Taking a Closer Look at Vanadium Tetrachloride
Historical Development
People started experimenting with vanadium compounds over a hundred years ago, searching for new ways to transform minerals into useful products. Chemists discovered vanadium tetrachloride after breakthroughs with mineral acids and metallic ores. European labs, in the late 1800s, saw rapid progress as researchers isolated new chlorides from vanadium. Methods advanced from crude distillation and improvised glassware to carefully controlled synthesis, with vanadium tetrachloride emerging alongside other halide compounds. The ability to isolate and work with this bright, red-brown liquid pushed forward organometallic chemistry and opened up pathways for new industrial catalysts. After the turn of the century, vanadium tetrachloride became part of academic and industrial toolkits, allowing researchers to lay the foundation for a wide range of modern chemical processes.
Product Overview
Vanadium tetrachloride is a thin liquid at room temperature with a notable deep red color. Unlike other chloride compounds that show up as powders or dull crystals, this one stands out right on the shelf — fleeing from moisture and air. Factories frequently rely on this chemical to bring vanadium into organic synthesis, polymer production, and metal refining. Its lone vanadium atom, linked to four chlorine atoms, makes it both reactive and somewhat aggressive. Scientists often reach for it as a starting material when building up more complex vanadium-based catalysts, especially in olefin polymerization or fine chemical synthesis. In the right hands, this volatile liquid becomes a Swiss army knife for shifting oxidation states and introducing vanadium in ways that simpler vanadium salts just can't match.
Physical and Chemical Properties
Vanadium tetrachloride presents itself with a boiling point near 154 °C and freezes only at −24 °C. As soon as the bottle opens, its pungent fumes fill the air — a reminder of its readiness to react. The compound weighs in with a molar mass of about 192.75 grams per mole. Put a drop on a metal surface and it eats away, corroding many standard metals and leaving stains that are tough to scrub out. Its density, a bit greater than water, means it settles in glass containers and that’s usually where it has to stay. Chemists respect its moisture sensitivity: a stray bit of humidity triggers violent hydrolysis, popping out clouds of hydrogen chloride gas and a messy, sticky residue. The compound's readiness to go from the +4 to +5 or +3 oxidation states underpins its value in catalysis and redox transformations.
Technical Specifications and Labeling
Legitimate vendors label vanadium tetrachloride as VCl4. Shipping labels warn of corrosive action and toxic vapors, with hazard codes tying back to both inhalation and severe reactivity with water. Typically, purity levels run above 98%, tracked by gas chromatography and titration. Laboratories look for minimal contamination by VCl3 or VCl5. Standard bottles come in amber glass, sometimes under vacuum seals, and always feature clear handling protocols. Safety Data Sheets detail thresholds for exposure, first aid measures, and emergency containment steps. Regulatory codes such as CAS 7632-51-1 crop up on inventory management logs and international shipping manifests, ensuring clarity from the warehouse to the hood.
Preparation Method
The classic route for preparing vanadium tetrachloride involves a direct reaction between vanadium metal and dry chlorine gas at elevated temperatures, usually around 160-200 °C. This process proceeds briskly in sealed apparatus, demanding stringent moisture control from start to finish. Any drop of water spells trouble — not just for the yield, but for safety too. Chemists also sometimes start with vanadium pentoxide (V2O5), blending it with carbon and flowing chlorine through a hot tube. This reduces and chlorinates the oxide to generate vanadium tetrachloride, which can be caught by condensation in chilled receivers. Only careful manipulation blocks side products from creeping in, and every run has to navigate the balance between maximum conversion and minimum byproducts.
Chemical Reactions and Modifications
Vanadium tetrachloride lends itself to a variety of powerful reactions. Its high oxidation state gives it a hand in oxidation and substitution reactions. Rattling a bottle of VCl4 near water or alcohol means trouble — hydrolysis sparks off vanadium oxychloride and clouds of HCl, leaving glass etched and operators on high alert. Its redox potential makes it a workhorse in organic synthesis; it slips into alkene polymerizations as a co-catalyst, especially for making polyethylenes and polypropylenes. Researchers tinker with ligand exchange, swapping chlorides for alkoxides, amines, or phosphines to push the compound into custom coordination complexes. Reductive chemistry brings in reducing agents to lower the vanadium oxidation state, paving the way for the stepwise creation of VCl3 or even vanadium metal. Each reaction draws on the twin features of volatility and reactivity, making careful laboratory technique a must.
