with sensitive stomachs mention mild headaches or nausea after eating very strong artificial vanilla. That said, I’ve shared vanilla treats at events with all ages and have heard almost no complaints.
Who Should Stay Cautious?
Some rare medical situations deserve caution. A condition called vanillin intolerance, separate from a true allergy, leads to flushing, headaches, or stomach upset. This happens in a small subgroup. My cousin once asked about vanillin after her child, who has multiple food sensitivities, broke out in hives after a packaged cake. After checking, it seemed more likely the cake’s preservatives caused the flare, not vanillin. People with many allergies or who react to perfumes might avoid products containing vanillin just in case, but the risk remains low.
Safer Choices and Transparency
We all want to trust what’s in our food. Reading ingredient lists helps, especially for those with complicated allergies. Reputable food producers provide clear allergen labeling and typically specify whether vanillin comes from natural or artificial sources. Companies have started using more traceability in their flavor supply as well.
What Can Help?
Anyone worried about food reactions can keep a symptom diary. Doctors sometimes request patients cut out processed foods for a while, then re-introduce each item one by one to watch for any changes. Consulting a knowledgeable allergist gives peace of mind, especially for families coping with lots of sensitivities. Modern food safety rules already keep an eye on known risks, but those with special needs should communicate with brands and healthcare providers for the best results.
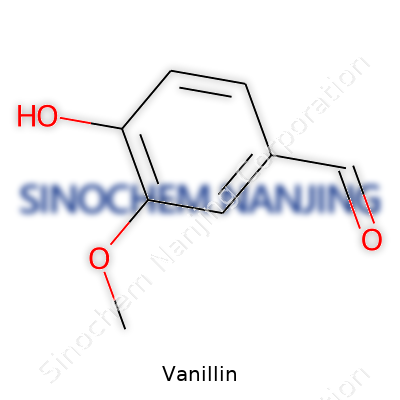
| Names | |
| Preferred IUPAC name | 4-hydroxy-3-methoxybenzaldehyde |
| Other names |
Vanilla Methyl vanillin Vanillic aldehyde 3-Methoxy-4-hydroxybenzaldehyde p-Vanillin |
| Pronunciation | /ˈvæn.ɪ.lɪn/ |
| Identifiers | |
| CAS Number | 121-33-5 |
| Beilstein Reference | 1209220 |
| ChEBI | CHEBI:18344 |
| ChEMBL | CHEMBL42020 |
| ChemSpider | 880 |
| DrugBank | DB09462 |
| ECHA InfoCard | 100.005.537 |
| EC Number | 2.1.1.42 |
| Gmelin Reference | 41212 |
| KEGG | C01432 |
| MeSH | D014636 |
| PubChem CID | 1183 |
| RTECS number | YO8400000 |
| UNII | JAI93QX3SP |
| UN number | UN3077 |
| CompTox Dashboard (EPA) | DTXSID7020182 |
| Properties | |
| Chemical formula | C8H8O3 |
| Molar mass | 152.15 g/mol |
| Appearance | white to pale yellow crystalline powder |
| Odor | Vanilla-like |
| Density | 1.06 g/cm3 |
| Solubility in water | 10 g/L (20 °C) |
| log P | 1.21 |
| Vapor pressure | 0.001 hPa (25 °C) |
| Acidity (pKa) | 7.4 |
| Basicity (pKb) | 7.88 |
| Magnetic susceptibility (χ) | -8.63×10⁻⁶ |
| Refractive index (nD) | 1.609 |
| Viscosity | 1.18 mPa·s (25 °C) |
| Dipole moment | 2.75 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 145.5 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -504.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -3219 kJ/mol |
| Pharmacology | |
| ATC code | A16AX10 |
| Hazards | |
| Main hazards | May cause respiratory irritation. May cause skin irritation. May cause eye irritation. |
| GHS labelling | GHS02, GHS07 |
| Pictograms | GHS07 |
| Signal word | Warning |
| Hazard statements | H302, H317 |
| Precautionary statements | P210, P261, P264, P271, P272, P273, P301+P312, P302+P352, P305+P351+P338, P330, P337+P313, P362+P364, P370+P378, P403+P233, P501 |
| NFPA 704 (fire diamond) | 2-1-0 |
| Flash point | 158°C |
| Autoignition temperature | 350 °C |
| Lethal dose or concentration | LD50 (oral, rat): 1580 mg/kg |
| LD50 (median dose) | LD50 (median dose) of Vanillin: "1580 mg/kg (oral, rat) |
| PEL (Permissible) | 10 mg/m³ |
| REL (Recommended) | 100 mg/kg |
| Related compounds | |
| Related compounds |
Ethylvanillin Isovanillin Vanillic acid Guaiacol Acetovanillone |