Synonyms and Product Names
While vanadium tetrachloride remains the preferred term among academic chemists, catalogs and Material Safety Data Sheets sometimes list it as vanadium(IV) chloride. Other synonyms include vanadium chloride, Vanadin(IV)-chloride (in Europe), or tetrachlorovanadium. International databases sometimes abbreviate it to VCl4. For customs or international trade, records often pull the UN number 2934, ensuring clear identification during transit. These aliases do not reflect differences in composition; they're simply rooted in language, nomenclature preference, or import requirements.
Safety and Operational Standards
Laboratory staff treat VCl4 with the same level of caution as other nasty halogenated agents. Splash protection, heavy gloves, and chemical hoods form the front line of defense. The compound's fuming character means no one ever opens a bottle outside properly ventilated enclosures. Leak checks and double containment limit risk. Spilled VCl4 reacts with sweat or moisture from the air, so emergency showers and eye washes stay close at hand. Storage practices echo experience from generations of chemists: only in tightly sealed glass, only away from organics, bases, or water sources. Disposal requires neutralization and professional handling, as fumes wreak havoc on plant steelwork and local air quality. High-volume users invest in detection meters and vapor scrubbers. Regulatory bodies—OSHA, REACH, and local fire codes—enforce strict boundaries on workplace exposure, storage quantity, and transport, all grounded in documented accident histories and peer-reviewed toxicological reports.
Application Area
Industry leans on vanadium tetrachloride in polymerization zones, where it anchors catalytic cycles for synthetic rubbers and hard-wearing plastics. Chevron Philips, Dow, and Japanese chemical leaders install VCl4 dosing units right on the reactor floor for propylene and ethylene chemistry. Fine chemical manufacturers value its ability to introduce vanadium into custom catalysts, driving everything from hydrocarbon transformations to select pharmaceutical syntheses. Metallurgists bring in VCl4 to refine alloys, introduce trace vanadium into specialty steels, and help produce superconductors. Academic research teams push boundaries by designing new catalyst systems for environmentally friendly oxidation reactions, or for replacing precious metals in industrial redox cycles. VCl4 also plays a part in pigment manufacture, ceramics, and as a chemical intermediate that can unlock dozens of downstream vanadium products.
Research and Development
Research labs dig deep into the coordination chemistry of vanadium tetrachloride. Its fourfold symmetry and variable oxidation numbers let scientists design catalysts that churn out new pharmaceuticals, packaging materials, and smart functional polymers. The flurry of work in sustainable chemical processes often runs up against the stability-versus-reactivity equation, so researchers constantly tweak ligands and solvents to control VCl4's behavior. Some groups explore vanadium's role in energy storage, especially in redox flow batteries where VCl4 chemistry can be adapted to new electrolytes. Funding agencies push projects that promise to cut waste or enable recyclable production loops — the kind of targets that global green chemistry movements demand. The compound sits at the fulcrum of curiosity-driven basic science and real-world, financially motivated industrial development.
Toxicity Research
Toxicological data on vanadium tetrachloride warn about more than burns: inhaling fumes or mist saps lung function, stings eyes, and deals lasting damage to skin. The hydrolysis byproduct, HCl, racks up extra danger—especially in confined laboratory or plant spaces. Animal trials and epidemiological studies rank vanadium tetrachloride with other Group IVB salts in terms of acute and chronic risks. Acute doses in lab animals lead to bronchial spasms, tissue necrosis, and severe inflammation. Workers with repeated exposure to vanadium salts sometimes report bronchitis-like symptoms or chronic airway irritation. Testing on liver, kidney, and blood markers in animal models flags metabolic impacts that should push operators to minimize workplace exposures. Regulatory guidelines cap exposure levels and prompt health surveillance for large operators, even as the scientific community continues monitoring subtle long-term effects.
Future Prospects
Vanadium tetrachloride's future looks tied to new technology. As batteries step up to meet electric grid demands, vanadium's redox capacity may land it right at the center of energy storage. Improved catalysts could unlock greener routes to plastics, fuels, and polymers, meeting both industry expectations and social pressure for sustainability. Advanced research probes ways to recycle vanadium chlorides from spent catalysts, closing the loop and lessening supply chain stress. Questions remain about environmental fate, workplace safety, and long-term ecological risk—fields hungry for more data. Efforts to tame vanadium tetrachloride's hazards, through new containment methods or less volatile derivatives, still rank high on chemists' to-do lists. Staying nimble, the chemical sector keeps eyes on emerging regulations and next-generation applications, hoping this fiery red liquid continues to prove its versatility and value.
What are the main uses of Vanadium Tetrachloride?
What It Brings to Industry
Vanadium tetrachloride isn’t a household name. Still, sit down with a chemist or someone working with metals, and you’ll likely hear about it. This compound carries a lot of weight in the world of chemical reactions and metal production. Companies lean on vanadium tetrachloride to extract pure vanadium, an element that shapes alloy performance in steel. If you have benefited from lighter bridges, stronger car parts, or improved tools, chances are, vanadium played a part and this chemical helped make it possible.
Role in Catalyst Production
Another key role lies in the chemical industry. Producers use vanadium tetrachloride to manufacture catalysts, especially those used in creating sulfuric acid. Sulfuric acid runs the show in fertilizer production, batteries, and detergent manufacturing. The numbers speak for themselves: global sulfuric acid production crossed 250 million tonnes last year, according to World Bank data. Without robust catalysts, achieving this scale doesn’t happen. Vanadium tetrachloride helps form vanadium pentoxide, which boosts catalyst efficiency and cuts down on waste—a tangible benefit from start to finish.
Electronics and Material Science
Researchers turn to vanadium tetrachloride when they want thin films for electronic devices. Demand for smaller and more powerful smartphones, solar panels, and sensors grows every year. The compound helps deposit a precise layer of vanadium oxide, which acts as a switch or “gatekeeper” for electrical signals. Experience in a university lab taught me how small shifts in material choices ripple across whole technologies. Swapping one chemical for another changes conductivity, durability, and safety in devices touching our daily lives. When vanadium tetrachloride shows up, it can mean devices last longer or work faster.
Challenges and Solutions in Handling
Despite its advantages, vanadium tetrachloride requires careful handling. It reacts strongly with air and moisture, releasing hydrochloric acid fumes and heat. I remember visiting a plant where an accidental spill shut down a section for days; workers had to neutralize acid and treat minor burns. Incidents like this highlight a need for better safety training, airtight storage, and robust emergency protocols. Manufacturers investing in updated safety equipment and clear labeling can reduce risks. There’s a lot of wisdom in learning from past accidents instead of repeating them.
Environmental Considerations
Chemicals like vanadium tetrachloride don’t vanish without a trace. Unchecked, they contaminate water, soil, and air. Strict controls and neutralization before disposal don’t just follow the law—they protect communities near industrial centers. Some newer plants are installing closed-loop systems that capture fumes and recycle byproducts. With a rising call for responsible manufacturing, these solutions matter as much as the chemistry itself.
Pushing Forward With Responsibility
Vanadium tetrachloride opens doors for innovation across big industries. Its uses boost steel, drive large-scale chemical production, and open up possibilities for electronics. Responsible storage, rigorous staff training, and updated disposal methods keep the benefits while limiting the risks. As technology and demand shift, so does the way we handle such powerful compounds. New solutions often shine brightest under the pressure of necessity.
What is the chemical formula and structure of Vanadium Tetrachloride?
Understanding the Formula
In a basic chemistry class, you’ll find that formulas give structure to compounds that sound intimidating. Vanadium tetrachloride looks complex, but at its core, it comes down to a simple combination: one atom of vanadium and four atoms of chlorine. Chemically, it’s written as VCl4. This arrangement comes from the innate behavior of vanadium when it reacts with chlorine gas at elevated temperatures—there is a direct result of each vanadium atom pairing up with four chlorines.
Getting to Know Its Structure
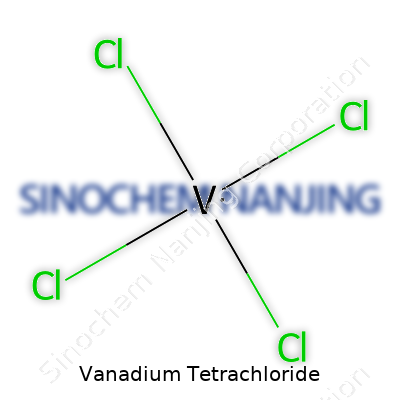
The way these atoms come together matters more than most people realize. In VCl4, vanadium sits at the center, bonding directly to four chlorine atoms in a symmetrical configuration. If you drew it with sticks and balls, it would show a tetrahedral structure, looking a lot like a tripod with four legs. Each chlorine comes off at an angle, which nobody set out to design—it’s just the natural result of their electron arrangements.
This shape helps vanadium tetrachloride stay as a liquid under room temperature if you seal it away from moisture. You wouldn’t want to open a bottle without gloves; the fumes can be tough on the lungs, and it stains the air with a deep, reddish-brown color. I remember working in a university lab and cracking open a tiny bottle—instantly, the room filled with a pungent smell, forcing everyone to the safety shower. That incident drove home the value of respecting even a few letters scrawled on a chemical label.
Why It Stands Out
Vanadium tetrachloride draws attention in industrial settings and scientific research for good reason. It serves as an important starting material for getting pure vanadium metal, which finds its way into specialty steel and aerospace applications. Chemists also use it to prepare other vanadium compounds. If you ever visit a catalyst manufacturing facility, you’ll find VCl4 on the list of backbone chemicals that help kickstart reactions—especially those in the production of plastics or sulfuric acid.
The science checks out on its stability, at least in a dry bottle. Bring it near water, and the chlorine reacts fast, giving off clouds of hydrochloric acid and making a mess in the fume hood. Labs store it under strict controls for good reason—too much moisture, and the chlorides escape into the air. Handling it safely isn’t just a best practice, it’s a necessity if you want to avoid a ruined experiment, a corroded workspace, or in some cases, a trip to the emergency room.
Tackling Safety and Environmental Concerns
The hazards attached to vanadium tetrachloride remind people that even everyday scientific pursuits can risk health and the environment. The regulatory agencies outline strict rules for its transport and disposal. Any accidental spill means calling in a professional cleanup team. Growing awareness around chemical safety in workplaces has reduced the number of mishaps involving VCl4. Wearing airtight goggles, gloves, and working in properly ventilated enclosures now count as routine, not extra, care for anyone expecting to handle this substance regularly.
To decrease risks, chemical suppliers have improved packaging and labeling. Some research labs swap it for less reactive alternatives or use smaller, diluted quantities. Government programs now train students and workers in better hazard management, contributing to fewer injuries every year. It comes down to combining up-to-date equipment with proper habits—simple tools and straightforward precautions solving what might otherwise become emergencies.
Final Thoughts on Responsible Use
Vanadium tetrachloride’s formula, VCl4, marks a meeting point between pure chemistry and practical safety. Knowledge, respect, and preparedness mean researchers can keep unleashing its potential without putting health or the environment on the line. It isn’t fear that keeps vanadium tetrachloride under control—it’s consistent, responsible action.
What are the storage and handling requirements for Vanadium Tetrachloride?
The Challenge of Storing a Reactive Chemical
Vanadium tetrachloride isn’t something you just toss on a shelf. Anyone who works in a lab knows that some chemicals act more like ticking time bombs than docile powders or liquids. I remember a chemist friend joking that certain bottles demand the same respect as a loaded mousetrap. Vanadium tetrachloride lands right in that category. This deep red liquid reacts fiercely with moisture. A trace of water—think steamy air in summer, a raindrop on your glove—unleashes a violent, lung-biting plume of hydrogen chloride gas. Anyone with a nose still remembers that sting long after leaving the fume hood.
Practical Precautions: Keeping It Dry Is Non-Negotiable
If you store vanadium tetrachloride in a regular plastic bottle, you end up with leaks, corrosion, or broken glass. The chemical chews through many common materials, including certain plastics, so lab supply shops recommend bottles made from glass or specific resistant plastics with proper seals. Even then, the container doesn't rest on an open shelf. Dry, cool, and absolutely moisture-free storage wins the day. A desiccator or a tightly sealed dry box earns its keep with this compound. Humidity sneaks through even the smallest cracks, so scientists use desiccants—think silica gel or activated alumina—to guard against water vapor.
Ventilation Is Not Optional
Lab veterans know fume hoods aren’t just expensive furniture. I recall watching someone set up a transfer outside the hood for speed—he regretted it within seconds. Vanadium tetrachloride’s vapors attack the throat and eyes before anyone can finish saying, “What’s that smell?” Proper handling goes hand-in-hand with good ventilation. Airflow whisks away the dangerous fumes, making a fume hood more of a lifeline than a luxury. It’s also wise to have well-maintained gas sensors and a plan for accidental releases. Time wasted fumbling for the emergency manual could cost real harm.
Personal Protection Matters More Than Looks
Gloves, splash-proof goggles, and lab coats feel like overkill until that first splash or puff. Disposable nitrile gloves often work better than latex, as vanadium tetrachloride can cruise right through natural rubber. Heavy-duty face shields count as serious insurance. My old professor always said, “Dress for the job you don’t want to encounter.” His advice holds up when handling this type of chemical.
Labeling and Inventory: Respect the Risk
Every container in a professional lab wears clear labels. When vanadium tetrachloride sits on a shelf, labels should broadcast its dangers—corrosive, moisture-reactive, and toxic. Mixing it up with other bottles could mean disaster, so every transfer, even from bottle to small reaction flask, gets logged in the record book. Tracking inventory helps spot missing or leaky samples before a problem escalates.
Serious Spills and Waste Disposal
Nobody wants to play firefighter with spilled vanadium tetrachloride. Every responsible lab pairs clear guidance with absorbent spill kits built for acidic chemicals. Waste disposal isn’t a “toss it down the drain” job: professional hazardous waste services pick up and neutralize leftovers. Regulators lay down harsh penalties, but real risk to health and the environment keeps strict routines in place, even for small amounts.
Room for Improvement
Lab managers and safety teams should review protocols every year. Training refreshers keep even seasoned researchers alert. Suppliers might improve packaging with tamper-resistant caps or integrated desiccants, and digital inventory systems warn faster when a container ages past its prime. In the end, trust is earned by safe habits built day-by-day—not by trusting luck or shortcuts.
What safety precautions should be taken when working with Vanadium Tetrachloride?
Understanding the Risks
Vanadium tetrachloride grabs immediate attention with its deep red color and dense fumes. Exposure brings a real risk of severe burns and lung damage. I spent years in laboratories, and I still get a punch in the gut remembering the sting of chlorine leaks—VCl4 smells even sharper and eats through clothing quickly. Anyone around this stuff cannot rely on instincts alone; training and protection matter. Both OSHA and the National Institute for Occupational Safety and Health list vanadium compounds among their chemical hazards for good reason.
The Right Kind of Protection
No one in their right mind touches vanadium tetrachloride without proper gear. Regular lab coats and exam gloves don’t cut it. You need chemical-resistant aprons, butyl rubber gloves, and splash-proof goggles. I’ve seen goggles fog up fast, so face shields help. Fume hoods are non-negotiable. Even a small drop in open air releases clouds of corrosive gas. Portable gas detectors pick up leaks before you smell them, a trick that has saved plenty of noses and lungs. Breathing in the fumes feels like inhaling bleach: instant pain followed by deep, raspy coughing. Respirators with acid gas cartridges give an extra layer of security when handling larger volumes or cleaning up spills.
Storage and Handling
Vanadium tetrachloride reacts fast with moisture. It bursts into a choking white mist. It eats holes in shelf liners and corrodes metal cabinets. Glass bottles with tight PTFE-lined caps slow down leaks. Store these in ventilated corrosives cabinets, away from heat, water, and even humidity. My old chemistry supervisor kept a spill kit with calcium carbonate, not sawdust or paper towels—these catch fire. I learned the hard way to replace caps immediately after use and keep bottles out of reach except during weighing or transfer. Never pipette by mouth or try ‘quick fixes’ for drips; that led to emergency showers in the past. Clean spills with neutralizers, not water, since water makes fumes worse. Keep a written protocol at eye level near storage areas, with emergency contacts and quick instructions.
Training and Medical Response
New team members should shadow experienced staff for weeks before solo work. Videos and quizzes only go so far; most mistakes happen fast, in split seconds. Refresher drills for chemical spills and eyewash use should happen every month. Back in graduate school, we paired up—no one ever worked alone after midnight, since tiredness causes careless moves. Medical checks for workers who handle vanadium tetrachloride do more than just reassure—they catch lung changes and kidney stress before they become real damage. Keep first aid kits close, with cool water for rinsing skin or eyes, and make sure staff know the route to the nearest emergency room. Time counts more than perfection after an incident.
Respect the Chemical
Working with vanadium tetrachloride calls for careful habits, not just fancy gear. Double-check transfers, measure slowly, and keep your gear clean. Listen to your coworkers’ reminders; they may save you from a painful mistake. Over the years, I’ve found that clear protocols, regular drills, and keeping the material locked down tight give the best shot at safety. These approaches help protect health, maintain clean records, and keep the lab open for business.
How should Vanadium Tetrachloride be disposed of safely?
What Makes Vanadium Tetrachloride a Challenge
Vanadium Tetrachloride, a deep red liquid, carries a reputation for trouble. It fumes hard in humid air, creates hydrochloric acid vapor when exposed to water, and quickly corrodes anything unprepared for its temperament. Over the years, I’ve seen labs take shortcuts, and the fallout always outweighs the convenience. One small spill creates a choking cloud. A single mistake in draining can lead to months of environmental headaches.
Health and Environmental Hazards
Touching or inhaling the fumes brings on burns and respiratory distress. The fluid eats through organic tissue and metal alike. If dumped down drains or tossed in regular waste, it maps out a quick route to local water supplies. Both hydrochloric acid and vanadium ions—products of its breakdown—call for special caution. Human error carries steep costs. The EPA has flagged heavy metals like vanadium as persistent toxins. They don’t break down easily and build up in soil and water, cycling into food and putting kids and workers at risk.
What’s Required for Safe Disposal
Hunting for easy fixes isn’t realistic. Under US law, only licensed hazardous waste handlers can accept this kind of waste, and for good reason. Untrained staff with protective gloves aren’t enough. The Department of Transportation and EPA have both dragged companies into court and levied fines for cutting corners. At my first job, a rushed intern poured some leftover vanadium tetrachloride into organic solvent waste. The safety shower ran the rest of the afternoon, and the repairs stuck with us for weeks.
Deactivation: The Critical First Step
Labs that use vanadium tetrachloride often practice chemical deactivation before sending it off-site. Deactivation means turning the chemical into something less aggressive. The usual route involves slowly adding it to a large stirred tank of ice-cold water, making sure local ventilation handles the acid vapors, then neutralizing the formed acids with sodium carbonate or a similar base. If done wrong, foam and heat shoot up, pushing clouds of hydrochloric acid every direction. Skilled chemists trained for hazard control oversee this process. A city’s wastewater system cannot handle surprises like this—in one case, the acid corroded pipes beneath a university chemistry building, leading to a midnight scramble.
Professional Transport and Disposal
After neutralization, remaining solids and liquids go into approved, labeled chemical waste containers. Licensed professionals haul them off using specialized sealed containers. These pros don’t just drive the barrels out back. They follow shipping regulations set out by the Department of Transportation and hand off records of every step. Modern disposal firms break down the neutralized waste further or send it for secure burial at chemical waste dumps built to isolate heavy metals.
What Can Be Improved?
Some chemists and lab managers wait until a shock or accident forces a real disposal plan. Regular training, clear labeling, and emergency drills make a difference. Lab techs, grad students, and even custodians should feel comfortable identifying and reporting spills. Local fire departments carry resources for hazardous materials—partnerships help, but these crews need to know what chemicals rest in local storage before emergencies hit.
As tighter rules continue to push for transparency, good habits prove more useful than any complex device. Building a routine around routinized, verified disposal creates a safer workplace. Safe disposal of vanadium tetrachloride always starts with respecting its risks, and ignoring those lessons never ends well.
| Names | |
| Preferred IUPAC name | tetrachlorovanadium(IV) |
| Other names |
Vanadium(IV) chloride Vanadium chloride Vanadium tetrachloride (VCl4) |
| Pronunciation | /vəˈneɪdiəm ˌtɛtəˈklɔːraɪd/ |
| Identifiers | |
| CAS Number | 7632-51-1 |
| 3D model (JSmol) | `JSmol.loadInline("data/model/vanadium-tetrachloride.xyz")` |
| Beilstein Reference | 3588252 |
| ChEBI | CHEBI:30442 |
| ChEMBL | CHEMBL1201801 |
| ChemSpider | 57926 |
| DrugBank | DB14546 |
| ECHA InfoCard | 100.032.464 |
| EC Number | 231-712-8 |
| Gmelin Reference | Gmelin Reference: 15862 |
| KEGG | C18639 |
| MeSH | D014636 |
| PubChem CID | 24413 |
| RTECS number | YW2800000 |
| UNII | IKO2P7X8E4 |
| UN number | 2857 |
| Properties | |
| Chemical formula | VCl4 |
| Molar mass | 195.75 g/mol |
| Appearance | Reddish-brown liquid |
| Odor | Pungent odor |
| Density | 1.86 g/mL at 25 °C |
| Solubility in water | Decomposes |
| log P | -0.9 |
| Vapor pressure | 53 mmHg (20°C) |
| Acidity (pKa) | -2. |
| Basicity (pKb) | -4.7 |
| Magnetic susceptibility (χ) | +11000.0e-6 cm³/mol |
| Refractive index (nD) | 1.787 |
| Viscosity | 1.6 cP (20 °C) |
| Dipole moment | 1.61 D |
| Thermochemistry | |
| Std molar entropy (S⦵298) | 274.8 J·mol⁻¹·K⁻¹ |
| Std enthalpy of formation (ΔfH⦵298) | -539.6 kJ/mol |
| Std enthalpy of combustion (ΔcH⦵298) | -604.1 kJ·mol⁻¹ |
| Pharmacology | |
| ATC code | V03AE04 |
| Hazards | |
| Main hazards | Toxic if swallowed, inhaled, or in contact with skin; causes severe burns and eye damage; corrosive to metals; releases toxic chlorine gas on contact with water. |
| GHS labelling | GHS05, GHS06 |
| Pictograms | GHS05,GHS06 |
| Signal word | Danger |
| Hazard statements | H301 + H331: Toxic if swallowed or if inhaled. H314: Causes severe skin burns and eye damage. H410: Very toxic to aquatic life with long lasting effects. |
| Precautionary statements | H260, H314, H318, H330, H400, P210, P222, P231+P232, P260, P261, P264, P271, P280, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P320, P363, P370+P378, P391, P403+P233, P405, P501 |
| NFPA 704 (fire diamond) | 3-0-2 |
| Autoignition temperature | 154 °C (309 °F; 427 K) |
| Lethal dose or concentration | LD50 oral rat 333 mg/kg |
| LD50 (median dose) | LD50 (median dose): Oral rat 333 mg/kg |
| NIOSH | CN2975000 |
| PEL (Permissible) | 1 mg/m³ |
| REL (Recommended) | Glovebag |
| IDLH (Immediate danger) | 100 mg/m3 |
| Related compounds | |
| Related compounds |
Vanadium(III) chloride Vanadium(V) oxytrichloride Vanadium oxychloride |

